Human serum albumin (HSA) is the most abundant carrier protein in the human body. Competition for the same binding site between different ligands can lead to an increased active concentration or a faster elimination of one or both ligands. Indomethacin and quercetin both bind to the binding site located in the IIA subdomain. To determine the nature of the HSA-indomethacin-quercetin interactions, spectrofluorometric, docking, molecular dynamics studies, and quantum chemical calculations were performed. The results show that the indomethacin and quercetin binding sites do not overlap. Moreover, the presence of quercetin does not influence the binding constant and position of indomethacin in the pocket. However, binding of quercetin is much more favorable in the presence of indomethacin, with its position and interactions with HSA significantly changed. These results provide a new insight into drug-drug interactions, which can be important in situations when displacement from HSA or other proteins is undesirable or even desirable. This principle could also be used to deliberately prolong or shorten the xenobiotics' half-life in the body, depending on the desired outcomes.
- human serum albumin
- quercetin
- indomethacin
- pharmacokinetic interactions
- fluorescence spectroscopy
- docking
- molecular dynamics
- quantum chemistry
Introduction
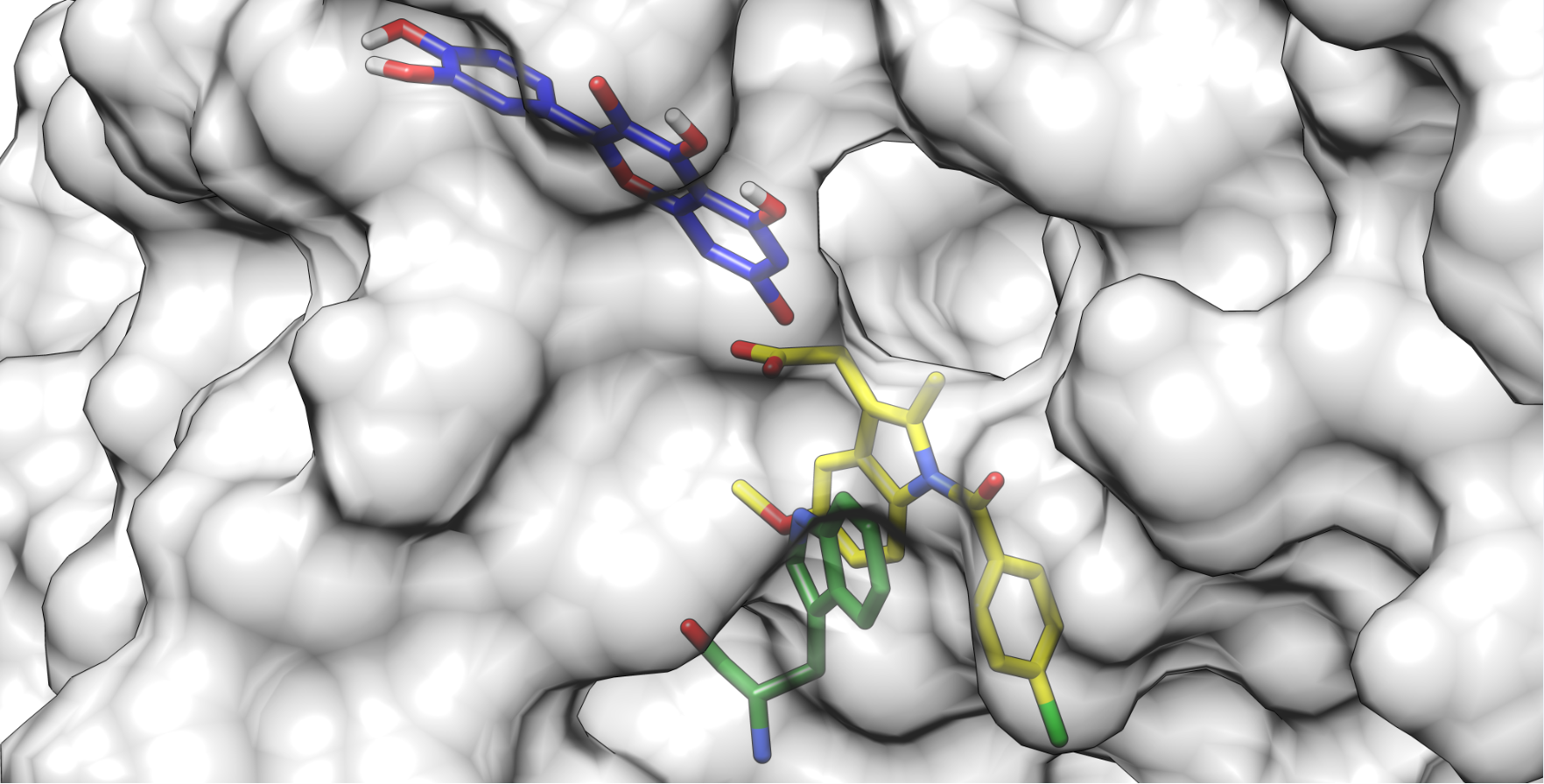
Figure 1. Quercetin anion at Position 7 (blue) docked to a crystallographic structure of indomethacin (yellow) bound to HSA. The Trp214 residue is depicted in green.
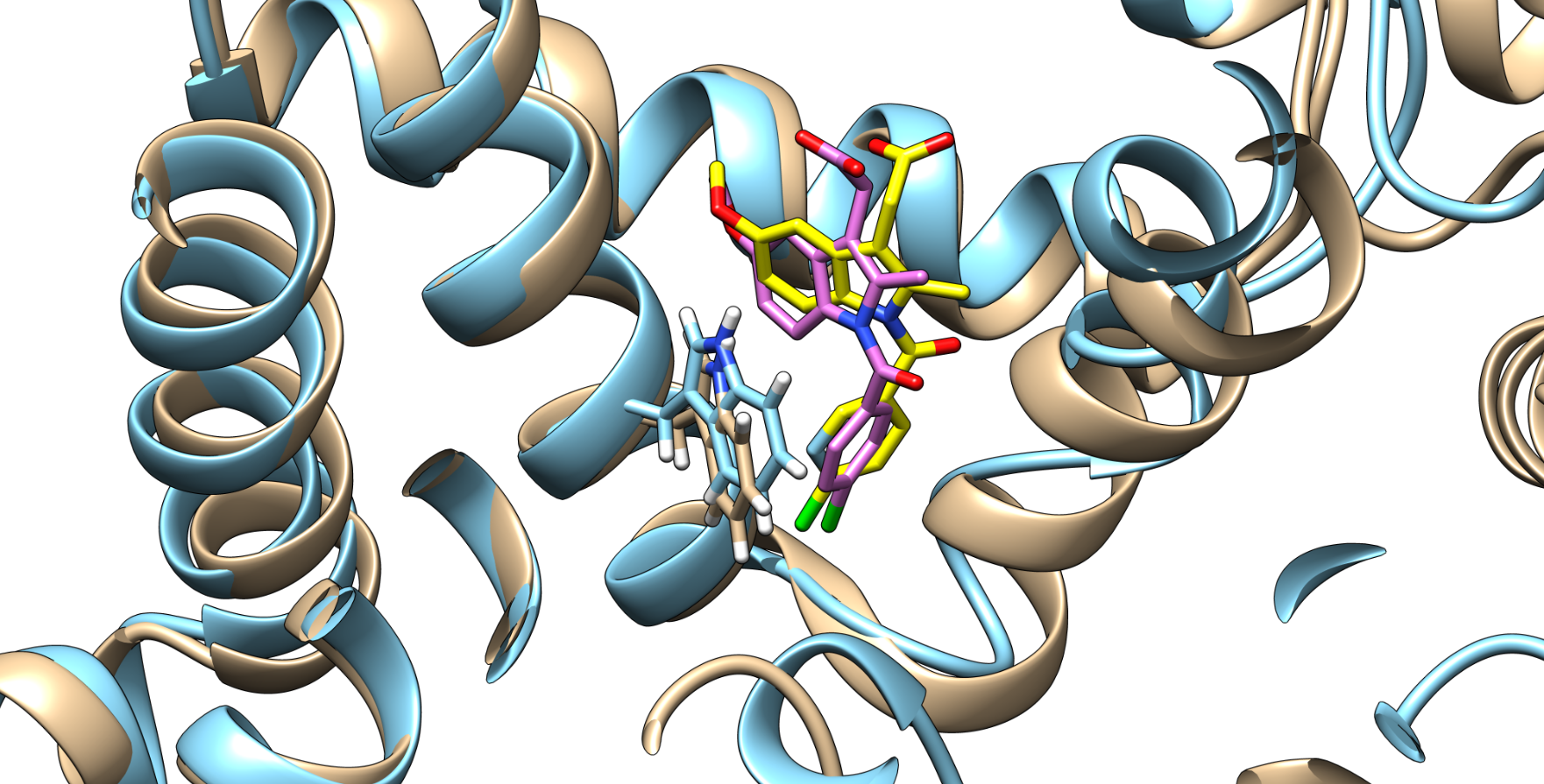
Figure 2. Indomethacin and HSA conformations before (indomethacin is depicted in pink and HSA in brown) and after (indomethacin is depicted in yellow and HSA in light blue) quercetin binding (quercetin is not shown), with the Trp214 shown.
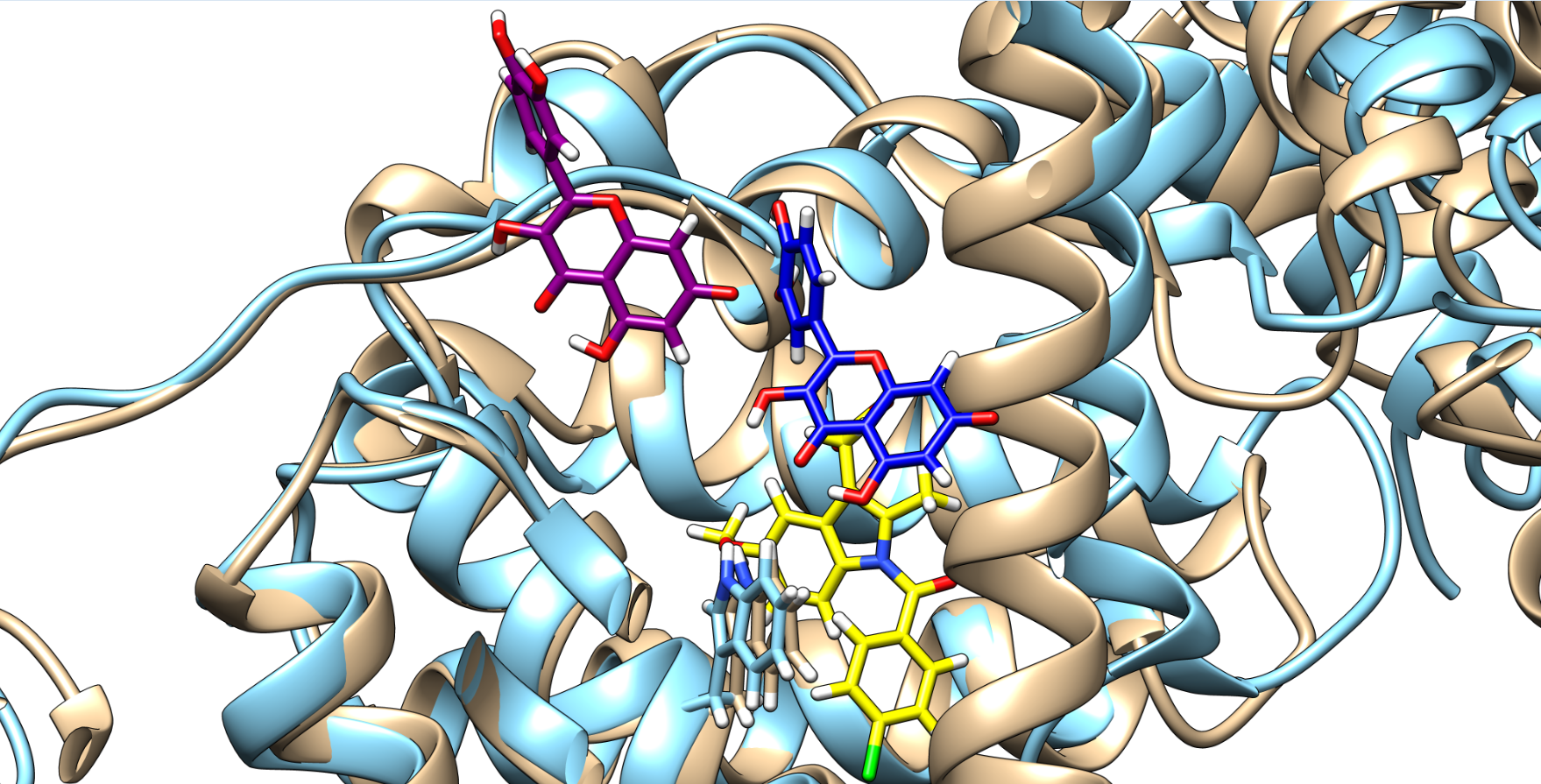
Figure 3. Quercetin poses and HSA conformation in the absence (quercetin is depicted in purple and HSA in brown) and in the presence of indomethacin (quercetin is depicted in blue, HSA in light blue, and indomethacin in yellow), with the Trp214 shown