CD24 aThis entry is adapted from 10.3390/biomedicines10051175.
Ind its ligand Siglec-10 were described as an innatethe past decade, immune checkpoint in carcinoma. Here, we investigated this axis in B-cell lymphoma by assessing CD24 expression and evaluating pro-phagocytic effects of CD24 antibody treatment in comparison to hallmark immune checkpoint CD47. In mantle cell lymphoma (MCL) and follicular lymphoma patients, high mRNA expression of CD24 correlated with poor overall survival, whereas CD47 expression did not. Conversely, CD24 expression did not correlate with survival in diffuse large B-cell lymphoma (DLBCL), whereas CD47 did. CD24 was also highly expressed on MCL cell lines, where treatment with CD24 antibody clones SN3 or ML5 potently induced phagocytosis, with SN3 yielding >90% removal of MCL cells and triggering phagocytosis of primary patient-derived MCL cells by autologous macrophages. Treatment with CD24 mAb was superior to CD47 mAb in MCL and was comparable in magnitude to the effect observed in carcinoma lines. Reversely, CD24 mAb treatment was less effective than CD47 mAb treatment in DLBCL. Finally, phagocytic activity of clone SN3 appeared at least partly independent of antibody-dependent cellular phagocytosis (ADCP), suggesting CD24/Siglec-10 checkpoint activity, whereas clone ML5 solely induced ADCP. In conclusion,hibitors (ICIs) that re-activate adaptive immunity have transformed the treatment paradigm in various cancer types. More recently, ICIs on innate immune cells have also gained prominence as therapeutic targets, being CD47 the hallmark ICI in the clinic. Lately, CD24 was also described as an innate immune checkpoint with apparent significance in several solid cancer types. In this entry, we discuss the role of CD24 is an immunoas a therapeutic target of potential clinical relevance for MCL, but not DLBCL, with a particular focus on mantle cell lymphoma (MCL) and diffuse-large B cell lymphoma (DLBCL).
- mantle cell lymphoma
- CD24
- immunotherapy
- immune checkpoint
- phagocytosis
Introduction
Results
CD24 Is Expressed in Several B-Cell Lymphomas, Being Most Highly Expressed in MCL, Where It Correlates with Poor Prognosis in Contrast to Hallmark Immune Checkpoint CD47
CD24 is a well-established marker overexpressed in various carcinomas, where it is associated with poor survival. Furthermore, CD24 is a B lineage marker that is also expressed at the mRNA level in several types of NHL, such as MCL, FL, BL, and DLBCL, albeit with a large range of expression (F1. Introduction
CD24 is a small, heavily glycosylated protein attached to the cell membrane by a glycosyl-phosphatidylinositol (GPI) anchor. It has been extensively studied in the context of cancer biology, with it being defined as a cancer stem cell marker in various malignancies, such as breast[1], pancreas[2], and ovarian carcinoma[3]. CD24-based cancer immunotherapy was originally reported decades ago for a subgroup of patients with B-lymphoproliferative disorders [4]. More recently, in a hallmark 2019 report, CD24 was also described as an innate immune checkpoint with apparent significance in several solid cancer types[5]. Specifically, CD24 checkpoint activity relayed anti-phagocytic signals to phagocytes through its interaction with Siglec-10, a lectin expressed on tumor-associated macrophages (TAMs). Accordingly, CD24 blockade using a monoclonal antibody (mAb) induced macrophage-mediated phagocytosis of breast, ovarian, and pancreas cell lines in vitro and inhibited tumor growth of xenografted breast cancer cell line in an NSG mouse model [5].
On other hand, CD24 is also reportedly expressed in various hematological malignancies, including non-Hodgkin B-cell lymphomas (NHLs)[6]. Thus, CD24 may be also an immune checkpoint relevant for B cell-NHL. Of note, certain types of NHLs such as mantle cell lymphoma (MCL) or follicular lymphoma (FL), retained CD24 expression in contrast to healthy counterparts [7], with MCL being a more aggressive lymphoma that comprises about 3% to 10% of total NHL cases [8], and not curable with conventional chemoimmunotherapy [9].
2. CD24 as a Potential Immunotherapeutic Target for Mantle Cell Lymphoma
The potential of CD24 mAb treatment and checkpoint targeting was previously preclinically investigated for pancreas, ovarian, and breast carcinoma[5]. In this study (10.3390/biomedicines10051175.), we focused on NHLs where CD24 mRNA expression was similarly high in follicular lymphoma (FL), burkitt lymphoma (BL), MCL, and DLBCL in line with previous studies[6][10]. At protein level, MCL showed the highest CD24 expression, the reason why we decided to focus our work in MCL. Specifically, antibody-mediated targeting of CD24 robustly enhanced the phagocytic uptake of MCL cells yielding over 90% removal of CD24 expressing MCL cancer cells. Phagocytic uptake upon CD24 mAb treatment was significantly greater than upon treatment with the CD47 antibody InhibRx, both in cell lines and primary patient-derived blasts in an autologous setting. In line with this, high expression of CD24, but not CD47, correlated with poor OS in MCL and FL, whereas the opposite was found for DLBCL. The robust increase in phagocytosis upon CD24 mAb treatment was not limited to MCL and was also detected in a panel of carcinoma cell lines expressing CD24. Reversely, only low levels of phagocytosis were observed upon treatment with CD24 mAb in DLBCL, with the CD47 antibody InhibRx having superior effects. Moreover, MCL blasts expressed more CD24 than B cells from healthy donors. Apart from B cells, CD24 is also highly expressed in almost all human tissues (ATLAS database) and other hematological cells [11]. Nevertheless, phagocytosis of healthy PBMCs by autologous macrophages did not increase upon treatment with CD24 mAbs in our experiments. Still, off-target effects of CD24 mAb treatment must be carefully considered in the design of CD24-based immunotherapy. These results highlight the potential of targeting CD24 in MCL, which remains incurable with conventional chemoimmunotherapy [12].
After showing the potential of targeting CD24 for the treatment of MCL, the mechanism behind these mAb-mediated phagocytosis was further investigated. Two different murine antibody clones (SN3 IgG1 and ML5 IgG2a) potently induced phagocytosis of MCL cells, so CD24/Siglec-10 checkpoint activity could exist in MCL similar to previously showed in carcinoma. In the original report on SN3 in carcinoma, its potential checkpoint activity was not evaluated using an F(ab’)2 preparation, thus raising the question of whether the observed activity of this antibody is due to checkpoint inhibitor activity or Fc-mediated phagocytosis. The phagocytic activity of both clones was abrogated when an F(ab’)2 preparation was used, suggesting that these antibodies triggered antibody-dependent cellular phagocytosis (ADCP) as the main mechanism of action. However, treatment with the complete SN3 mAb in the presence of high concentrations of Fc blocker solution still yielded high levels of phagocytic uptake. Similarly, alternative blocking of the Fc domain of clone SN3, with a goat anti-mouse F(ab’)2, also did not negatively impact phagocytic activity. With both approaches, the pro-phagocytic activity of clone ML5 was abrogated. Thus, in these assays, SN3 appeared to have Fc-independent activity. Thus, both proofs to support and disprove checkpoint inhibitor activity of antibody SN3 and/or Siglec-10-CD24 interaction were uncovered, highlighting the need for further investigation into the underlying mechanism (e.g., by constructing a human IgG4 or IgG2 variant).
Higure 1
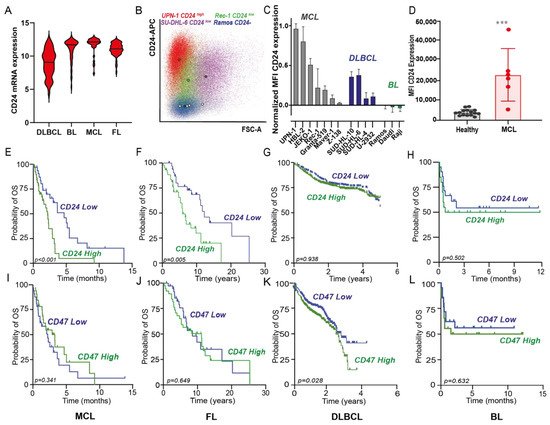
Figure 1. CD24 expression in several non-Hodgkin’s lymphomas (NHL) and correlation with survival. (A) CD24 mRNA expression (2log) in 4 subtypes of NHL, namely diffuse large B-cell lymphoma (DLBCL; n = 94), Burkitt lymphoma (BL; n = 58), mantle cell lymphoma (MCL; n = 42), and follicular lymphoma (FL; n = 64). (B) Dot-plot diagram of forward scatter vs. CD24 expression of representative cell line panel, illustrating high CD24 expression (UPN-1, red), low CD24 expression (Rec-1, green; SU-DHL-6, violet), or lack of CD24 expression (Ramos, blue) compared to isotype controls (gray). The colored circles indicate the mean value of the population. (C) Surface CD24 expression (mean ± SD of corrected MFI values, n = 3) in MCL, DLBCL and BL cell lines. Expression was normalized to the highest value, corresponding CD24 expression of UPN-1. (D) Surface CD24 expression (MFI values) of MCL blast cells (CD45+CD19+CD5+) (n = 6) and healthy donors (n = 13) (CD45+CD19+) cells. Mean ± SD, Student’s t-test, *** = p < 0.001 (E–H) Kaplan–Meier plots for CD24 and (I–L) CD47 expression in an MCL patient data set (n= 71) (E,I), an FL patient data set (n = 77) (F,J), a DLBCL patient data set (n = 1017) (G,K), and BL patient data set (n = 41) (H,L); log-rank test performed in all cases, with p < 0.05 considered significant.
CD24 Is a Target for Reactivation of Phagocytosis in MCL, with a Superior Effect Than CD47 Antibody Treatment
To delineate the potential role of CD24 as a “don’t eat me” signal and therapeutic target for B-NHL, mixed macrophage/cancer cell cultures were subjected to CD24 antibody treatment. Hereto, firstly MCL was selected based on the high CD24 expression and association with survival. Secondly, M2c-differentiated macrophages were selected as these expressed higher levels of the CD24 ligand Siglec-10 than M1 macrophages (Figure 2A). Using confocal fluorescence microscopy, single fluorescently labeled and CD24-positive UPN-1 cells were clearly detected inside M2c macrophages after 3 h of co-culture in control conditions (Figure 2B, left panel). Upon treatment with CD24-mIgG1 antibody (clone SN3), prominent phagocytosis of cancer cells by M2c macrophages was detected with engulfment of several cancer cells per single macrophage in some donors (Figure 2B, middle panel). Similar treatment with CD47 blocking antibody InhibRx, as the ‘benchmark’ innate immune checkpoint, did trigger a clear increase in phagocytosis compared to untreated conditions, but the number of macrophages with engulfed cancer cells and the number of cancer cells per macrophage were lower than upon CD24 antibody treatment (Figure 2B, right panel). Subsequent quantification of phagocytosis using flow cytometry confirmed these findings, with ~30% phagocytosis upon control treatment that was increased to ~95% phagocytosis upon CD24 antibody treatment (Figure 2C). In contrast, treatment with CD47 antibody only increased phagocytosis to ~45% (Figure 2C). The level of phagocytosis induction upon CD24 antibody treatment was already maximal at the dose of 1 µg/mL (Figure 2D, red diamonds). Treatment with CD47 antibody increased phagocytosis only by 15–20% compared to medium control and also reached its maximal level at 1 µg/mL (Figure 2D, blue diamonds). In line with expectations, treatment with CD24 mAb SN3 induced lower levels of phagocytosis of UPN1 cells by M1 macrophages, although InhibRx similarly induced a lower level of phagocytosis in M1 macrophages (Figure 2E).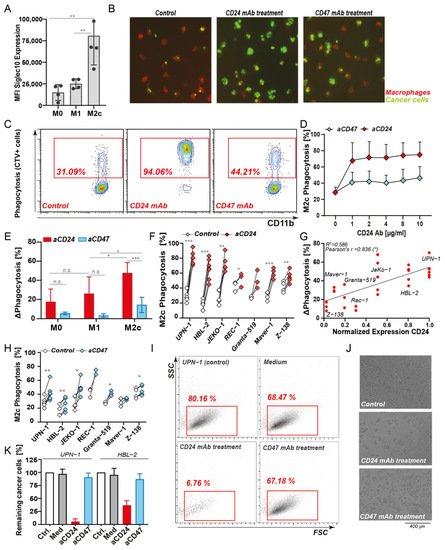
Figure 2. Phagocytic uptake and clearance of MCL cell lines upon CD24 and CD47 antibody treatment. (A) Levels of Siglec10 expression in M0, M1, and M2c type macrophages. (B) Microscopy images of macrophage-mediated phagocytosis of UPN-1 cells in M2c mixed culture (control, left), CD24 antibody SN3 (middle) or CD47 mAb InhibRx treatment (right), with macrophages in red (CD11b) and cancer cells in green (CSFE). (C) Flow cytometry analysis of UPN-1 phagocytosis with cell trace violet (CTV)-stained cancer cells and macrophages stained with CD11b-APC. Level of phagocytosis was determined as the percentage of CD11b+CTV+ cells. (D) Dose range of CD24 antibody SN3 and CD47 antibody InhibRx treatment in UPN-1 cells. (E) Phagocytosis levels of UPN-1 cells by M0, M1, and M2c-polarized macrophages upon SN3 and InhibRx treatment. (F) Percentage of phagocytosis upon CD24 SN3 antibody treatment vs. medium control of several MCL cell lines by M2c macrophages (of at least 3 independent donors). (G) Correlation between CD24 expression and increase in phagocytosis upon CD24 SN3 treatment in MCL cell lines. CD24 expression was normalized to the highest value, corresponding to UPN-1 CD24 expression. Pearson’s r = 0.836 (*) and linear regression p < 0.05. (H) Percentage of phagocytosis upon CD47 InhibRx treatment vs. medium control by M2c macrophages (of at least 3 independent donors) in several MCL cell lines. (I) Flow cytometry diagram showing the percentage of remaining UPN1 cell line after 2 h phagocytosis. (J) Bright-field microscopy pictures showing remaining UPN-1 cells after o/n co-culture with macrophages. (K) Percentage of remaining UPN-1 and HBL-2 cells in different experimental conditions (cancer cells only (Ctrl), mixed culture with medium control (Med), CD24 antibody SN3, and CD47 antibody InhibRx. All averaged values represent the mean ± SD of at least 3 independent experiments. Unless specified otherwise, statistics were performed using a two-sided paired Student’s t-test. ns: not significant; * p < 0.05; ** p < 0.01; *** p < 0.001.
CD24 mAb Treatment Increased Phagocytosis of Primary MCL Blasts by Autologous Macrophages and PMNs, but Did Not Induce High Level of Phagocytosis of Healthy Cells
Treatment of primary MCL blasts with CD24 mAb SN3 increased phagocytosis by autologous M2c macrophages by an average of 24%, whereas treatment with CD47 mAb InhibRx almost did not induce (3%) phagocytosis (Figure 3A). Further, the combination of CD24 mAb SN3 with RTX augmented RTX-mediated phagocytosis, whereas the combination with CD47 mAb InhibRx did not significantly potentiate activity (Figure 3A). When the same experiments were performed with PBMCs from healthy donors and autologous M2c macrophages, only an average of 6% increase in phagocytosis upon CD24 mAb treatment was observed, similar to CD47 mAb InhibRx (Figure 3B). Although not significant, CD24 antibody treatment clearly induced a higher phagocytic response in MCL compared to healthy samples (Figure 3C). In contrast, no significant difference was obtained for CD47 InhibRx treatment between MCL and healthy samples (Figure 3C). Combinatorial treatment with CD24 mAb SN3 and RTX did induce up to 5% phagocytosis of healthy PBMCs, but this effect was slightly lower than the combination of RTX and CD47 treatment (Figure 3B). In contrast, when primary MCL blasts were mixed with healthy donor-derived M2c macrophages, a similar potentiating effect to that obtained with primary autologous macrophages was detected (Figure 3D). Of note, this anti-cancer pro-phagocytic effect was also observed when primary patient-derived blasts were mixed with autologous PMNs, where SN3 treatment-induced up to 30% trogocytosis of primary PBMCs containing MCL blasts (Figure 3E). In contrast, InhibRx treatment did not significantly increase trogocytosis of MCL blasts (Figure 3E). Further, both CD24 and CD47 antibody treatment did not induce trogocytosis of healthy autologous PBMCs (Figure 3F). Thus, treatment of MCL with CD24 antibody-induced phagocytosis of CD24+ MCL cells alone and in combination with RTX, whereas this treatment minimally induced phagocytosis of healthy cells.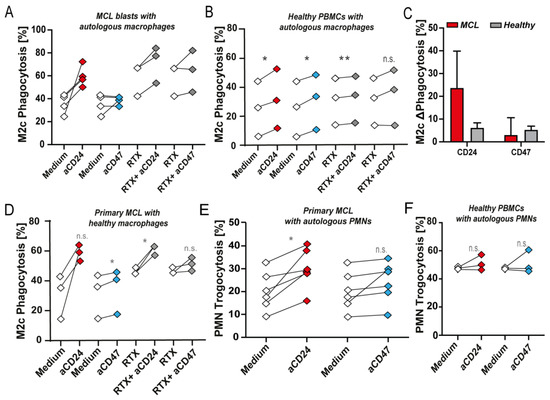
Figure 3. CD24 and CD47 antibody-mediated phagocytosis of MCL and healthy blasts by primary patient-derived autologous and allogeneic healthy donor macrophages. (A) Phagocytosis of MCL blasts by autologous macrophages upon CD24 mAb SN3 or CD47 mAb InhibRx treatment (n = 4) and combination treatment with RTX (n = 3). (B) Phagocytosis of healthy PBMCs by autologous macrophages upon CD24 mAb SN3 or CD47 mAb InhibRx treatment and in combination with RTX. (n = 3). (C) Comparison between phagocytosis levels induced upon CD24 and CD47 antibody treatment of MCL blasts (red) and healthy PBMCs (gray). (D) Phagocytosis of MCL blasts co-cultured with macrophages from healthy donors upon treatment with CD24 mAb SN3 or CD47 mAb InhibRx and in combination with RTX. (n = 3). (E) Trogocytosis of MCL blasts by autologous PMNs upon treatment with CD24 mAb SN3 or CD47 mAb InhibRx (n = 5). Statistical testing was performed using paired Student’s t-test. (F) Trogocytosis of healthy PBMCs by autologous PMNs upon treatment with CD24 mAb SN3 or CD47 mAb InhibRx (n = 3). Statistical testing was performed using paired Student’s t-test. Where indicated, n.s. = non-significant, * = p < 0.05; ** = p < 0.01.
CD24 Antibody-Mediated Phagocytosis Is Superior to CD47 Checkpoint Targeting in MCL and Carcinoma, but Not in DLBCL
The prominent pro-phagocytic effect of CD24 but not CD47 antibody treatment on MCL cells suggested that CD24 is a potential novel target for therapy in MCL. However, targeting of the SIRPα/CD47 checkpoint did yield prominent clinical activity in a clinical trial in DLBCL and FL in combination with CD20 antibody rituximab. Notably, SN3 antibody treatment of DLBCL cell lines that expressed CD24, such as SU-DHL-10 and SU-DHL-6, did increase phagocytosis, albeit only up to 30% (Figure 4A). Moreover, and in contrast to MCL, CD24 antibody treatment of the DLBCL cell line panel induced lower levels of phagocytosis than treatment with CD47 mAb InhibRx in most cell lines, with the exception of SU-DHL-6 (Figure 4B). Confocal fluorescence microscopy also revealed low levels of engulfment of SU-DHL-6 by M2c macrophages upon CD24 mAb SN3 treatment, whereas CD47 mAb InhibRx treatment led to slightly higher phagocytic uptake (Figure 4C). Of note, expression levels of CD24 in both SU-DHL-10 and SU-DHL-6 are similar to those found in CD24low MCL cell lines (Figure 1C) and CD24 expression did not correlate with CD24 antibody-induced phagocytosis in DLBCL cell lines (Figure 4D). Thus, CD24 mAb treatment was inferior to CD47 mAb treatment in activating innate immune responses in DLBCL cell lines.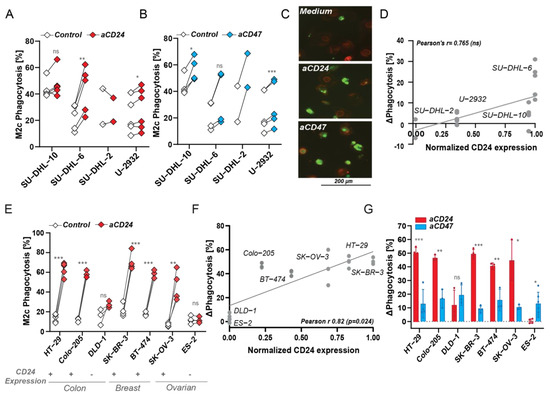
Figure 4. CD24 expression levels positively correlated with CD24 antibody-induced phagocytosis. (A) Percentage of phagocytosis upon CD24 SN3 antibody treatment vs. medium control by M2c macrophages (independent donors) in several DLBCL cell lines. Paired t-test. (B) Percentage of phagocytosis upon CD47 InhibRx antibody treatment vs. medium control by M2c macrophages (independent donors) in several DLBCL cell lines. Paired t-test. (C) Confocal microscopy images of phagocytosis of SU-DHL-6 where macrophages are in red and cancer cells are visible in green. (D) Correlation between CD24 expression and increase in phagocytosis upon CD24 SN3 treatment in DLBCL cell lines. CD24 expression was normalized to the highest value, corresponding to UPN-1 CD24 expression. Pearson’s r = 0.765 (ns) and linear regression p > 0.05. (E) Percentage of phagocytosis upon CD24 SN3 treatment vs. medium control by M2c macrophages (independent donors) in several colon (HT-29, Colo-205, DLD-1), breast (SK-BR-3, BT-474), and ovarian (SK-OV-3, ES-2) cell lines. Any effect was observed in CD24 negative cell lines (DLD-1, ES-2). Paired t-test. (F) Correlation between CD24 expression and increase in phagocytosis upon CD24 SN3 treatment in several colon (HT-29, Colo-205, DLD-1), breast (SK-BR-3, BT-474), and ovarian (SK-OV-3, ES2) cell lines. CD24 expression was normalized to the highest value, corresponding to SKBR3 CD24 expression. Pearson’s r = 0.82 (*) and linear regression p < 0.05. (G) Delta increase in phagocytosis compared to medium control upon CD24 and CD47 antibody treatment in several colon (HT-29, Colo-205, DLD-1), breast (SK-BR-3, BT-474), and ovarian (SK-OV-3, ES-2) cell lines. Student’s t-test. Where indicated, n.s. = non-significant, * = p < 0.05; ** = p < 0.01; *** = p < 0.001.
Induction of Phagocytosis of CD24 Expressing Cells Is Only in Part an Effect of Breaking CD24-Siglec-10 ‘Don’t Eat Me’ Signaling
In an effort to delineate the requisites and characteristics of CD24-based (re)activation of innate immunity, SN3 antibody clone (a mouse IgG1 binding to glycosylated epitope) and clone ML5 (a mouse IgG2a binding to the CD24 protein core) were first compared to their pro-phagocytic effect. The pro-phagocytic activity of mAb treatment may not only stem from the reported CD24/Siglec-10 checkpoint inhibition but may also stem from opsonization and induction of antibody-dependent cellular phagocytosis (ADCP). Indeed, when UPN-1 cells pretreated with mAbs were mixed with macrophages in the presence of high concentrations of Fc blocker to prevent ADCP-mediated activity, ML5-induced phagocytosis was abrogated (Figure 5A,B), similar to blocking effects observed for RTX (Figure 5B), a human IgG1 antibody that induces an ADCP [45]. Specifically, the delta increase in phagocytosis decreased from up to 60% without Fc blocker to <10% when Fc blocker solution was used for both UPN-1 and HBL-2 (Figure 5B,C). However, SN3-induced phagocytosis of UPN-1 was not significantly inhibited in the same setting (Figure 5A), with a non-significant <10% variation in the delta increase in phagocytosis upon inclusion of Fc blocker solution, for both UPN-1 and HBL-2 (Figure 5B,C). This data suggests that SN3 functions as a CD24 checkpoint inhibitor independent of functional mIgG1 activity, whereas ML5 activity is merely due to the opsonization of cancer cells, in agreement with its IgG2a backbone [46]. Indeed, mIgG1 is reported to not effectively trigger ADCP, with, e.g., also no phagocytosis detected upon treatment with CD19 mIgG1 antibody (Figure 5D). In line with this mode-of-action for SN3, the blocking of Fc-FcγR-dependent interaction using a goat F(ab’)2 anti-mouse IgG1 (Fc specific), as an alternate way to block SN3 IgG interaction with FcR, only decreased phagocytosis upon SN3 treatment by ~11% and ~9% for UPN-1 and HBL-2, respectively (Figure 5E). Thus, SN3 appears to have ADCP-independent activity reminiscent of checkpoint inhibition. However, in apparent contradiction, a direct F(ab’)2 preparation of SN3, generated by Ficin-mediated cleavage of the mIgG1 Fc, did not have significant pro-phagocytic effects on UPN-1 and HBL-2 (Figure 5F). Notably, the SN3 F(ab’)2 did block the binding of APC-labeled CD24 antibody to HBL-2 cells, indicating the preparation was functionally active. Further, the combination of SN3 F(ab’)2 with 1 ng/mL of RTX did not augment RTX-induced phagocytosis levels. Thus, although most evidence pointed to SN3-mediated innate checkpoint activity that was FcR-independent, conflicting results were obtained using SN3 F(ab’)2.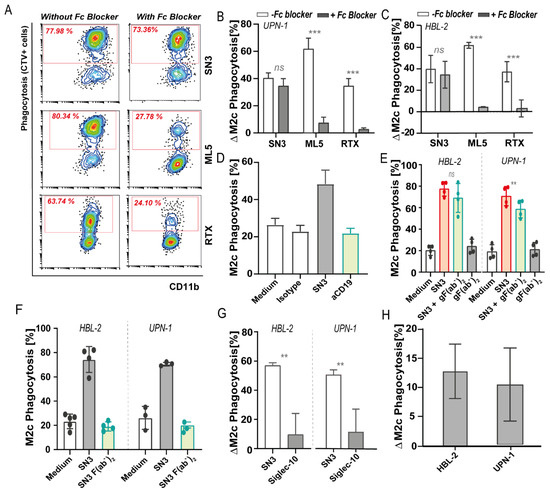
Figure 5. CD24 antibody treatment with two different murine anti-CD24 clones (SN3 and ML5) and Siglec-10 disruption of CD24/Siglec10 axis. (A) Flow cytometry diagrams showing the percentage of phagocytosis of UPN-1 cells upon CD24 antibody treatment with SN3 and ML5 clones by M2c macrophages in the presence or absence of high concentrations of Fc blocker. Rituximab (RTX) (anti-CD20) antibody was used as a control. (B) Increase in phagocytosis compared to medium control of UPN-1 cells upon CD24 antibody treatment with SN3 and ML5 antibody clones by M2c macrophages in the presence (gray) or absence (white) of high concentrations of Fc blocker. (C) Increase in phagocytosis compared to medium control of HBL-2 cells upon CD24 antibody treatment with SN3 and ML5 antibody clones by M2c macrophages in the presence (gray) or absence (white) of high concentrations of Fc blocker. n = 3, mean ± SD. n = 3, mean ± SD. Paired t-test, with p < 0.05 significant. (D) Phagocytosis of HBL-2 cells upon anti-CD24 (1 µg/mL, red) or anti-CD19 (10 µg/mL, green) treatment, being both mouse IgG1 antibodies. (E) Phagocytosis of HBL-2 and UPN-1 cells upon anti-CD24 treatment (SN3 clone) (red), same antibody pre-incubated with goat F(ab’)2 against mouse IgG Fc domain (gF(ab’)2) (green) or the goat F(ab’)2 alone (gray). Paired t-test, with p < 0.05 significant. (F) Phagocytosis levels of HBL-2 and UPN-1 cells upon CD24 SN3 treatment with the complete antibody (red) or F(ab’)2 preparations of the same antibody clone (SN3) (green). (G) Phagocytic uptake by M2c macrophages upon CD24 treatment with recombinant human Siglec-10 (His tagged) protein of HBL-2 and UPN-1 cells. (H) Increase in phagocytosis compared to medium control of HBL-2 and UPN-1 cells co-cultured with M2c macrophages previously incubated with 10 µg/mL of anti-Siglec-10 antibodies (5G6 clone). n = 3, mean ± SD. Where indicated, n.s. = non-significant, ** = p < 0.01; *** = p < 0.001.
Discussion
In the current study, CD24 was identified as a potential target for immunotherapy of MCL. Specifically, antibody-mediated targeting of CD24 robustly enhanced the phagocytic uptake of MCL cells yielding over 90% removal of CD24 expressing MCL cancer cells. Phagocytic uptake upon CD24 mAb treatment was significantly greater than upon treatment with the CD47 antibody InhibRx, both in cell lines and primary patient-derived blasts in an autologous setting. In line with this, high expression of CD24, but not CD47, correlated with poor OS in MCL and FL, whereas the opposite was found for DLBCL. The robust increase in phagocytosis upon CD24 mAb treatment was not limited to MCL and was also detected in a panel of carcinoma cell lines expressing CD24. Reversely, only low levels of phagocytosis were observed upon treatment with CD24 mAb in DLBCL, with the CD47 antibody InhibRx having superior effects. CD24-based cancer immunotherapy was originally reported decades ago for a subgroup of patients with B-lymphoproliferative disorders [47,48]. Recently, the interest in therapeutic targeting of CD24 was revitalized by its reported innate immune checkpoint function in carcinoma [19]. Specifically, in preclinical studies, the CD24 antibody clone SN3 was reported to block CD24/Siglec-10 inhibitory signaling and potentiate macrophage-mediated phagocytosis of carcinoma cells [19]. Correspondingly, treatment with the same antibody clone as well as a second murine CD24 antibody, clone ML5, here potently induced phagocytosis of MCL cells, an effect possibly related to checkpoint inhibition. Although newly uncovered in the context of cancer, such immune checkpoint activity of the CD24/Siglec-10 axis on innate immune cells has been well established in the context of apoptotic cell removal and infection [49]. As antibody clone SN3 potently induced phagocytosis of MCL cells, CD24/Siglec-10 checkpoint activity could exist in MCL similar to carcinoma. However, treatment with an F(ab’)2 preparation of this antibody failed to potentiate phagocytosis of MCL cells. In the original report on SN3 in carcinoma, its potential checkpoint activity was not evaluated using an F(ab’)2 preparation, thus raising the question of whether the observed activity of this antibody is due to checkpoint inhibitor activity or Fc-mediated. Notably, the phagocytic activity of clone ML5 was similarly abrogated when an F(ab’)2 preparation was used, which suggests that for both of these antibodies, the induction of antibody-dependent cellular phagocytosis (ADCP) may be the main mechanism of action. This would be in line with macrophage-dependent ADCP as reported in the initial clinical trial of combination treatment with murine CD24 and CD21 mAbs of B-cell lymphoma patients [47,48]. However, treatment with the complete SN3 mAb in the presence of high concentrations of Fc blocker solution still yielded high levels of phagocytic uptake. Similarly, alternative blocking of the Fc domain of clone SN3, with a goat anti-mouse F(ab’)2, also did not negatively impact phagocytic activity. With both approaches, the pro-phagocytic activity of clone ML5 was abrogated. Thus, in these assays, SN3 appeared to have Fc-independent activity. Thus, both proofs to support and disprove checkpoint inhibitor activity of antibody SN3 and/or Siglec-10-CD24 interaction were uncovered, highlighting the need for further investigation into the underlying mechanism (e.g., by constructing a human IgG4 or IgG2 variant). Notably, both clones SN3 and ML5 in our experiments effectively blocked Siglec-10/CD24 interaction, suggesting that the blocking of this interaction does not confer checkpoint inhibitor activity per se. In line with this, no significant correlation was found between CD24 mAb treatment response and Siglec-10 expression in a previous study [19], and, in our study, treatment with Siglec-10:Fc only induced only a modest increase in phagocytosis of 15%, compared to the 60% observed with CD24 mAb treatment. Nevertheless, studies using SIGLEC10 knock-out (KO) cells did yield a significant increase in the phagocytic ability of macrophages [19]. Since Siglec-10 triggers inhibitory ITIM-mediated signaling in macrophages [50], knocking out this inhibitory molecule may also have direct activatory and immunomodulatory effects on macrophages by itself. Interestingly, in this respect, high Siglec-10 levels in hepatocellular cancer patients correlated with significantly higher expression of inhibitory receptor genes such as PD1, TIM3, CTLA-4, or LAG-3 in a more recent study [51]. Moreover, blocking of Siglec-10 in TAMs decreased the expression of these immunosuppressive molecules. Thus, the fact that SIGLEC10 KO cells showed higher phagocytosis levels might also be due to alterations in the immunoregulatory profile of macrophages. Reversely, CD24 KO in cancer cells also significantly increased the level of cancer cell phagocytosis and reduced tumor outgrowth in vivo [19], which was taken as support for a role as an immune checkpoint in carcinoma. However, depletion of CD24 by siRNA in cell lines from different cancer types decreases cell proliferation rates, causes a variation in the actin cytoskeleton, and induces apoptosis in single cancer cell cultures [52]. Such alterations might be responsible for macrophage uptake after CD24 KO and be independent of Siglec10/CD24 interaction. CD24 KO in the MCL cell line UPN-1 resulted in cell death (data not shown), which is in line with the reported induction of apoptosis in immature CD24 KO B cells [53,54], demonstrating that altering the expression of CD24 has serious repercussions in these cells. Indeed, CD24 has also been reported as a cancer stem cell marker, with CD24 regulating PI3K/Akt [55], STAT, or FAK signaling pathways [56]. Thus, cell-intrinsic signaling alterations may underlie (part of) the effect of CD24 KO on phagocytic removal. Similarly, CD24 mAb treatment might trigger cell-intrinsic signaling effects. In the literature, mAb-mediated cross-linking induced apoptosis in BL cells via glycolipid-enriched membrane (GEM)-mediated signaling [30]. However, a 60% increase in phagocytic uptake was detected after only 2 h of incubation, which contrasts with the longer times typically required to bring intracellular signaling into play [57]. Correspondingly, no increase in PS exposure was observed upon treatment with CD24 mAb in our experiments within the time frame of the phagocytosis experiments. Finally, Siglec-10 is not the only reported macrophage ligand for CD24, with Siglec-5 [28], P-Selectins [58], L-selectins, or L1CAM being reported as well [59]. Interaction of CD24 with these other ligands and inhibition thereof by antibody treatment should also be considered. Altogether, it seems warranted to conclude that the phagocytosis detected upon CD24 antibody treatment is not solely due to the inhibition of CD24/Siglec-10 interaction and alternative mechanisms might be at play. Therefore, further efforts are needed to evaluate the mechanism behind CD24 mAb treatment, which might vary from clone to clone. The potential of CD24 mAb treatment and checkpoint targeting was previously preclinically investigated for pancreas, ovarian, and breast carcinoma [19]. Here, we focused on NHL where CD24 mRNA expression was similarly high in FL, BL, MCL, and DLBCL in line with previous studies [32,60]. However, CD24 expression only correlated with poor survival in FL and MCL. Notably, a previous study analyzed the prognostic significance of CD24 expression only in a mixed cohort of different types of NHL, but not within FL or MCL individually [60]. Unfortunately, further studies in FL could not be performed due to a lack of cell lines and patient material, but at the protein level, MCL cell lines expressed by far the highest CD24 surface levels. Moreover, MCL blasts expressed more CD24 than B cells from healthy donors. Apart from B cells, CD24 is also highly expressed in almost all human tissues (ATLAS database) and other hematological cells [10]. Nevertheless, phagocytosis of healthy PBMCs by autologous macrophages did not increase upon treatment with CD24 mAbs in our experiments. Still, off-target effects of CD24 mAb treatment must be carefully considered in the design of CD24-based immunotherapy. These results highlight the potential of targeting CD24 in MCL, which remains incurable with conventional chemoimmunotherapy [35]. The effect of CD24 mAb treatment in MCL and other NHL samples was compared to CD47 InhibRx antibody, with CD47 being the hallmark innate immune checkpoint. Interestingly, and despite the high CD47 expression found in the MCL samples, CD24 mAb treatment yielded a significantly higher effect than CD47 InhibRx antibody. Reversely, CD47 mAb treatment triggered significantly higher induction of phagocytosis of DLBCL cell lines than CD24 mAb (with the exception of SU-DHL-6). In line with this, the combination of CD47 antagonistic antibody with rituximab has already yielded complete responses in relapsed and refractory DLBCL patients [9]. This lack of efficacy of CD24 mAb in inducing phagocytosis in DLBCL is also in agreement with previous studies, where CD24 mRNA expression in ABC-DLBCL patients correlated with better R-CHOP treatment response [38]. In contrast to DLBCL, the effect of CD47 mAb in MCL was minimal compared to the significantly higher response induced by the treatment with CD24 mAb. Correspondingly, CD47 expression did not correlate with poor prognosis in MCL. Thus, CD47 seems to be a promising therapeutic target only in specific forms of B-cell lymphoma, such as DLBCL, in which it also correlated with poor prognosis, as detailed in our previous work [43]. CD24 mAb treatment might be a therapeutic option for some B-cell malignancies where current checkpoints such as CD47 may be less effective. Finally, in contrast to CD47, whose ligand SIRPα is highly expressed in both M2 and M1 subtypes of macrophages, Siglec-10, the ligand of CD24 is expressed at the highest level in the M2c subtype. However, anti-tumoral activity is mostly attributed to the M1 macrophage type [61,62], with, e.g., the antitumor effect of CD47 mAb in mice being attributed to macrophage-mediated antigen presentation, likely through M1 macrophages, and activation of adaptive T cell immunity [63]. For CD24 mAb treatment, pro-phagocytic activity was strongest in M2c macrophages, which are typically reported to promote anti-inflammatory and pro-tumoral responses in the tumor microenvironment (TME) [61,62]. Thus, preferential activation of M2c phagocytosis may not be desirable in the end. Notably, CD24 mAb treatment also induced an, albeit more reduced, effect in M1 macrophages. Therefore, and regardless of the final mechanism behind CD24 mAb treatment, the ability of macrophages to present tumor-associated antigens and activate the adaptive immune response upon CD24 antibody treatment should be investigated to shed more light on the nature of the final immune response and its clinical implications.Conclusions
CD24 mAb treatment in vitro enhanced phagocytic removal of CD24-positive MCL cell lines and primary autologous MCL blasts. This effect of CD24 mAb treatment was more potent than treatment with CD47 InhibRx mAb in MCL, but not in DLBCL. In agreement with this, high CD24 expression correlated with reduced OS in MCL (and FL) but not DLBCL. Thus, CD24 mAb treatment may represent an alternative therapeutic approach for MCL, although the relative importance of the CD24/Siglec-10 interaction as a “don’t eat me” signal remains to be elucidated.- , 2179–2188. [
- ] [
- CrossRef
- ]
- Cendrowicz, E.; Jacob, L.; Greenwald, S.; Tamir, A.; Pecker, I.; Tabakman, R.; Ghantous, L.; Tamir, L.; Kahn, R.; Avichzer, J.; et al. DSP107 combines inhibition of CD47/SIRPalpha axis with activation of 4-1BB to trigger anti-cancer immunity. J. Exp. Clin. Cancer Res. 2022, 41, 97. [Google Scholar] [CrossRef]
- Jalil, A.R.; Andrechak, J.C.; Discher, D.E. Macrophage checkpoint blockade: Results from initial clinical trials, binding analyses, and CD47-SIRPalpha structure-function. Antib. Ther. 2020, 3, 80–94. [Google Scholar] [CrossRef]
- Sikic, B.I.; Lakhani, N.; Patnaik, A.; Shah, S.A.; Chandana, S.R.; Rasco, D.; Colevas, A.D.; O’Rourke, T.; Narayanan, S.; Papadopoulos, K.; et al. First-in-Human, First-in-Class Phase I Trial of the Anti-CD47 Antibody Hu5F9-G4 in Patients With Advanced Cancers. J. Clin. Oncol. 2019, 37, 946–953. [Google Scholar] [CrossRef]
- Tseng, D.; Volkmer, J.P.; Willingham, S.B.; Contreras-Trujillo, H.; Fathman, J.W.; Fernhoff, N.B.; Seita, J.; Inlay, M.A.; Weiskopf, K.; Miyanishi, M.; et al. Anti-CD47 antibody-mediated phagocytosis of cancer by macrophages primes an effective antitumor T-cell response. Proc. Natl. Acad. Sci. USA 2013, 110, 11103–11108. [Google Scholar] [CrossRef]
- Advani, R.; Flinn, I.; Popplewell, L.; Forero, A.; Bartlett, N.L.; Ghosh, N.; Kline, J.; Roschewski, M.; LaCasce, A.; Collins, G.P.; et al. CD47 Blockade by Hu5F9-G4 and Rituximab in Non-Hodgkin’s Lymphoma. N. Engl. J. Med. 2018, 379, 1711–1721. [Google Scholar] [CrossRef]
- Barkal, A.A.; Weiskopf, K.; Kao, K.S.; Gordon, S.R.; Rosental, B.; Yiu, Y.Y.; George, B.M.; Markovic, M.; Ring, N.G.; Tsai, J.M.; et al. Engagement of MHC class I by the inhibitory receptor LILRB1 suppresses macrophages and is a target of cancer immunotherapy. Nat. Immunol. 2018, 19, 76–84. [Google Scholar] [CrossRef]
- Chen, H.M.; van der Touw, W.; Wang, Y.S.; Kang, K.; Mai, S.; Zhang, J.; Alsina-Beauchamp, D.; Duty, J.A.; Mungamuri, S.K.; Zhang, B.; et al. Blocking immunoinhibitory receptor LILRB2 reprograms tumor-associated myeloid cells and promotes antitumor immunity. J. Clin. Investig.
- ] [
- PubMed
- ]
- Pallegar, N.K.; Ayre, D.C.; Christian, S.L. Repression of CD24 surface protein expression by oncogenic Ras is relieved by inhibition of Raf but not MEK or PI3K.
- Front. Cell Dev. Biol.
- 2015
- ,
- 3
- , 47. [
- Google Scholar
- ] [
- CrossRef
- ]
- Bretz, N.P.; Salnikov, A.V.; Perne, C.; Keller, S.; Wang, X.; Mierke, C.T.; Fogel, M.; Erbe-Hofmann, N.; Schlange, T.; Moldenhauer, G.; et al. CD24 controls Src/STAT3 activity in human tumors.
- Cell Mol. Life Sci.
- 2012
- ,
- 69
- , 3863–3879. [
- Google Scholar
- ] [
- CrossRef
- ]
- Lan, H.; Li, W.; Jiang, H.; Yang, Y.; Zheng, X. Intracellular signaling transduction pathways triggered by a well-known anti-GHR monoclonal antibody, Mab263, in vitro and in vivo.
- Int. J. Mol. Sci.
- 2014
- ,
- 15
- , 20538–20554. [
- Google Scholar
- ] [
- CrossRef
- ]
- Tchernychev, B.; Furie, B.; Furie, B.C. Peritoneal macrophages express both P-selectin and PSGL-1. J. Cell Biol. 2003, 163, 1145–1155. [Google Scholar] [CrossRef]
- Finas, D.; Huszar, M.; Agic, A.; Dogan, S.; Kiefel, H.; Riedle, S.; Gast, D.; Marcovich, R.; Noack, F.; Altevogt, P.; et al. L1 cell adhesion molecule (L1CAM) as a pathogenetic factor in endometriosis. Hum. Reprod. 2008, 23, 1053–1062. [Google Scholar
CD24 Is Expressed in Several B-Cell Lymphomas, Being Most Highly Expressed in MCL, Where It Correlates with Poor Prognosis in Contrast to Hallmark Immune Checkpoint CD47
CD24 Is a Target for Reactivation of Phagocytosis in MCL, with a Superior Effect Than CD47 Antibody Treatment in both cell lines and
CD24 mAb Treatment Increased Phagocytosis of Primary MCL Blasts by Autologous Macrophages and PMNs, but Did Not Induce High Level of Phagocytosis of Healthy Cells
-
CD24 Antibody-Mediated Phagocytosis Is Superior to CD47 Checkpoint Targeting in MCL and Carcinoma, but Not in DLBCL
-
Induction of Phagocytosis of CD24 Expressing Cells Is Only in Part an Effect of Breaking CD24-Siglec-10 ‘Don’t Eat Me’ Signaling
Refere
3. Concelusions
- Robert, C. A decade of immune-checkpoint inhibitors in cancer therapy. Nat. Commun. 2020, 11, 3801. [Google Scholar] [CrossRef] [PubMed]
- Lentz, R.W.; Colton, M.D.; Mitra, S.S.; Messersmith, W.A. Innate Immune Checkpoint Inhibitors: The Next Breakthrough in Medical Oncology? Mol. Cancer Ther. 2021, 20, 961–974. [Google Scholar] [CrossRef] [PubMed]
- Cendrowicz, E.; Sas, Z.; Bremer, E.; Rygiel, T.P. The Role of Macrophages in Cancer Development and Therapy. Cancers 2021, 13, 1946. [Google Scholar] [CrossRef] [PubMed]
- Tong, B.; Wang, M. CD47 is a novel potent immunotherapy target in human malignancies: Current studies and future promises. Future Oncol. 2018, 14
- 2018
- ,
- 128
- , 5647–5662. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.S.; Gao, F.H. Molecular Mechanism of Tumor Cell Immune Escape Mediated by CD24/Siglec-10. Front. Immunol. 2020, 11, 1324. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Ma, H.; Zhang, J.; Zhu, L.; Wang, C.; Yang, Y. Unraveling the roles of CD44/CD24 and ALDH1 as cancer stem cell markers in tumorigenesis and metastasis. Sci. Rep. 2017, 7, 13856. [Google Scholar] [CrossRef] [PubMed]
- Salaria, S.; Means, A.; Revetta, F.; Idrees, K.; Liu, E.; Shi, C. Expression of CD24, a Stem Cell Marker, in Pancreatic and Small Intestinal Neuroendocrine Tumors. Am. J. Clin. Pathol. 2015, 144, 642–648. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Kim, D.; Kim, D.K.; Choi, K.U.; Suh, D.S.; Kim, J.H. Therapeutic Strategies for Targeting Ovarian Cancer Stem Cells. Int. J. Mol. Sci. 2021, 22, 5059. [Google Scholar] [CrossRef]
- Overdevest, J.B.; Knubel, K.H.; Duex, J.E.; Thomas, S.; Nitz, M.D.; Harding, M.A.; Smith, S.C.; Frierson, H.F.; Conaway, M.; Theodorescu, D. CD24 expression is important in male urothelial tumorigenesis and metastasis in mice and is androgen regulated. Proc. Natl. Acad. Sci. USA 2012, 109, E3588–E3596. [Google Scholar] [CrossRef]
- Altevogt, P.; Sammar, M.; Huser, L.; Kristiansen, G. Novel insights into the function of CD24: A driving force in cancer. Int. J. Cancer 2021, 148, 546–559. [Google Scholar] [CrossRef]
- Fang, X.; Zheng, P.; Tang, J.; Liu, Y. CD24: From A to Z. Cell Mol. Immunol. 2010, 7, 100–103. [Google Scholar] [CrossRef]
- Barkal, A.A.; Brewer, R.E.; Markovic, M.; Kowarsky, M.; Barkal, S.A.; Zaro, B.W.; Krishnan, V.; Hatakeyama, J.; Dorigo, O.; Barkal, L.J.; et al. CD24 signalling through macrophage Siglec-10 is a target for cancer immunotherapy. Nature 2019, 572, 392–396. [Google Scholar] [CrossRef]
- Kwon, M.J.; Han, J.; Seo, J.H.; Song, K.; Jeong, H.M.; Choi, J.S.; Kim, Y.J.; Lee, S.H.; Choi, Y.L.; Shin, Y.K. CD24 Overexpression Is Associated with Poor Prognosis in Luminal A and Triple-Negative Breast Cancer. PLoS ONE 2015, 10, e0139112. [Google Scholar] [CrossRef]
- Zhang, P.; Zheng, P.; Liu, Y. Amplification of the CD24 Gene Is an Independent Predictor for Poor Prognosis of Breast Cancer. Front. Genet. 2019, 10, 560. [Google Scholar] [CrossRef] [PubMed]
- Tarhriz, V.; Bandehpour, M.; Dastmalchi, S.; Ouladsahebmadarek, E.; Zarredar, H.; Eyvazi, S. Overview of CD24 as a new molecular marker in ovarian cancer. J. Cell Physiol. 2019, 234, 2134–2142. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.L.; Guo, C.R.; Su, W.Y.; Chen, Y.X.; Xu, J.; Fang, J.Y. CD24 Overexpression Related to Lymph Node Invasion and Poor Prognosis of Colorectal Cancer. Clin. Lab. 2018, 64, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Kay, R.; Rosten, P.M.; Humphries, R.K. CD24, a signal transducer modulating B cell activation responses, is a very short peptide with a glycosyl phosphatidylinositol membrane anchor. J. Immunol. 1991, 147, 1412–1416. [Google Scholar] [PubMed]
- Lingwood, D.; Simons, K. Lipid rafts as a membrane-organizing principle. Science 2010, 327, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Aigner, S.; Ruppert, M.; Hubbe, M.; Sammar, M.; Sthoeger, Z.; Butcher, E.C.; Vestweber, D.; Altevogt, P. Heat stable antigen (mouse CD24) supports myeloid cell binding to endothelial and platelet P-selectin. Int. Immunol. 1995, 7, 1557–1565. [Google Scholar] [CrossRef]
- Myung, J.H.; Gajjar, K.A.; Pearson, R.M.; Launiere, C.A.; Eddington, D.T.; Hong, S. Direct measurements on CD24-mediated rolling of human breast cancer MCF-7 cells on E-selectin. Anal. Chem. 2011, 83, 1078–1083. [Google Scholar] [CrossRef]
- Kristiansen, G.; Machado, E.; Bretz, N.; Rupp, C.; Winzer, K.J.; Konig, A.K.; Moldenhauer, G.; Marme, F.; Costa, J.; Altevogt, P. Molecular and clinical dissection of CD24 antibody specificity by a comprehensive comparative analysis. Lab. Investig. 2010, 90, 1102–1116. [Google Scholar] [CrossRef]
- Zhou, Q.; Wu, Y.; Nielsen, P.J.; Liu, Y. Homotypic interaction of the heat-stable antigen is not responsible for its co-stimulatory activity for T cell clonal expansion. Eur. J. Immunol. 1997, 27, 2524–2528. [Google Scholar] [CrossRef]
- Suzuki, T.; Kiyokawa, N.; Taguchi, T.; Sekino, T.; Katagiri, Y.U.; Fujimoto, J. CD24 induces apoptosis in human B cells via the glycolipid-enriched membrane domains/rafts-mediated signaling system. J. Immunol. 2001, 166, 5567–5577. [Google Scholar] [CrossRef]
- Mensah, F.F.K.; Armstrong, C.W.; Reddy, V.; Bansal, A.S.; Berkovitz, S.; Leandro, M.J.; Cambridge, G. CD24 Expression and B Cell Maturation Shows a Novel Link With Energy Metabolism: Potential Implications for Patients With Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Front. Immunol. 2018, 9, 2421. [Google Scholar] [CrossRef] [PubMed]
- Shapira, S.; Kazanov, D.; Mdah, F.; Yaakobi, H.; Herishanu, Y.; Perry, C.; Avivi, I.; Itchaki, G.; Shacham-Abulafia, A.; Raanani, P.; et al. Feasibly of CD24/CD11b as a Screening Test for Hematological Malignancies. J. Pers. Med. 2021, 11, 724. [Google Scholar] [CrossRef] [PubMed]
- Lancaster-Shorts, K.; Pirruccello, S. CD24 Expression in Follicular Lymphoma: An Alternative B-Cell Marker in Therapy Selected, Recurrent Lymphoma. Grad. Med. Educ. Res. J. 2020, 2, 65. [Google Scholar] [CrossRef]
- Jain, P.; Wang, M. Mantle cell lymphoma: 2019 update on the diagnosis, pathogenesis, prognostication, and management. Am. J. Hematol. 2019, 94, 710–725. [Google Scholar] [CrossRef] [PubMed]
- Maddocks, K. Update on mantle cell lymphoma. Blood 2018, 132, 1647–1656. [Google Scholar] [CrossRef] [PubMed]
- Hanel, W.; Epperla, N. Emerging therapies in mantle cell lymphoma. J. Hematol. Oncol. 2020, 13, 79. [Google Scholar] [CrossRef] [PubMed]
- Susanibar-Adaniya, S.; Barta, S.K. 2021 Update on Diffuse large B cell lymphoma: A review of current data and potential applications on risk stratification and management. Am. J. Hematol. 2021, 96, 617–629. [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.Y.; Li, H.B.; Zhang, Y.; Shen, D.; Liu, P.; Che, Y.Q. CD24 Contributes to Treatment Effect in ABC-DLBCL Patients with R-CHOP Resistance. Pharmgenom. Pers. Med. 2021, 14, 591–599. [Google Scholar] [CrossRef]
- Ma, M.C.J.; Tadros, S.; Bouska, A.; Heavican, T.; Yang, H.; Deng, Q.; Moore, D.; Akhter, A.; Hartert, K.; Jain, N.; et al. Subtype-specific and co-occurring genetic alterations in B-cell non-Hodgkin lymphoma. Haematologica 2022, 107, 690–701. [Google Scholar] [CrossRef]
- Blenk, S.; Engelmann, J.C.; Pinkert, S.; Weniger, M.; Schultz, J.; Rosenwald, A.; Muller-Hermelink, H.K.; Muller, T.; Dandekar, T. Explorative data analysis of MCL reveals gene expression networks implicated in survival and prognosis supported by explorative CGH analysis. BMC Cancer 2008, 8, 106. [Google Scholar] [CrossRef]
- Glas, A.M.; Kersten, M.J.; Delahaye, L.J.; Witteveen, A.T.; Kibbelaar, R.E.; Velds, A.; Wessels, L.F.; Joosten, P.; Kerkhoven, R.M.; Bernards, R.; et al. Gene expression profiling in follicular lymphoma to assess clinical aggressiveness and to guide the choice of treatment. Blood 2005, 105, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Dave, S.S.; Fu, K.; Wright, G.W.; Lam, L.T.; Kluin, P.; Boerma, E.J.; Greiner, T.C.; Weisenburger, D.D.; Rosenwald, A.; Ott, G.; et al. Molecular diagnosis of Burkitt’s lymphoma. N. Engl. J. Med. 2006, 354, 2431–2442. [Google Scholar] [CrossRef] [PubMed]
- Bouwstra, R.; He, Y.; de Boer, J.; Kooistra, H.; Cendrowicz, E.; Fehrmann, R.S.N.; Ammatuna, E.; Zu Eulenburg, C.; Nijland, M.; Huls, G.; et al. CD47 Expression Defines Efficacy of Rituximab with CHOP in Non-Germinal Center B-cell (Non-GCB) Diffuse Large B-cell Lymphoma Patients (DLBCL), but Not in GCB DLBCL. Cancer Immunol. Res. 2019, 7, 1663–1671. [Google Scholar] [CrossRef] [PubMed]
- Fadok, V.A.; de Cathelineau, A.; Daleke, D.L.; Henson, P.M.; Bratton, D.L. Loss of phospholipid asymmetry and surface exposure of phosphatidylserine is required for phagocytosis of apoptotic cells by macrophages and fibroblasts. J. Biol. Chem. 2001, 276, 1071–1077. [Google Scholar] [CrossRef] [PubMed]
- VanDerMeid, K.R.; Elliott, M.R.; Baran, A.M.; Barr, P.M.; Chu, C.C.; Zent, C.S. Cellular Cytotoxicity of Next-Generation CD20 Monoclonal Antibodies. Cancer Immunol. Res. 2018, 6, 1150–1160. [Google Scholar] [CrossRef] [PubMed]
- Invivogen. <review-ImmunoglobulinG-invivogen.pdf>. 2011. Available online: https://www.invivogen.com/review-antibody-generation (accessed on 29 April 2022).
- Waldmann, T.A. Monoclonal antibodies in diagnosis and therapy. Science 1991, 252, 1657–1662. [Google Scholar] [CrossRef] [PubMed]
- Benkerrou, M.; Jais, J.P.; Leblond, V.; Durandy, A.; Sutton, L.; Bordigoni, P.; Garnier, J.L.; Le Bidois, J.; Le Deist, F.; Blanche, S.; et al. Anti-B-cell monoclonal antibody treatment of severe posttransplant B-lymphoproliferative disorder: Prognostic factors and long-term outcome. Blood 1998, 92, 3137–3147. [Google Scholar] [CrossRef]
- Chen, G.Y.; Tang, J.; Zheng, P.; Liu, Y. CD24 and Siglec-10 selectively repress tissue damage-induced immune responses. Science 2009, 323, 1722–1725. [Google Scholar] [CrossRef]
- Forgione, R.E.; Di Carluccio, C.; Guzman-Caldentey, J.; Gaglione, R.; Battista, F.; Chiodo, F.; Manabe, Y.; Arciello, A.; Del Vecchio, P.; Fukase, K.; et al. Unveiling Molecular Recognition of Sialoglycans by Human Siglec-10. iScience 2020, 23, 101231. [Google Scholar] [CrossRef]
- Xiao, N.; Zhu, X.; Li, K.; Chen, Y.; Liu, X.; Xu, B.; Lei, M.; Xu, J.; Sun, H.C. Blocking siglec-10(hi) tumor-associated macrophages improves anti-tumor immunity and enhances immunotherapy for hepatocellular carcinoma. Exp. Hematol. Oncol. 2021, 10, 36. [Google Scholar] [CrossRef]
- Smith, S.C.; Oxford, G.; Wu, Z.; Nitz, M.D.; Conaway, M.; Frierson, H.F.; Hampton, G.; Theodorescu, D. The metastasis-associated gene CD24 is regulated by Ral GTPase and is a mediator of cell proliferation and survival in human cancer. Cancer Res. 2006, 66, 1917–1922. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, P.J.; Lorenz, B.; Muller, A.M.; Wenger, R.H.; Brombacher, F.; Simon, M.; von der Weid, T.; Langhorne, W.J.; Mossmann, H.; Kohler, G. Altered erythrocytes and a leaky block in B-cell development in CD24/HSA-deficient mice. Blood 1997, 89, 1058–1067. [Google Scholar] [CrossRef] [PubMed]
- Ayre, D.C.; Christian, S.L. CD24: A Rheostat That Modulates Cell Surface Receptor Signaling of Diverse Receptors. Front. Cell Dev. Biol. 2016, 4, 146. [Google Scholar] [CrossRef
- ] [CrossRef]
- Went, P.; Dellas, T.; Bourgau, C.; Maurer, R.; Augustin, F.; Tzankov, A.; Dirnhofer, S. [Expression profile and prognostic significance of CD24, p53 and p21 in lymphomas. A tissue microarray study of over 600 non-Hodgkin lymphomas]. Dtsch. Med. Wochenschr. 2004, 129, 2094–2099. [Google Scholar] [CrossRef]
- Yao, Y.; Xu, X.H.; Jin, L. Macrophage Polarization in Physiological and Pathological Pregnancy. Front. Immunol. 2019, 10, 792. [Google Scholar] [CrossRef]
- Porta, C.; Riboldi, E.; Ippolito, A.; Sica, A. Molecular and epigenetic basis of macrophage polarized activation. Semin. Immunol. 2015, 27, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Iribarren, K.; Buque, A.; Mondragon, L.; Xie, W.; Levesque, S.; Pol, J.; Zitvogel, L.; Kepp, O.; Kroemer, G. Anticancer effects of anti-CD47 immunotherapy in vivo. Oncoimmunology 2019, 8, 1550619. [Google Scholar] [CrossRef] [PubMed]
CD24 mAb treatment in vitro enhanced phagocytic removal of CD24-positive MCL cell lines and primary autologous MCL blasts. This effect of CD24 mAb treatment was more potent than treatment with CD47 InhibRx mAb in MCL, but not in DLBCL. In agreement with this, high CD24 expression correlated with reduced OS in MCL (and FL) but not DLBCL. Thus, CD24 mAb treatment may represent an alternative therapeutic approach for MCL, although the relative importance of the CD24/Siglec-10 interaction as a “don’t eat me” signal remains to be elucidated.
References
- Wenzhe Li; Huailei Ma; Jin Zhang; Ling Zhu; Chen Wang; Yanlian Yang; Unraveling the roles of CD44/CD24 and ALDH1 as cancer stem cell markers in tumorigenesis and metastasis. Scientific Reports 2017, 7, 1-15, 10.1038/s41598-017-14364-2.
- Safia N Salaria; Anna Means; Frank Revetta; Kamran Idrees; Eric Liu; Chanjuan Shi; Expression of CD24, a Stem Cell Marker, in Pancreatic and Small Intestinal Neuroendocrine Tumors.. American Journal of Clinical Pathology 2015, 144, 642-8, 10.1309/AJCPMZY5P9TWNJJV.
- Wookyeom Yang; Dasol Kim; Dae Kim; Kyung Choi; Dong Suh; Jae Kim; Therapeutic Strategies for Targeting Ovarian Cancer Stem Cells. International Journal of Molecular Sciences 2021, 22, 5059, 10.3390/ijms22105059.
- Thomas A. Waldmann; Monoclonal Antibodies in Diagnosis and Therapy. Science 1991, 252, 1657-1662, 10.1126/science.2047874.
- Amira A. Barkal; Rachel E. Brewer; Maxim Markovic; Mark Kowarsky; Sammy A. Barkal; Balyn Zaro; Venkatesh Krishnan; Jason Hatakeyama; Oliver Dorigo; Layla J. Barkal; et al.Irving L. Weissman CD24 signalling through macrophage Siglec-10 is a target for cancer immunotherapy. Nature 2019, 572, 392-396, 10.1038/s41586-019-1456-0.
- Shiran Shapira; Dina Kazanov; Fatin Mdah; Hadas Yaakobi; Yair Herishanu; Chava Perry; Irit Avivi; Gilad Itchaki; Adi Shacham-Abulafia; Pia Raanani; et al.Mori Hay-LevyGal AigerJacob MashiahShahar Lev-AriNadir Arber Feasibly of CD24/CD11b as a Screening Test for Hematological Malignancies. Journal of Personalized Medicine 2021, 11, 724, 10.3390/jpm11080724.
- Katrina Lancaster-Shorts Et Al.; Follow; CD24 Expression in Follicular Lymphoma: An Alternative B-Cell Marker in Therapy Selected, Recurrent Lymphoma. Graduate Medical Education Research Journal 2020, 2, 65, 10.32873/unmc.dc.gmerj.2.1.065.
- Preetesh Jain; Michael Wang; Mantle cell lymphoma: 2019 update on the diagnosis, pathogenesis, prognostication, and management. American Journal of Hematology 2019, 94, 710-725, 10.1002/ajh.25487.
- Kami Maddocks; Update on mantle cell lymphoma. Blood 2018, 132, 1647-1656, 10.1182/blood-2018-03-791392.
- P. Went; T. Dellas; C. Bourgau; R. Maurer; F. Augustin; A. Tzankov; S. Dirnhofer; Expressionsmuster und prognostische Bedeutung von CD24, p53 und p21 in Lymphomen. DMW - Deutsche Medizinische Wochenschrift 2004, 129, 2094-2099, 10.1055/s-2004-831850.
- Amira A. Barkal; Kipp Weiskopf; Kevin S. Kao; Sydney R. Gordon; Benyamin Rosental; Ying Y. Yiu; Benson M. George; Maxim Markovic; Nan G. Ring; Jonathan Tsai; et al.Kelly McKennaPo Yi HoRobin Z. ChengJames Y. ChenLayla J. BarkalAaron RingIrving L. WeissmanRoy L. Maute Engagement of MHC class I by the inhibitory receptor LILRB1 suppresses macrophages and is a target of cancer immunotherapy. Nature Immunology 2017, 19, 76-84, 10.1038/s41590-017-0004-z.
- Kami Maddocks; Update on mantle cell lymphoma. Blood 2018, 132, 1647-1656, 10.1182/blood-2018-03-791392.
