Cardiovascular disease is the leading cause of death and disability, accounting for approximately one in three deaths worldwide. It is the leading cause of death in the United States; coronary artery disease (CAD) and ischemic heart disease (IHD) remain the leading causes of death attributable to cardiovascular disease in men and women. The incidence of atherosclerotic cardiovascular disease is increasing due to an increase in cardiac risk factors such as obesity, diabetes, and hypercholesterolemia in addition to an aging population. However, age-adjusted mortality rates are decreasing, likely due to improved medical therapies allowing patients to live longer with IHD. Although cardiovascular disease is more common in men, women with CAD have a worse short and long-term prognosis. In addition, women with symptoms of angina or who have had an abnormal cardiac stress test are less likely to be referred for additional diagnostic testing and initiated on guideline directed medical therapies.
- ischemic heart disease
- noninvasive imaging
- stress testing cardiovascular disease in women
1. Etiologies
2. Noninvasive Imaging Modalities
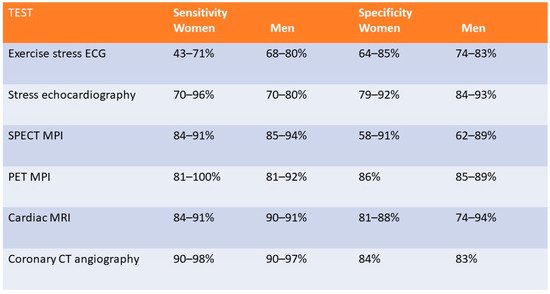
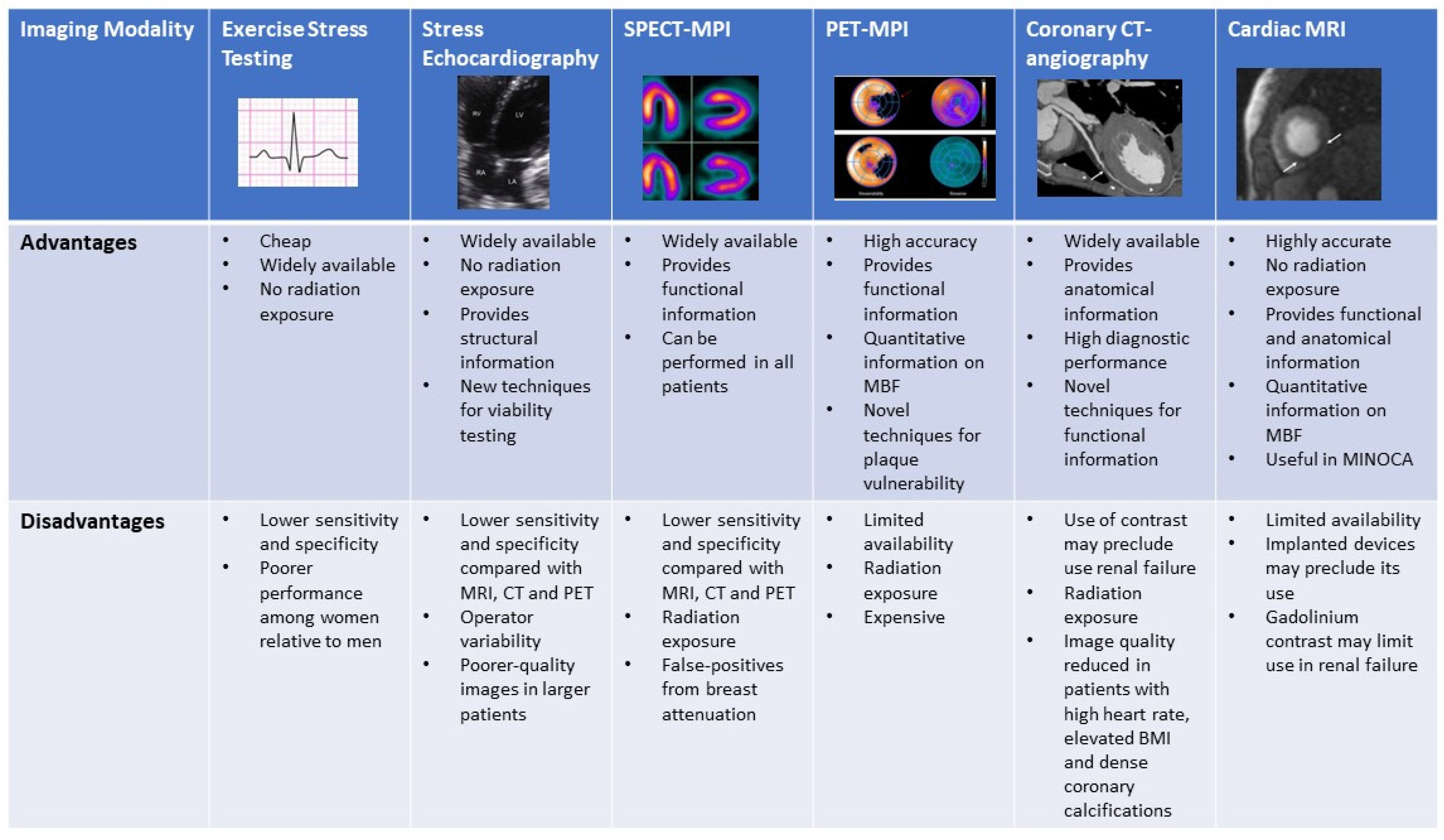
3. Exercise Stress Testing
4. Exercise and Dobutamine Stress Echocardiography
5. SPECT- Myocardial Perfusion Imaging (Exercise and Pharmacological)
6. Stress Positron Emission Tomography
7. Coronary CT Angiography
8. Stress Cardiac Magnetic Resonance Imaging
9. Safety of Non-Invasive Imaging Modalities for Ischemia in Women and Pregnancy
References
- Strong, J.P.; Malcom, G.T.; Newman, W.P.; Oalmann, M.C. Early Lesions of Atherosclerosis in Childhood and Youth: Natural History and Risk Factors. J. Am. Coll. Nutr. 1992, 11 (Suppl. 1), S51–S54.
- Pasupathy, S.; Tavella, R.; Beltrame, J.F. Myocardial Infarction with Nonobstructive Coronary Arteries (MINOCA). Circulation 2017, 135, 1490–1493.
- Tamis-Holland, J.E.; Jneid, H.; Reynolds, H.R.; Agewall, S.; Brilakis, E.S.; Brown, T.M.; Lerman, A.; Cushman, M.; Kumbhani, D.J.; Arslanian-Engoren, C.; et al. Contemporary Diagnosis and Management of Patients with Myocardial Infarction in the Absence of Obstructive Coronary Artery Disease: A Scientific Statement From the American Heart Association. Circulation 2019, 139, e891–e908.
- Safdar, B.; Spatz, E.S.; Dreyer, R.P.; Beltrame, J.F.; Lichtman, J.H.; Spertus, J.A.; Reynolds, H.R.; Geda, M.; Bueno, H.; Dziura, J.D.; et al. Presentation, Clinical Profile, and Prognosis of Young Patients with Myocardial Infarction with Nonobstructive Coronary Arteries (MINOCA): Results From the VIRGO Study. J. Am. Heart. Assoc. 2018, 7, e009174.
- Dreyer, R.P.; Tavella, R.; Curtis, J.P.; Wang, Y.; Pauspathy, S.; Messenger, J.; Rumsfeld, J.S.; Maddox, T.M.; Krumholz, H.M.; Spertus, J.A.; et al. Myocardial infarction with non-obstructive coronary arteries as compared with myocardial infarction and obstructive coronary disease: Outcomes in a Medicare population. Eur. Heart J. 2020, 41, 870–878.
- Brieger, D.; Eagle, K.A.; Goodman, S.G.; Steg, P.G.; Budaj, A.; White, K.; Montalescot, G. Acute Coronary Syndromes without Chest Pain, An Underdiagnosed and Undertreated High-Risk Group: Insights From The Global Registry of Acute Coronary Events. Chest 2004, 126, 461–469.
- Khan, N.A. Sex Differences in Acute Coronary Syndrome Symptom Presentation in Young Patients. JAMA Intern. Med. 2013, 173, 1863–1871.
- Vaccarino, V.; Rathore, S.S.; Wenger, N.K.; Frederick, P.D.; Abramson, J.L.; Barron, H.V.; Manhapra, A.; Mallik, S.; Krumholz, H.M. Sex and Racial Differences in the Management of Acute Myocardial Infarction, 1994 through 2002. N. Engl. J. Med. 2005, 353, 671–682.
- Murthy, V.L.; Naya, M.; Taqueti, V.R.; Foster, C.R.; Gaber, M.; Hainer, J.; Dorbala, S.; Blankstein, R.; Rimoldi, O.; Camici, P.G.; et al. Effects of Sex on Coronary Microvascular Dysfunction and Cardiac Outcomes. Circulation 2014, 129, 2518–2527.
- Kligfield, P.; Lauer, M.S. Exercise Electrocardiogram Testing. Circulation 2006, 114, 2070–2082.
- Lewis, J.F.; McGorray, S.; Lin, L.; Pepine, C.J.; Chaitman, B.; Doyle, M.; Etdmundowicz, D.; Sharaf, B.L.; Merz, C.N.B. Exercise treadmill testing using a modified exercise protocol in women with suspected myocardial ischemia: Findings from the National Heart, Lung and Blood Institute–sponsored Women’s Ischemia Syndrome Evaluation (WISE). Am. Heart J. 2005, 149, 527–533.
- Kwok, Y.; Kim, C.; Grady, D.; Segal, M.; Redberg, R. Meta-analysis of exercise testing to detect coronary artery disease in women. Am. J. Cardiol. 1999, 83, 660–666.
- Linhart, J.W.; Laws, J.G.; Satinsky, J.D. Maximum Treadmill Exercise Electrocardiography in Female Patients. Circulation 1974, 50, 1173–1178.
- Hlatky, M.A.; Pryor, D.B.; Harrell, F.E.; Califf, R.M.; Mark, D.B.; Rosati, R.A. Factors affecting sensitivity and specificity of exercise electrocardiography: Multivariable analysis. Am. J. Med. 1984, 77, 64–71.
- Weiner, D.A.; Ryan, T.J.; McCabe, C.H.; Kennedy, J.W.; Schloss, M.; Tristani, F.; Chaitman, B.R.; Fisher, L.D. Exercise Stress Testing. N. Engl. J. Med. 1979, 301, 230–235.
- Blankstein, R.; Ahmed, W.; Bamberg, F.; Rogers, I.S.; Schlett, C.L.; Nasir, K.; Fontes, J.D.; Tawakol, A.; Brady, T.J.; Nagurney, J.T.; et al. Comparison of Exercise Treadmill Testing with Cardiac Computed Tomography Angiography Among Patients Presenting to the Emergency Room with Chest Pain. Circ. Cardiovasc. Imaging 2012, 5, 233–242.
- Shaw, L.J.; Mieres, J.H.; Hendel, R.H.; Boden, W.E.; Gulati, M.; Veledar, E.; Hachamovitch, R.; Arrighi, J.A.; Merz, C.N.B.; Gibbons, R.J.; et al. Comparative Effectiveness of Exercise Electrocardiography with or without Myocardial Perfusion Single Photon Emission Computed Tomography in Women with Suspected Coronary Artery Disease. Circulation 2011, 124, 1239–1249.
- Smiseth, O.A.; Torp, H.; Opdahl, A.; Haugaa, K.H.; Urheim, S. Myocardial strain imaging: How useful is it in clinical decision making? Eur. Heart J. 2016, 37, 1196–1207.
- Hoffmann, R.; Altiok, E.; Heussen, N.; Hanrath, P.; Nowak, B.; Kaiser, H.-J.; Büll, U.; Kühl, H. Strain rate measurement by doppler echocardiography allows improvedassessment of myocardial viability inpatients with depressed left ventricular function. J. Am. Coll. Cardiol. 2002, 39, 443–449.
- Pellikka, P.A.; Arruda-Olson, A.; Chaudhry, F.A.; Chen, M.H.; Marshall, J.E.; Porter, T.R.; Sawada, S.G. Guidelines for Performance, Interpretation, and Application of Stress Echocardiography in Ischemic Heart Disease: From the American Society of Echocardiography. J. Am. Soc. Echocardiogr. 2020, 33, 1–41.e8.
- Kim, C.; Kwok, Y.S.; Heagerty, P.; Redberg, R. Pharmacologic stress testing for coronary disease diagnosis: A meta-analysis. Am. Heart J. 2001, 142, 934–944.
- Mieres, J.H.; Shaw, L.J.; Arai, A.; Budoff, M.J.; Flamm, S.D.; Hundley, W.G.; Marwick, T.H.; Mosca, L.; Patel, A.; Quinones, M.A.; et al. Role of Noninvasive Testing in the Clinical Evaluation of Women with Suspected Coronary Artery Disease. Circulation 2005, 111, 682–696.
- Roger, V.L.; Pellikka, P.A.; Bell, M.R.; Chow, C.W.H.; Bailey, K.R.; Seward, J.B. Sex and Test Verification Bias. Circulation 1997, 95, 405–410.
- Dionisopoulos, P.N.; Collins, J.D.; Smart, S.C.; Knickelbine, T.A.; Sagar, K.B. The value of dobutamine stress echocardiography for the detection of coronary artery disease in women. J. Am. Soc. Echocardiogr. 1997, 10, 811–817.
- Arruda-Olson Adelaide, M.; Juracan Eldyn, M.; Mahoney Douglas, W.; McCully Robert, B.; Roger Veronique, L.; Pellikka Patricia, A. Prognostic value of exercise echocardiographyin 5798 patients: Is there a gender difference? J. Am. Coll. Cardiol. 2002, 39, 625–631.
- Woodward, W.; Dockerill, C.; McCourt, A.; Upton, R.; O’Driscoll, J.; Balkhausen, K.; Chandrasekaran, B.; Firoozan, S.; Kardos, A.; Wong, K.; et al. Real-world performance and accuracy of stress echocardiography: The EVAREST observational multi-centre study. Eur. Heart J.-Cardiovasc. Imaging 2022, 23, 689–698.
- Gowd, B.M.P.; Heller, G.V.; Parker, M.W. Stress-only SPECT myocardial perfusion imaging: A review. J. Nucl. Cardiol. 2014, 21, 1200–1212.
- Santana-Boado, C.; Candell-Riera, J.; Castell-Conesa, J.; Aguadé-Bruix, S. Diagnostic accuracy of technetium-99m-MIBI myocardial SPECT in women and men. J. Nucl. Med. 1998, 39, 751.
- Kong, B.A.; Shaw, L.; Miller, D.D.; Chaitman, B.R. Comparison of accuracy for detecting coronary artery disease and side-effect profile of dipyridamole thallium-201 myocardial perfusion imaging in women versus men. Am. J. Cardiol. 1992, 70, 168–173.
- Amanullah, A.M.; Berman, D.S.; Hachamovitch, R.; Kiat, H.; Kang, X.; Friedman, J.D. Identification of Severe or Extensive Coronary Artery Disease in Women by Adenosine Technetium-99m Sestamibi SPECT. Am. J. Cardiol. 1997, 80, 132–137.
- Iskandar, A.; Limone, B.; Parker, M.W.; Perugini, A.; Kim, H.; Jones, C.; Calamari, B.; Coleman, C.; Heller, G.V. Gender differences in the diagnostic accuracy of SPECT myocardial perfusion imaging: A bivariate meta-analysis. J. Nucl. Cardiol. 2013, 20, 53–63.
- Hachamovitch, R.; Berman, D.S.; Kiat, H.; Bairey Merz, C.N.B.; Cohen, I.; Cabico, J.; Friedman, J.; Germano, G.; Van Train, K.F.; Dialmond, G.A. Effective risk stratification using exercise myocardial perfusion SPECT in women: Gender-related differences in prognostic nuclear testing. J. Am. Coll. Cardiol. 1996, 28, 34–44.
- Sharir, T.; Germano, G.; Kavanagh, P.B.; Lai, S.; Cohen, I.; Lewin, H.C.; Frietdman, J.D.; Zellweger, M.J.; Bermaln, D.S. Incremental Prognostic Value of Post-Stress Left Ventricular Ejection Fraction and Volume by Gated Myocardial Perfusion Single Photon Emission Computed Tomography. Circulation 1999, 100, 1035–1042.
- Mieres, J. A report of the American Society of Nuclear Cardiology Task Force on Women and Heart Disease (writing group on perfusion imaging in women). J. Nucl. Cardiol. 2003, 10, 95–101.
- Taqueti, V.R.; Dorbala, S.; Wolinsky, D.; Abbott, B.; Heller, G.V.; Bateman, T.M.; Mietres, J.H.; Phillips, L.M.; Wenger, N.K.; Shaw, L.J. Myocardial perfusion imaging in women for the evaluation of stable ischemic heart disease—State-of-the-evidence and clinical recommendations. J. Nucl. Cardiol. 2017, 24, 1402–1426.
- Berrington De Gonzalez, A.; Kim, K.-P.; Smith-Bindman, R.; McAreavey, D. Myocardial Perfusion Scans. Circulation 2010, 122, 2403–2410.
- Murthy, V.L.; Bateman, T.M.; Beanlands, R.S.; Berman, D.S.; Borges-Neto, S.; Chareonthaitawee, P.; Cerqueira, M.D.; deKemp, R.A.; DePuey, E.G.; Dilsizian, V.; et al. Clinical Quantification of Myocardial Blood Flow Using PET: Joint Position Paper of the SNMMI Cardiovascular Council and the ASNC. J. Nucl. Cardiol. 2018, 25, 269–297.
- Nakazato, R.; Berman, D.S.; Alexanderson, E.; Slomka, P. Myocardial perfusion imaging with PET. Imaging Med. 2013, 5, 35–46.
- Bateman, T.M.; Heller, G.V.; McGhie, A.I.; Friedman, J.D.; Case, J.A.; Bryngelson, J.R.; Hertenstein, G.K.; Moutray, K.L.; Reid, K.; Cullom, S.J. Diagnostic accuracy of rest/stress ECG-gated Rb-82 myocardial perfusion PET: Comparison with ECG-gated Tc-99m sestamibi SPECT. J. Nucl. Cardiol. 2006, 13, 24–33.
- Cerqueira, M.D.; Allman, K.C.; Ficaro, E.P.; Hansen, C.L.; Nichols, K.J.; Thompson, R.C.; Van Decker, W.A.; Yakovlevitch, M. Recommendations for reducing radiation exposure in myocardial perfusion imaging. J. Nucl. Cardiol. 2010, 17, 709–718.
- Sampson Uchechukwu, K.; Dorbala, S.; Limaye, A.; Kwong, R.; Di Carli Marcelo, F. Diagnostic Accuracy of Rubidium-82 Myocardial Perfusion Imaging with Hybrid Positron Emission Tomography/Computed Tomography in the Detection of Coronary Artery Disease. J. Am. Coll. Cardiol. 2007, 49, 1052–1058.
- Nandalur, K.R.; Dwamena, B.A.; Choudhri, A.F.; Nandalur, S.R.; Reddy, P.; Carlos, R.C. Diagnostic Performance of Positron Emission Tomography in the Detection of Coronary Artery Disease: A Meta-analysis. Acad. Radiol. 2008, 15, 444–451.
- Sdringola, S.; Nakagawa, K.; Nakagawa, Y.; Yusuf, S.W.; Boccalandro, F.; Mullani, N.; Haynie, M.; Hess, M.J.; Gould, K.L. Combined intense lifestyle and pharmacologic lipid treatment further reduce coronary events and myocardial perfusion abnormalities compared with usual-care cholesterol-lowering drugs in coronary artery disease. J. Am. Coll. Cardiol. 2003, 41, 263–272.
- Figueroa, A.L.; Subramanian, S.S.; Cury, R.C.; Truong, Q.A.; Gardecki, J.A.; Tearney, G.J.; Hoffmann, U.; Brady, T.J.; Tawakol, A. Distribution of inflammation within carotid atherosclerotic plaques with high-risk morphological features: A comparison between positron emission tomography activity, plaque morphology, and histopathology. Circ. Cardiovasc. Imaging 2012, 5, 69–77.
- Tarkin, J.M.; Joshi, F.R.; Evans, N.R.; Chowdhury, M.M.; Figg, N.L.; Shah, A.V.; Starks, L.T.; Martin-Garrido, A.; Manavaki, R.; Yu, E.; et al. Detection of Atherosclerotic Inflammation by 68Ga-DOTATATE PET Compared to FDG PET Imaging. J. Am. Coll. Cardiol. 2017, 69, 1774–1791.
- Joshi, N.V.; Vesey, A.T.; Williams, M.C.; Shah, A.S.V.; Calvert, P.A.; Craighead, F.H.M.; Yeoh, S.E.; Wallace, W.; Salter, D.; Fletcher, A.M.; et al. 18F-fluoride positron emission tomography for identification of ruptured and high-risk coronary atherosclerotic plaques: A prospective clinical trial. Lancet 2014, 383, 705–713.
- Joseph, P.; Tawakol, A. Imaging atherosclerosis with positron emission tomography. Eur. Heart J. 2016, 37, 2974–2980.
- Bhalla, S. Coronary Computed Tomography Angiography of Spontaneous Coronary Artery Dissection: A Case Report and Review of the Literature. Am. J. Case Rep. 2015, 16, 130–135.
- Truong, Q.A.; Rinehart, S.; Abbara, S.; Achenbach, S.; Berman, D.S.; Bullock-Palmer, R.; Carrascosa, P.; Chinnaiyan, K.M.; Dey, D.; Ferencik, M.; et al. Coronary computed tomographic imaging in women: An expert consensus statement from the Society of Cardiovascular Computed Tomography. J. Cardiovasc. Comput. Tomogr. 2018, 12, 451–466.
- Abdelrahman Khaled, M.; Chen Marcus, Y.; Dey Amit, K.; Virmani, R.; Finn Aloke, V.; Khamis Ramzi, Y.; Choi, A.D.; Min, J.K.; Williams, M.C.; Buckler, A.J.; et al. Coronary Computed Tomography Angiography From Clinical Uses to Emerging Technologies. J. Am. Coll. Cardiol. 2020, 76, 1226–1243.
- Haase, R.; Schlattmann, P.; Gueret, P.; Andreini, D.; Pontone, G.; Alkadhi, H.; Hausleiter, J.; Garcia, M.J.; Leschka, S.; Meijboom, W.B.; et al. Diagnosis of obstructive coronary artery disease using computed tomography angiography in patients with stable chest pain depending on clinical probability and in clinically important subgroups: Meta-analysis of individual patient data. BMJ 2019, 365, l1945.
- Arbab-Zadeh, A.; Di Carli, M.F.D.; Cerci, R.; George, R.T.; Chen, M.Y.; Dewey, M.; Niinuma, H.; Vavere, A.L.; Betoko, A.; Plotkin, M.; et al. Accuracy of Computed Tomographic Angiography and Single-Photon Emission Computed Tomography–Acquired Myocardial Perfusion Imaging for the Diagnosis of Coronary Artery Disease. Circ. Cardiovasc. Imaging 2015, 8, e003533.
- Chow, B.J.; Abraham, A.; Wells, G.A.; Chen, L.; Ruddy, T.D.; Yam, Y.; Govas, N.; Galbraith, P.D.; Dennie, C.; Beanlands, R.S. Diagnostic accuracy and impact of computed tomographic coronary angiography on utilization of invasive coronary angiography. Circ. Cardiovasc. Imaging 2009, 2, 16–23.
- Williams, M.C.; Moss, A.J.; Dweck, M.; Adamson, P.D.; Alam, S.; Hunter, A.; Shah, A.S.; Pawade, T.; Weir-McCall, J.R.; Roditi, G.; et al. Coronary Artery Plaque Characteristics Associated with Adverse Outcomes in the SCOT-HEART Study. J. Am. Coll. Cardiol. 2019, 73, 291–301.
- Neglia, D.; Rovai, D.; Caselli, C.; Pietila, M.; Teresinska, A.; Aguadé-Bruix, S.; Pizzi, M.N.; Todiere, G.; Gimelli, A.; Schroeder, S.; et al. Detection of Significant Coronary Artery Disease by Noninvasive Anatomical and Functional Imaging. Circ. Cardiovasc. Imaging 2015, 8, e002179.
- Douglas, P.S.; Hoffmann, U.; Patel, M.R.; Mark, D.B.; Al-Khalidi, H.R.; Cavanaugh, B.; Cole, J.; Dolor, R.J.; Fordyce, C.B.; Huang, M.; et al. Outcomes of Anatomical versus Functional Testing for Coronary Artery Disease. N. Engl. J. Med. 2015, 372, 1291–1300.
- Jug, B.; Gupta, M.; Papazian, J.; Li, D.N.; Tsang, J.; Bhatia, H.; Karlsbetrg, R.; Budoff, M. Diagnostic performance of 64-slice multidetector coronary computed tomographic angiography in women. J. Nucl. Cardiol. 2012, 19, 1154–1161.
- Nous, F.M.A.; Geisler, T.; Kruk, M.B.P.; Alkadhi, H.; Kitagawa, K.; Vliegenthart, R.; Hell, M.M.; Hausleiter, J.; Nguyen, P.K.; Budde, R.P.; et al. Dynamic Myocardial Perfusion CT for the Detection of Hemodynamically Significant Coronary Artery Disease. JACC Cardiovasc. Imaging 2022, 15, 75–87.
- Taylor, C.A.; Fonte, T.A.; Min, J.K. Computational Fluid Dynamics Applied to Cardiac Computed Tomography for Noninvasive Quantification of Fractional Flow Reserve. J. Am. Coll. Cardiol. 2013, 61, 2233–2241.
- Patel, A.R.; Salerno, M.; Kwong, R.Y.; Singh, A.; Heydari, B.; Kramer, C.M. Stress Cardiac Magnetic Resonance Myocardial Perfusion Imaging: JACC Review Topic of the Week. J. Am. Coll. Cardiol. 2021, 78, 1655–1668.
- Thomson, L.E.J.; Wei, J.; Agarwal, M.; Haft-Baradaran, A.; Shufelt, C.; Mehta, P.K.; Gill, E.B.; Johnson, B.D.; Kenkre, T.; Handberg, E.M.; et al. Cardiac Magnetic Resonance Myocardial Perfusion Reserve Index Is Reduced in Women with Coronary Microvascular Dysfunction. Circ. Cardiovasc. Imaging 2015, 8, e002481.
- Woolen, S.A.; Shankar, P.R.; Gagnier, J.J.; Maceachern, M.P.; Singer, L.; Davenport, M.S. Risk of Nephrogenic Systemic Fibrosis in Patients with Stage 4 or 5 Chronic Kidney Disease Receiving a Group II Gadolinium-Based Contrast Agent. JAMA Intern. Med. 2020, 180, 223.
- Barker, D.; Mason, G.; Schlosshan, D.; McLoughlin, H.; Tan, L.-B. Maximal exercise testing can be safely used to assess pregnant cardiac patients. Am. J. Obstet. Gynecol. 2005, 193, S80.
- Colletti, P.M.; Lee, K.H.; Elkayam, U. Cardiovascular imaging of the pregnant patient. AJR Am. J. Roentgenol. 2013, 200, 515–521.
