Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Changlin Allen Zheng and Version 2 by Conner Chen.
Boron and boron compounds have been extensively studied together in the history and development of lithium batteries, which are crucial to decarbonization in the automotive industry and beyond. As early as the year 2000, lithium bis(oxalato)borate, also known as LiBOB, was first reported for synthesis and use in electrolytes of LIBs. Because of the merits of being halide-free and more thermally stable than LiPF6, LiBOB has been proven beneficial either as a direct replacement for LiPF6 or as an electrolyte additive (2 wt%) in a nickel cobalt aluminum (NCA)—graphite system.
- boron
- boron compounds
- boric acid
- lithium battery
- lithium-ion batteries
1. Introduction to Boron and Lithium
During decarbonization, the trend of electrification continues to intensify and will drive the demand for batteries, especially for electric vehicles (EVs), where lithium plays a critical role. The challenge to improve battery performance has made boron, in various forms of compounds, a research topic in relation to lithium-ion batteries (LIBs) for decades.
Boron and lithium are similar elements in some ways. They are both considered light elements and less abundant in both present crustal concentrations and, indeed, in the universe. Their formation requires a further enrichment process that involves continental crust growth [1][2][1,2]. Furthermore, the extraction of lithium and boron together is common in existing mining operations and some prospective greenfield projects. Application wise, both elements could be used as flux in the making of vitreous materials, such as glass.
Why is boron used in batteries? Boron is a unique element in many respects. Firstly, boron is lightest element of the “metalloids”, which separates metals and non-metals in the periodic table. The fact that many boron compounds are electron deficient means that they have Lewis acidic characteristics. The four covalent bonds and the variety in the molecular structures of its compounds result in a wide range of chemical properties [3]. Consequently, its applications in urbanization are extremely diverse: glass and ceramics, oil and gases, polymer fire retardants, wood preservatives, fertilizers in agriculture, and many more. The benefit of using boron is often multifaceted. For instance, when sodium borate pentahydrate is used in fiberglass, it brings a unique combination of benefits to both the melting process and fiber properties; when disodium octaborate tetrahydrate (DOT) is used as an indoor wood preservative, it offers deep penetration into wood and residential benefits (such as low mammal toxicity).
2. Boron Used in Electrolytes
Battery mainly consists of four major components, which are cathodes, anodes, the electrolyte, and separators. Figure 1 shows the schematics of these components in conventional Li ion batteries (LIBs) and the movement of electrons, ions, and current flow under charging and discharging condition.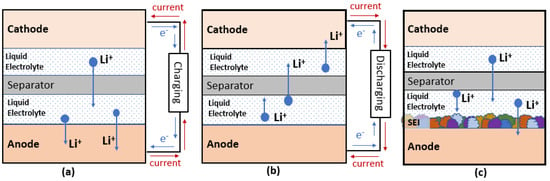
Figure 1.
Schematic diagram of LIBs for (
a
) charging (
b
) discharging and (
c
) formation of SEI.
