Neuroendocrine neoplasms (NEN) consist of a very heterogenous group of tumors, contributing to large differences in patients’ disease burden, symptomatology, clinical and objective responses to different treatments, and prognosis. Liver-directed treatments for NELM can be divided into two categories: ablative localized treatments, e.g., radiofrequency ablation (RFA) or microwave ablation (MWA); or trans-arterial treatments, e.g., trans-arterial (bland) embolization (TAE), trans-arterial chemoembolization (TACE) and trans-arterial radioembolization (TARE). The latter technique is also known as selective internal radiation therapy (SIRT). Radioembolization is a more commonly used and simplified term but also a misnomer. Contrary to TAE and TACE, the primary effect is not to embolize vasculature and induce ischemia but to deliver high doses of radiation to tumor tissue via trans-arterial implantation.
- NEN
- radioembolization
- SIRT
- neuroendocrine tumor
1. Introduction
In accordance with the most recent WHO/ENETS criteria, tumor grading is the most common denominator for survival: grade 1 and 2 neuroendocrine tumors (G1-/G2NET) are regarded as well- to moderately differentiated tumors with a low Ki67 index (<3% and 3–20%, respectively); grade 3 NET (G3NET) as well- or moderately differentiated NET with high Ki67 index >20%; and neuroendocrine carcinomas (NEC) as poorly differentiated and with highly proliferative tumors (Ki67 index is most commonly >55%) [1][2][3][1,2,3]. However, within its heterogeneity, a well-established negative factor for survival for NEN patients is the presence of neuroendocrine liver metastases (NELM) [4]. Unfortunately, at diagnosis, 21% of all G1NET, 30% of all G2NET and 50% of all G3NET already have distant metastases, of which the liver is the most commonly affected [5][6][5,6]. In the presence of NELM, Frilling et al. provided an easy method to categorize liver involvement into three groups, based on tumor distribution in the liver [7]: from a ‘simple pattern’ (NELM involves 1–2 liver segments) to a ‘complex pattern’ (extensive unilobar disease with limited disease in the contralateral lobe) to a ‘diffuse pattern’ (bilobar or miliary disease). Whereas these ‘simple’ and ‘complex’ patterns allow surgical resection, the ‘diffuse’ pattern does not. Unfortunately, 60–70% of patients with NELM reside in the ‘diffuse pattern’ group, illustrating the clinical need for liver-directed treatments in light of the limited systemic options for NENs.
Within trans-arterial treatments for NELM, radioembolization has gained a lot of attention over the last decade and reports high tumor objective response rates and limited toxicities [8][10]. As illustrated by the European Neuroendocrine Tumor Society (ENETS) guideline from 2016 and the European Society for Medical Oncology (ESMO) guideline from 2020, the role of radioembolization has extended, including early application as a tumor debulking treatment or as a salvage treatment in selected cases, after the failure of systemic treatments [4][9][4,11]. As NEN and NELM development are highly variable between individuals, the application of radioembolization needs to be determined on a case-by-case basis through discussions by multidisciplinary tumor boards (MDT).
2. How Is Radioembolization Performed?
| Clinical Assessment | Laboratory Testing | Imaging Work-Up | |
|---|---|---|---|
| Minimal | |||
| ECOG performance score | Bilirubin, ALP, AST, ALT, albumin | gdMRI/CECT for intrahepatic tumor load | 1 |
| Signs of hepatic dysfunction (Child–Pugh score) | Creatinine, eGFR | Early-phase CECT for arterial vasculature | |
| NET hormone-related symptoms | Tumor markers (e.g., CgA, gastrin) | ||
| Additional | |||
| In selected cases, Fibroscan or gastroscopy to assess esophageal varices | Hb, hematocrit, WBC, platelets | SSTR-PET/CT for total body tumor load | 1 |
| Coagulation (e.g., Prothrombin time or INR) | FDG-PET/CT for tumor grade distinction, excluding aggressive disease. |
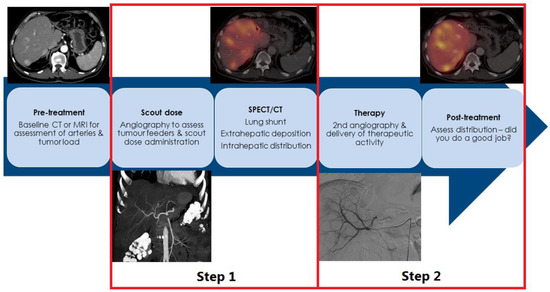
3. SIRT in NEN: Salvage Setting
Table 2 summarizes the most important scientific evidence available to date for salvage radioembolization.
| Year | N | ORR * | DCR * | PFS | OS | REILD | |||
|---|---|---|---|---|---|---|---|---|---|
| % | % | Months | Months | n (%) | |||||
| Devcic et al. | † [8] | [10] | 2014 | 435 | 50 | 86 | NR | 28.5 | NR |
| Peker et al. [13] | Peker et al. [18] | 2015 | 38 | 46 | 83 | NR | 39 | 0 | |
| Barbier et al. [14] | Barbier et al. [19] | 2016 | 54 | 54 | 94 | NR | 34.8 | 1 (1.8) | |
| Braat et al. [10] | Braat et al. [9] | 2019 | 244 | 16 | 91 | NR | 31 | 2 (0.8) | |
| 43 | 91 | ||||||||
| Schaarschmidt et al. [15] | Schaarschmidt et al. [20] | 2022 | 297 | 41.3 | 83.5 | 15.9 | 30.6 | 2 (0.8) | |
| Wong et al. [16] | Wong et al. [21] | 2022 | 170 | 36 | 69 | 25 | 33 | 1 (0.6) |
4. Radioembolization in Earlier Lines or Combinations Treatments
As the liver is the most commonly affected organ for NEN metastases, independent of tumor origin and often the only affected organ after the resection of a primary tumor, the application of radioembolization in an earlier line of the disease is increasingly debated. As shown in the current ENETS and ESMO guidelines, liver-directed treatments should be considered in specific cases [4][9][4,11]. Schaarschmidt et al. showed a median hepatic PFS of 18.6 months and median global PFS of 18.8 months, which is slightly better than the results obtained in a salvage setting (Table 2). Logically, prolonged median OS was found (44.8 vs. 30.6 months) in the group treated in the second-line compared to the salvage setting group [15][20]. The mainstay in the treatment of metastatic disease resides in the use of systemic treatments, most commonly somatostatin analogs (SSA), peptide receptor radionuclide therapy (PRRT) and chemotherapy [4][9][4,11]. In patients with significant intrahepatic tumor burden or aggressive disease, systemic treatments tend to have less prolonged effects [17][18][22,23]. Combining a systemic and/or targeted treatment with a liver-directed treatment seems logical to boost the benefit for patients suffering from high intrahepatic tumor burden or patients with mainly NELM (liver-only or so-called “liver dominant disease”). The most recent study by Braat et al. (“HEPAR PLuS”) combined PRRT with 166Ho-radioembolization, by adding radioembolization within 20 weeks after the fourth cycle of PRRT [19][28]. The scholarauthors concluded that the combination was safe and effective. REILD was encountered in one patient. However, due to the heterogeneity of the group and the selection bias introduced by patient inclusion after the completion of PRRT, a patient population with a poor prognosis was selected. Five out of thirty-one (17%) of the included patients already failed PRRT (with only intrahepatic progressive disease). Nonetheless, high ORR, both RECIST 1.1 and mRECIST, and durable responses during the first year in follow-up were reported, thus the combination seems promising, and no loss in quality of life was reported (Figure 24).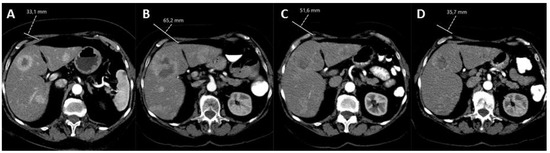
5. Conclusions
Hepatic radioembolization is safe and effective as a monotreatment in NEN. Based on current evidence, the exact application of radioembolization in NEN care remains unknown, and the scientific debate on suggested long-term toxicities remains unresolved. The application of radioembolization should be considered on a case-by-case basis through multidisciplinary discussion. Upcoming clinical and technical developments in the field will ensure a more promising role for radioembolization in NEN care.
