The sympathetic nervous system (SNS) originates in the ventral brainstem, where sympathetic premotor neurons are found. They are found predominantly in the rostral ventrolateral medulla (RVLM) and in the rostral ventromedial medulla (RVMM). These neurons project to the intermediolateral nucleus (IML, also known as the sympathetic preganglionic nucleus), which then projects to the dorsal root ganglia (DRG) for terminal output to peripheral organs which control heart rate, blood pressure, respiration, glycemia, vigilance and other physiological responses. When negative emotions are induced under chronic stress, the sympathetic nervous system is continuously activated and increases the release of catecholamines (such as epinephrine and norepinephrine). In a spontaneous colon tumor model, ablation of sympathetic premotor neurons in APCmin/+ mice reduces the number of polyps in the mouse intestine. Sympathetic denervation also leads to decreased tumorigenesis in a spontaneous prostate tumor mouse model. These results suggest that loss of SNS function may slow tumorigenesis.
- cancer
- central nervous system
- sympathetic nervous system
- adrenergic receptor
1. Stress Activates SNS-Related Neural Circuits
2. Sympathetic Nerve Fibers Release Neurotransmitters to Promote Tumor Progression
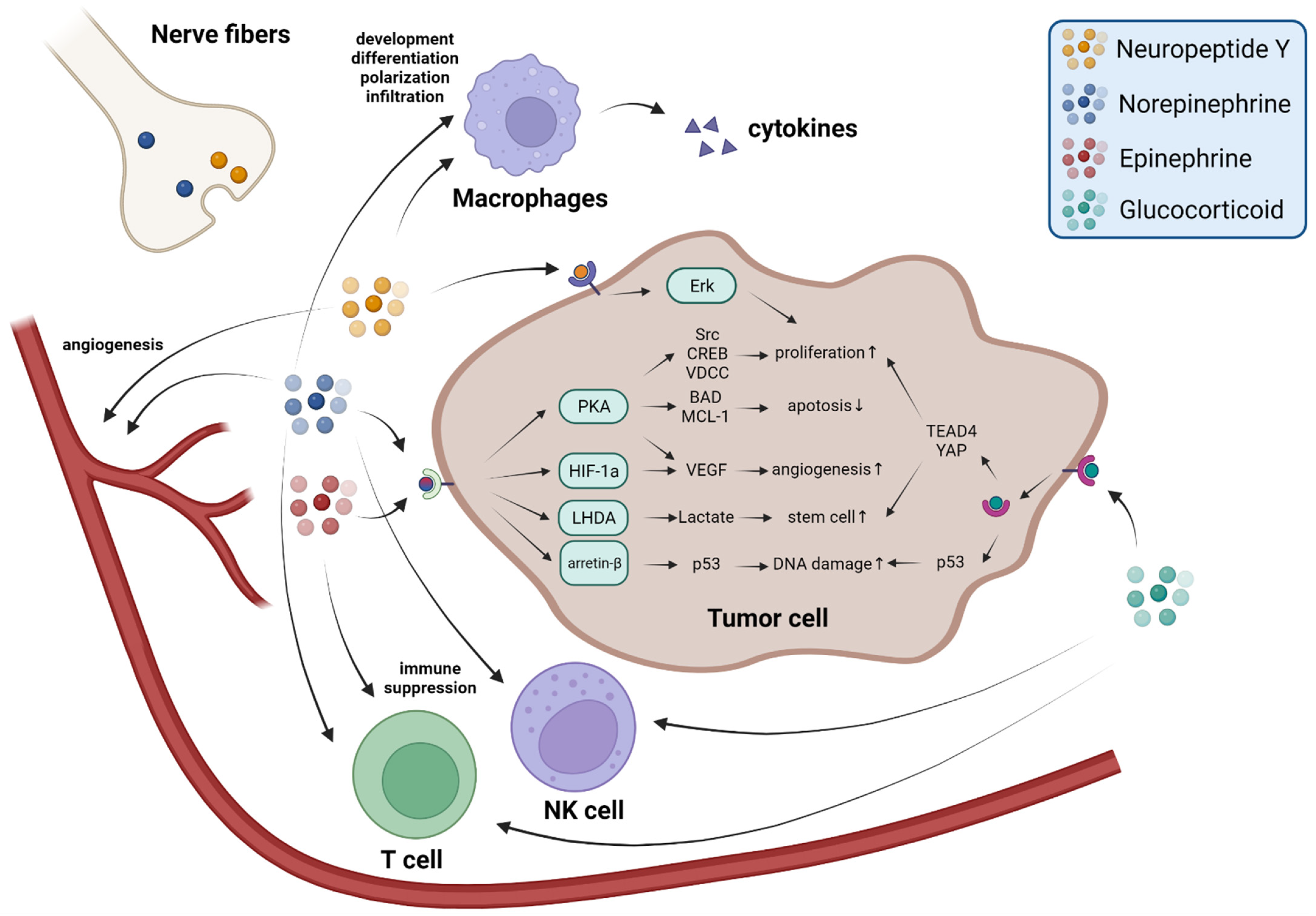
3. The Adrenal Medulla Secretes Epinephrine to Promote Tumor Progression
References
- Zhang, Z.; Li, Y.; Lv, X.; Zhao, L.; Wang, X. VLM catecholaminergic neurons control tumor growth by regulating CD8+ T cells. Proc. Natl. Acad. Sci. USA 2021, 118, e2103505118.
- Zhao, Z.; Wang, L.; Gao, W.; Hu, F.; Zhang, J.; Ren, Y.; Lin, R.; Feng, Q.; Cheng, M.; Ju, D.; et al. A Central Catecholaminergic Circuit Controls Blood Glucose Levels during Stress. Neuron 2017, 95, 138–152.e5.
- Calhoon, G.G.; Tye, K.M. Resolving the neural circuits of anxiety. Nat. Neurosci. 2015, 18, 1394–1404.
- Zhang, W.H.; Zhang, J.Y.; Holmes, A.; Pan, B.X. Amygdala Circuit Substrates for Stress Adaptation and Adversity. Biol. Psychiatry 2021, 89, 847–856.
- Yoshikawa, E.; Matsuoka, Y.; Yamasue, H.; Inagaki, M.; Nakano, T.; Akechi, T.; Kobayakawa, M.; Fujimori, M.; Nakaya, N.; Akizuki, N.; et al. Prefrontal cortex and amygdala volume in first minor or major depressive episode after cancer diagnosis. Biol. Psychiatry 2006, 59, 707–712.
- Leschak, C.J.; Dutcher, J.M.; Haltom, K.E.B.; Breen, E.C.; Bower, J.E.; Eisenberger, N.I. Associations between amygdala reactivity to social threat, perceived stress and C-reactive protein in breast cancer survivors. Soc. Cogn. Affect. Neurosci. 2020, 15, 1056–1063.
- Muscatell, K.A.; Eisenberger, N.I.; Dutcher, J.M.; Cole, S.W.; Bower, J.E. Links between inflammation, amygdala reactivity, and social support in breast cancer survivors. Brain Behav. Immun. 2016, 53, 34–38.
- Saha, S. Role of the central nucleus of the amygdala in the control of blood pressure: Descending pathways to medullary cardiovascular nuclei. Clin. Exp. Pharmacol. Physiol. 2005, 32, 450–456.
- Xiao, Q.; Zhou, X.; Wei, P.; Xie, L.; Han, Y.; Wang, J.; Cai, A.; Xu, F.; Tu, J.; Wang, L. A new GABAergic somatostatin projection from the BNST onto accumbal parvalbumin neurons controls anxiety. Mol. Psychiatry 2021, 26, 4719–4741.
- Gomes-de-Souza, L.; Costa-Ferreira, W.; Oliveira, L.A.; Benini, R.; Crestani, C.C. Cannabinoid receptor type 1 in the bed nucleus of the stria terminalis modulates cardiovascular responses to stress via local N-methyl-D-aspartate receptor/neuronal nitric oxide synthase/soluble guanylate cyclase/protein kinase G signaling. J. Psychopharmacol. 2020, 34, 429–440.
- Oliveira, L.A.; Gomes-de-Souza, L.; Benini, R.; Crestani, C.C. Control of cardiovascular responses to stress by CRF in the bed nucleus of stria terminalis is mediated by local NMDA/nNOS/sGC/PKG signaling. Psychoneuroendocrinology 2018, 89, 168–176.
- Nasimi, A.; Kafami, M. Vasopressin and sympathetic system mediate the cardiovascular effects of the angiotensin II in the bed nucleus of the stria terminalis in rat. Neurosci. Res. 2016, 108, 34–39.
- Kono, Y.; Yokota, S.; Fukushi, I.; Arima, Y.; Onimaru, H.; Okazaki, S.; Takeda, K.; Yazawa, I.; Yoshizawa, M.; Hasebe, Y.; et al. Structural and functional connectivity from the dorsomedial hypothalamus to the ventral medulla as a chronological amplifier of sympathetic outflow. Sci. Rep. 2020, 10, 13325.
- Ulrich-Lai, Y.M.; Herman, J.P. Neural regulation of endocrine and autonomic stress responses. Nat. Rev. Neurosci. 2009, 10, 397–409.
- Heydendael, W.; Sengupta, A.; Beck, S.; Bhatnagar, S. Optogenetic examination identifies a context-specific role for orexins/hypocretins in anxiety-related behavior. Physiol. Behav. 2014, 130, 182–190.
- Borniger, J.C.; Walker II, W.H.; Surbhi; Emmer, K.M.; Zhang, N.; Zalenski, A.A.; Muscarella, S.L.; Fitzgerald, J.A.; Smith, A.N.; Braam, C.J.; et al. A Role for Hypocretin/Orexin in Metabolic and Sleep Abnormalities in a Mouse Model of Non-metastatic Breast Cancer. Cell Metab. 2018, 28, 118–129.e5.
- Brechner, T.; Motyka, D.; Sherman, J. Growth enhancement of prolactin-sensitive mammary tumor by periaqueductal gray stimulation. Life Sci. 1983, 32, 525–530.
- Ni, H.D.; Xu, L.S.; Wang, Y.; Li, H.; An, K.; Liu, M.; Liu, Q.; Deng, H.; He, Q.; Huang, B.; et al. Astrocyte activation in the periaqueductal gray promotes descending facilitation to cancer-induced bone pain through the JNK MAPK signaling pathway. Mol. Pain 2019, 15, 1744806919831909.
- Li, P.; Zhang, Q.; Xiao, Z.; Yu, S.; Yan, Y.; Qin, Y. Activation of the P2X7 receptor in midbrain periaqueductal gray participates in the analgesic effect of tramadol in bone cancer pain rats. Mol. Pain 2018, 14, 1744806918803039.
- Benarroch, E.E. Descending monoaminergic pain modulation: Bidirectional control and clinical relevance. Neurology 2008, 71, 217–221.
- Yin, W.; Mei, L.; Sun, T.; Wang, Y.; Li, J.; Chen, C.; Farzinpour, Z.; Mao, Y.; Tao, W.; Li, J.; et al. A Central Amygdala-Ventrolateral Periaqueductal Gray Matter Pathway for Pain in a Mouse Model of Depression-like Behavior. Anesthesiology 2020, 132, 1175–1196.
- de Menezes, R.C.; Zaretsky, D.V.; Fontes, M.A.; DiMicco, J.A. Cardiovascular and thermal responses evoked from the periaqueductal grey require neuronal activity in the hypothalamus. J. Physiol. 2009, 587 Pt 6, 1201–1215.
- Adhikari, A.; Lerner, T.N.; Finkelstein, J.; Pak, S.; Jennings, J.H.; Davidson, T.J.; Ferenczi, E.; Gunaydin, L.A.; Mirzabekov, J.J.; Ye, L.; et al. Basomedial amygdala mediates top-down control of anxiety and fear. Nature 2015, 527, 179–185.
- Schaeuble, D.; Packard, A.E.B.; McKlveen, J.M.; Morano, R.; Fourman, S.; Smith, B.L.; Scheimann, J.R.; Packard, B.A.; Wilson, S.P.; James, J.; et al. Prefrontal Cortex Regulates Chronic Stress-Induced Cardiovascular Susceptibility. J. Am. Heart Assoc. 2019, 8, e014451.
- Pastor, V.; Medina, J.H. Medial prefrontal cortical control of reward- and aversion-based behavioral output: Bottom-up modulation. Eur. J. Neurosci. 2021, 53, 3039–3062.
- Whitton, A.E.; Treadway, M.T.; Pizzagalli, D.A. Reward processing dysfunction in major depression, bipolar disorder and schizophrenia. Curr. Opin. Psychiatry 2015, 28, 7–12.
- Ben-Shaanan, T.L.; Schiller, M.; Azulay-Debby, H.; Korin, B.; Boshnak, N.; Koren, T.; Krot, M.; Shakya, J.; Rahat, M.A.; Hakim, F.; et al. Modulation of anti-tumor immunity by the brain’s reward system. Nat. Commun. 2018, 9, 2723.
- Xu, X.R.; Xiao, Q.; Hong, Y.C.; Liu, Y.H.; Liu, Y.; Tu, J. Activation of dopaminergic VTA inputs to the mPFC ameliorates chronic stress-induced breast tumor progression. CNS Neurosci. Ther. 2021, 27, 206–219.
- Magnon, C.; Hall, S.J.; Lin, J.; Xue, X.; Gerber, L.; Freedland, S.J.; Frenette, P.S. Autonomic nerve development contributes to prostate cancer progression. Science 2013, 341, 1236361.
- Ayala, G.E.; Dai, H.; Powell, M.; Li, R.; Ding, Y.; Wheeler, T.M.; Shine, D.; Kadmon, D.; Thompson, T.; Miles, B.J.; et al. Cancer-related axonogenesis and neurogenesis in prostate cancer. Clin. Cancer Res. 2008, 14, 7593–7603.
- Kamiya, A.; Hayama, Y.; Kato, S.; Shimomura, A.; Shimomura, T.; Irie, K.; Kaneko, R.; Yanagawa, Y.; Kobayashi, K.; Ochiya, T. Genetic manipulation of autonomic nerve fiber innervation and activity and its effect on breast cancer progression. Nat. Neurosci. 2019, 22, 1289–1305.
- Thaker, P.H.; Han, L.Y.; Kamat, A.A.; Arevalo, J.M.; Takahashi, R.; Lu, C.; Jennings, N.B.; Armaiz-Pena, G.; Bankson, J.A.; Ravoori, M.; et al. Chronic stress promotes tumor growth and angiogenesis in a mouse model of ovarian carcinoma. Nat. Med. 2006, 12, 939–944.
- Gidron, Y.; Russ, K.; Tissarchondou, H.; Warner, J. The relation between psychological factors and DNA-damage: A critical review. Biol. Psychol. 2006, 72, 291–304.
- Flint, M.S.; Baum, A.; Episcopo, B.; Knickelbein, K.Z.; Liegey Dougall, A.J.; Chambers, W.H.; Jenkins, F.J. Chronic exposure to stress hormones promotes transformation and tumorigenicity of 3T3 mouse fibroblasts. Stress 2013, 16, 114–121.
- Hara, M.R.; Kovacs, J.J.; Whalen, E.J.; Rajagopal, S.; Strachan, R.T.; Grant, W.; Towers, A.J.; Williams, B.; Lam, C.M.; Xiao, K.; et al. A stress response pathway regulates DNA damage through beta2-adrenoreceptors and beta-arrestin-1. Nature 2011, 477, 349–353.
- Reeder, A.; Attar, M.; Nazario, L.; Bathula, C.; Zhang, A.; Hochbaum, D.; Roy, E.; Cooper, K.L.; Oesterreich, S.; Davidson, N.E.; et al. Stress hormones reduce the efficacy of paclitaxel in triple negative breast cancer through induction of DNA damage. Br. J. Cancer 2015, 112, 1461–1470.
- Renz, B.W.; Takahashi, R.; Tanaka, T.; Macchini, M.; Hayakawa, Y.; Dantes, Z.; Maurer, H.C.; Chen, X.; Jiang, Z.; Westphalen, C.B.; et al. beta2 Adrenergic-Neurotrophin Feedforward Loop Promotes Pancreatic Cancer. Cancer Cell 2018, 33, 75–90.e7.
- Armaiz-Pena, G.N.; Allen, J.K.; Cruz, A.; Stone, R.L.; Nick, A.M.; Lin, Y.G.; Han, L.Y.; Mangala, L.S.; Villares, G.J.; Vivas-Mejia, P.; et al. Src activation by beta-adrenoreceptors is a key switch for tumour metastasis. Nat. Commun. 2013, 4, 1403.
- Jang, H.J.; Boo, H.J.; Lee, H.J.; Min, H.Y.; Lee, H.Y. Chronic Stress Facilitates Lung Tumorigenesis by Promoting Exocytosis of IGF2 in Lung Epithelial Cells. Cancer Res. 2016, 76, 6607–6619.
- Zahalka, A.H.; Arnal-Estape, A.; Maryanovich, M.; Nakahara, F.; Cruz, C.D.; Finley, L.W.S.; Frenette, P.S. Adrenergic nerves activate an angio-metabolic switch in prostate cancer. Science 2017, 358, 321–326.
- Sloan, E.K.; Priceman, S.J.; Cox, B.F.; Yu, S.; Pimentel, M.A.; Tangkanangnukul, V.; Arevalo, J.M.; Morizono, K.; Karanikolas, B.D.; Wu, L.; et al. The sympathetic nervous system induces a metastatic switch in primary breast cancer. Cancer Res. 2010, 70, 7042–7052.
- Armaiz-Pena, G.N.; Gonzalez-Villasana, V.; Nagaraja, A.S.; Rodriguez-Aguayo, C.; Sadaoui, N.C.; Stone, R.L.; Matsuo, K.; Dalton, H.J.; Previs, R.A.; Jennings, N.B.; et al. Adrenergic regulation of monocyte chemotactic protein 1 leads to enhanced macrophage recruitment and ovarian carcinoma growth. Oncotarget 2015, 6, 4266–4273.
- Heidt, T.; Sager, H.B.; Courties, G.; Dutta, P.; Iwamoto, Y.; Zaltsman, A.; von Zur Muhlen, C.; Bode, C.; Fricchione, G.L.; Denninger, J.; et al. Chronic variable stress activates hematopoietic stem cells. Nat. Med. 2014, 20, 754–758.
- Qin, J.F.; Jin, F.J.; Li, N.; Guan, H.T.; Lan, L.; Ni, H.; Wang, Y. Adrenergic receptor beta2 activation by stress promotes breast cancer progression through macrophages M2 polarization in tumor microenvironment. BMB Rep. 2015, 48, 295–300.
- Zukowska-Grojec, Z.; Neuropeptide, Y. A novel sympathetic stress hormone and more. Ann. N. Y. Acad. Sci. 1995, 771, 219–233.
- Medeiros, P.J.; Al-Khazraji, B.K.; Novielli, N.M.; Postovit, L.M.; Chambers, A.F.; Jackson, D.N. Neuropeptide Y stimulates proliferation and migration in the 4T1 breast cancer cell line. Int. J. Cancer 2012, 131, 276–286.
- Lu, C.; Everhart, L.; Tilan, J.; Kuo, L.; Sun, C.C.; Munivenkatappa, R.B.; Jonsson-Rylander, A.C.; Sun, J.; Kuan-Celarier, A.; Li, L.; et al. Neuropeptide Y and its Y2 receptor: Potential targets in neuroblastoma therapy. Oncogene 2010, 29, 5630–5642.
- Ekstrand, A.J.; Cao, R.; Bjorndahl, M.; Nystrom, S.; Jonsson-Rylander, A.C.; Hassani, H.; Hallberg, B.; Nordlander, M.; Cao, Y. Deletion of neuropeptide Y (NPY) 2 receptor in mice results in blockage of NPY-induced angiogenesis and delayed wound healing. Proc. Natl. Acad. Sci. USA 2003, 100, 6033–6038.
- Lee, E.W.; Michalkiewicz, M.; Kitlinska, J.; Kalezic, I.; Switalska, H.; Yoo, P.; Sangkharat, A.; Ji, H.; Li, L.; Michalkiewicz, T.; et al. Neuropeptide Y induces ischemic angiogenesis and restores function of ischemic skeletal muscles. J. Clin. Investig. 2003, 111, 1853–1862.
- Chandrasekharan, B.; Nezami, B.G.; Srinivasan, S. Emerging neuropeptide targets in inflammation: NPY and VIP. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 304, G949–G957.
- Wurtman, R.J. Control of epinephrine synthesis by the pituitary and adrenal cortex: Possible role in the pathophysiology of chronic stress. Recent Adv. Biol. Psychiatry 1966, 9, 359–368.
- Feher, J. The Adrenal Medulla and Integration of Metabolic Control. In Quantitative Human Physiology; Academic Press: Cambridge, MA, USA, 2012; pp. 916–923.
- Zhang, X.; Zhang, Y.; He, Z.; Yin, K.; Li, B.; Zhang, L.; Xu, Z. Chronic stress promotes gastric cancer progression and metastasis: An essential role for ADRB2. Cell Death Dis. 2019, 10, 788.
- Hassan, S.; Karpova, Y.; Baiz, D.; Yancey, D.; Pullikuth, A.; Flores, A.; Register, T.; Cline, J.M.; D’Agostino, R., Jr.; Danial, N.; et al. Behavioral stress accelerates prostate cancer development in mice. J. Clin. Investig. 2013, 123, 874–886.
- Hassan, S.; Pullikuth, A.; Nelson, K.C.; Flores, A.; Karpova, Y.; Baiz, D.; Zhu, S.; Sui, G.; Huang, Y.; Choi, Y.A.; et al. beta2-adrenoreceptor Signaling Increases Therapy Resistance in Prostate Cancer by Upregulating MCL1. Mol. Cancer Res. 2020, 18, 1839–1848.
- Cui, B.; Luo, Y.; Tian, P.; Peng, F.; Lu, J.; Yang, Y.; Su, Q.; Liu, B.; Yu, J.; Luo, X.; et al. Stress-induced epinephrine enhances lactate dehydrogenase A and promotes breast cancer stem-like cells. J. Clin. Investig. 2019, 129, 1030–1046.
- Shan, T.; Ma, J.; Ma, Q.; Guo, K.; Guo, J.; Li, X.; Li, W.; Liu, J.; Huang, C.; Wang, F.; et al. beta2-AR-HIF-1alpha: A novel regulatory axis for stress-induced pancreatic tumor growth and angiogenesis. Curr. Mol. Med. 2013, 13, 1023–1034.
- Muthuswamy, R.; Okada, N.J.; Jenkins, F.J.; McGuire, K.; McAuliffe, P.F.; Zeh, H.J.; Bartlett, D.L.; Wallace, C.; Watkins, S.; Henning, J.D.; et al. Epinephrine promotes COX-2-dependent immune suppression in myeloid cells and cancer tissues. Brain Behav. Immun. 2017, 62, 78–86.
