The alterations of platelet function in metabolic disorders contribute to increase the tendency to thrombus formation playing a major role in the increased risk of atherothrombotic events in patients with cardiometabolic risk factors. Obesity, classically a condition of insulin resistance, dyslipidemia, impaired glucose homeostasis and hypertension are conditions associated with a number of platelet abnormalities that lead to "angrier" platelets, resulting in increased platelet activation and aggregability. Here we summarize the role of these clinical settings on promoting the biochemical imbalance towards platelet hyperactivation, and provide some platelet indices and biomarkers that may be useful to identify a prothrombotic phenotype.
- platelets
- thrombosis
- metabolic syndrome
- insulin resistance
- nitric oxide
- obesity
1. Introduction
Several lines of evidence suggest a strong correlation between metabolic disorders and hemodynamic such as obesity, dyslipidemia, diabetes, hypertension, and cardiovascular (CV) diseases (CVD), with endothelial dysfunction as the initial step toward atherothrombosis (Figure 1). Oxidative stress and a chronic low-grade of inflammation may be considered a “common soil” able to create a feed-forward cycle that can deeply influence the development of a prothrombotic tendency of these metabolic abnormalities.
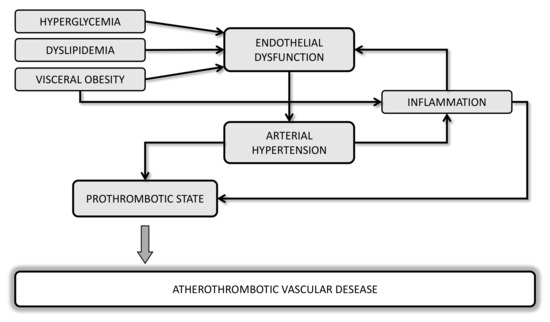
Figure 1. Potential mechanisms linking cardiometabolic disorders and atherothrombotic vascular diseases.
2. Influence of Cardiometabolic Risk Factors on Platelet Function
One of the first epidemiological studies showing the causal relationship between obesity and CVD was the Framingham Heart Study [
[3]
[6]
[11]
[12]
This entry provides an overview of changes in platelet function occurring in metabolic and hemodynamic disorders mainly characterizing the MS, all with an impact on the risk of CV morbidity and mortality owing to atherothrombotic events. Many of impairments in platelets converge on oxidative stress with release of oxidation products, which have a causal link to platelet hyperaggregability and hyperactivation. The excess of adipose tissue of the trunk and/or abdomen has a strong impact on vascular complications, through the production of PMPs and mediators with paracrine and endocrine actions, which influence platelet response. Platelet indices and biomarkers of platelet activation may have useful clinical value through the whole journey of cardiometabolic diseases for prediction and risk assessment of thrombotic risk. Different methodological approaches for platelet (dys) function investigations are now available and each-one based on different operating principles. However, few assays are able to assess “all in one device” platelet aggregation and activation pathways and standardization and quality controls are still limited despite several efforts.
This review provides an overview of changes in platelet function occurring in metabolic and hemodynamic disorders mainly characterizing the MS, all with an impact on the risk of CV morbidity and mortality owing to atherothrombotic events. Many of impairments in platelets converge on oxidative stress with release of oxidation products, which have a causal link to platelet hyperaggregability and hyperactivation. The excess of adipose tissue of the trunk and/or abdomen has a strong impact on vascular complications, through the production of PMPs and mediators with paracrine and endocrine actions, which influence platelet response. Platelet indices and biomarkers of platelet activation may have useful clinical value through the whole journey of cardiometabolic diseases for prediction and risk assessment of thrombotic risk. Different methodological approaches for platelet (dys) function investigations are now available and each-one based on different operating principles. However, few assays are able to assess “all in one device” platelet aggregation and activation pathways and standardization and quality controls are still limited despite several efforts. Insulin resistance, a condition frequently associated with obesity, with or without hyperglycemia, dyslipidemia, and hypertension alters a number of distinct aspects of hemostasis responsible for platelets more prone to aggregate to agonists and less responsive to platelet inhibitors. However, weight reduction is a powerful measure to restore a physiological platelet function in obese subjects.References
- Shanti Mendis; The Contribution of the Framingham Heart Study to the Prevention of Cardiovascular Disease: A Global Perspective. Progress in Cardiovascular Diseases 2010, 53, 10-14, 10.1016/j.pcad.2010.01.001.
- Johan Ärnlöv; Erik Ingelsson; Johan Sundström; Lars Lind; Impact of Body Mass Index and the Metabolic Syndrome on the Risk of Cardiovascular Disease and Death in Middle-Aged Men. Circulation 2010, 121, 230-236, 10.1161/circulationaha.109.887521.
- Mente, A.; Yusuf, S.; Islam, S.; McQueen, M.J.; Tanomsup, S.; Onen, C.L.; Rangarajan, S.; Gerstein, H.C.; Anand, S.S.; INTERHEART Investigators Metabolic syndrome and risk of acute myocardial infarction a case-control study of 26,903 subjects from 52 countries. J. Am. Coll. Cardiol. 2010, , 55, , 2390–2398..
- Salvatore Novo; A. Peritore; F.P. Guarneri; Egle Corrado; F. Macaione; S. Evola; Giuseppina Novo; Metabolic syndrome (MetS) predicts cardio and cerebrovascular events in a twenty years follow-up. A prospective study. Atherosclerosis 2012, 223, 468-472, 10.1016/j.atherosclerosis.2012.05.018.
- KaziM Sheikh; Metabolic syndrome and stroke.. Stroke 2008, 39, , 10.1161/STROKEAHA.108.523837.
- Gijs H. Goossens; The Metabolic Phenotype in Obesity: Fat Mass, Body Fat Distribution, and Adipose Tissue Function. Obesity Facts 2017, 10, 207-215, 10.1159/000471488.
- Tarantino Giovanni; Carmine Finelli; Annamaria Colao; Domenico Capone; Marianna Tarantino; Ernesto Grimaldi; Donato Chianese; Saverio Gioia; Fabrizio Pasanisi; Franco Contaldo; Francesco Scopacasa; Silvia Savastano; Are hepatic steatosis and carotid intima media thickness associated in obese patients with normal or slightly elevated gamma-glutamyl-transferase?. Journal of Translational Medicine 2012, 10, 50-50, 10.1186/1479-5876-10-50.
- McGill, H.C.; McMahan, C.A.; Herderick, E.E.; Zieske, A.W.; Malcom, G.T.; Tracy, R.E.; Strong, J.P. Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Research Group Obesity accelerates the progression of coronary atherosclerosis in young men. Circulation 2002, 105, 2712–2718.
- Luc F. Van Gaal; Ilse L. Mertens; Christophe E. De Block; Mechanisms linking obesity with cardiovascular disease. Nature 2006, 444, 875-880, 10.1038/nature05487.
- Olgun Göktaş; Canan Ersoy; Ilker Ercan; Fatma Ezgi Can; General and abdominal obesity prevelances and their relations with metabolic syndrome components.. Pakistan Journal of Medical Sciences 2019, 35, 945-950, 10.12669/pjms.35.4.235.
- K.G.M.M. Alberti; Paul Z Zimmet; Who Consultation; Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus. Provisional report of a WHO Consultation. Diabetic Medicine 1998, 15, 539-553, 10.1002/(sici)1096-9136(199807)15:7<539::aid-dia668>3.0.co;2-s.
- Isabella Russo; The Prothrombotic Tendency in Metabolic Syndrome: Focus on the Potential Mechanisms Involved in Impaired Haemostasis and Fibrinolytic Balance. Scientifica 2012, 2012, 525374, 10.6064/2012/525374.
