Vasculogenic erectile dysfunction (ED) due to endothelial dysfunction and atherosclerosis of penile arteries is the most common cause of ED, especially in men over fifty. Cell-based regenerative therapies include platelet-rich plasma (PRP), both heterologous and autologous stem cell therapy (SCT), and peripheral blood mononuclear cells (PBMNC), highlighting the role played by immune cell populations, which may represent the new frontier of vasculogenic erectile dysfunction treatment.
- immune cells
- erectile dysfunction
- cell therapy
1. Introduction
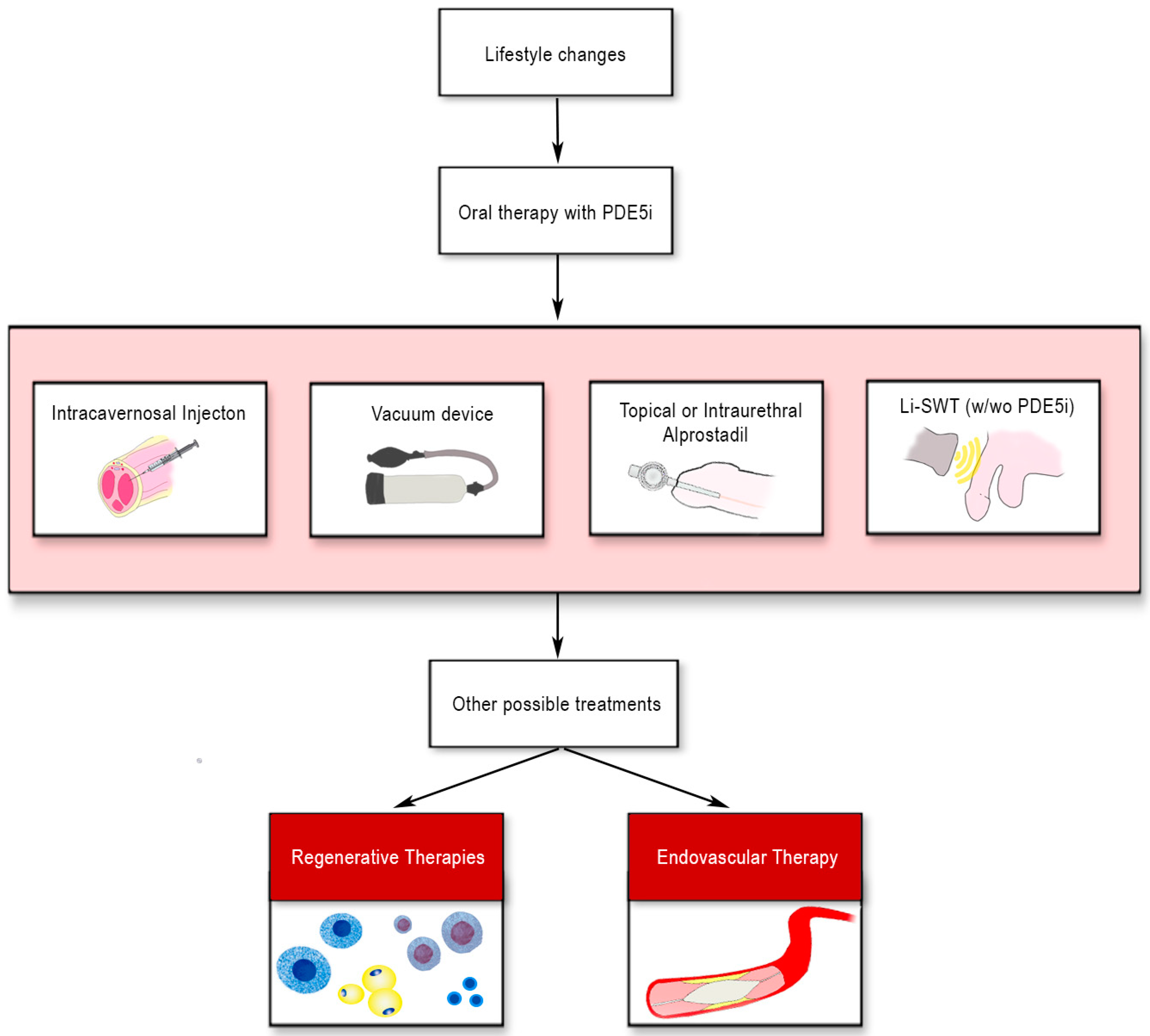
2. Erectile Dysfunction in Patients Affected by Diabetes
3. Stem Cell Therapy and Erectile Dysfunction
SCs are undifferentiated cells capable of unlimited proliferation, multi-differentiation potency, and perpetual self-renewal. The specific mechanisms underlying the effectiveness of SCs in the treatment of ED are not yet understood. Since pre-clinical studies have shown that few stem cells can be detected after transplantation, and almost no direct evidence supports the theory that transplanted stem cells have differentiated into vascular endothelial cells, smooth muscle cells, or nerves, the main mechanisms of action of stem cell transplantation would seem to be related to their paracrine action [30][24].
Moreover, preclinical research has shown that SCs exert their therapeutic effects on the basis of active factors contained in their secretions that can act as messengers. Indeed, the effect of SCs has been shown to persist after their disappearance, and even cell-free treatments have shown benefits [31,32][25][26]. Bioactive factors may represent a future treatment option for ED due to their pro-angiogenic, anti-inflammatory, anti-apoptotic, and anti-fibrotic properties [31,32][25][26].
Notably, the peripheral blood mononuclear cell (PBMNC) secretome differs only slightly from the stem cell secretome in its ability to promote cell proliferation [33][27]. These active paracrine factors, represented by different types of protein molecules, lipid mediators, microRNAs, and exosomes, underlie the regenerative effects of both stem cells and PBMNCs. The proteins are mainly growth factors, cytokines, and chemokines (e.g., CXCL8, CXCL5, CXCL1, CCL5, and VEGF). Lipids and especially oxidised phospholipids (e.g., PLPC-OOH, PAPC-OOH, SGPC, and PGPC) have also shown pleiotropic biological effects, such as neoangiogenesis, but also inflammation modulation by acting on Toll-like receptors (TLRs) and neutrophil granulocytes. Exosomes that arise intracellularly may contain a mixture of proteins, lipids, messenger RNA (mRNA), and micro RNA (miRNA). Due to the complexity of cellular paracrine activity, other factors also play an active role in this process, but these are the biological factors that have been most extensively studied both in vivo and in vitro [34][28]. Apoptotic PBMNCs have been shown to induce angiogenesis and vasodilation, enhance re-epithelialisation, promote macrophage polarization, and modulate the immune system through their paracrine factors. Since the isolation and cultivation of stem cells is not easy, while the secretome is easier to obtain, the latter could take on a central role in regenerative therapy [34][28] (Figure 32).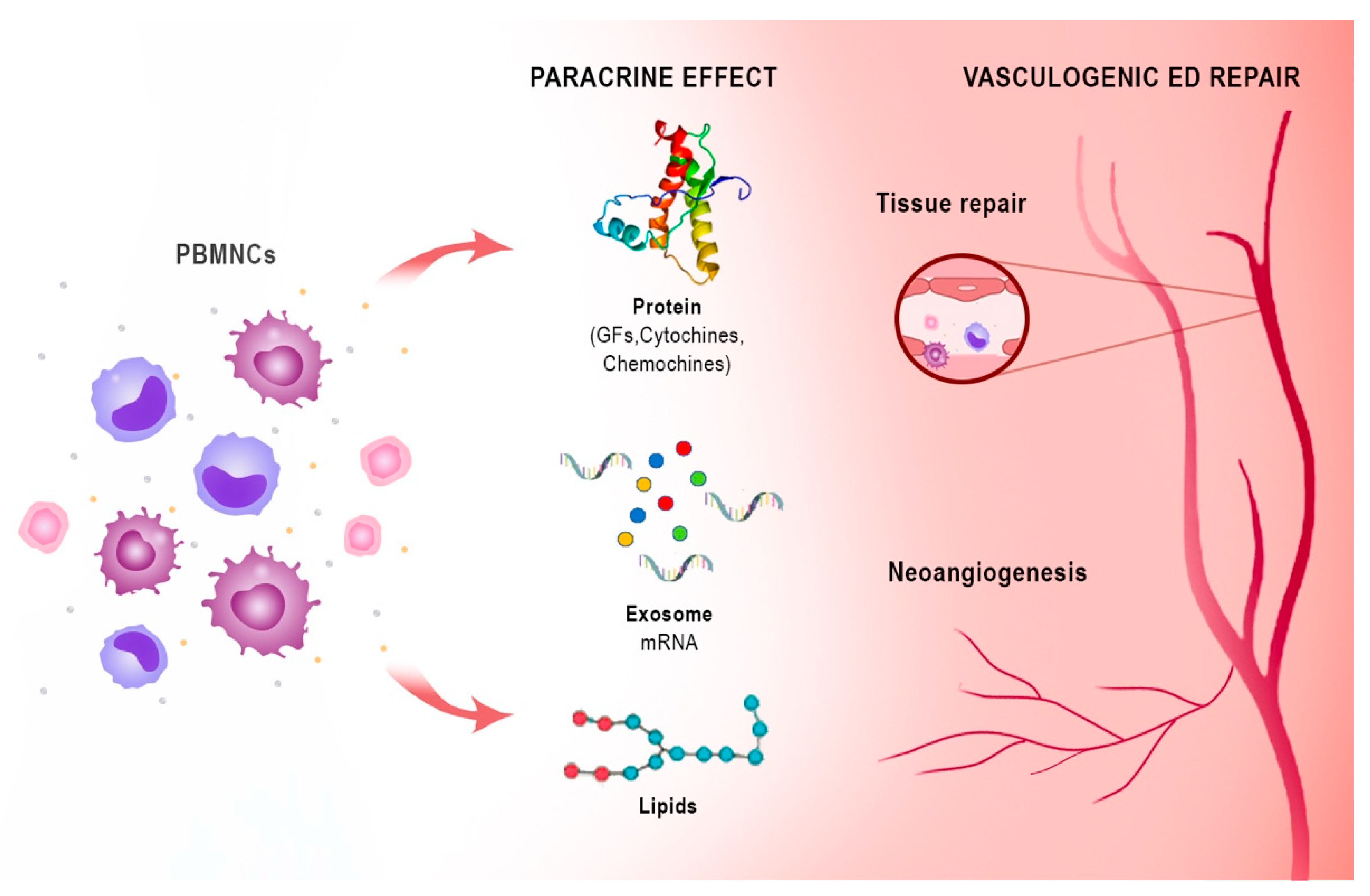
4. Platelet-Rich Plasma Therapy
5. Harnessing the Immune System for Tissue Repair and Regeneration
Tissue healing and regeneration is a complex, organized, spatiotemporal process involving a plethora of cell subsets, the action of which is strictly regulated to obtain an effective tissue [61][31]. Heart disease, critical limb ischemia, diabetic foot, and severe musculoskeletal disorders require new therapeutic strategies to repair damaged tissue, especially considering an aging population where diabetes and obesity have reached gigantic proportions. Clinical trials using adult stem cells to regenerate damaged heart tissue are ongoing, notwithstanding the questions of efficacy and scarcity of understanding of the mechanism of action and of the biological effects [62][32]. The rationale for adult stem cell therapy clinical trials to repair damaged heart tissue is derived from animal studies that showed a limited but reproducible recovery in cardiac function after ischaemic injury [63][33]. Vagnozzi ander co-authorsal. proved that after cells implant after ischaemia–reperfusion injury, although heart function was improved, it was not correlated with the production of new cardiomyocytes [64][34]. On the contrary, two different types of adult stem cell bone marrow mononuclear cells (BM-MNCs), which were the most heavily used stem cell type used in clinical trials, and cardiac mesenchymal cells from the heart that express the receptor tyrosine kinase c-Kit), improved heart function through an acute sterile immune response due to strong recruitment of specific macrophage populations (CCR2+ and CX3CR1+). Moreover, both intracardiac injection of killed stem cell or zymosan, a non-cellular and potent activator of the innate immune response, induced an analogous local macrophage accumulation that provided functional recovery after ischemic damage. Vagnozzi et al. proved that this selective macrophage response acted in multiple fashions altering the activity of the cardiac fibroblasts, reducing in the border zone the extracellular matrix content, and increasing the mechanical asset of the damaged area. These data showed that cardiac cell therapy’s functional benefit was due to the immune system’s acute inflammatory wound-healing response. Interesting to notice, and surprisingly, a zymosan immune-based response maintained its effect for a longer time as opposed to stem-cell therapies.6. Harness Peripheral Blood Mononuclear Cells Angiogenic Potency: From Critical Limb Ischemia to ED
PBMNCs based on monocytes/macrophages and lymphocytes are an innovative autologous cell therapy that have shown angiogenesis potency and tissue regeneration in no-option critical limb patients and in diabetic foot patients [80,81,82,83][35][36][37][38] (Figure 32). The detailed mechanism of action of PBMNCs is beyond the scope of this review and is adequately described in a review on autologous cell therapy [46]. BrieBriefly, the angiogenic and arteriogenic ability of monocytes/macrophages is well known and extensively described [84,85,86,87][39][40][41][42]. It has also been observed that monocytes/macrophages are able to repair cerebrovascular ruptures in haemorrhagic strokes due to their ability to physically adhere to rupture sites and generate mechanical traction [88][43]. Krishnasamy et al. demonstrated that Notch signalling recruits macrophage differentiation and maturation from monocytes, promoting arteriogenesis and tissue repair in ischaemic tissue [89][44]. A point-of-care (POC) device has recently shown reasonable efficacy in therapeutic angiogenesis both in vivo and in vitro. It is a device based on selective filtration of peripheral blood and is intended for intraoperative use in human cell therapy to produce fresh autologous PBMNCs. [97,98][45][46]. PBMNCs produced with this device (Hematrate Blood Filtration system—Cook Regentec) have shown promising results in several clinical trials, including studies in diabetic patients [81,99,100][36][47][48].7. Conclusions
Recommended ED treatments frequently do not achieve adequate results, particularly in diabetic patients. Regenerative therapies, including platelet-rich plasma (PRP) and stem cell therapy (SCT), are starting to be used for ED treatment as an adjunct or alternative therapy, although on a limited number of patients. PRP delivers an autologous sample rich in growth factors to damaged tissue. PRP studies have shown an increased erectile function recovery and preservation of cavernous nerve axons on animal models; however, studies with PRP in humans are very limited. SCT has been used in diabetic patients and post-prostatectomy ED with mixed results in clinical trials, although SCT treatments improved erectile rigidity and functionality. Still, there is a lack of evidence to support the efficacy of these treatments. The scenario seems similar to the initial enthusiasm for cell therapy in ischemic heart disease, which was dampened by less than brilliant results in the clinic despite promising efficacy data in animal models.References
- Salonia, A.; Bettocchi, C.; Boeri, L.; Capogrosso, P.; Carvalho, J.; Cilesiz, N.C.; Cocci, A.; Corona, G.; Dimitropolous, K.; Gül, M.; et al. European Association of Urology Guidelines on Sexual and Reproductive Health-2021 Update: Male Sexual Dysfunction. Eur. Urol. 2021, 25, 333–357.
- Burnett, A.L.; Nehra, A.; Breau, R.H.; Culkin, D.J.; Faraday, M.M.; Hakim, L.S.; Heidelbaugh, J.; Khera, M.; McVary, K.T.; Miner, M.M.; et al. Erectile Dysfunction: AUA Guideline. J. Urol. 2018, 200, 633–641.
- McMahon, C.N.; Smith, C.J.; Shabsigh, R. Treating erectile dysfunction when PDE5 inhibitors fail. BMJ 2006, 332, 589–592.
- Penson, D.F.; Latini, D.M.; Lubeck, D.P.; Wallace, K.L.; Henning, J.M.; Lue, T.F. Do impotent men with diabetes have more severe erectile dysfunction and worse quality of life than the general population of impotent patients? Results from the Exploratory Comprehensive Evaluation of Erectile Dysfunction (ExCEED) database. Diabetes Care 2003, 26, 1093–1099.
- De Angelis, L.; Marfella, M.A.; Siniscalchi, M.; Marino, L.; Nappo, F.; Giugliano, F.; De Lucia, D.; Giugliano, D. Erectile and endothelial dysfunction in Type II diabetes: A possible link. Diabetologia 2001, 44, 1155–1160.
- Chitaley, K. Type 1 and Type 2 diabetic-erectile dysfunction: Same diagnosis (ICD-9), different disease? J. Sex. Med. 2009, 6 (Suppl. 3), 262–268.
- Chitaley, K.; Kupelian, V.; Subak, L.; Wessells, H. Diabetes, obesity and erectile dysfunction: Field overview and research priorities. J. Urol. 2009, 182 (Suppl. 6), S45–S50.
- Feldman, H.A.; Goldstein, I.; Hatzichristou, D.G.; Krane, R.J.; McKinlay, J.B. Impotence and its medical and psychosocial correlates: Results of the Massachusetts Male Aging Study. J Urol. 1994, 151, 54–61.
- Lehman, T.P.; Jacobs, J.A. Etiology of diabetic impotence. J. Urol. 1983, 129, 291–294.
- Koncz, L.; Balodimos, M.C. Impotence in diabetes mellitus. Med. Times 1970, 98, 159–170.
- Whitehead, E.D.; Klyde, B.J. Diabetes-related impotence in the elderly. Clin. Geriatr. Med. 1990, 6, 771–795.
- Ganz, P.; Vita, J.A. Testing endothelial vasomotor function: Nitric oxide, a multipotent molecule. Circulation 2003, 108, 2049–2053.
- Kim, N.; Azadzoi, K.M.; Goldstein, I.; Saenz de Tejada, I. A nitric oxide-like factor mediates nonadrenergic-noncholinergic neurogenic relaxation of penile corpus cavernosum smooth muscle. J. Clin. Investig. 1991, 88, 112–118.
- Cellek, S.; Rodrigo, J.; Lobos, E.; Fernández, P.; Serrano, J.; Moncada, S. Selective nitrergic neurodegeneration in diabetes mellitus—A nitric oxide-dependent phenomenon. Br. J. Pharmacol. 1999, 128, 1804–1812.
- Brownlee, M.; Cerami, A.; Vlassara, H. Advanced glycosylation end products in tissue and the biochemical basis of diabetic complications. N. Engl. J. Med. 1988, 318, 1315–1321.
- Melis, M.R.; Argiolas, A. Role of central nitric oxide in the control of penile erection and yawning. Prog. Neuropsychopharmacol. Biol. Psychiatry 1997, 21, 899–922.
- Cartledge, J.J.; Eardley, I.; Morrison, J.F. Nitric oxide-mediated corpus cavernosal smooth muscle relaxation is impaired in ageing and diabetes. BJU Int. 2001, 87, 394–401.
- Saenz de Tejada, I.; Goldstein, I.; Azadzoi, K.; Krane, R.J.; Cohen, R.A. Impaired neurogenic and endothelium-mediated relaxation of penile smooth muscle from diabetic men with impotence. N. Engl. J. Med. 1989, 320, 1025–1030.
- Bivalacqua, T.J.; Champion, H.C.; Usta, M.F.; Cellek, S.; Chitaley, K.; Webb, R.C.; Lewis, R.L.; Mills, T.M.; Hellstrom, W.J.; Kadowitz, P.J. RhoA/Rho-kinase suppresses endothelial nitric oxide synthase in the penis: A mechanism for diabetes-associated erectile dysfunction. Proc. Natl. Acad. Sci. USA 2004, 101, 9121–9126.
- Vita, J.A.; Keaney, J.F., Jr. Endothelial function: A barometer for cardiovascular risk? Circulation 2002, 106, 640–642.
- Newsholme, P.; Haber, E.P.; Hirabara, S.M.; Rebelato, E.L.; Procopio, J.; Morgan, D.; Oliveira-Emilio, H.C.; Carpinelli, A.R.; Curi, R. Diabetes associated cell stress and dysfunction: Role of mitochondrial and non-mitochondrial ROS production and activity. J. Physiol. 2007, 583 Pt 1, 9–24.
- Yan, S.F.; Ramasamy, R.; Schmidt, A.M. Mechanisms of disease: Advanced glycation end-products and their receptor in inflammation and diabetes complications. Nat. Clin. Pract. Endocrinol. Metab. 2008, 4, 285–293.
- Cignarelli, A.; Genchi, V.A.; D’Oria, R.; Giordano, F.; Caruso, I.; Perrini, S.; Natalicchio, A.; Laviola, L.; Giorgino, F. Role of Glucose-Lowering Medications in Erectile Dysfunction. J. Clin. Med. 2021, 10, 2501.
- Shan, H.; Chen, F.; Zhang, T.; He, S.; Xu, L.; Wei, A. Stem cell therapy for erectile dysfunction of cavernous nerve injury rats: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0121428.
- Sun, D.Z.; Abelson, B.; Babbar, P.; Damaser, M.S. Harnessing the mesenchymal stem cell secretome for regenerative urology. Nat. Rev. Urol. 2019, 16, 363–375.
- Sun, C.; Lin, H.; Yu, W.; Li, X.; Chen, Y.; Qiu, X.; Wang, R.; Dai, Y. Neurotrophic effect of bone marrow mesenchymal stem cells for erectile dysfunction in diabetic rats. Int. J. Androl. 2012, 35, 601–607.
- Korf-Klingebiel, M.; Kempf, T.; Sauer, T.; Brinkmann, E.; Fischer, P.; Meyer, G.P.; Ganser, A.; Drexler, H.; Wollert, K.C. Bone marrow cells are a rich source of growth factors and cytokines: Implications for cell therapy trials after myocardial infarction. Eur. Heart J. 2008, 29, 2851–2858.
- Beer, L.; Mildner, M.; Gyöngyösi, M.; Ankersmit, H.J. Peripheral blood mononuclear cell secretome for tissue repair. Apoptosis 2016, 21, 1336–1353.
- Ding, X.G.; Li, S.W.; Zheng, X.M.; Hu, L.Q.; Hu, W.L.; Luo, Y. The effect of platelet-rich plasma on cavernous nerve regeneration in a rat model. Asian J. Androl. 2009, 11, 215–221.
- Wu, C.C.; Wu, Y.N.; Ho, H.O.; Chen, K.C.; Sheu, M.T.; Chiang, H.S. The neuroprotective effect of platelet-rich plasma on erectile function in bilateral cavernous nerve injury rat model. J. Sex. Med. 2012, 9, 2838–2848.
- Shanley, L.C.; Mahon, O.R.; Kelly, D.J.; Dunne, A. Harnessing the innate and adaptive immune system for tissue repair and regeneration: Considering more than macrophages. Acta Biomater. 2021, 133, 208–221.
- Epstein, J.A. A Time to Press Reset and Regenerate Cardiac Stem Cell Biology. JAMA Cardiol. 2019, 4, 95–96.
- Tompkins, B.A.; Balkan, W.; Winkler, J.; Gyöngyösi, M.; Goliasch, G.; Fernández-Avilés, F.; Hare, J.M. Pre-clinical Studies of Stem Cell Therapy for Heart Disease. Circ. Res. 2018, 122, 1006–1020.
- Vagnozzi, R.J.; Maillet, M.; Sargent, M.A.; Khalil, H.; Johansen, A.K.Z.; Schwanekamp, J.A.; York, A.J.; Huang, V.; Nahrendorf, M.; Sadayappan, S.; et al. An acute immune response underlies the benefit of cardiac stem cell therapy. Nature 2020, 577, 405–409.
- Forbes, S.J.; Rosenthal, N. Preparing the ground for tissue regeneration: From mechanism to therapy. Nat. Med. 2014, 20, 857–869.
- Dubsky, M.; Jirkovska, A.; Bem, R.; Fejfarova, V.; Pagacova, L.; Sixta, B.; Varga, M.; Langkramer, S.; Sykova, E.; Jude, E.B. Both autologous bone marrow mononuclear cell and peripheral blood progenitor cell therapies similarly improve ischaemia in patients with diabetic foot in comparison with control treatment. Diabetes Metab. Res. Rev. 2013, 29, 369–376.
- Persiani, F.; Paolini, A.; Camilli, D.; Mascellari, L.; Platone, A.; Magenta, A.; Furgiuele, S. Peripheral Blood Mononuclear Cells Therapy for Treatment of Lower Limb Ischemia in Diabetic Patients: A Single-Center Experience. Ann. Vasc. Surg. 2018, 53, 190–196.
- De Angelis, B.; Gentile, P.; Orlandi, F.; Bocchini, I.; Di Pasquali, C.; Agovino, A.; Gizzi, C.; Patrizi, F.; Scioli, M.G.; Orlandi, A.; et al. Limb rescue: A new autologous-peripheral blood mononuclear cells technology in critical limb ischemia and chronic ulcers. Tissue Eng. Part C Methods 2015, 21, 423–435.
- Fantin, A.; Vieira, J.M.; Gestri, G.; Denti, L.; Schwarz, Q.; Prykhozhij, S.; Peri, F.; Wilson, S.W.; Ruhrberg, C. Tissue macrophages act as cellular chaperones for vascular anastomosis downstream of VEGF-mediated endothelial tip cell induction. Blood 2010, 116, 829–840.
- Fung, E.; Helisch, A. Macrophages in collateral arteriogenesis. Front. Physiol. 2012, 3, 353.
- Barnett, F.H.; Rosenfeld, M.; Wood, M.; Kiosses, W.B.; Usui, Y.; Marchetti, V.; Aguilar, E.; Friedlander, M. Macrophages form functional vascular mimicry channels in vivo. Sci. Rep. 2016, 6, 36659.
- Gurevich, D.B.; Severn, C.E.; Twomey, C.; Greenhough, A.; Cash, J.; Toye, A.M.; Mellor, H.; Martin, P. Live imaging of wound angiogenesis reveals macrophage orchestrated vessel sprouting and regression. EMBO J. 2018, 37, e97786.
- Liu, C.; Wu, C.; Yang, Q.; Gao, J.; Li, L.; Yang, D.; Luo, L. Macrophages Mediate the Repair of Brain Vascular Rupture through Direct Physical Adhesion and Mechanical Traction. Immunity 2016, 44, 1162–1176.
- Krishnasamy, K.; Limbourg, A.; Kapanadze, T.; Gamrekelashvili, J.; Beger, C.; Häger, C.; Lozanovski, V.J.; Falk, C.S.; Napp, L.C.; Bauersachs, J.; et al. Blood vessel control of macrophage maturation promotes arteriogenesis in ischemia. Nat. Commun. 2017, 8, 952.
- Rigato, M.; Monami, M.; Fadini, G.P. Autologous Cell Therapy for Peripheral Arterial Disease: Systematic Review and Meta-Analysis of Randomized, Nonrandomized, and Noncontrolled Studies. Circ. Res. 2017, 120, 1326–1340.
- Spaltro, G.; Straino, S.; Gambini, E.; Bassetti, B.; Persico, L.; Zoli, S.; Zanobini, M.; Capogrossi, M.C.; Spirito, R.; Quarti, C.; et al. Characterization of the Pall Celeris system as a point-of-care device for therapeutic angiogenesis. Cytotherapy 2015, 17, 1302–1313.
- Huang, P.P.; Yang, X.F.; Li, S.Z.; Wen, J.C.; Zhang, Y.; Han, Z.C. Randomised comparison of G-CSF-mobilized peripheral blood mononuclear cells versus bone marrow-mononuclear cells for the treatment of patients with lower limb arteriosclerosis obliterans. Thromb. Haemost. 2007, 98, 1335–1342.
- Dubský, M.; Jirkovská, A.; Bem, R.; Fejfarová, V.; Pagacová, L.; Nemcová, A.; Sixta, B.; Chlupac, J.; Peregrin, J.H.; Syková, E.; et al. Comparison of the effect of stem cell therapy and percutaneous transluminal angioplasty on diabetic foot disease in patients with critical limb ischemia. Cytotherapy 2014, 16, 1733–1738.
