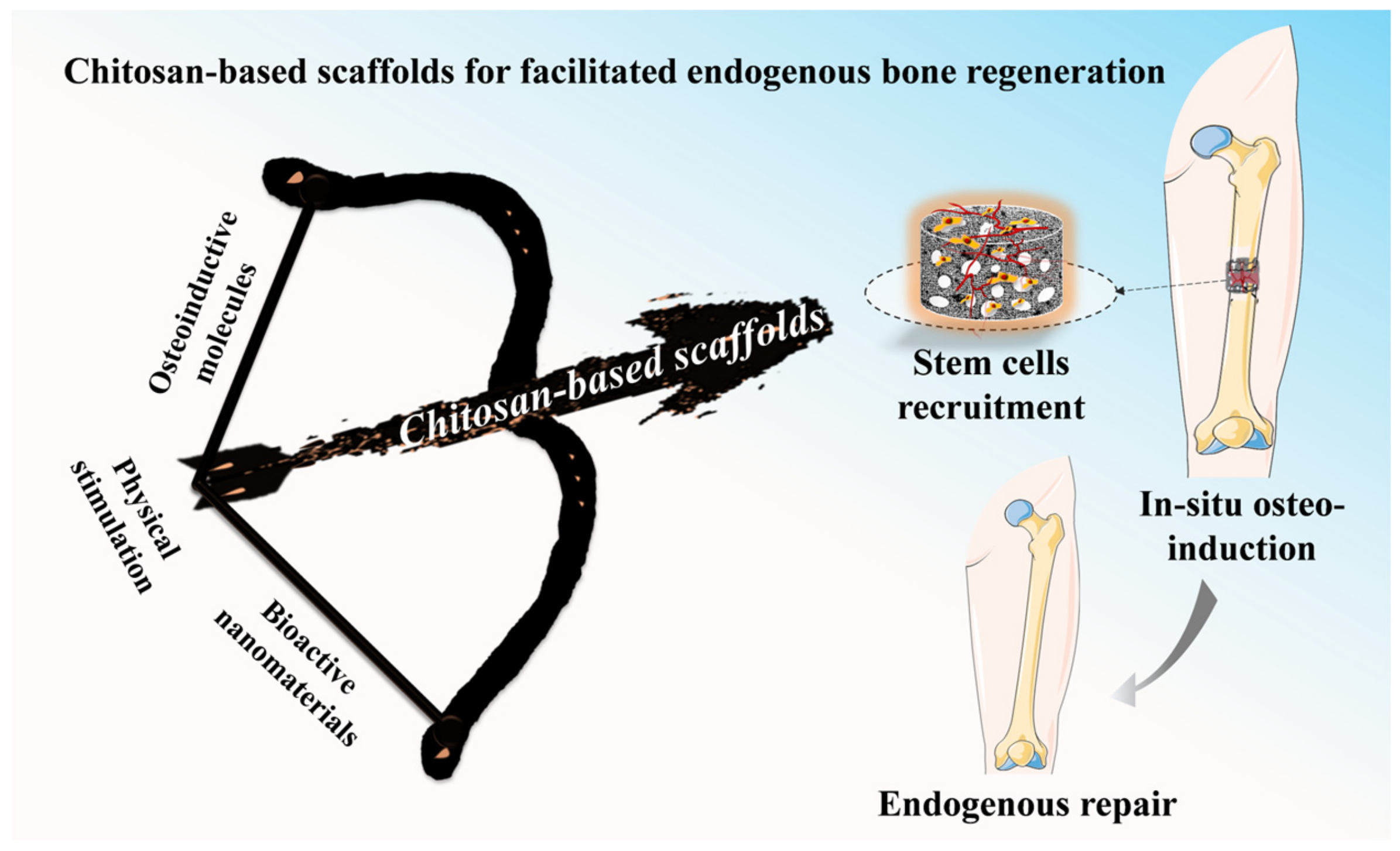
In comparison with the bone tissue engineering (BTE) strategy, the facilitated endogenous tissue engineering (FEBTE) strategy as a novel practical approach tries to eliminate time-consuming and costly tedious process: tissue harvest, cell isolation and ex vivo co-culture with a scaffold. Based on this, the FEBTE strategy as a facile and effective strategy, is booming in bone tissue regeneration. Particularly, chitosan (CS)-based scaffolds with versatile qualities including good biocompatibility, biodegradability, and tunable physicochemical and biological properties could recruit endogenous stem cells homing and differentiation towards lesion areas during the process of bone repair.
In comparison with the bone tissue engineering (BTE) strategy, the facilitated endogenous tissue engineering (FEBTE) strategy as a novel practical approach tries to eliminate time-consuming and costly tedious process: tissue harvest, cell isolation and ex vivo co-culture with a scaffold. Based on this, the FEBTE strategy as a facile and effective strategy, is booming in bone tissue regeneration. Particularly, chitosan (CS)-based scaffolds with versatile qualities including good biocompatibility, biodegradability, and tunable physicochemical and biological properties could recruit endogenous stem cells homing and differentiation towards lesion areas during the process of bone repair.
- facilitated endogenous tissue engineering
- chitosan
- bone repair
- bioactive scaffold
- functional design
1. Introduction
2. Bone Repair Strategies
2.1. Traditional Bone Tissue Engineering
Although auto/allogeneic transplantations, as the golden standard in the clinical setting, have achieved positive results in critical-sized bone defects repair, the limited donor sources, immunogenicity and the risk of infection caused by the second surgical procedure limit their widespread development [44][45]. In order to overcome the drawbacks of auto/allogeneic transplantations, the emergence of BTE has been warmly welcomed in the last three decades [46]. Briefly, the BTE strategy mainly consists of three parts: a scaffold, cells and growth factors [47]. As shown in Figure 2, the procedure begins with the isolation and harvest of target autologous tissues, then resuscitation and expansion of stem cells in specific culture equipment. After reaching a sufficient number, the cells are seeded into a prefabricated scaffold for co-cultivation in vitro. Simultaneously, suitable growth factors and nutrients are continuously added to provide beneficial conditions. Finally, the differentiated new tissues are implanted into the patient’s lesions to further interact with the host tissue, and finally promote defect tissue healing in vivo [48][49][50].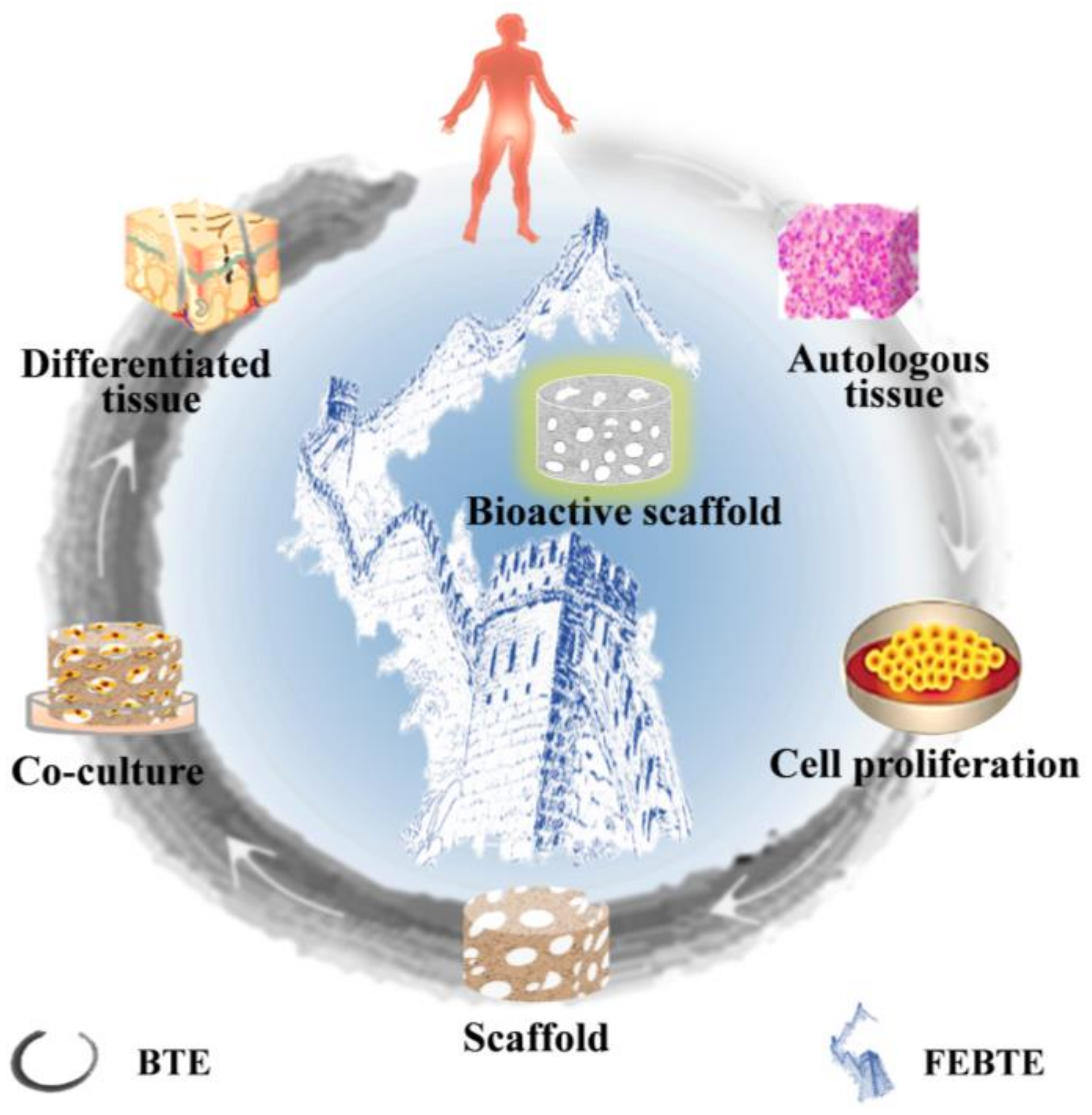
2.2. Facilitated Endogenous Bone Tissue Engineering
The issues of complexity and high cost need to be addressed if BTE is to avoid becoming an expensive therapy available only to the wealthy. Specifically, wresearchers need to develop large-scale automated and replicable production systems as alternatives to labor-intensive production process. In view of the organism’s own repair potential, the biological microenvironment is used as a bioreactor to simplify the tedious process of BTE and finally achieve bone healing in situ. Therefore, the facilitated endogenous bone tissue engineering (FEBTE) strategy has emerged [11]. In comparison with the BTE strategy, the FEBTE strategy as a novel practical approach tries to eliminate time-consuming and costly tedious process: tissue harvest, cell isolation and ex vivo co-culture with a scaffold. Interestingly, this strategy only requires the implantation of a bioactive scaffold into the bone defect sites to induce the defect tissue repair by itself (Figure 2). The bioactive scaffold functions as a “gravitational field” to attract and positively recruit endogenous stem cells and growth factors to the damage site, and then promotes stem cells proliferation and osteogenic differentiation, thereby repairing bone defects [15]. Therefore, based on the convenience and cost-effectiveness of the FEBTE strategy, it has successfully attracted extensive attention in the field of scientific research [14][52]. Among them, the most important and key point is to construct a bone repair scaffold with excellent osteo-inductivity.
References
- Farokhi, M.; Mottaghitalab, F.; Samani, S.; Shokrgozar, M.A.; Kundu, S.C.; Reis, R.L.; Fatahi, Y.; Kaplan, D.L. Silk fibroin/hydroxyapatite composites for bone tissue engineering. Biotechnol. Adv. 2018, 36, 68–91.
- Wu, M.; Zhao, Y.; Jiang, H.; Xu, X.; Wang, D.; Xu, X.; Zhou, Y.; Tan, H.; Ding, C.; Li, J. Self-Organized Spatiotemporal Mineralization of Hydrogel: A Simulant of Osteon. Small 2022, 18, e2106649.
- Liu, Y.; Liu, S.; Luo, D.; Xue, Z.; Yang, X.; Gu, L.; Zhou, Y.; Wang, T. Hierarchically Staggered Nanostructure of Mineralized Collagen as a Bone-Grafting Scaffold. Adv. Mater. 2016, 28, 8740–8748.
- Li, X.; Wei, L.; Li, J.; Shao, J.; Yi, B.; Zhang, C.; Liu, H.; Ma, B.; Ge, S. Multifunctional SDF-1-loaded hydroxyapatite/polylactic acid membranes promote cell recruitment, immunomodulation, angiogenesis, and osteogenesis for biomimetic bone regeneration. Appl. Mater. Today 2021, 22, 100942.
- Koons, G.L.; Diba, M.; Mikos, A.G. Materials design for bone-tissue engineering. Nat. Rev. Mater. 2020, 5, 584–603.
- Qu, H.; Fu, H.; Han, Z.; Sun, Y. Biomaterials for bone tissue engineering scaffolds: A review. RSC Adv. 2019, 9, 26252–26262.
- Oryan, A.; Alidadi, S.; Moshiri, A.; Maffulli, N. Bone regenerative medicine: Classic options, novel strategies, and future directions. J. Orthop. Surg. Res. 2014, 9, 18.
- Bose, S.; Roy, M.; Bandyopadhyay, A. Recent advances in bone tissue engineering scaffolds. Trends Biotechnol. 2012, 30, 546–554.
- Amini, A.R.; Laurencin, C.T.; Nukavarapu, S.P. Bone tissue engineering: Recent advances and challenges. Crit. Rev. Biomed. Eng. 2012, 40, 363–408.
- Evans, C.H.; Palmer, G.D.; Pascher, A.; Porter, R.; Kwong, F.N.; Gouze, E.; Gouze, J.N.; Liu, F.; Steinert, A.; Betz, O.; et al. Facilitated endogenous repair: Making tissue engineering simple, practical, and economical. Tissue Eng. 2007, 13, 1987–1993.
- Yao, Q.; Liu, Y.; Tao, J.; Baumgarten, K.M.; Sun, H. Hypoxia-Mimicking Nanofibrous Scaffolds Promote Endogenous Bone Regeneration. ACS Appl. Mater. Interfaces 2016, 8, 32450–32459.
- Chen, J.; Zhang, Y.; Pan, P.; Fan, T.; Chen, M.; Zhang, Q. In situ strategy for bone repair by facilitated endogenous tissue engineering. Colloids Surf. B Biointerfaces 2015, 135, 581–587.
- Wang, Q.; Tang, Y.; Ke, Q.; Yin, W.; Zhang, C.; Guo, Y.; Guan, J. Magnetic lanthanum-doped hydroxyapatite/chitosan scaffolds with endogenous stem cell-recruiting and immunomodulatory properties for bone regeneration. J. Mater. Chem. B 2020, 8, 5280–5292.
- Zhao, Y.; Chen, J.; Zou, L.; Xu, G.; Geng, Y. Facile one-step bioinspired mineralization by chitosan functionalized with graphene oxide to activate bone endogenous regeneration. Chem. Eng. J. 2019, 378, 122174.
- Collins, M.N.; Ren, G.; Young, K.; Pina, S.; Reis, R.L.; Oliveira, J.M. Scaffold fabrication technologies and structure/function properties in bone tissue engineering. Adv. Funct. Mater. 2021, 31, 2010609.
- Turnbull, G.; Clarke, J.; Picard, F.; Riches, P.; Jia, L.; Han, F.; Li, B.; Shu, W. 3D bioactive composite scaffolds for bone tissue engineering. Bioact. Mater. 2018, 3, 278–314.
- Wang, S.; Yang, Y.; Zhao, Z.; Wang, X.; Mikos, A.G.; Qiu, Z.; Song, T.; Sun, X.; Zhao, L.; Zhang, C.; et al. Mineralized Collagen-Based Composite Bone Materials for Cranial Bone Regeneration in Developing Sheep. ACS Biomater. Sci. Eng. 2017, 3, 1092–1099.
- Wang, S.; Zhao, Z.; Yang, Y.; Mikos, A.G.; Qiu, Z.; Song, T.; Cui, F.; Wang, X.; Zhang, C. A high-strength mineralized collagen bone scaffold for large-sized cranial bone defect repair in sheep. Regen. Biomater. 2018, 5, 283–292.
- Torgbo, S.; Sukyai, P. Bacterial cellulose-based scaffold materials for bone tissue engineering. Appl. Mater. Today 2018, 11, 34–49.
- Gambari, L.; Amore, E.; Raggio, R.; Bonani, W.; Barone, M.; Lisignoli, G.; Grigolo, B.; Motta, A.; Grassi, F. Hydrogen sulfide-releasing silk fibroin scaffold for bone tissue engineering. Mater. Sci. Eng. C 2019, 102, 471–482.
- Park, S.H.; Park, J.Y.; Ji, Y.B.; Ju, H.J.; Min, B.H.; Kim, M.S. An injectable click-crosslinked hyaluronic acid hydrogel modified with a BMP-2 mimetic peptide as a bone tissue engineering scaffold. Acta Biomater. 2020, 117, 108–120.
- Janmohammadi, M.; Nazemi, Z.; Salehi, A.O.M.; Seyfoori, A.; John, J.V.; Nourbakhsh, M.S.; Akbari, M. Cellulose-based composite scaffolds for bone tissue engineering and localized drug delivery. Bioact. Mater. 2023, 20, 137–163.
- Deshpande, R.; Shukla, S.; Sayyad, R.; Salunke, S.; Nisal, A.; Venugopalan, P. Silk fibroin and ceramic scaffolds: Comparative in vitro studies for bone regeneration. Bioeng.Transl. Med. 2021, 6, e10221.
- Abdallah, M.M.; Fernández, N.; Matias, A.A.; Bronze, M.d.R. Hyaluronic acid and Chondroitin sulfate from marine and terrestrial sources: Extraction and purification methods. Carbohydr. Polym. 2020, 243, 116441.
- Pakizeh, M.; Moradi, A.; Ghassemi, T. Chemical extraction and modification of chitin and chitosan from shrimp shells. Eur. Polym. J. 2021, 159, 110709.
- Hu, D.; Ren, Q.; Li, Z.; Zhang, L. Chitosan-based biomimetically mineralized composite materials in human hard tissue repair. Molecules 2020, 25, 4785.
- Levengood, S.L.; Zhang, M. Chitosan-based scaffolds for bone tissue engineering. J. Mater. Chem. B 2014, 2, 3161–3184.
- LogithKumar, R.; KeshavNarayan, A.; Dhivya, S.; Chawla, A.; Saravanan, S.; Selvamurugan, N. A review of chitosan and its derivatives in bone tissue engineering. Carbohydr. Polym. 2016, 151, 172–188.
- Tian, Y.; Wu, D.; Wu, D.; Cui, Y.; Ren, G.; Wang, Y.; Wang, J.; Peng, C. Chitosan-Based Biomaterial Scaffolds for the Repair of Infected Bone Defects. Front. Bioeng. Biotechnol. 2022, 10, 899760.
- Querido, D.; Vieira, T.; Ferreira, J.L.; Henriques, C.; Borges, J.P.; Silva, J.C. Study on the incorporation of chitosan flakes in electrospun polycaprolactone scaffolds. Polymers 2022, 14, 1496.
- Xu, T.; Yang, H.; Yang, D.; Yu, Z.Z. Polylactic acid nanofiber scaffold decorated with chitosan islandlike topography for bone tissue engineering. ACS Appl. Mater. Interfaces 2017, 9, 21094–21104.
- Amirian, J.; Tripathi, G.; Kang, H.-J.; Lee, B.-T. Porous BMP-2 immobilized PLGA/Glycol chitosan scaffold with enhanced hydrophilicity, mineralization and osteogenesis. Mater. Lett. 2022, 308, 131140.
- Tao, F.; Cheng, Y.; Tao, H.; Jin, L.; Wan, Z.; Dai, F.; Xiang, W.; Deng, H. Carboxymethyl chitosan/sodium alginate-based micron-fibers fabricated by emulsion electrospinning for periosteal tissue engineering. Mater. Des. 2020, 194, 108849.
- Yang, Y.; Ao, H.; Wang, Y.; Lin, W.; Yang, S.; Zhang, S.; Yu, Z.; Tang, T. Cytocompatibility with osteogenic cells and enhanced in vivo anti-infection potential of quaternized chitosan-loaded titania nanotubes. Bone Res. 2016, 4, 16027.
- Cui, Z.-K.; Kim, S.; Baljon, J.J.; Wu, B.M.; Aghaloo, T.; Lee, M. Microporous methacrylated glycol chitosan-montmorillonite nanocomposite hydrogel for bone tissue engineering. Nat. Commun. 2019, 10, 3523.
- Zhang, S.; Zhao, G.; Ma, W.; Song, Y.; Huang, C.; Xie, C.; Chen, K.; Li, X. The root-like chitosan nanofiber porous scaffold cross-linked by genipin with type I collagen and its osteoblast compatibility. Carbohydr. Polym. 2022, 285, 119255.
- Menon, A.H.; Soundarya, S.P.; Sanjay, V.; Chandran, S.V.; Balagangadharan, K.; Selvamurugan, N. Sustained release of chrysin from chitosan-based scaffolds promotes mesenchymal stem cell proliferation and osteoblast differentiation. Carbohydr. Polym. 2018, 195, 356–367.
- Stevanović, M.; Djošić, M.; Janković, A.; Kojić, V.; Vukašinović-Sekulić, M.; Stojanović, J.; Odović, J.; Crevar Sakač, M.; Kyong Yop, R.; Mišković-Stanković, V. Antibacterial graphene-based hydroxyapatite/chitosan coating with gentamicin for potential applications in bone tissue engineering. J. Biomed. Mater. Res. A 2020, 108, 2175–2189.
- Ding, T.; Kang, W.; Li, J.; Yu, L.; Ge, S. An in situ tissue engineering scaffold with growth factors combining angiogenesis and osteoimmunomodulatory functions for advanced periodontal bone regeneration. J. Nanobiotechnol. 2021, 19, 247.
- Zeng, R.; Tu, M.; Liu, H.; Zhao, J.; Zha, Z.; Zhou, C. Preparation, structure, drug release and bioinspired mineralization of chitosan-based nanocomplexes for bone tissue engineering. Carbohydr. Polym. 2009, 78, 107–111.
- Brun, P.; Zamuner, A.; Battocchio, C.; Cassari, L.; Todesco, M.; Graziani, V.; Iucci, G.; Marsotto, M.; Tortora, L.; Secchi, V.; et al. Bio-Functionalized Chitosan for Bone Tissue Engineering. Int. J. Mol. Sci. 2021, 22, 5916.
- Zhao, Y.; Fan, T.; Chen, J.; Su, J.; Zhi, X.; Pan, P.; Zou, L.; Zhang, Q. Magnetic bioinspired micro/nanostructured composite scaffold for bone regeneration. Colloids Surf. B 2019, 174, 70–79.
- Patel, A.; Zaky, S.H.; Schoedel, K.; Li, H.; Sant, V.; Beniash, E.; Sfeir, C.; Stolz, D.B.; Sant, S. Design and evaluation of collagen-inspired mineral-hydrogel nanocomposites for bone regeneration. Acta Biomater. 2020, 112, 262–273.
- Cao, S.; Zhao, Y.; Hu, Y.; Zou, L.; Chen, J. New perspectives: In-situ tissue engineering for bone repair scaffold. Compos. B Eng. 2020, 202, 108445.
- Wang, S.; Yang, Y.; Koons, G.L.; Mikos, A.G.; Qiu, Z.; Song, T.; Cui, F.; Wang, X. Tuning pore features of mineralized collagen/PCL scaffolds for cranial bone regeneration in a rat model. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 106, 110186.
- Crane, G.M.; Ishaug, S.L.; Mikos, A.G. Bone tissue engineering. Nat. Med. 1995, 1, 1322–1324.
- Santin, M. 14—Bone tissue engineering. In Bone Repair Biomaterials; Planell, J.A., Best, S.M., Lacroix, D., Merolli, A., Eds.; Woodhead Publishing Series in Biomaterials: Brighton, UK, 2009; Volume 14, pp. 378–422.
- Brown, J.L.; Kumbar, S.G.; Laurencin, C.T. Chapter II.6.7—Bone tissue engineering. In Biomaterials Science, 3rd ed.; Ratner, B.D., Hoffman, A.S., Schoen, F.J., Lemons, J.E., Eds.; Academic Press: Cambridge, MA, USA, 2013; pp. 1194–1214.
- Datta, S.; Rameshbabu, A.P.; Bankoti, K.; Roy, M.; Gupta, C.; Jana, S.; Das, A.K.; Sen, R.; Dhara, S. Decellularized bone matrix/oleoyl chitosan derived supramolecular injectable hydrogel promotes efficient bone integration. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 119, 111604.
- Bouchie, A. Tissue engineering firms go under. Nat. Biotechnol. 2002, 20, 1178–1179.
- Yang, H.; Wang, S.; Bian, H.; Xing, X.; Yu, J.; Wu, X.; Zhang, L.; Liang, X.; Lu, A.; Huang, C. Extracellular matrix-mimicking nanofibrous chitosan microspheres as cell micro-ark for tissue engineering. Carbohydr. Polym. 2022, 292, 119693.
- Raftery, R.M.; Woods, B.; Marques, A.L.P.; Moreira-Silva, J.; Silva, T.H.; Cryan, S.-A.; Reis, R.L.; O’Brien, F.J. Multifunctional biomaterials from the sea: Assessing the effects of chitosan incorporation into collagen scaffolds on mechanical and biological functionality. Acta Biomater. 2016, 43, 160–169.
