One of the earliest hallmarks of plant immune response is production of reactive oxygen species (ROS) in different subcellular compartments, which regulate plant immunity. A suitable equilibrium, which is crucial to prevent ROS overaccumulation leading to oxidative stress, is maintained by salicylic acid (SA), a chief regulator of ROS. However, ROS not only act downstream of SA signaling, but are also proposed to be a central component of a self-amplifying loop that regulates SA signaling as well as the interaction balance between different phytohormones. The exact role of this crosstalk, the position where SA interferes with ROS signaling and ROS interferes with SA signaling and the outcome of this regulation, depend on the origin of ROS but also on the pathosystem. The precise spatiotemporal regulation of organelle-specific ROS and SA levels determine the effectiveness of pathogen arrest and is therefore crucial for a successful immune response. However, the regulatory interplay behind still remains poorly understood, as up until now, the role of organelle-specific ROS and SA in hypersensitive response (HR)-conferred resistance has mostly been studied by altering the level of a single component. In order to address these aspects, a sophisticated combination of research methods for monitoring the spatiotemporal dynamics of key players and transcriptional activity in plants is needed and will most probably consist of biosensors and precision transcriptomics.
- plant immune response
- reactive oxygen species
- salicylic acid
- reactive oxygen species–salicylic acid crosstalk
- programmed cell death
- hypersensitive-response-conferred resistance
- RBOH NADPH oxidases
- chloroplastic redox state
- biosensors
- precision transcrip
1. Reactive Oxygen Species as One of the Earliest Hallmarks of Plant Immune Response
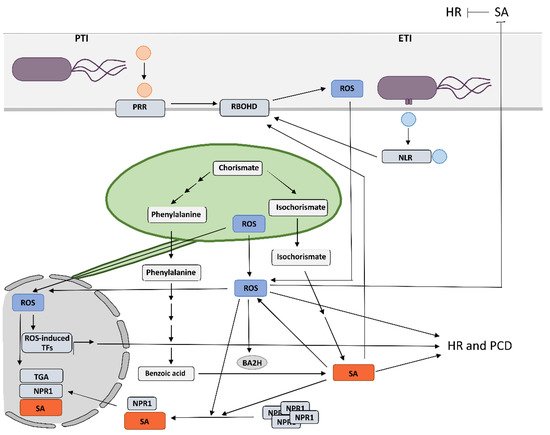
2. Crosstalk between RBOHD-Derived Reactive Oxygen Species and Salicylic Acid in Programmed Cell Death and Resistance
3. Chloroplastic Reactive Oxygen Species Play a Role in the Signaling for Programmed Cell Death and Induce SA-Dependent Transcription of Immune Genes
A growing body of evidence supports a central role of chloroplasts as integrators of environmental signals and key defense organelles, as they host biosynthesis of several key defense-related molecules, including SA and ROS, and are therefore primary sites for the biosynthesis and transmission of pro-defense signals during plant immune responses [7][47][48][12,52,53]. Increases in chloroplastic ROS concentration have been observed in different incompatible plant–pathogen interactions [49][54], and, in addition, the results of several studies suggest the involvement of chloroplastic ROS in the signaling for and/or execution of HR cell death in HR-conferred resistance [11][50][51][52][53][54][55][16,55,56,57,58,59,60]. However, its exact role during HR still remains largely elusive. Zurbriggen et al. (2009) suggested that ROS generated in chloroplasts during non-host interaction are essential for the progression of PCD, but do not contribute to the induction of pathogenesis-related genes or other signaling components of the response, including SA signaling [51][56]. Similarly, Yao and Greenberg did not detect an increase in the expression of genes from SA-mediated signaling in the acd2 mutants that show spontaneous light-dependent PCD and chloroplastic H2O2 increase [56][61]. In contrast, Straus et al. suggested that chloroplastic ROS acts as a flexible spatiotemporal integration point leading to opposite SA signaling reactions in infected and surrounding tissue to control the propagation of PCD [52][57]. Predominance of chloroplast superoxide over H2O2 drives PCD in infected tissue and RBOHD-regulated restriction of PCD in the surrounding tissue. When the equilibrium is through SA synthesis shifted towards H2O2 production, this results in runaway PCD [52][57]. Interestingly, Ochsenbein et al. (2006) also observed that chloroplastic singlet oxygen activates SA-mediated signaling, although SA was not required for a singlet-oxygen-mediated cell death [57][62]. In potato HR, ROS generated in the chloroplasts around the cell death zone are involved in SA-independent execution of cell death and SA-dependent immune signaling, which are spatially regulated [11][58][16,63]. Moreover, recent evidence suggests that chloroplastic ROS might, in addition to signaling in HR cell death, also be involved in controlling plant immune responses by reprogramming transcription of genes involved in response to pathogen attack as one of the retrograde signals [57][59][60][61][62][63][64][62,64,65,66,67,68,69]. Transmission of pro-defense signals is facilitated by direct connections between chloroplasts and other organelles [65][66][70,71]. It has been suggested that stromules, which are extensions in the form of fluid-filled tubules, containing soluble components of the compartments, could facilitate this transmission by enabling more targeted and stronger signal transmission [67][72]. Stromules are induced by a variety of biotic and abiotic stresses and are involved in retrograde signaling after pathogen invasion, light stress, or movement of chloroplasts within the cell [68][69][70][73,74,75]. However, their direct role in immunity is still largely unresolved [63][68]. In potato HR-conferred resistance against PVY, stromule formation is induced in close proximity to the cells with oxidized chloroplasts [58][63]. Since Stonebloom et al. (2012) showed that cell-to-cell transport is negatively regulated by an oxidative shift in chloroplasts, while reductive shift in chloroplasts causes increased cell-to-cell transport, these results could indicate on the potential role of stromules in the signaling for HR-conferred resistance [71][76]. This is further supported by the fact that stromule formation is induced on the front of virus multiplication zone and is tightly spatiotemporaly regulated by SA signaling [58][63]. Another type of phenomena that indicate the role of stromules in signaling are the connections of stromules with the plasma membrane, mitochondria, and the nucleus, suggesting that the direct transfer of proteins and metabolites between these organelles and the apoplast could occur [72][77].4. Reactive Oxygen Species of Different Source and Type Induce Diverse Transcriptional Responses
Distinct subcellular compartments produce different ROS types [73][78], which could all regulate gene expression [74][79]. Although each organelle could, in theory, locally manage its own ROS homeostasis, ROS and related signaling intermediates are also involved in interorganellar communication [6][72][75][9,77,80]. For example, chloroplast-derived redox signals could be first integrated in the cytosol or directly transferred to the nucleus, either through physical nucleus–chloroplast interaction or via stromules, in order to control retrograde signaling [76][81]. Only a small number of proteins targeted to the chloroplast have been also identified in the nucleus to function as retrograde signal transducers in response to biotic and abiotic stresses, some of them being WHIRLY1, the PHD-type transcription factor PTM, and NUCLEAR RECEPTOR INTERACTING PROTEIN 1 (NRIP1) [77][82]. For example, WHIRLY1 has been proposed to convey the redox status in chloroplasts to the nucleus in a SA-dependent manner [78][83]. While localization of WHIRLY1 and PTM in both chloroplasts and the nucleus have been shown, the way in which their translocation from chloroplasts to the nucleus occurs is still not known [79][80][84,85]. On the other hand, Caplan et al. suggested that after TMV inoculation, the NRIP1 protein is translocated from the chloroplast to the nucleus via stromules [69][74]. In the nucleus, the control of gene expression depends mainly on the activity of transcription factors (TFs) that interact with oxidative-stress-responsive cis-regulatory elements within the gene promoters. It has been reported that the transcripts generated by increased intracellular H2O2 levels encode proteins of diverse functional categories including TFs, protein kinases, heat shock proteins, glutathione S-transferases (GSTs), UDP-glucuronosyltransferases (UGTs), and cytochrome P450 monooxygenases (CYPs). Upregulated TFs belong to different stress-related TFs’ families, including WRKY, AP2/ERF, MYB, NAC, heat shock factor (HSF), and ZAT [73][78]. Interestingly, studies have shown that the transcriptional response to apoplastic ROS produced during oxidative burst has little similarity to the effect of chloroplastic ROS [6][9]. The roles of individual ROS species from different organelles in transcriptional responses have been studied [81][86], but since the signaling pathways are connected, multiple effects of different organelle-specific ROS on transcriptional response have to be addressed.5. Tools for Studying Redox State with High Spatiotemporal Resolution with Focus on Cytoplasmic and Chloroplastic Redox State in Hypersensitive Response
Despite the new insights that have been brought into the role of redox mechanisms in plant defense response, still one of the major challenges is to monitor local, subcellular, and global ROS dynamics with high selectivity, sensitivity, and spatiotemporal resolution that allow for quantification [82][87]. Small organic-molecule-based probes have been originally used to measure ROS in plants; however, they have significant limitation as they do not allow for spatiotemporal resolution, since the fluorescence changes as a result of ROS presence are irreversible [83][84][88,89]. Nondestructive real-time measuring of redox state in plants with high spatial and temporal resolution is feasible since Jiang et al. reported the use of redox-sensitive green fluorescent protein (roGFP) [85][90] (Figure 2). Measurement of roGFP fluorescence intensity following excitation with two different wavelengths enables the calculation of the ratio between reduced and oxidized roGFP and thus the determination of the redox state on the cellular or organelle level. The biosensors have been modified by the addition of signal sequences to target them to different subcellular organelles, while two variants, roGFP1 and roGFP2, with different excitation and emission spectra, were developed to allow for optimal selection according to the redox state in the particular organelle. By fusing the peroxisomal targeting peptide sequence SKL, per-roGFP1 and px-roGFP2 were targeted to peroxisomes [86][87][91,92]. mt-roGFP1 and mt-roGFP2 are available for measuring the redox state in mitochondria due to the fusion with mitochondrial localization signal peptide from the tobacco b-ATPase [85][87][90,92], while er-roGFP2 was constructed by fusing roGFP2 with the endoplasmic reticulum (ER) retention signal peptide HDEL for following the redox state in ER [88][93]. Finally, cp-GFP2, pt-roGFP2, and chl-roGFP2 were constructed by adding plastid-targeting signal peptide TKTP, coding sequence for RuBisCo small subunit transit peptide, or the first 74 amino acids from PRXa to roGFP2 coding sequence, respectively, for measuring change of redox state in chloroplasts [71][89][90][76,94,95]. Since pt-roGFP is targeted to the chloroplast outer plastid envelope membrane, it also allows for the imaging of stromules formation [68][73]. The above-mentioned sensors were used in tobacco, Arabidopsis, and potato subjected to different developmental and environmental stresses [68][71][85][86][87][88][89][90][91][92][93][94][73,76,90,91,92,93,94,95,96,97,98,99], some of them also in HR [58][95][96][63,100,101]. roGFP2 expressed in the cytosol senses the redox potential of the cellular glutathione buffer via glutaredoxin (GRX) as a mediator of reversible electron flow between glutathione and roGFP2 [88][93]. To facilitate specific real-time equilibration between roGFP2 and the glutathione redox couple, fusion constructs with human glutaredoxin 1 (GRX1) Grx1-roGFP2 and roGFP2-Grx2 were generated [97][98][99][102,103,104]. Moreover, two roGFP derivatives, roGFP2-iL and roGFP1-iX, with different midpoint potential and excitation properties, were developed to further extend the range of suitable probes [100][105]. Another group of genetically encoded sensors that detect H2O2 levels instead of measuring glutathione redox potential was also developed. roGFP2-Orp1, based on a redox relay between the GPX-like enzyme oxidant receptor peroxidase-1 (Orp1) from Saccharomyces cerevisiae and roGFP2 was generated for sensing transient changes in H2O2 [93][98]. Another sensor that reports on local alterations in H2O2 concentrations exploits the H2O2-sensitive bacterial transcription factor OxyR for its response and was named HyPer [101][102][106,107]. The above-mentioned types of sensors were also used to follow redox potential and H2O2 in the cytoplasm during HR [103][104][105][106][107][108][108,109,110,111,112,113] or were further upgraded to follow redox potential in different cellular compartments, including chloroplasts [109][114]. Using HyPer2, Exposito-Rodriguez et al. showed that in photosynthetic Nicotiana benthamiana epidermal cells, exposure to high light increased H2O2 production in chloroplast stroma, cytosol, and nuclei, suggesting direct H2O2 transfer from chloroplasts to nuclei [110][115]. Other genetically encoded redox and H2O2 sensors are summarizeviewed in [82][111][87,116]. The most recent ones are biosensor CROST for measurements of the thioredoxin redox state in chloroplasts [112][117], FRET-based biosensors, and ROS regulated promoter–FP fusions [82][113][87,118].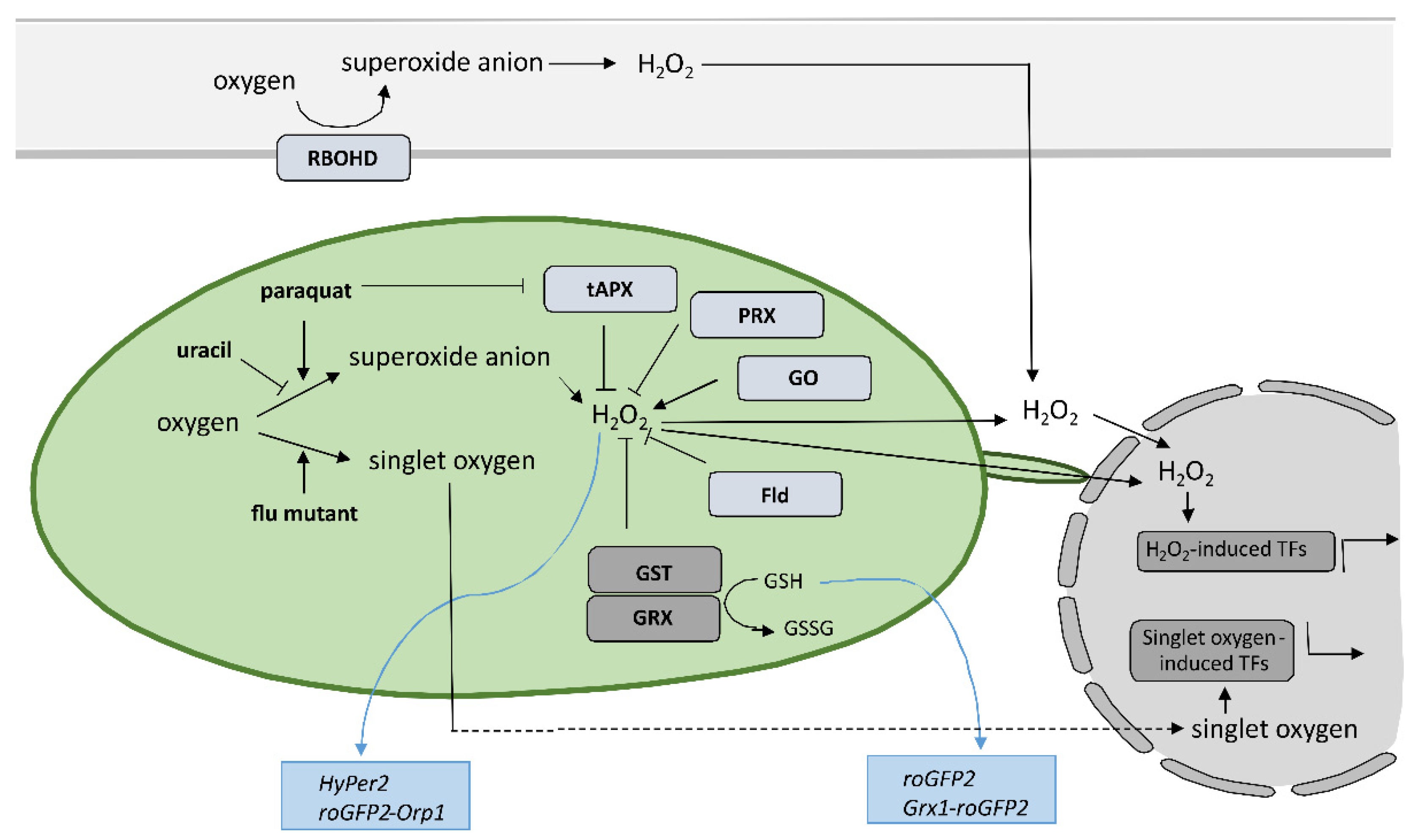
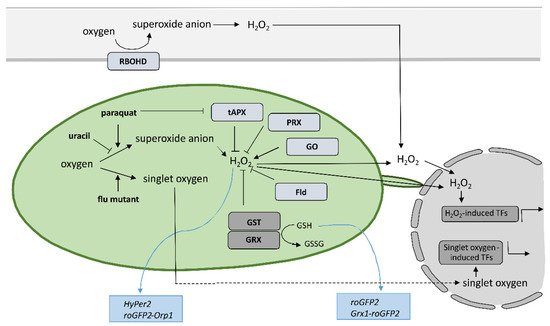
6. Conclusions
The results of the above-mentioned studies suggest that the precise spatiotemporal regulation of key players, including organelle-specific ROS and SA levels, determines the effectiveness of pathogen arrest and is therefore crucial for a successful immune response. The change of SA and ROS levels and other key players alter the rate of cell-to-cell and systemic pathogen spread, rate of cell death induction, and spatial transcriptional response, leading to susceptibility or resistance. ScholarsWe suggest that only a coordinated and intertwined action of all main components enable effective immune response. However, the specific interactions between them and the regulatory interplay behind still remain poorly understood, as up until now, the role of organelle-specific ROS and SA in HR-conferred resistance has only been studied by altering the level of a single component. In order to address these aspects, a sophisticated combination of research methods for monitoring the spatiotemporal dynamics of key players and transcriptional activity in plants is needed. The precise sampling of tissue sections surrounding the HR-PCD, with spatial resolution and suitable for transcriptomics analyses [11][16], in combination with the use of biosensors [130][135], could enable identification of novel key players and could unravel the interconnectivity of immune signaling components. Such an approach could therefore present a step forward in studying the resistance response.
