Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 4 by Conner Chen and Version 3 by Conner Chen.
It has been estimated that between 30 and 50 per cent of all injuries that take place throughout participation in a sport are the consequence of soft tissue injuries, and muscle injuries are the primary cause of physical disability. Due to the produced effects, manual therapy combined with high-intensity laser therapy (Hilt) or capacitive and resistive electrical transfer (TECAR) therapy, or even all three therapies used in a treatment plan, can provide optimal muscle recovery in a shorter time; therefore, the use of combined and not isolated recovery procedures is a better alternative in muscle recovery.
- TECAR therapy
- high-intensity laser therapy
- manual therapy
1. Manual Therapy vs. TECAR vs. HILT Therapy
Manual treatment is applied in treating muscle disorders in approximately every recovery program. Its effectiveness is debated, requiring much faster and more efficient recovery methods. Capacitive and resistive electrical transfer (TECAR), a type of diathermy, has recently been created as a form of deep thermotherapy and is used in sports medicine [1]. This device provides radio frequency energy, passing between an active and an inactive electrode and generating heat inside the body [2]. This therapy has shown that TECAR therapy is more efficient than a warm packet at increasing blood circulation, a conventional form of thermotherapy commonly used in clinical practice. Improving blood circulation plays an essential role in improving muscle recovery after fatigue. Thus, TECAR therapy can effectively improve muscle recovery after fatigue, which leads to maintaining and improving muscle flexibility [1].
Although manual therapy has been used to treat various muscle ailments since ancient times, for better and faster recovery of these ailments, more muscular stimulation is needed to have the desired effect (Figure 1). TECAR therapy combines manual therapy with deep thermotherapy, based on high-frequency electric currents, allowing for faster recovery of the affected muscles [3]. Due to the positive effects on muscle tissue, TECAR therapy seems to be a more helpful alternative in treating muscle disorders than conventional manual therapy, where cellular metabolism is not as strongly stimulated, and vasodilation is not as intense [4].
Laser treatment is a pain-free, non-invasive therapy that can be used to treat various clinical conditions. Laser therapy has significantly reduced acute and chronic pain, rheumatoid arthritis, chronic osteoarthritis, carpal tunnel syndrome, fibromyalgia, knee injuries, shoulder pain, and postoperative pain [5][6]. The reduction in pain after laser treatment results from its anti-inflammatory effects, the increased microcirculation and stimulation of immune processes, nerve regeneration, and the secretion of β-endorphins [6]. These properties of high-frequency laser therapy can influence the healing and regeneration of muscle tissue, which is recommended in most muscle conditions, both in athletes and non-athletes. Compared to TECAR therapy, HILT requires a much shorter treatment period, but its use with manual therapy does not seem as effective as using manual therapy with TECAR therapy because, as mentioned, the duration of treatment is short. The device does not allow a massage to be performed on the treated area. However, manipulations, massages, active or passive mobilisations, or other manual therapy techniques may be performed before or after HILT treatment (Figure 1).
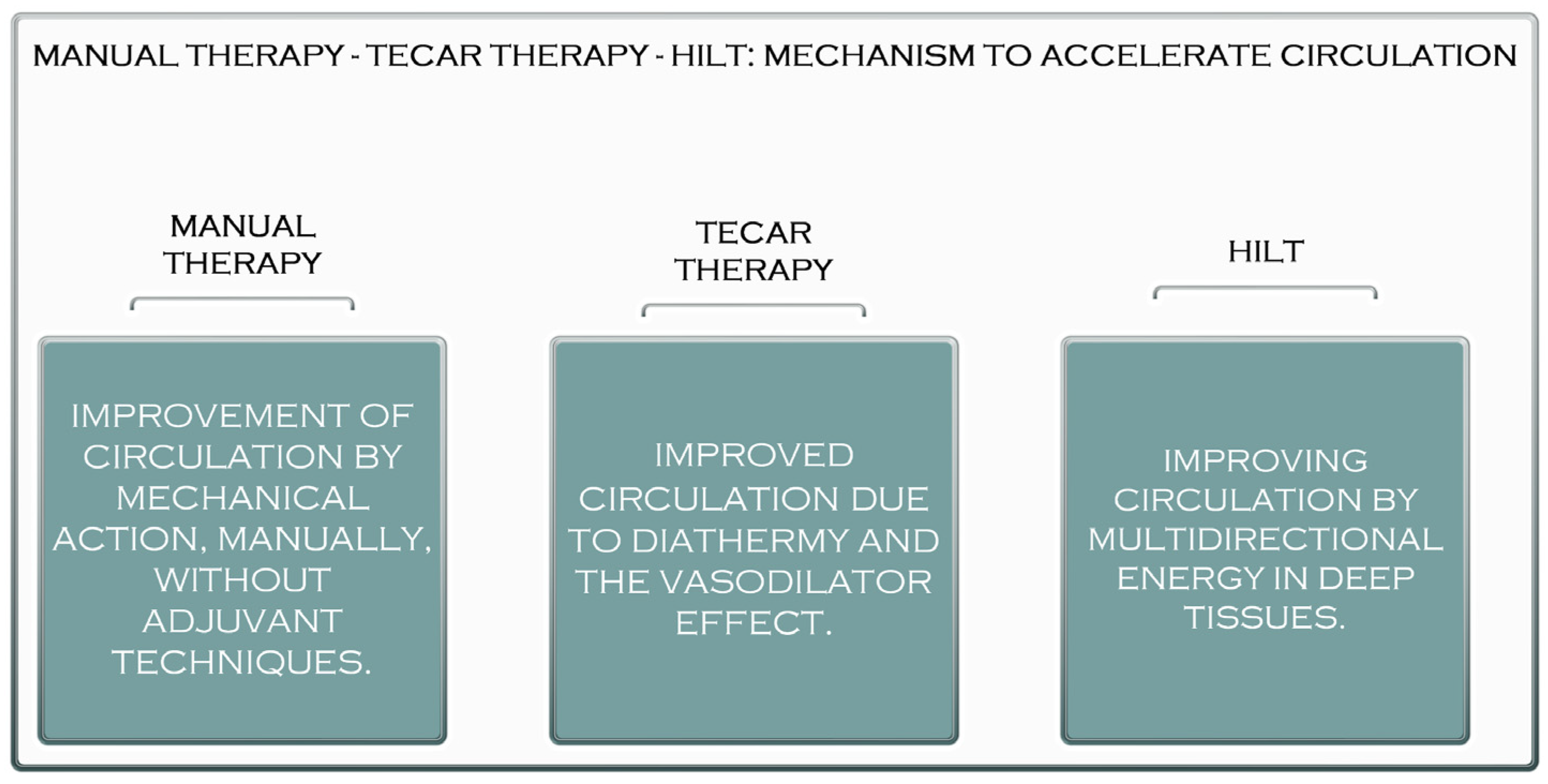
Figure 1. The acceleration mechanism of manual therapy, TECAR therapy, and HILT.
2. The Benefits of Manual Therapy in the Treatment of Muscle Diseases
Manual therapy is a technique used in physical therapy that involves using the hands to apply a force for therapeutic purposes. Manual therapy includes a wide range of therapeutic procedures such as massages, joint mobilisations/manipulations, myofascial releases, nerve manipulations, counter-stress, and acupressure (Figure 32).
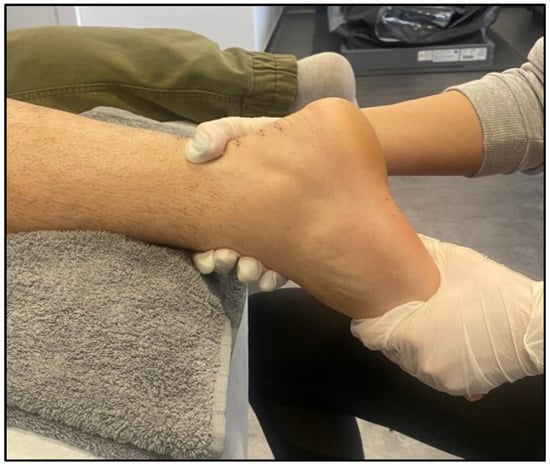
Figure 2. Manual therapy after Achilles tendinopathy.
Although manual therapy can be considered today as an old technique used by physical therapists, it remains one of the basic techniques of recovery used by physiotherapists in most recovery programs [7][8][9].
Manual therapy has been used to treat individuals suffering from various diseases since ancient times. The use of hands for healing dates back to the Old Testament and appears to have been advocated in the fifth century B.C. Hippocrates suggested the application of manual therapy, including traction prone to associated manipulation of the spine. He proved the efficiency of several manual approaches and proposed modifications to force delivery characteristics such as direction, pace, and frequency [10] (Figure 3).
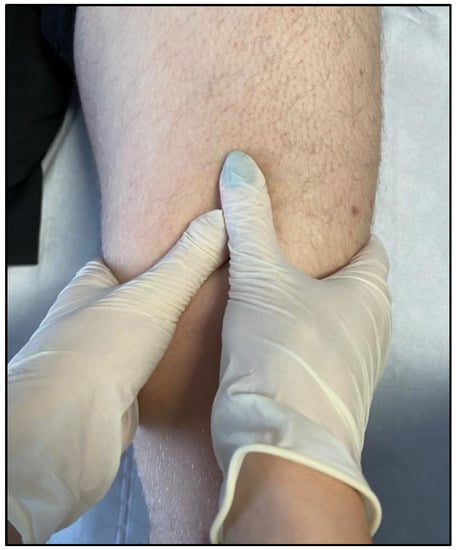
Figure 3. Relaxing massage of the thigh muscles (quadriceps).
Currently, manual treatment is applied in treating patients with various conditions, including musculoskeletal disorders, joint dysfunctions, spinal disorders, lymphedema, musculotendinous junction disorders, cystic fibrosis, and, most importantly, after immobilisation [11].
Among the essential benefits of manual therapy are modulating pain, increasing joint mobility, especially in stiff joints and increasing range of motion, accelerating tissue repair, improving tissue stability and extensibility, reducing soft tissue inflammation, reducing muscle tension, inducing relaxation, facilitating movement, and preparing segments for exercise therapy [12]
3. The Benefits of HILT Therapy in the Treatment of Muscle Disorders
As previously stated, laser treatment is a non-invasive treatment with minimal risk of adverse effects [13].
High-intensity laser treatment (HILT) is primarily employed in physio-kinetic therapy therapeutic regimens [14][15][16]. The primary distinction between HILT therapy and low-intensity laser therapy is that more giant beams (power > 500 mW) are irradiated to penetrate deeper, bringing a desired large amount of multidirectional energy into the deep tissues in a short time [17][18].
Moreover, the application techniques, the treatment time, and the cost of the device are different between these two generations of laser therapy [19][20]. However, High-Intensity Laser Therapy seems to have multiple benefits in the therapy of musculoskeletal disorders, precisely due to the effects produced in the tissues and also due to the reduction in recovery time [21][22][23].
HILT provides high tissue energy, and its optical energy forms dynamic vibrations. It then generates photochemical effects, such as increased mitochondrial oxidation, and it facilitates the formation of adenosine triphosphate (ATP) and ultimately leads to the rapid absorption of oedema and the elimination of exudates by increasing metabolism and blood circulation [24][25].
HILT has its own photomechanical, photothermal, and photochemical properties and has many therapeutic effects, including oedema reduction, analgesic effects, and physical stimulation [26]. Another benefit of HILT is its greater penetration strength and depth into deep tissues [27].
High-intensity laser treatment (HILT) has lately been applied in fundamental research and clinical rehabilitation practice with promising results (Watt-level performance) (high-intensity laser, Class IV laser) [28][29].
The high-intensity laser is usually used in two ways—pulsed and continuous [30]. Each mode impacts the tissue differently and triggers different therapeutic effects. The general therapeutic effects are bio-stimulation, pain relief, anti-inflammatory effects, superficial thermal effects, and muscle relaxation [31].
The high-intensity laser delivers energy to the cells, stimulates cellular metabolism, and promotes quicker resorption of the pro-inflammatory mediators [32].
Decreasing the concentration of pro-inflammatory mediators improves capillary permeability, resulting in the total eradication of inflammation and a speedier return to daily activities and sports. The high-intensity laser can provide extremely brief heartbeats at a maximum repetition rate [33]. This property can create real pressure. Pressure waves are transported through the tissue, stimulating the free nerve endings. According to the pain control mechanism, the mechanical stimulation of free nerve terminals causes their inhibition and, as a result, pain reduction [34]. The high-intensity laser has an immediate and long-lasting analgesic effect. The energy transferred by the continuous emission of HILT to the tissue causes superficial hyperthermia and vasodilation in the treated area [35]. As the blood perfusion increases, more blood passes through the treated area, and the muscles relax. In painful indications related to muscles, such as muscle injuries and muscle contracture, the patient feels immediate relief from discomfort triggered by muscle tension, and an amplitude of movement increases immediately [36].
Bio-stimulation means stimulating the body to help enhance healing and recovery at the cellular level. Mitochondria in cells metabolise oxygen. A cascade of respiratory enzymes processes oxygen and delivers it to ATP synthase, which synthesises the body’s energy source—ATP. As a result of the quicker exchange of oxygen and metabolites caused by laser irradiation, more oxygen atoms reach the mitochondria. Mitochondria are further stimulated to synthesise ATP faster. ATP allows faster R.N.A. and D.N.A. synthesis and leads to faster recovery, faster healing, and reduced oedema in the treated area [37].
Class IV laser therapy (High-Intensity Laser Therapy) can be used to treat a range of ailments, including knee, hip, and ankle osteoarthritis; rheumatoid arthritis; shoulder pain impact syndromes [38]; hip or shoulder bursitis; lumbar disc degeneration; herniated disc sciatica; tendonitis; lateral and medial epicondylitis; plantar fasciitis; and a variety of muscle disorders [39]. Compared to other procedures used in medical recovery, high-frequency laser therapy has a limited range of contraindications, but they must be considered before starting therapy using this type of laser [40].
Pregnancy is one of the absolute contraindications of HILT therapy, and although, to date, there are no documents indicating that laser therapy is harmful to a pregnant woman or her child, to be safe and to prevent any problems, the use of this procedure in pregnancy is not recommended [41].
Another contraindication to HILT is cancer. Therefore, the use of laser therapy in a patient who has cancer or is suspected of having cancer is not recommended.
There are several relative contraindications, which each physiotherapist takes into account in the use of HILT therapy, including:
- Thyroid disorders: The thyroid is known to be sensitive to light, and although it has not yet been found to harm the thyroid gland, caution and careful dosing are recommended when using the laser [42].
- Clotting problems: Laser therapy affects blood clotting, so its use for patients with such problems should be consulted with a specialist or even replaced with another procedure [43].
- Children: Although there is no contraindication to the use of the laser for children, its dose should be adjusted according to weight [44].
4. The Benefits of TECAR Therapy in the Treatment of Muscle Diseases
TECAR treatment is a type of non-invasive electrothermal treatment classified as deep thermotherapy, based on the planning of electric tides in the field of radiofrequency, constituting a monopolar capacitive resistive radiofrequency of 448 kHz [45][46][47][48].
TECAR or capacitive-resistive electric transfer (CRet) is often used in the treatment of muscle, joint, and tendon injuries in traumatology and sports [47][49][50]. CRet is a non-invasive electrothermal treatment characterised as deep thermotherapy based on the application of electric flows in the radiofrequency range of 300 kHz to 1.2 MHz [51][52][53]. Unlike superficial thermotherapy, which has a relatively limited ability to penetrate muscle tissue [54][55], CRet may create heat in deep muscle tissue, hence increasing haemoglobin saturation [56]. Applying an electromagnetic field of around 0.5 MHz to the human body is responsible for this form of therapy’s physiological effects. It has been discovered that an increase in blood perfusion is associated with a rise in body temperature, and other effects, such as cell proliferation, seem to be primarily connected to the current flow [57][58]. It has been demonstrated that cell growth begins at 0.00005 A per square millimetre [49][58].
TECAR therapy is characterised by its quick action and is employed in high-performance sports because it speeds up the recovery process [59][60]. Thermal changes in the neuromotor arrangement caused by TECAR treatment generate vasodilation, minimise muscle spasms, speed up cellular metabolism, and increase the extensibility of soft tissues [61][62].
TECAR therapy is available in capacitive (C.A.P.) and resistive (R.E.S.) modes. These modes are often given with a number of immaculate medical steel probes (electrodes) that have been manufactured [63]. According to TECAR, depending on the strength of the treated tissue, the two treatment techniques generate various tissue responses. When a cushioning ceramic coating functions as a dielectric mechanism (C.A.P.) on the active electrode, energy transfer generates only heat to the superficial layers of tissue, with selective action on soft tissues with low impedance (rich in water), e.g., adipose matter, muscles, and the lymphatic system [59].
The resistive mode targets denser tissues with more fat and fibre (such as bones, ligaments, and tendons). High-frequency waves penetrate deep into tissues and cause a rise in exchange and temperature, and recent studies have shown the effects of radiofrequency on skin microcirculation and intramuscular blood flow [64][65] (Figure 4).
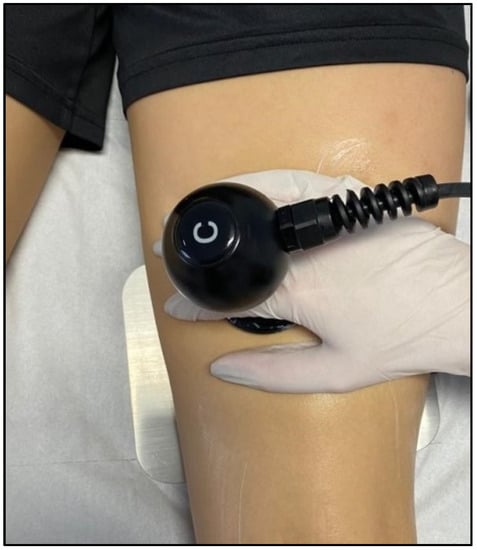
Figure 4. TECAR therapy in the treatment of hamstring muscles.
It should be noted that this new technology is a valuable tool in the therapy of different pathologies compared to other therapies used in recovery in terms of the presence and/or absence of specific positive effects while having distinctive features compared to conventional treatments [66]. It is guaranteed that TECAR therapy provides a balance between the therapist’s manual capacity and the unique energy that this technology emanates from the tissues, thus giving therapists and patients much more satisfactory results [67]. Among its positive effects is better blood circulation, which removes inflammatory catabolites [68]. TECAR therapy also substantially improves blood circulation in the peritendinous region and increases haemoglobin saturation [69]. In addition, this therapy warms deep tissues, stimulates vascularity, promotes the repose of tissues (particularly muscle tissue), and stimulates drainage (oedema and hematoma) [70]. Based on these properties, TECAR treatment is employed in most orthopaedic pathologies before the start of rehabilitation exercises [71].
Among the physiological effects of TECAR therapy are increased extensibility of collagen tissue due to reduced viscosity; reduced discomfort because of anti-irritant behaviour or release of endorphins; reduced spasms and muscle contractions due to reduced activity of secondary efferents; faster dissociation of oxygen because of more available haemoglobin [72], accompanied by a reduction in the activation energy of important chemical and metabolic reactions; vasodilation with increased local blood flow, contributing to the replenishment of oxygen and nutrients; the elimination of catabolites; and accelerated reabsorption of hemorrhagic masses [73].
References
- Elias, G.P.; Varley, M.C.; Wyckelsma, V.L.; McKenna, M.J.; Minahan, C.L.; Aughey, R.J. Effects of water immersion on post training recovery in Australian footballers. Int. J. Sports Physiol. Perform. 2012, 7, 357–366.
- Leeder, J.; Gissane, C.; van Someren, K.; Gregson, W.; Howatson, G. Cold water immersion and recovery from strenuous exercise: A meta-analysis. Br. J. Sports Med. 2012, 46, 233–240.
- Osti, R.; Pari, C.; Salvatori, G.; Massari, L. Tri-length laser therapy associated to TECAR therapy in the treatment of low-back pain in adults: A preliminary report of a prospective case series. Lasers Med. Sci. 2015, 30, 407–412.
- Pereira, W.M.; Ferreira, L.A.; Rossi, L.P.; Kerpers, I.I.; Grecco, S.L.A.; de Paula, A.R.J.; Oliveira, C.S. Influence of heat on fatigue and electromyographic activity of the biceps brachii muscle. J. Bodyw. Mov. Ther. 2011, 15, 478–484.
- Hayashi, K.; Arai, Y.C.; Ikemoto, T.; Nishihara, M.; Suzuki, S.; Hirakawa, T.; Matsuo, S.; Kobayashi, M.; Haruta, M.; Kawabata, Y.; et al. Predictive factors for the outcome of multidisciplinary treatments in chronic low back pain at the first multidisciplinary pain center of Japan. J. Phys. Ther. Sci. 2015, 27, 2901–2905.
- Alayat, M.S.; Atya, A.M.; Ali, M.M.; Shosha, T.M. Long-term effect of high-intensity laser therapy in the treatment of patients with chronic low back pain: A randomised blinded placebo-controlled trial. Lasers Med. Sci. 2014, 29, 1065–1073.
- Pillastrini, P.; Gardenghi, I.; Bonetti, F.; Capra, F.; Guccione, A.; Mugnai, R.; Violante, F.S. An updated overview of clinical guidelines for chronic low back pain management in primary care. Jt. Bone Spine 2012, 79, 176–185.
- Hernández-Bule, M.L.; Paíno, C.L.; Trillo, M.Á.; Úbeda, A. Electric stimulation at 448 kHz promotes proliferation of human mesenchymal stem cells. Cell Physiol. Biochem. 2014, 34, 1741–1755.
- Kato, S.; Saitoh, Y.; Miwa, N. Repressive effects of a capacitive-resistive electric transfer (CRet) hyperthermic apparatus combined with provitamin C on intracellular lipid-droplets formation in adipocytes. Int. J. Hyperth. 2013, 29, 30–37.
- Cheng, H.; Huang, Y.; Yue, H.; Fan, Y. Electrical Stimulation Promotes Stem Cell Neural Differentiation in Tissue Engineering. Stem Cells Int. 2021, 2021, 6697574.
- Oostendorp, R.A.B. Credibility of manual therapy is at stake “Where do we go from here?”. J. Man Manip. Ther. 2018, 26, 189–192.
- Reid, D.; Cook, C.; Sizer, P.S. Is orthopedic manipulative physical therapy not fashionable anymore? Lessons learned from 2016 IFOMPT meeting and future directions. J. Man Manip. Ther. 2017, 25, 1–2.
- Babatunde, O.O.; Jordan, J.L.; Van der Windt, D.A.; Hill, J.C.; Foster, N.E.; Protheroe, J. Effective treatment options for musculoskeletal pain in primary care: A systematic overview of current evidence. PLoS ONE 2017, 12, e0178621.
- Kim, G.J.; Choi, J.; Lee, S.; Jeon, C.; Lee, K. The effects of high intensity laser therapy on pain and function in patients with knee osteoarthritis. J. Phys. Ther. Sci. 2016, 28, 3197–3199.
- Conforti, M.; Fachinetti, G.P. High power laser therapy treatment compared to simple segmental physical rehabilitation in whiplash injuries (1° and 2° grade of the Quebec Task Force classification) involving muscles and ligaments. Muscles Ligaments Tendons J. 2013, 3, 106–111.
- Ezzati, K.; Fekrazad, R.; Raoufi, Z. The effects of photo-biomodulation therapy on post-surgical pain. J. Lasers Med. Sci. 2019, 10, 79–85.
- Thabet, A.A.E.; Elsodany, A.M.; Battecha, K.H.; Alshehri, M.A.; Refaat, B. High-intensity laser therapy versus pulsed electromagnetic field in the treatment of primary dysmenorrhea. J. Phys. Ther. Sci. 2017, 29, 1742–1748.
- Thabet, A.A.E.; Mahran, H.G.; Ebid, A.A.; Alshehri, M.A. Effect of pulsed high intensity laser therapy on delayed caesarean section healing in diabetic women. J. Phys. Ther. Sci. 2018, 30, 570–575.
- Pekyavas, N.O.; Baltaci, G. Short-term effects of high-intensity laser therapy, manual therapy, and Kinesio taping in patients with subacromial impingement syndrome. Lasers Med. Sci. 2016, 31, 1133–1141.
- Ordahan, B.; Karahan, A.Y.; Kaydok, E. The effect of high-intensity versus low-level laser therapy in the management of plantar fasciitis: A randomised clinical trial. Lasers Med. Sci. 2018, 33, 1363–1369.
- Taradaj, J.; Rajfur, K.; Shay, B.; Rajfur, J.; Ptaszkowski, K.; Walewicz, K.; Dymarek, R.; Sopel, M.; Rosińczuk, J. Photo-biomodulation using high—Or low-level laser irradiations in patients with lumbar disc degenerative changes: Disappointing outcomes and remarks. Clin. Interv. Aging 2018, 13, 1445–1455.
- Song, H.J.; Seo, H.J.; Lee, Y.; Kim, S.K. Effectiveness of high-intensity laser therapy in the treatment of musculoskeletal disorders: A systematic review and meta-analysis of randomised controlled trials. Medicine 2018, 97, e13126.
- Thoomes, E.J.; Scholten-Peeters, W.; Koes, B.; Falla, D.; Verhagen, A.P.; Clin, J. The effectiveness of conservative treatment for patients with cervical radiculopathy: A systematic review. Pain 2013, 29, 1073–1086.
- Kheshie, A.R.; Alayat, M.S.; Ali, M.M. High-intensity versus low-level laser therapy in the treatment of patients with knee osteoarthritis: A randomised controlled trial. Lasers Med. Sci. 2014, 29, 1371–1376.
- Yıldırıım, M.A.; Uçar, D.; Öneş, K. Comparison of therapeutic duration of therapeutic ultrasound in patients with knee osteoarthritis. J. Phys. Ther. Sci. 2015, 27, 3667–3670.
- Boyraz, I.; Yildiz, A.; Koc, B.; Sarman, H. Comparison of high-intensity laser therapy and ultrasound treatment in the patients with lumbar discopathy. Biomed. Res. Int. 2015, 2015, 304328.
- Zielińska, P.; Nicpoń, J.; Kiełbowicz, Z.; Soroko, M.; Dudek, K.; Zaborski, D. Effects of high intensity laser therapy in the treatment of tendon and ligament injuries in performance horses. Animals 2020, 10, 1327.
- Ezzati, K.; Laakso, E.L.; Salari, A.; Hasannejad, A.; Fekrazad, R.; Aris, A. The beneficial effects of high-intensity laser therapy and co-interventions on musculoskeletal pain management: A systematic review. J. Lasers Med. Sci. 2020, 11, 81–90.
- Abdelbasset, W.K.; Nambi, G.; Alsubaie, S.F.; Abodonya, A.M.; Saleh, A.K.; Ataalla, N.N.; Ibrahim, A.A.; Tantawy, S.A.; Kamel, D.M.; Verma, A.; et al. A randomised comparative study between high-intensity and low-level laser therapy in the treatment of chronic nonspecific low back pain. Evid. Based Complement. Altern. Med. 2020, 2020, 1350281.
- Pellegrino, R.; Paolucci, T.; Brindisino, F.; Mondardini, P.; Di Iorio, A.; Moretti, A.; Iolascon, G. Effectiveness of High-Intensity Laser Therapy Plus Ultrasound-Guided Peritendinous Hyaluronic Acid Compared to Therapeutic Exercise for Patients with Lateral Elbow Tendinopathy. J. Clin. Med. 2022, 11, 5492.
- Alayat, M.S.; El Soudany, A.M.; El Fiky, A.A. Efficacy of high—And low-level laser therapy in the treatment of Bell’s palsy: A randomised double-blind placebo-controlled trial. Lasers Med. Sci. 2014, 29, 335–342.
- Alayat, M.S.; El Soudany, A.M.; Ali, M.E. Efficacy of multiwave locked system laser on pain and function in patients with chronic neck pain: A randomised placebo-controlled trial. Photomed. Laser Surg. 2017, 35, 450–455.
- Alayat, M.S.; Aly, T.H.A.; Elsayed, A.E.M.; Fadil, A.S.M. Efficacy of pulsed Nd:YAG laser in the treatment of patients with knee osteoarthritis: A randomised controlled trial. Lasers Med. Sci. 2017, 32, 503–511.
- Thabet, A.A.E.; Alshehri, M.A. Effect of pulsed high-intensity laser therapy on pain, adhesions, and quality of life in women having endometriosis: A randomized controlled trial. Photomed. Laser Surg. 2018, 36, 363–369.
- Tkocz, P.; Matusz, T.; Kosowski, Ł.; Walewicz, K.; Argier, Ł.; Kuszewski, M.; Hagner-Derengowska, M.; Ptaszkowski, K.; Dymarek, R.; Taradaj, J. A Randomised-Controlled Clinical Study Examining the Effect of High-Intensity Laser Therapy (HILT) on the Management of Painful Calcaneal Spur with Plantar Fasciitis. J. Clin. Med. 2021, 10, 4891.
- Alayat, M.S.M.; Abdel-Kafy, E.M.; Elsoudany, A.M.; Helal, O.F.; Alshehri, M.A. Efficacy of high intensity laser therapy in the treatment of male with osteopenia or osteoporosis: A randomised placebo-controlled trial. J. Phys. Ther. Sci. 2017, 29, 1675–1679.
- Ebid, A.A.; El-Kafy, E.M.; Alayat, M.S. Effect of pulsed Nd:YAG laser in the treatment of neuropathic foot ulcers in children with spina bifida: A randomised controlled study. Photomed. Laser Surg. 2013, 31, 565–570.
- Wyszynska, J.; Bal-Bochenska, M. Efficacy of high-intensity laser therapy in treating knee osteoarthritis: A first systematic review. Photomed. Laser Surg. 2018, 36, 343–353.
- Cotler, H.; Chow, R.T.; Hamblin, M.R.; Carroll, J. The use of low-level laser therapy (LLLT) for musculoskeletal pain. MOJ Orthop. Rheumatol. 2015, 2, 00068.
- Tortorici, S.; Messina, P.; Scardina, G.A. Effectiveness of low-level laser therapy on pain intensity after lower third molar extraction. Int. J. Clin. Dent. 2019, 12, 357–367.
- Chow, R.; Armati, P.; Laakso, E.L.; Bjordal, J.M.; Baxter, G.D. Inhibitory effects of laser irradiation on peripheral mammalian nerves and relevance to analgesic effects: A systematic review. Photomed. Laser Surg. 2011, 29, 365–381.
- Holanda, V.M.; Chavantes, M.C.; Wu, X.; Anders, J.J. The mechanistic basis for photo-biomodulation therapy of neuropathic pain by near infrared laser light. Lasers Surg. Med. 2017, 49, 516–524.
- Ketz, A.K.; Byrnes, K.R.; Grunberg, N.E. Characterization of macrophage/microglial activation and effect of photo-biomodulation in the spared nerve injury model of neuropathic pain. Pain Med. 2017, 18, 932–946.
- Thabet, A.A.; Ebid, A.A.; El-Boshy, M.E.; Almuwallad, A.O.; Hudaimoor, E.A.; Alsaeedi, F.E.; Alsubhi, R.H.; Almatrook, R.H.; Aljifry, R.F.; Alotaibi, S.H.; et al. Pulsed high-intensity laser therapy versus low level laser therapy in the management of primary dysmenorrhea. J. Phys. Ther. Sci. 2021, 33, 695–699.
- Collins, C.K.; Masaracchio, M.; Brismée, J.-M. The future of orthopedic manual therapy: What are we missing? J. Man Manip. Ther. 2017, 25, 169–171.
- Coronado, R.A.; Bialosky, J.E. Manual physical therapy for chronic pain: The complex whole is greater than the sum of its parts. J. Man Manip. Ther. 2017, 25, 115–117.
- Duñabeitia, I.; Arrieta, H.; Torres-Unda, J.; Gil, J.; Santos-Concejero, J.; Gil, S.M.; Irazusta, J.; Bidaurrazaga-Letona, I. Effects of a capacitive-resistive electric transfer therapy on physiological and biomechanical parameters in recreational runners: A randomised controlled crossover trial. Phys. Ther. Sport 2018, 32, 227–234.
- Diego, I.M.A.; Fernández-Carnero, J.; Val, S.L.; Cano-de-la-Cuerda, R.; Calvo-Lobo, C.; Piédrola, R.M.; Oliva, L.C.L.; Rueda, F.M. Analgesic effects of a capacitive-resistive monopolar radiofrequency in patients with myofascial chronic neck pain: A pilot randomised controlled trial. Rev. Assoc. Med. Bras. 2019, 65, 156–164.
- Rodríguez-Sanz, J.; López-De-Celis, C.; Hidalgo-García, C.; Canet-Vintró, M.; Fanlo-Mazas, P.; Pérez-Bellmunt, A. Temperature and current flow effects of different electrode placement in shoulder capacitive-resistive electric transfer applications: A cadaveric study. BMC Musculoskelet. Disord. 2021, 22, 139.
- Sousa, L.D.S.-D.; Sanchez, C.T.; Maté-Muñoz, J.L.; Hernández-Lougedo, J.; Barba, M.; Lozano-Estevan, M.D.C.; Garnacho-Castaño, M.V.; García-Fernández, P. Application of Capacitive-Resistive Electric Transfer in Physiotherapeutic Clinical Practice and Sports. Int. J. Environ. Res. Public Health 2021, 18, 12446.
- Beltrame, R.; Ronconi, G.; Ferrara, P.E.; Salgovic, L.; Vercelli, S.; Solaro, C.; Ferriero, G. Capacitive and resistive electric transfer therapy in rehabilitation: A systematic review. Int. J. Rehabil. Res. 2020, 43, 291–298.
- Nakamura, M.; Sato, S.; Kiyono, R.; Yahata, K.; Yoshida, R.; Kasahara, K.; Konrad, A. The Effect of Capacitive and Resistive Electric Transfer Intervention on Delayed-Onset Muscle Soreness Induced by Eccentric Exercise. Int. J. Environ. Res. Public Health 2022, 19, 5723.
- López-De-Celis, C.; Hidalgo-García, C.; Pérez-Bellmunt, A.; Fanlo-Mazas, P.; González-Rueda, V.; Tricás-Moreno, J.M.; Ortiz, S.; Rodríguez-Sanz, J. Thermal and non-thermal effects off capacitive-resistive electric transfer application on the Achilles tendon and musculotendinous junction of the gastrocnemius muscle: A cadaveric study. BMC Musculoskelet. Disord. 2020, 21, 46.
- Ostrowski, J.; Herb, C.C.; Scifers, J.; Gonzalez, T.; Jennings, A.; Breton, D. Comparison of muscle temperature increases produced by moist hot pack and ThermoStim probe. J. Sport Rehabil. 2019, 28, 459–463.
- Martínez-Pizarro, S. Transferencia eléctrica capacitiva y resistiva para mitigar el dolor . Rehabilitacion 2020, 54, 221–222.
- Tashiro, Y.; Hasegawa, S.; Yokota, Y.; Nishiguchi, S.; Fukutani, N.; Shirooka, H.; Tasaka, S.; Matsushita, T.; Matsubara, K.; Nakayama, Y.; et al. Effect of capacitive and resistive electric transfer on haemoglobin saturation and tissue temperature. Int. J. Hyperth. 2017, 33, 696–702.
- Hernández-Bule, M.L.; Trillo, M.Á.; Úbeda, A. Molecular mechanisms underlying antiproliferative and differentiating responses of hepatocarcinoma cells to subthermal electric stimulation. PLoS ONE 2014, 9, e84636.
- Chen, C.; Bai, X.; Ding, Y.; Lee, I.S. Electrical stimulation as a novel tool for regulating cell behavior in tissue engineering. Biomater. Res. 2019, 23, 25.
- Paolucci, T.; Pezzi, L.; Centra, M.A.; Porreca, A.; Barbato, C.; Bellomo, R.G.; Saggini, R. Effects of capacitive and resistive electric transfer therapy in patients with painful shoulder impingement syndrome: A comparative study. J. Int. Med. Res. 2020, 48, 300060519883090.
- Bito, T.; Tashiro, Y.; Suzuki, Y.; Kajiwara, Y.; Zeidan, H.; Kawagoe, M.; Sonoda, T.; Nakayama, Y.; Yokota, Y.; Shimoura, K.; et al. Acute effects of capacitive and resistive electric transfer (CRet) on the Achilles tendon. Electromagn. Biol. Med. 2019, 38, 48–54.
- Wostyn, V. La Tecarthérapie Appliquée a la Kinésithérapie: Evaluation de Leffet Antalgique Immédiat; Institut de Formation on Masso-Kinésithérapie: Reims, France, 2015.
- Clijsen, R.; Leoni, D.; Schneebeli, A.; Cescon, C.; Soldini, E.; Li, L.; Barbero, M. Does the application of TECAR therapy affect temperature and perfusion of skin and muscle microcirculation? A pilot feasibility study on healthy subjects. J. Altern. Complement. Med. 2020, 26, 147–153.
- López-de-Celis, C.; Rodríguez-Sanz, J.; Hidalgo-García, C.; Cedeño-Bermúdez, S.A.; Zegarra-Chávez, D.; Fanlo-Mazas, P.; Pérez-Bellmunt, A. Thermal and Current Flow Effects of a Capacitive-Resistive Electric Transfer Application Protocol on Chronic Elbow Tendinopathy. A Cadaveric Study. Int. J. Environ. Res. Public Health 2021, 18, 1012.
- Masiero, S.; Pignataro, A.; Piran, G.; Duso, M.; Mimche, P.; Ermani, M.; Del Felice, A. Short-wave diathermy in the clinical management of musculoskeletal disorders: A pilot observational study. Int. J. Biometeorol. 2020, 64, 981–988.
- Ozen, S.; Doganci, E.B.; Ozyuvali, A.; Yalcin, A.P. Effectiveness of continuous versus pulsed short-wave diathermy in the management of knee osteoarthritis: A randomised pilot study. Casp. J. Intern Med. 2019, 10, 431–438.
- Koller, T. Mechanosensitive Aspects of Cell Biology in Manual Scar Therapy for Deep Dermal Defects. Int. J. Mol. Sci. 2020, 21, 2055.
- Kumaran, B.; Watson, T. Thermal build-up, decay and retention responses to local therapeutic application of 448 kHz capacitive resistive monopolar radiofrequency: A prospective randomised crossover study in healthy adults. Int. J. Hyperth. 2015, 31, 883–895.
- Priego-Quesada, J.I.; De la Fuente, C.; Kunzler, M.R.; Perez-Soriano, P.; Hervás-Marín, D.; Carpes, F.P. Relationship between Skin Temperature, Electrical Manifestations of Muscle Fatigue, and Exercise-Induced Delayed Onset Muscle Soreness for Dynamic Contractions: A Preliminary Study. Int. J. Environ. Res. Public Health 2020, 17, 6817.
- Notarnicola, A.; Maccagnano, G.; Gallone, M.F.; Covelli, I.; Tafuri, S.; Moretti, B. Short term efficacy of capacitive resistive diathermy therapy in patients with low back pain: A prospective randomised controlled trial. J. Biol. Regul. Homeost. Agents 2017, 31, 509–515.
- Yokota, Y.; Tashiro, Y.; Suzuki, Y. Effect of capacitive and resistive electric transfer on tissue temperature, muscle flexibility and blood circulation. J. Nov. Physiother. 2017, 30, 719–725.
- Niajalili, M.; Sedaghat, M.; Reazasoltani, A.; Akbarzade Baghban, A.R.; Naimi, S.S. Effect of Capacitive Tecar Therapy on Foot Pain and Tactile Sensation in Patients with Type 2 Diabetes. Arch. Rehab. 2020, 21, 304–319.
- Du, J.; Zhen, G.; Chen, H.; Zhang, S.; Qing, L.; Yang, X.; Lee, G.; Mao, H.Q.; Jia, X. Optimal electrical stimulation boosts stem cell therapy in nerve regeneration. Biomaterials 2018, 181, 347–359.
- Zaretsky, D.V.; Romanovsky, A.A.; Zaretskaia, M.V.; Molkov, Y.I. Tissue oxidative metabolism can increase the difference between local temperature and arterial blood temperature by up to 1.3 °C: Implications for brain, brown adipose tissue, and muscle physiology. Temperature 2018, 5, 22–35.
More
