Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Beatrix Zheng and Version 1 by Ekateina Kozuharova.
Some of the most effective anticancer compounds are still derived from plants since the chemical synthesis of chiral molecules is not economically efficient. Rapid discovery of lead compounds with pronounced biological activity is essential for the successful development of novel drug candidates. The genus Astragalus L. is the largest in the family Fabaceae (syn. Leguminosae), with more than 3500 species. Astragalus, excluding Astracantha (formerly Astragalus subgenus Tragacantha), has a world total of ca. 2500 species, of which ca. 500 are in the Americas. Many of the species have conservation status “vulnerable” or “critically endangered”.
- plant anticancer compounds
- in vitro production
- saponins
- flavonoids
- alkaloids
- Astragalus
1. Ethnobotanical Data of Astragalus Species Used against Cancer
Astragalus mongholicus Bunge is the accepted name of A. membranaceus var. mongholicus (Bunge) P.G. Xiao often referred to as A. membranaceu [28][1] is a key plant in Chinese Traditional Medicine used mainly as a Qi (Chi) tonic [29,30][2][3] but also prescribed against cancer [30][3]. A study found that A. membranaceus is an ingredient in 172 of the 200 analyzed Chinese herbal formulae [31][4].
2. Secondary Metabolites of Astragalus Species Anticancer Properties
In recent years, progress in phytochemical studies has been made on Astragalus species due to their effects as immunostimulants or anticancer agents [45,47,49,50,51,52,53,54,55,56,57,58][5][6][7][8][9][10][11][12][13][14][15][16].
Many Astragalus species contain cycloartane saponins–astragalosides, which are cycloastragenol derivatives. Some saponins isolated from representatives of the genus are based on an oleanane skeleton [51,53][9][11]. The high intake of flavonoids is generally associated with a reduced risk of neoplasms [59,60][17][18]. Over 160 different flavonoids of 90 species of Astragalus have been isolated and identified as revised previously [52,53][10][11]. Polysaccharides have been shown to play a role in immune modulation. Astraglucanes have been isolated from roots and rhizomes of A. mongholicus (syn. A. membranaceus) [42][19] and other species. The polysaccharide fraction contains highly branched, predominantly 1,3-β-glucans. These products find their application as an aid in radiation antineoplastic therapy and chemotherapy, as well as in the treatment and prevention of bacterial and viral infections [61][20].
Different Astragalus extracts have been shown to increase resistance to the immunosuppressive effects of chemotherapy drugs while stimulating macrophages to produce interleukin-6 and tumor necrosis factor (TNF). Human clinical trials demonstrated a substantial increase in survival rates when extracts from Astragalus plants are given to cancer patients receiving chemo- or radiotherapies. They have also increased IgA, IgC, and interferon production in humans [51][9]. Astragaloside IV inhibited the development of non-small cell lung cancer by inhibiting the Akt/GSK-3p/p-catenin signaling pathway. It also increased the expression of Bax (a cell death marker) while decreasing the expression of Bcl-2 (anti-apoptotic protein). This demonstrates the importance of astragaloside IV as a potential antitumor agent [62][21].
A. angustifolius is an endemic Bulgarian species that has been reported to contain cyclosiversigenin (cycloastragenol), siversigenin (astragenol), and soyasapogenol B [51][9]. The antiproliferative activity of compounds isolated from A. angustifolius in cervical (HeLa), human lung (H-446), human colon (HT-29) cancer, and human monocyte lymphoma (U937) cell lines are examined [63][22], and only 3-O-[α-L-rhamnopyranosyl-(1→2)-β-D-xylopyranosyl-(1→2)-β-D-glucuronopyranosyl]-3β,22β,24-trihydroxyolean-12-en-29-oic acid possessed weak cytotoxicity against HeLa.
Another endemic Bulgarian plant is A. aitosensis which afforded 5,6-dehydro-6-desoxyastragenol [51][9].
Recently, a novel and unusual for the genus Astragalus group of compounds, flavoalkaloids, is identified in A. monspessulanus subsp. monspessulanus. Before, they were known only as aglycones (Figure 1). One novel quercetin tetraglycoside and eight known flavonoids are isolated as well [53][11]. Also, from the aerial parts of this species, two saponins are reported [64][23]. Two rare flavonoids with an unusual hydroxymethylglutaric acid as a moiety: quercetin-3-O-α-L-rhamnopyranosyl-(1→2)-[6-O-(3-hydroxy-3-methylglutaryl)-β-D-galactopyranoside and kaempferol-3-O-α-L-rhamnopyranosyl-(1→2)-[6-O-(3-hydroxy-3-methylglutaryl)-β-D-galactopyranoside are isolated from the aerial parts of A. monspessulanus subsp. illyricus (Figure 1) [65][24].
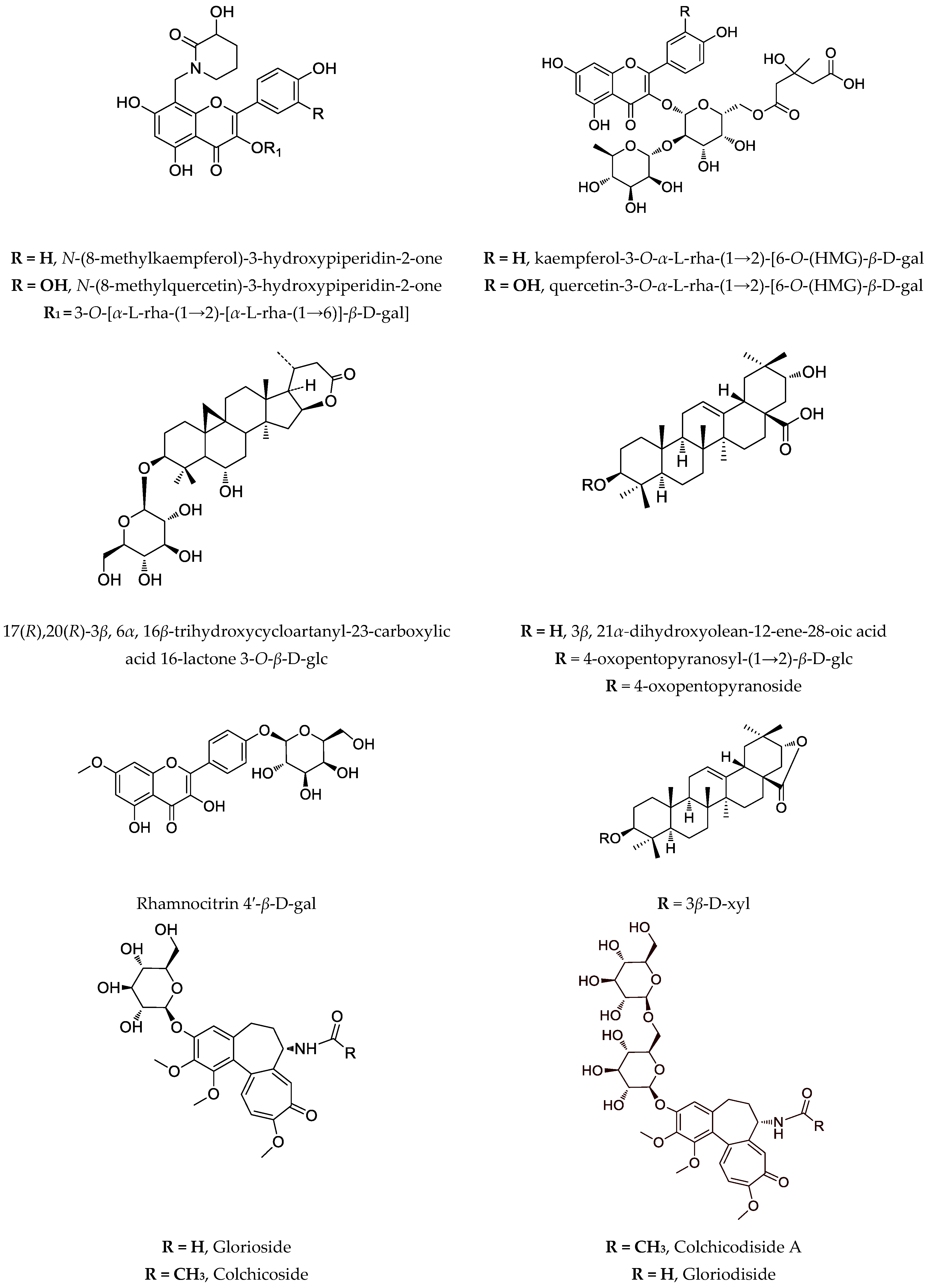
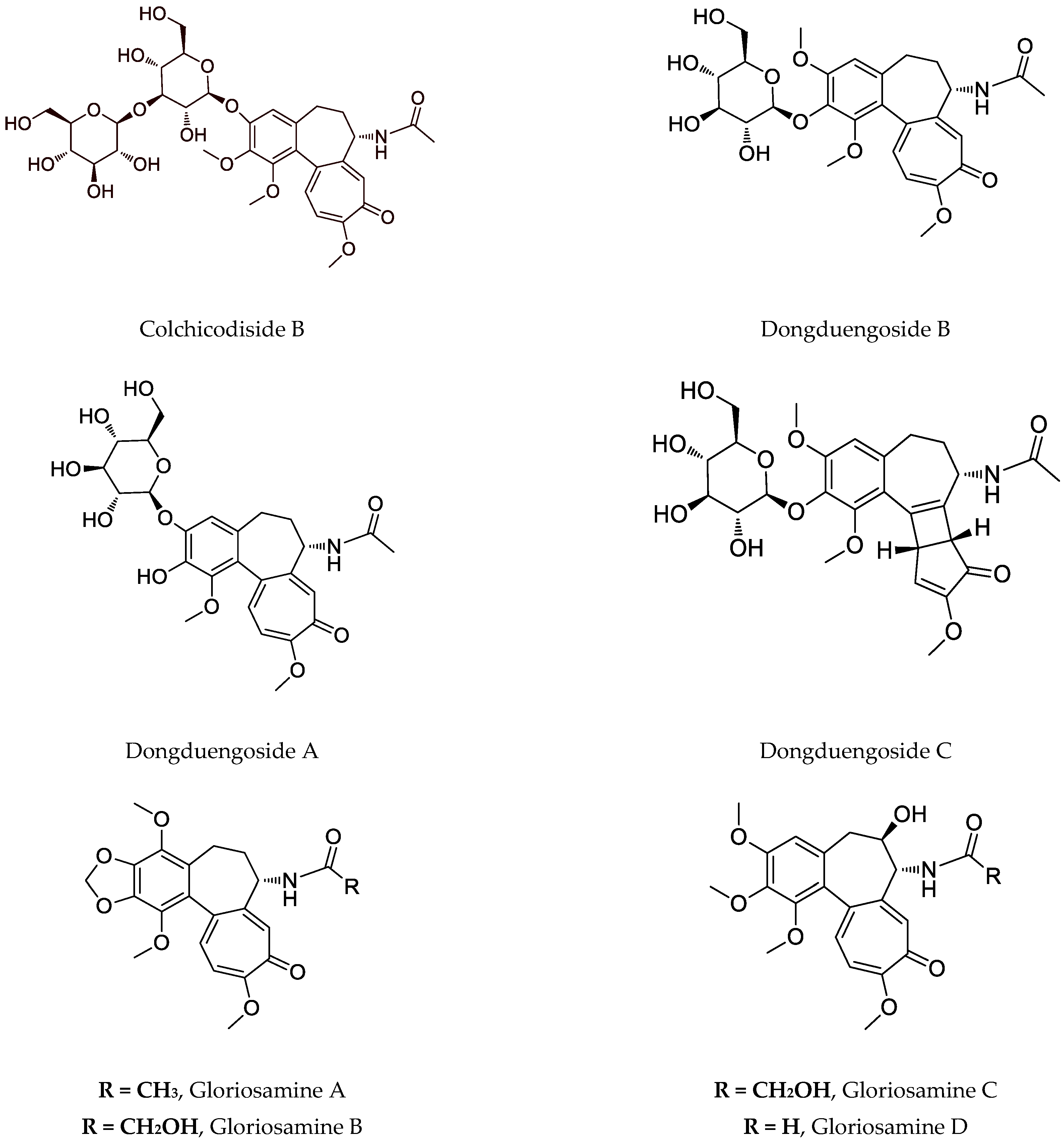
Figure 1.
Notable compounds, isolated from
Astragalus
and
Gloriosa
species.
Phytochemical investigation of A. glycyphyllos led to the isolation of six saponins, and their structures are partially elucidated [66,67][25][26]. After acid hydrolysis of a saponin mixture obtained from the aerial parts of the plant, soyasapogenol B and 3β,22β,24-trihidroxyolean-12-en-19-one are identified [68][27]. Cycloartane saponins askenoside C and F [68][27] and 17(R), 20(R)-3β,6α,16β-trihydroxycycloartanyl-23-carboxylic acid 16-lactone 3-O-β-D-glucopyranoside are later isolated from the species [69][28]. Several known flavonoids, including the rare camelliaside A, are also identified [53,69][11][28]. The antineoplastic activity in vitro of the saponin-containing fractions obtained from wild-grown and cultivated A. glycyphyllos, respectively, were tested in a panel of human tumor cell lines of different origin and characteristics. A standard MTT-based protocol for assessing cell viability was used. Both fractions inhibited tumor cell growth in a dose-dependent manner. However, according to the calculated IC50 value, the fraction obtained from the in vitro shoot cultures showed relatively superior cytotoxic activity compared to that of the wild-type species in all of the screened tumor cell lines (our unpublished data). In vitro cultures of A. glycyphyllos could be an alternative way to produce saponins, with promising antineoplastic activity.
Three saponins are isolated from A. corniculatus: two of them with an aglycone 3β,21α-dihydoxyolaean-12-ene-28-oic acid, and the third–with its corresponding lactone [22,87][29][30]. A series of studies demonstrated that a purified saponin fraction containing these compounds had a protective effect against the invasiveness of bone marrow carcinoma (Graffi myeloid tumor) in hamsters. Administration of the saponin mixture increased the number, migration, and phagocytic index of peritoneal macrophages and blood polymorphonuclear leukocytes in animals with implanted tumors. Also, due to the hamster treatment with the mixture, an increased mitogenic response to phytohemagglutinin and lipopolysaccharides is observed, i.e., the saponins have an immunostimulatory effect [88,89,90][31][32][33].
Rhamnocitrin 4′-β-D-galactopyranoside and a mixture of two saponins are isolated from aerial parts of A. hamosus and investigated for antiproliferative activity on SKW-3 cells. Significant apoptosis-induction activity is proved for the saponin mixture compared to the flavonoid glycoside at equal concentrations. After co-administration of rhamnocitrin 4′-β-D-galactopyranoside, with Cisplatin and Gentamicin, there is significant protection of human kidney cells HEK-293T against the cytotoxic effects of nephrotoxic drugs [75][34]. The same mixture of two saponins is examined on cell lines HL-60, HL-60/Dox, SKW-3, RPMI-8226, U-266, and OPM-2 [75][34]. The saponins caused concentration-dependent suppression of the proliferative activity of malignantly transformed cells. These data are confirmed by an ELISA test evaluating apoptosis-specific DNA fragmentation. The significance of the transcription factor NFκB, as well as the mitochondrial protein Bcl-xL for the antitumor activity of the saponin mixture, is established. Selective cytotoxic activity of saponins in cell lines originating from breast cancer is demonstrated. The saponin mixture showed cytotoxicity concerning both cell lines and clearly demonstrated inhibitory properties against the mitochondrial anti-apoptotic protein Bcl-xL. This gives a reason to believe that unlocking the internal pathway of apoptosis by suppressing the expression of BclxL is a part of the mechanism of action of the saponins. Furthermore, the mixture of two saponins showed no cytotoxic effect on the non-malignant cell line MCF-10A, which originated from the mammary gland, suggesting that it exhibited selective malignant cell toxicity and may be the subject of further studies [91][35].
3. Biotechnology of Astragalus Species
Many in vitro cultures are established not only to increase the production of important secondary metabolites in selected Astragalus plants but also to preserve the endemic and/or endangered species. In general, the most investigated species both in phytochemical and biotechnological means is A. mongholicus (syn A. membranaceus) [92][36].
3.1. Cell Culture
The active substances from wild and field-grown plants usually have different quality and quantity and vary depending on the environmental conditions. The diseases and the application of pesticides further reduce the quality of the plant material. In vitro plant cultures surmount these problems as environmental conditions affecting the metabolism of plants can be precisely controlled. Working with tissue cells dramatically decreases preparation time, processing, and storage costs associated with traditional plant approaches [93][37]. There are several advantages to producing secondary metabolites in plant cell culture compared to in vivo cultivation. Production can be more predictable, reliable, and independent from unpredictably changing climatic conditions. Isolation of the phytochemical metabolites can be more rapid and efficient than extraction from the whole plant. Interfering compounds in the wild plant can be avoided in tissue cultures. Cell cultures can produce phytochemicals in large volumes.
Astragalus genus possesses characteristics that make it significant for in vitro cultivation. Reducing natural supplies due to excessive collection is already present and, therefore, industrial or consumer interest. Due to their complex structures, saponins, flavonoids, and polysaccharides are still most efficiently produced by the plants. There are a lot of problems involved with this production method. Variable qualities and quantities of the plant material, plants that need to grow several years before being ready for harvesting (Astragalus roots), and the over-collecting of some species (A. mongholicus, syn. A. membranaceus), A. angustifolius, A. missouriensis, A. thracicus, A. aitosensis, etc.) are just a few of the problems connected with the production of these natural products. Therefore, tissue cultures might be explored as an alternative production method [7][38].
3.2. Effects of Medium Composition and Growth Regulators
The flavonoid biosynthesis in cell cultures of A. missouriensis and A. angustifolius is strongly suppressed by 2,4-dichlorophenoxyacetic acid (2,4-D). The higher concentrations of 2,4-D decreased the content of flavonoids. In this respect, 1-naphthaleneacetic acid (NAA) is weaker than 2,4-D. Therefore, flavonoid production is inhibited by adding 2,4-D to the medium. In all tested concentrations of cytokinins, such as 6-benzylaminopurine (BAP) or kinetin (Kn), under the light cultivation regimen, induction of flavonoid production in cell suspension cultures is achieved. Kinetin was found to be the most effective. The sucrose levels of the medium have a tremendous impact on saponins production. The growth and the saponins production are favored by the higher concentrations of sucrose. The investigation was carried out on A. membranaceus hairy roots cultures (HR) with different concentrations of sucrose in the MS medium. A basic MS medium supplemented with 2% sucrose increases overall saponins yield but inhibits the growth of HR. The growth of Astragalus-HR is promoted by high concentration (6%), but the yield of saponins remains very low. The optimal medium for both yield and growth is supplemented with 4% sucrose [94][39].
3.3. Effects of End-Product Inhibition
Because phytosterols (campesterol, stigmasterol, and β-sitosterol) are present in the hairy roots of Astragalus species, the effect on the total saponin production of these substances is examined. Since the biosynthetic route of saponins and that of phytosterols are branched at 2,3-epoxyscvalen, it may be possible to increase the production of saponins by end-product inhibition. In the experiments, contents of saponins after adding 0.2 mg/mL of β-sitosterol in MS liquid medium without ammonium nitrate are examined at different stages of growth. The saponin content (total saponins) in the hairy roots induced by Agrobacterium rhizogenes LBA 9402 reached 5.25% of dry wt on day 28 of cultivation. When β-sitosterol is added to the culture media of these hairy roots, astragaloside production is remarkably increased to 7.13% of dry wt and led to an increase of 36% of the total saponin content in comparison with the control. From these data, β-sitosterol seems to behave as an inhibitor in the biosynthetic route when the amount added is relatively large. These results prove that the hairy root cultures of Astragalus mongholicus can be a valuable alternative for overproducing cycloartane saponins compared with the whole plant. Using a selected high productive clone, inducing by Agrobacterium rhizogenes LBA 9402, optimized culture medium (MS without ammonium nitrate), and end-product inhibition, a relatively high saponin production can be achieved [94,95][39][40].
3.4. Genetic Transformation of Astragalus Species by Agrobacterium Rhizogenes
In a specific soil, A. rhizogenes can induce a certain type of “hairy roots” culture, which can be maintained without phytohormones in the medium [96][41]. Usually, four different bacterial strains are used—TR 105, R 1601, ATCC 15834, and LBA 9402. With the same Agrobacterium strain, the susceptibility of Astragalus species to infection is highly variable. Some species of the genus (A. membranaceus, A. mongholicus, A. monspessulanus) have more difficulties establishing transformed roots. In some plants (A. englerianus, A. mongholicus, A. missouriensis, A. sulcatus), a callus is formed initially, and transformed roots subsequently emerge from it. Still, in others (A. glycyphyllos, A. hamosus, A. boeticus), a profusion of roots appear directly at the site of inoculation [70][42].
3.5. In Vitro Production of Secondary Metabolite
Optimization of cultures and production conditions has been achieved to produce flavonoids from cell cultures of different Astragalus species: A. monspessulanus, A. aitosensis, A. missouriensis, A. edulis, A. hamosus, and A. angustifolius [53,65][11][24]. Flavonoids within complex plant tissues can be more difficult to separate in an intact polymeric form than cell culture-derived flavonoids. A novel vehicle for depth investigation of flavonoids individually represents the production of these compounds in uniform plant-cell culture systems.
All in vitro cultures of A. missouriensis and A. angustifolius produced flavonoids. Quercetin is the main aglycone identified in the in vitro cell suspension in both free and bound forms (as glycosides). The main flavonoid glycosides are isoquercitrine and quercitrine. Rutin and hyperoside have also been detected. The maximum total amount of flavonoids, 1.78% for A. angustifolius (unpublished results) and 1.34% for A. missouriensis, is achieved after optimization of the production medium [97][43].
4. Characteristics of Target Gloriosa Species
Genus Gloriosa (Liliaceae) includes 12 species and, despite its taxonomic complication, was found to be monophyletic [127][44]. Few of them are of commercial, pharmaceutical, or ethnobotanical interest.
4.1. Ethnobotaical Data of
Gloriosa
Species Used against Cancer
Gloriosa superba is one of the plants used as an antidote against snakebite in the Southern part of Tamilnadu, India [128][45], and several drops of extract of this plant are rubbed onto the cuts and wounds in Rajouri and Poonch districts of Jammu and Kashmir, India [129][46]. It has wide application in folk medicine in tropical Africa and Asia, such as abdominal and general pain, anthelminthic and antiparasitic, leprosy, leucorrhea, mental illness, skin diseases, ulcers, etc. [130][47]. But also, in traditional applications in Asia and Africa, in addition to diseases such as gout, scrofula, antipyretic, anthelmintic, purgative, and antiabortive activity, anticancer use is indicated [131][48]. This activity is well confirmed in recent pharmacological tests against pancreatic cancer [79][49], colon cancer [84][50], and other cancer cells [132][51]. Due to the boom in harvesting and export trade, some populations of G. superba are on the edge of extinction [127][44].
4.2. Secondary Metabolites of
Gloriosa
Species with Anticancer Properties
The main secondary metabolite is colchicine, which has anticancer activity but its toxicity profile is not acceptable. Several studies suggested the cytotoxic activity of semisynthetic derivatives of colchicine and thiocolchicoside; thus, the reported IC50 values have no relevance to naturally occurring tropolones [133,134,135,136][52][53][54][55]. Gene expression, as well as cytotoxic effects of colchicine in human gastric cancer ASG and NCI-N87 cell lines, are evaluated. It was found that only 6 ng/mL of colchicine had the desired antiproliferative effect on both lines. Interestingly, the gene regulation of those cells is affected in the same manner as the stated concentration leading to apoptosis [137][56]. Despite this compound, the interesting colchicinoids such as gloriosamine A-D are isolated from the aerial part of G. rothschildiana [86][57].
Nowadays, the focus on G. superba is also due to the presence of glycosylated colchicine derivatives, especially colchicoside, which is considered ten times less toxic than colchicine, as shown in Table 2 [79][49]. Recently, four novel colchicinoids named N-deacetyl-N-formyl-3-de-O-methylcolchicine-3-O-β-D-glucopyranoside (glorioside), 3-de-O-methylcolchicine-3-O-β-D-glucopyranosyl-(1→6)-3-O-β-D-glucopyranoside (colchicodiside A), N-deacetyl-N-formyl-3-de-O-methylcolchicine-3-O-β-D-glucopyranosyl-(1→6)-3-O-β-D-glucopyranoside (gloriodiside) and 3-de-O-methylcolchicine-3-O-β-D-glucopyranosyl-(1→3)-3-O-β-D-glucopyranoside (colchicodiside B) are isolated from the seeds of the species [81][58]. Also, from a seedless pot of Thai origin, G. superba was identified in novel glycosylated colchicinoids–dongduengoside A-C, and colchicine, 2-demethilcolchicine, colchicoside, and luteolin 7-O-β-D-glucopyranoside [82][59]. Some colchicinoids are obtained using biotechnological approaches. Riva et al. (1997) describe that β-1,4-galactosyltransferase catalyzes galactosylation of colchicoside, and Pišvejcová et al. (2006) describe the influence of various parameters on the activity of β-1,4-galactosyltransferase derived from beef milk and the optimization of the conditions leading to the preparation of galactosylate and glycosylated colchicoside derivatives [138,139][60][61].
4.3. Biotechnology of
| Plant Species | Type | Compounds Isolated | Cytotoxicity on Cell Lines (IC50) | References |
|---|---|---|---|---|
| A. aitosensis | callus, suspension | cycloartane saponins, sterols, flavonoids | n.d. | [9] |
| aerial pars, wild grown * | 5,6-dehydro-6-desoxyastragenol | n.d. | [9] | |
| A. angustifolius | callus, suspension | cycloartane saponins, flavonoids | n.d. | [11] |
| aerial parts, wild grown * | β-sitosterol, cycloastragenol, astragenol, soyasapogenol B, 3-O-[α-L-rha-(1→2)-β-D-xyl-(1→2)-β-D-glc]-3β,22β,24-trihydroxyolean-12-en-29-oic acid |
n. d. HeLa (36 µM); HT-29 (50 µM) |
[22] [11] |
|
| A. asper | aerial parts, wild grown * | saponins, flavonoids | n.d. | [11] |
| A. boeticus | callus, suspension, hairy roots |
saponins, soyasapogenol B, β-sitosterol, flavonoids | n.d. | [42] |
| A. brachycera | hairy roots ** shoots ** |
cycloartane saponins, sterols | n.d. | [9] |
| A. canadensis | hairy roots | cycloartane saponins, cycloastragenol, astragenol, | n.d. | [9] |
| A. centralpinus | aerial parts, wild grown * | flavonoids | n.d. | [11] |
| A. corniculatus | aerial parts, wild grown *** | two oleanane type saponins and a corresponding lactone | Graffi tumour–in vivo, i.p., hamsters (50 mg/kg) ***; in vitro (20 µg/mL) *** | [62] |
| A. edulis | callus | quercetin, kaempferol, isorhamnetin, saponins | n.d. | [42] |
| A. englerianus | hairy roots | cycloartane saponins | n.d. | [9] |
| A. falcatus | hairy roots | cycloartane saponins | n.d. | [9] |
| A. glycypyllos | hairy roots **, callus shoots *** |
cycloastragenol, astragenol, soyasapogenol B epoxycycloartanes |
n.d. T-24 (125 µg/mL); CAL-29 (90 µg/mL); MJ (75 µg/mL); HUT-78 (78 µg/mL) |
[9] [11] [63] |
| aerial parts, wild grown *** | epoxycycloartanes | K-562 (50 µg/mL) ***; HL-60 (40 µg/mL) ***; BV-173 (70 µg/mL) *** | [64] | |
| aerial parts, wild grown *** | epoxycycloartanes | T-24 (168 µg/mL); CAL-29 (105 µg/mL); MJ (126 µg/mL); HUT-78 (87 µg/mL) | [63] | |
| aerial parts, wild grown | 17(R),20(R)-3β,6α,16β-trihydroxycycloartanyl-23-carboxylic acid 16-lactone 3-O-β-D-glucopyranoside | T-24 (66 µg/mL); CAL-29 (52 µg/mL); MJ (52 µg/mL); HUT-78 (18 µg/mL) | [65] | |
| A. hamosus | callus, suspension, hairy roots | saponins, soyasapogenol B, β-sitosterol, astragalin, rutin, isorhamnetin-3-O-glycoside | n.d. | [11][42] |
| aerial parts, wild grown *** | saponins | HL-60 (63 µg/mL); HL-60/Dox (25 µg/mL); SKW-3 (84 µg/mL) | [34] | |
| A. missouriensis | Callus **, suspension, hairy roots | isoquercitrin, quercitrin, rutin, hyperoside, saponins | n.d. | [42][62] |
| A. mongholicus (syn. A. membranaceus) | hairy roots **, shoots ** | astragalosides, β-sitosterol, stigmasterol, campesterol | n.d. | [9][63] |
| A. monspessulanus | aerial parts, wild grown * | flavoalkaloids, acylated flavonoids, flavonoids | n.d. | [24] |
| A. onobrychis | aerial parts, wild grown * | flavonoids, saponins | n.d. | [66] |
| A. oxyglotis | hairy roots | cycloartane saponins | n.d. | [9] |
| A. sesameus | Shoots ** | - | HL-60/Dox (87 µg/mL); SKW-3 (68 µg/mL) | |
| A. spruneri | aerial parts, wild grown * | flavonoids | n.d. | [67] |
| A. sulcatus | hairy roots | cycloartane saponins, sterols, swensonine | n.d. | [9] |
| A. thracicus | callus, suspension | saponins, flavonoids | n.d. | [11] |
| aerial parts, wild grown * | saponins, flavonoids | HT-29 (52 µg/mL); HL-60 (67 µg/mL); HL-60/Dox (53 µg/mL); SKW-3 (83 µg/mL) | [11] | |
| A. vesicarius ssp. carniolicus | callus | flavonoids | HL-60 (8.8 µg/mL) *; HL-60/Dox (11.8 µg/mL) * | [68] |
| callus | 5-hydroxy-7-methoxy-2′, 5′-dihydroxyisoflavone | HL-60 (38.9 µg/mL); HL-60/Dox (35.2 µg/mL) | [68] | |
| 5, 7-dihydroxy-4′-methoxyisoflavone | HL-60 (41.4 µg/mL); HL-60/Dox (42.4 µg/mL) | [68] | ||
| 7-methoxy-5-hydroxy-4′-methoxy-2′-hydroxyisoflavone | HL-60 (64.1 µg/mL); HL-60/Dox (41.8 µg/mL) | [68] | ||
| 8-pregnyl genistein | HL-60 (36.1 µg/mL); HL-60/Dox (36.1 µg/mL) | [68] | ||
| 5,7-dihydroxy-8-pregnyl-4′-methoxy-2′-hydroxyisoflavone | HL-60 (56.3 µg/mL); HL-60/Dox (56.8 µg/mL) | [68] | ||
| sophorophenolone | HL-60 (78.0 µg/mL); HL-60/Dox (63.0 µg/mL) | [68] | ||
| G. superba | seeds | colchicoside, colchicine, 3-O-demethylcolchicine | PANC-1, PANC02 (GS ++ 0.45–0.59 µg/mL) PANC02 (GS2B + 9.49 µg/mL) |
[49] [69] |
| glorioside, colchicodiside A, gloriodiside, colchicodiside B, colchicodiside C, dongduengoside A-C, colchicine, 2-demethilcolchicine, colchicoside and luteolin 7-O-β-D-glucopyranoside | DLA (29 µg #; 21 µg ##) | [58] [59] [70] |
||
| rhizomes | peptides | SW620 (n.d.) | [50] | |
| roots | colchicine | HT-29 (0.12 μg/mL *) | [71] | |
| G. rothschildiana | aerial parts | gloriosamine A-D, colchicine, colchiciline, colchifoline and N-deacetyl-N-formylcolchicine | - | [57] |
* Extract was tested; ** Extract from this culture was tested; *** Purified saponins’ mixture was tested; + GS2B, colchicine poor extract; ++ GS, G. superba total extract; # Methanolic extract of G. superba seeds; ## AgNPs; n.d., not defined.
4.3. Biotechnology of
Gloriosa
Species
The medicinal interest in G. superba and its nonstop over-exploitation are the main reasons to apply in vitro techniques for the conservation, production, and enhancement of secondary metabolites. The species has a very low rate of seed germination as well as seed production is quite low and uneconomical. One of the problems with commercial cultivation is the poor viability of the seeds [143][72]. Four or five vegetative cycles are necessary for the complete reproductive phase [144][73]. Commercially these plants are propagated using daughter corms with a week multiplication ratio (1:1), slow and insufficient for conservation of this species [145][74]. Thus, in vitro cultivation is needed to conserve this taxon, otherwise wthe researchers will lose it by 2020 [146][75]. Plant biotechnological approaches, such as in vitro mass multiplication, have been taken for the conservation, and various methods and techniques have been studied for the production and enhancement of secondary metabolites. An efficient protocol is established for in vitro micro-propagation using corm bud explant [147][76]. Extracts from buds inoculated at MS medium supplemented with different concentrations of 2,4-D (1.0–10.0 mg/L) and IAA (0.5–5.0 mg/L) indicated that higher concentrations of 2,4-D and IAA reduce the callus induction. The shoot initiation depends on the combination of cytokinins. Most shoots are obtained in the presence of 9.84 μM 2iP combined with 4.64 μM Kin after 21-day culturing. Sivakumar et al. (2019) developed an efficient protocol for in vitro mass propagation of G. superba using callus derived from non-dormant corm buds [148][77]. Medium supplemented with a combination of plant growth regulators such as BAP (1.5 mg/L), NAA (0.6 mg/L), and polyamine putrescine (15 mg/L) as secondary messengers in signaling pathways, induced maximum shoot buds (87.5). Within this restudyearch, optimal seed germination of 86% is also achieved when seeds are treated with 70% sulphuric acid for 2 min. Mahendran et al. (2018) initiated cell suspension cultures of G. superba with a callus derived from rhizomes cultivated on MS medium supplemented with 2.0 mg/L, 2,4-D, and 0.5 mg/L NAA [149][78].
References
- Xu, L.; Podlech, D. Astragalus Mongholicus Bunge. Flora China 2010, 10, 338–339, 343.
- Tierra, M.; Tierra, L. Chinese Traditional Herbal Medicine; Lotus Light Pub: Twin Lakes, WI, USA, 1998.
- Shahrajabian, M.H. A Review of Astragalus Species as Foodstuffs, Dietary Supplements, a Traditional Chinese Medicine and A Part of Modern Pharmaceutical Science. Appl. Ecol. Env. Res. 2019, 17, 13371–13382.
- Wang, S.F.; Wang, Q.; Jiao, L.J.; Huang, Y.L.; Garfield, D.; Zhang, J.; Xu, L. Astragalus-Containing Traditional Chinese Medicine, with and without Prescription Based on Syndrome Differentiation, Combined with Chemotherapy for Advanced Non-Small-Cell Lung Cancer: A Systemic Review and Meta-Analysis. Curr. Oncol. 2016, 23, 188–195.
- Auyeung, K.K.; Han, Q.-B.; Ko, J.K. Astragalus Membranaceus: A Review of Its Protection Against Inflammation and Gastrointestinal Cancers. Am. J. Chin. Med. 2016, 44, 1–22.
- Lin, S.; An, X.; Guo, Y.; Gu, J.; Xie, T.; Wu, Q.; Sui, X. Meta-Analysis of Astragalus-Containing Traditional Chinese Medicine Combined With Chemotherapy for Colorectal Cancer: Efficacy and Safety to Tumor Response. Front. Oncol. 2019, 9, 749.
- Cho, W.C. Immunomodulatory and Anti-Tumor Activities of Astragalus Ancient Herb—Modern Miracle; LAP LAMBERT Academic Publishing: Saarbrücken, Germany, 2009.
- Li, R.; Chen, W.; Wang, W.; Tian, W.; Zhang, X. Extraction, Characterization of Astragalus Polysaccharides and Its Immune Modulating Activities in Rats with Gastric Cancer. Carbohydr. Polym. 2009, 78, 738–742.
- Ionkova, I.; Shkondrov, A.; Krasteva, I.; Ionkov, T. Recent Progress in Phytochemistry, Pharmacology and Biotechnology of Astragalus Saponins. Phytochem. Rev. 2014, 13, 343–374.
- Li, X.; Qu, L.; Dong, Y.; Han, L.; Liu, E.; Fang, S.; Zhang, Y.; Wang, T. A Review of Recent Research Progress on the Astragalus Genus. Molecules 2014, 19, 18850–18880.
- Krasteva, I.; Shkondrov, A.; Ionkova, I.; Zdraveva, P. Advances in Phytochemistry, Pharmacology and Biotechnology of Bulgarian Astragalus Species. Phytochem. Rev. 2016, 15, 567–590.
- Zhou, R.; Chen, H.; Chen, J.; Chen, X.; Wen, Y.; Xu, L. Extract from Astragalus Membranaceus Inhibit Breast Cancer Cells Proliferation via PI3K/AKT/MTOR Signaling Pathway. BMC Complement. Altern. Med. 2018, 18, 83.
- Zhang, J.; Liu, L.; Wang, J.; Ren, B.; Zhang, L.; Li, W. Formononetin, an Isoflavone from Astragalus Membranaceus Inhibits Proliferation and Metastasis of Ovarian Cancer Cells. J. Ethnopharmacol. 2018, 221, 91–99.
- Li, W.; Song, K.; Wang, S.; Zhang, C.; Zhuang, M.; Wang, Y.; Liu, T. Anti-Tumor Potential of Astragalus Polysaccharides on Breast Cancer Cell Line Mediated by Macrophage Activation. Mater. Sci. Eng. C 2019, 98, 685–695.
- Li, S.; Sun, Y.; Huang, J.; Wang, B.; Gong, Y.; Fang, Y.; Liu, Y.; Wang, S.; Guo, Y.; Wang, H.; et al. Anti-Tumor Effects and Mechanisms of Astragalus Membranaceus (AM) and Its Specific Immunopotentiation: Status and Prospect. J. Ethnopharmacol. 2020, 258, 112797.
- Shen, L.; Gwak, S.R.; Cui, Z.Y.; Joo, J.C.; Park, S.J. Astragalus-Containing Chinese Herbal Medicine Combined With Chemotherapy for Cervical Cancer: A Systematic Review and Meta-Analysis. Front. Pharmacol. 2021, 12, 587021.
- Le Marchand, L. Cancer Preventive Effects of Flavonoids—A Review. Biomed. Pharmacother. 2002, 56, 296–301.
- Thomasset, S.C.; Berry, D.P.; Garcea, G.; Marczylo, T.; Steward, W.P.; Gescher, A.J. Dietary Polyphenolic Phytochemicals—Promising Cancer Chemopreventive Agents in Humans? A Review of Their Clinical Properties. Int. J. Cancer 2007, 120, 451–458.
- Council of Europe; European Pharmacopoeia Commission; European Directorate for the Quality of Medicines & Healthcare. European Pharmacopoeia; Council of Europe: Strasbourg, France, 2019.
- Zong, A.; Cao, H.; Wang, F. Anticancer Polysaccharides from Natural Resources: A Review of Recent Research. Carbohydr. Polym. 2012, 90, 1395–1410.
- Jia, L.; Lv, D.; Zhang, S.; Wang, Z.; Zhou, B. Astragaloside IV Inhibits the Progression of Non-Small Cell Lung Cancer Through the Akt/GSK-3β/β-Catenin Pathway. Ooncol. Res. 2019, 27, 503–508.
- Gülcemal, D.; Masullo, M.; Bedir, E.; Festa, M.; Karayıldırım, T.; Alankus-Caliskan, O.; Piacente, S. Triterpene Glycosides from Astragalus Angustifolius. Planta Med. 2012, 78, 720–729.
- Bourezzane, S.; Haba, H.; Long, C.; Benkhaled, M. Chemical Composition and Antioxidant Activity of Astragalus Monspessulanus L. Growing in Semiarid Areas of Algeria. J. Serb. Chem. Soc. 2018, 83, 31–38.
- Krasteva, I.; Bratkov, V.; Bucar, F.; Kunert, O.; Kollroser, M.; Kondeva-Burdina, M.; Ionkova, I. Flavoalkaloids and Flavonoids from Astragalus Monspessulanus. J. Nat. Prod. 2015, 78, 2565–2571.
- Elenga, P.; Nikolov, S.; Panova, D. Triterpene Glycosides and Sterols from Astragalus Glycyphyllos L. Pharmazie 1986, 41, 300.
- Elenga, P.; Nikolov, S.; Panova, D. Triterpene Glycosides from Astragalus Glycyphyllos L.—A New Natural Compound of the Overground Parts. Pharmazie 1987, 42, 422–423.
- Linnek, J.; Mitaine-Offer, A.; Miyamoto, T.; Lacaille-Dubois, M. Two Cycloartane-Type Glycosides from the Roots of Astragalus Glycyphyllos. Planta Med. 2008, 74, PB141.
- Shkondrov, A.; Krasteva, I.; Bucar, F.; Kunert, O.; Kondeva-Burdina, M.; Ionkova, I. A New Tetracyclic Saponin from Astragalus Glycyphyllos L. and Its Neuroprotective and HMAO-B Inhibiting Activity. Nat. Prod. Res. 2020, 34, 511–517.
- Isah, T.; Umar, S.; Mujib, A.; Sharma, M.P.; Rajasekharan, P.E.; Zafar, N.; Frukh, A. Secondary Metabolism of Pharmaceuticals in the Plant in Vitro Cultures: Strategies, Approaches, and Limitations to Achieving Higher Yield. Plant Cell Tissue Organ Cult. 2018, 132, 239–265.
- Krasteva, I.; Nikolov, S.; Kaloga, M.; Mayer, G. Triterpenoid Saponins from Astragalus Corniculatus. Z. Für. Nat. B 2006, 61, 1166–1169.
- Krasteva, I.; Nikolov, S.; Kaloga, M.; Mayer, G. A New Saponin Lactone from Astragalus Corniculatus. Nat. Prod. Res. 2007, 21, 941–945.
- Krasteva, I.N.; Toshkova, R.A.; Nikolov, S.D. Protective Effect of Astragalus Corniculatus Saponins against Myeloid Graffi Tumour in Hamsters. Phytother. Res. 2004, 18, 255–257.
- Toshkova, R.A.; Krasteva, I.N.; Wesselinova, D.W.; Nikolov, S.D. Influence of Purified Saponin Mixture from Astragalus Corniculatus Bieb. on Phagocytic Cells in Graffi-Tumor Bearing Hamsters. J. Ethnopharmacol. 2007, 109, 394–399.
- Krasteva, I.; Momekov, G.; Zdraveva, P.; Konstantinov, S.; Nikolov, S. Antiproliferative Effects of a Flavonoid and Saponins from Astragalus Hamosus against Human Tumor Cell Lines. Pharmacogn. Mag. 2008, 4, 269–272.
- Dineva, I.; Krasteva, I.; Berger, M.; Konstantinov, S. In Vitro Antineoplastic Activity of Some Cytoreductive Drugs versus New Compounds of Plant Origin. Intern. J. Curr. Chem. 2010, 1, 281–290.
- Du, M.; Wu, X.J.; Ding, J.; Hu, Z.B.; White, K.N.; Branford-White, C.J. Astragaloside IV and Polysaccharide Production by Hairy Roots of Astragalus Membranaceus in Bioreactors. Biotechnol. Lett. 2003, 25, 1853–1856.
- Wilson, S.A.; Roberts, S.C. Recent Advances towards Development and Commercialization of Plant Cell Culture Processes for the Synthesis of Biomolecules: Development and Commercialization of Plant Cell Culture. Plant Biotechnol. J. 2012, 10, 249–268.
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.-M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and Resupply of Pharmacologically Active Plant-Derived Natural Products: A Review. Biotechnol. Adv. 2015, 33, 1582–1614.
- Ionkova, I. Genetic Transformation in Astragalus spp. In Transgenic Medicinal Plants; Biotechnology in Agriculture and Forestry; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1999; pp. 55–72.
- Ionkova, I.; Kartnig, T.; Alfermann, W. Cycloartane Saponin Production in Hairy Root Cultures of Astragalus Mongholicus. Phytochemistry 1997, 45, 1597–1600.
- Giri, A.; Narasu, M.L. Transgenic Hairy Roots. Biotechnol. Adv. 2000, 18, 1–22.
- Ionkova, I.; Alfermann, A. Transformation of Astragalus Species by Agrobacterium Rhizogenes and Their Saponin Production. Planta Med. 1990, 56, 634–635.
- Ionkova, I. Optimization of Flavonoid Production in Cell Cultures of Astragalus Missouriensis Nutt. (Fabaceae). Pharmacogn. Mag. 2009, 5, 92–97.
- Maroyi, A. The Genus Gloriosa (Colchicaceae): Ethnobotany, Phylogeny and Taxonomy; Wageningen University: Wageningen, The Netherlands, 2012.
- Samy, R.P.; Thwin, M.M.; Gopalakrishnakone, P.; Ignacimuthu, S. Ethnobotanical Survey of Folk Plants for the Treatment of Snakebites in Southern Part of Tamilnadu, India. J. Ethnopharmacol. 2008, 115, 302–312.
- Shah, A.; Bharati, K.A.; Ahmad, J.; Sharma, M.P. New Ethnomedicinal Claims from Gujjar and Bakerwals Tribes of Rajouri and Poonch Districts of Jammu and Kashmir, India. J. Ethnopharmacol. 2015, 166, 119–128.
- Maroyi, A. Gloriosa Superba L. (Family Colchicaceae): Remedy or Poison? J. Med. Plants Res. 2011, 5, 6112–6121.
- Ade, R.; Rai, M.K. Review: Current Advances in Gloriosa Superba L. Biodiversitas 2009, 10, 210–214.
- Capistrano, R.; Vangestel, C.; Wouters, A.; Dockx, Y.; Pauwels, P.; Stroobants, S.; Apers, S.; Lardon, F.; Pieters, L.; Staelens, S. Efficacy Screening of Gloriosa Superba Extracts in a Murine Pancreatic Cancer Model Using 18F-FDG PET/CT for Monitoring Treatment Response. Cancer Biother. Radiopharm. 2016, 31, 99–109.
- Budchart, P.; Khamwut, A.; Sinthuvanich, C.; Ratanapo, S.; Poovorawan, Y.; T-Thienprasert, N.P. Partially Purified Gloriosa Superba Peptides Inhibit Colon Cancer Cell Viability by Inducing Apoptosis Through P53 Upregulation. Am. J. Med. Sci. 2017, 354, 423–429.
- Murugesan, A.K.; Pannerselvam, B.; Javee, A.; Rajenderan, M.; Thiyagarajan, D. Facile Green Synthesis and Characterization of Gloriosa Superba L. Tuber Extract-Capped Silver Nanoparticles (GST-AgNPs) and Its Potential Antibacterial and Anticancer Activities against A549 Human Cancer Cells. Environ. Nanotechnol. Monit. Manag. 2021, 15, 100460.
- Gelmi, M.L.; Mottadelli, S.; Pocar, D.; Riva, A.; Bombardelli, E.; De Vincenzo, R.; Scambia, G. N-Deacetyl-N-Aminoacylthiocolchicine Derivatives: Synthesis and Biological Evaluation on MDR-Positive and MDR-Negative Human Cancer Cell Lines. J. Med. Chem. 1999, 42, 5272–5276.
- Reuter, S.; Prasad, S.; Phromnoi, K.; Ravindran, J.; Sung, B.; Yadav, V.R.; Kannappan, R.; Chaturvedi, M.M.; Aggarwal, B.B. Thiocolchicoside Exhibits Anticancer Effects through Downregulation of NF-ΚB Pathway and Its Regulated Gene Products Linked to Inflammation and Cancer. Cancer Prev. Res. 2010, 3, 1462–1472.
- Shi, Q.; Chen, K.; Chen, X.; Brossi, A.; Verdier-Pinard, P.; Hamel, E.; McPhail, A.T.; Tropsha, A.; Lee, K.-H. Antitumor Agents. 183. Syntheses, Conformational Analyses, and Antitubulin Activity of Allothiocolchicinoids. J. Org. Chem. 1998, 63, 4018–4025.
- Balkrishna, A.; Das, S.K.; Pokhrel, S.; Joshi, A.; Verma, S.; Sharma, V.K.; Sharma, V.; Sharma, N.; Joshi, C.S. Colchicine: Isolation, LC–MS QTof Screening, and Anticancer Activity Study of Gloriosa Superba Seeds. Molecules 2019, 24, 2772.
- Lin, Z.-Y.; Kuo, C.-H.; Wu, D.-C.; Chuang, W.-L. Anticancer Effects of Clinically Acceptable Colchicine Concentrations on Human Gastric Cancer Cell Lines. Kaohsiung J. Med. Sci. 2016, 32, 68–73.
- Kitajima, M.; Tanaka, A.; Kogure, N.; Takayama, H. Four New Colchicinoids, Gloriosamines A–D, from Gloriosa Rothschildiana. Tetrahedron Lett. 2008, 49, 257–260.
- Zarev, Y.; Foubert, K.; Ionkova, I.; Apers, S.; Pieters, L. Isolation and Structure Elucidation of Glucosylated Colchicinoids from the Seeds of Gloriosa Superba by LC-DAD-SPE-NMR. J. Nat. Prod. 2017, 80, 1187–1191.
- Sahakitpichan, P.; Chimnoi, N.; Namsa-aid, A.; Panyadee, A.; Ruchirawat, S.; Kanchanapoom, T. Colchicinoid Glucosides from Seedless Pods of Thai Origin Gloriosa Superba. Phytochem. Lett. 2016, 16, 299–302.
- Riva, S.; Sennino, B.; Zambianchi, F.; Danieli, B.; Panza, L. Effect of Organic Cosolvents on the Stability and Activity of the β-1,4-Galactosyltransferase from Bovine Colostrum. Carbohydr. Res. 1997, 305, 525–531.
- Pišvejcová, A.; Rossi, C.; Hušáková, L.; Křen, V.; Riva, S.; Monti, D. β-1,4-Galactosyltransferase-Catalyzed Glycosylation of Sugar Derivatives: Modulation of the Enzyme Activity by α-Lactalbumin, Immobilization and Solvent Tolerance. J. Mol. Catal. B: Enzym. 2006, 39, 98–104.
- Georgieva, A.; Popov, G.; Shkondrov, A.; Toshkova, R.; Krasteva, I.; Kondeva-Burdina, M.; Manov, V. Antiproliferative and Antitumour Activity of Saponins from Astragalus Glycyphyllos on Myeloid Graffi Tumour. J. Ethnopharmacol. 2021, 267, 113519.
- Shkondrov, A.; Krasteva, I.; Ionkova, I.; Popova, P.; Zarev, Y.; Mihaylova, R.; Konstantinov, S. Production of Saponins from in Vitro Cultures of Astragalus Glycyphyllos and Their Antineoplastic Activity. Biotechnol. Biotechnol. Equip. 2019, 33, 1413–1418.
- Shkondrov, A. Phytochemical Investigation of Species from Genus Astragalus L. (Fabaceae); Medical University of Sofia: Sofia, Bulgaria, 2017.
- Mihaylova, R.; Shkondrov, A.; Aluani, D.; Ionkova, I.; Tzankova, V.; Krasteva, I. In Vitro Antitumour and Immunomodulating Activity of Saponins from Astragalus Glycyphyllos. Biotechnol. Biotechnol. Equip. 2021, 35, 1948–1955.
- Benbassat, N.; Nikolov, S. Flavonoids from Astragalus Onobrychis. Planta Med. 1995, 61, 100.
- Kondeva-Burdina, M.; Shkondrov, A.; Simeonova, R.; Vitcheva, V.; Krasteva, I.; Ionkova, I. In Vitro/in Vivo Antioxidant and Hepatoprotective Potential of Defatted Extract and Flavonoids Isolated from Astragalus Spruneri Boiss. (Fabaceae). Food Chem. Toxicol. 2018, 111, 631–640.
- Popova, P.; Zarev, Y.; Mihaylova, R.; Momekov, G.; Ionkova, I. Antiproliferative Activity of Extract from in Vitro Callus Cultures of Astragalus Vesicarius Ssp. Carniolicus (A. Kern.) Chater. Pharmacia 2021, 68, 217–221.
- Capistrano, I.R.; Vangestel, C.; Vanpachtenbeke, H.; Fransen, E.; Staelens, S.; Apers, S.; Pieters, L. Coadministration of a Gloriosa Superba Extract Improves the in Vivo Antitumoural Activity of Gemcitabine in a Murine Pancreatic Tumour Model. Phytomedicine 2016, 23, 1434–1440.
- Saradhadevi, M.; Gnanadesigan, M.; Kapildev, G.; Vasanth, D. Dataset on Antitumor Properties of Silver Nanoparticles from Gloriosa Superba (L.) Seed on Dalton Lymphoma Ascites (DLA) Tumor: Facile and Biocompatible Approach. Data Brief 2017, 14, 524–530.
- Manosroi, A.; Akazawa, H.; Akihisa, T.; Jantrawut, P.; Kitdamrongtham, W.; Manosroi, W.; Manosroi, J. In Vitro Anti-Proliferative Activity on Colon Cancer Cell Line (HT-29) of Thai Medicinal Plants Selected from Thai/Lanna Medicinal Plant Recipe Database “MANOSROI III. ” J. Ethnopharmacol. 2015, 161, 11–17.
- Mamatha, H.; Farooqi, A.A.; Joshi, S.S.; Prasad, T.G. Pollen studies in gloriosa superba linn. Acta Hortic. 1993, 331, 371–376.
- Samarajeewa, P.K. Clonal Propagation of Gloriosa Superba L. Indian J. Exp. Biol. 1993, 31, 719–720.
- Sivakumar, G.; Krishnamurthy, K.; Rajendran, T.D. Embryoidogenesis and Plant Regeneration from Leaf Tissue of Gloriosa Superba. Planta Med 2003, 69, 479–481.
- Jana, S.; Shekhawat, G.S. Critical Review on Medicinally Potent Plant Species: Gloriosa Superba. Fitoterapia 2011, 82, 293–301.
- Arumugam, A.; Gopinath, K. In Vitro Micropropagation Using Corm Bud Explants: An Endangered Medicinal Plant of Gloriosa Superba L. Asian J. Biotechnol. 2012, 4, 120–128.
- Sivakumar, S.; Siva, G.; Sathish, S.; Prem Kumar, G.; Vigneswaran, M.; Vinoth, S.; Kumar, T.S.; Sathishkumar, R.; Jayabalan, N. Influence of Exogenous Polyamines and Plant Growth Regulators on High Frequency in Vitro Mass Propagation of Gloriosa Superba L. and Its Colchicine Content. Biocatal. Agric. Biotechnol. 2019, 18, 101030.
- Mahendran, D.; Kavi Kishor, P.B.; Sreeramanan, S.; Venkatachalam, P. Enhanced Biosynthesis of Colchicine and Thiocolchicoside Contents in Cell Suspension Cultures of Gloriosa Superba L. Exposed to Ethylene Inhibitor and Elicitors. Ind. Crops Prod. 2018, 120, 123–130.
More
