Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Vivi Li and Version 1 by Roberto Sorrentino.
Dental professionals and patients are exposed to a high risk of COVID-19 infection, particularly in the prosthodontic practice, because of the bio-aerosol produced during teeth preparation with dental handpieces and the strict contact with oral fluids during impression making.
- coronavirus
- COVID-19
- SARS-CoV-2
- prosthodontics
- airborne
- dental impression
- pandemic
- prevention
1. Introduction
In December 2019, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), was first reported in Wuhan, Hubei province (China), and rapidly spread over 24 countries, leading the World Health Organization (WHO) to declare this severe pneumonia a global emergency on 30 January 2020 [1]. From 31 December 2019 to 17 June 2022, over 535,863,000 confirmed cases of coronavirus infectious disease-19 (COVID-19) have been reported, including approximately 6,315,000 deaths, and these numbers are increasing daily (Figure 1 and Figure 2) [2].
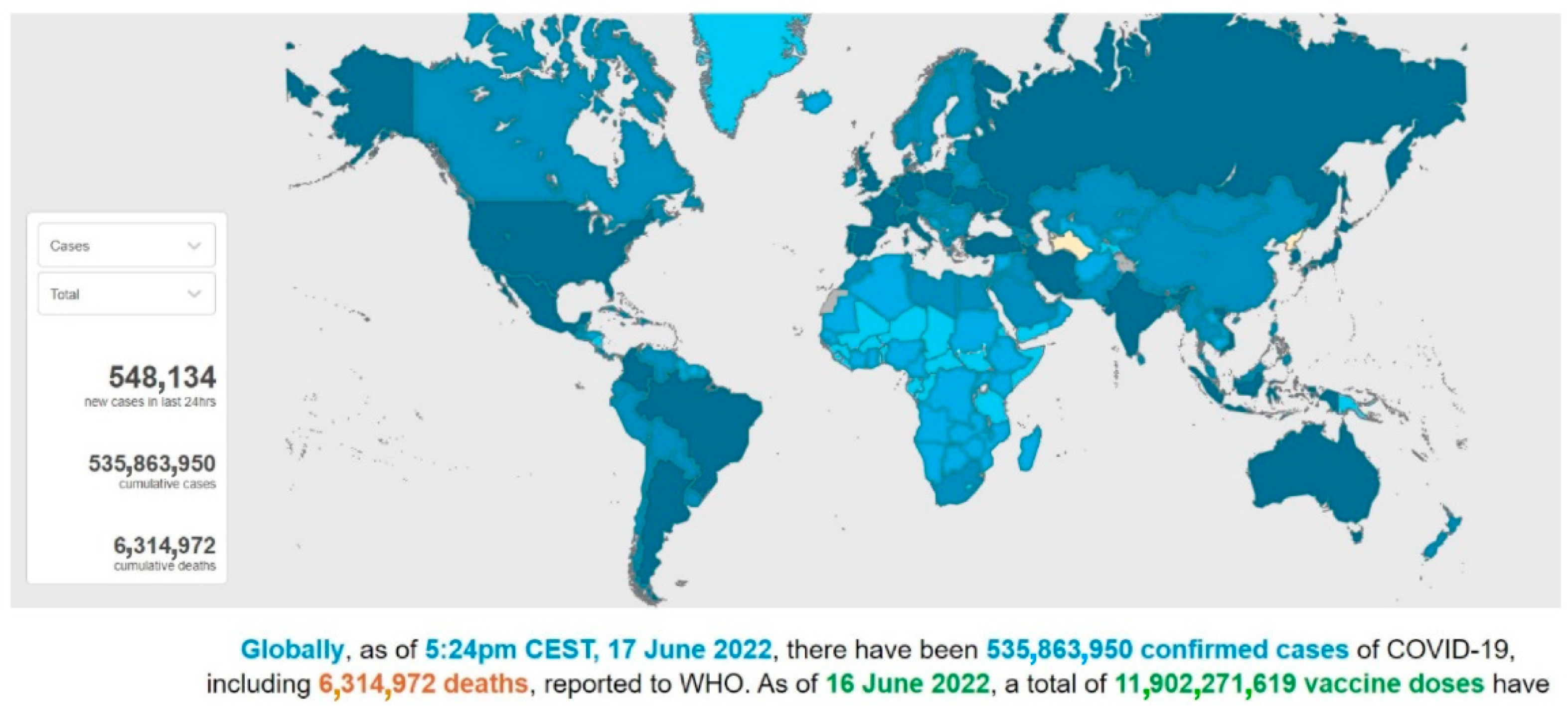
Figure 1. A screenshot of the interactive dashboard of COVID-19 global cases by the World Health Organization. This dashboard is continually updated and can be accessed at https://covid19.who.int/ (accessed on 17 June 2022).
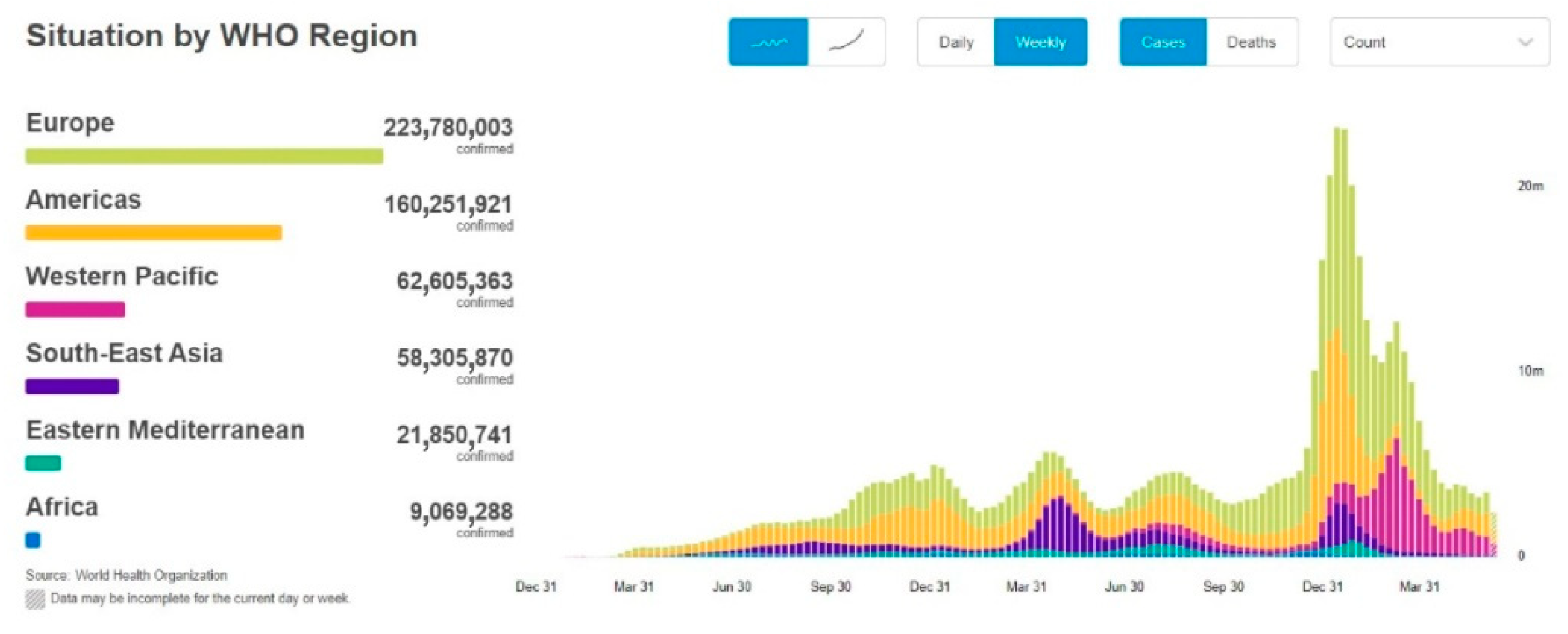
Figure 2. A screenshot of the COVID-19 situation by WHO Region. This dashboard is continually updated and can be accessed at https://covid19.who.int/ (accessed on 17 June 2022).
SARS-CoV-2, of the family Coronaviridae, is a single-stranded RNA beta coronavirus that is characterized by a diameter of 50–200 nm and probably originated from bats and pangolins [3]. It shares 85–92% nucleotide sequence homology with the pangolin coronavirus (CoV) genome and 96.2% nucleotide homology with bat CoV RaTG13, confirming the zoonotic origin of the virus [4]. The animal-to-human transmission event, the spillover phenomenon of SARS-CoV-2, can be plausibly imputed to the sale and killing of wildlife species at the Huanan Seafood Wholesale Market, where the initial cases of COVID-19 emerged [4]. The comprehension of the initial dynamics of the infection and the identification of the animal source of SARS-CoV-2 would help to prevent future new zoonosis by strengthening the control of food and hygiene within live animal markets.
Since the first COVID-19 infection, some variants of SARS-CoV-2 have been found [5]. These variants are adaptive mutations in the viral genome that can alter the virus’s pathogenic potential, and some of them were classified as variants of concern (VOCs) due to their public health implications [5,6][5][6]. In particular, VOCs have been associated with increased virulence or transmissibility, decreased neutralization by antibodies acquired through vaccination or natural infection, the ability to elude detection, and a reduction in therapeutic or vaccine efficiency [6]. Five SARS-CoV-2 VOCs have been recognized according to the WHO: Alpha (B.1.1.7, first report in the United Kingdom); Beta (B.1.351, first report in South Africa); Gamma (P.1, first report in Brazil); Delta (B.1.617.2, first report in India); and Omicron (B.1.1.529, first report in South Africa) [6]. Among the listed VOCs, the Omicron variant is the most severely altered, paving the path for increased transmissibility and partial resistance to COVID-19 vaccine-induced immunity (Figure 3 and Figure 4) [5,7,8][5][7][8].
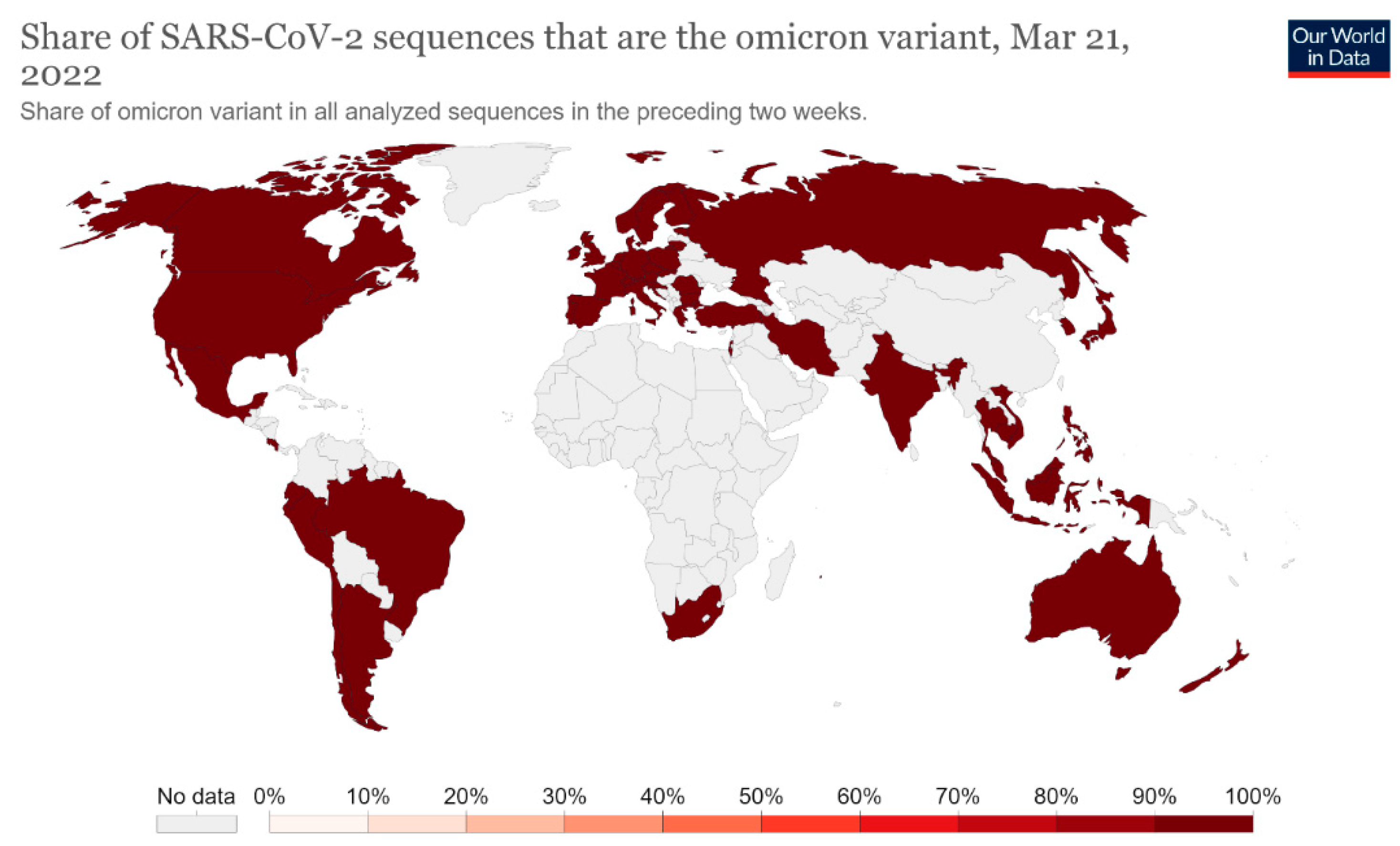
Figure 3. Share of SARS-CoV-2 sequences that are the omicron variant. This dashboard is continually updated and can be accessed at https://ourworldindata.org/grapher/covid-cases-omicron?country=GBR~FRA~BEL~DEU~ITA~ESP~USA~ZAF~BWA~AUS (accessed on 17 June 2022).
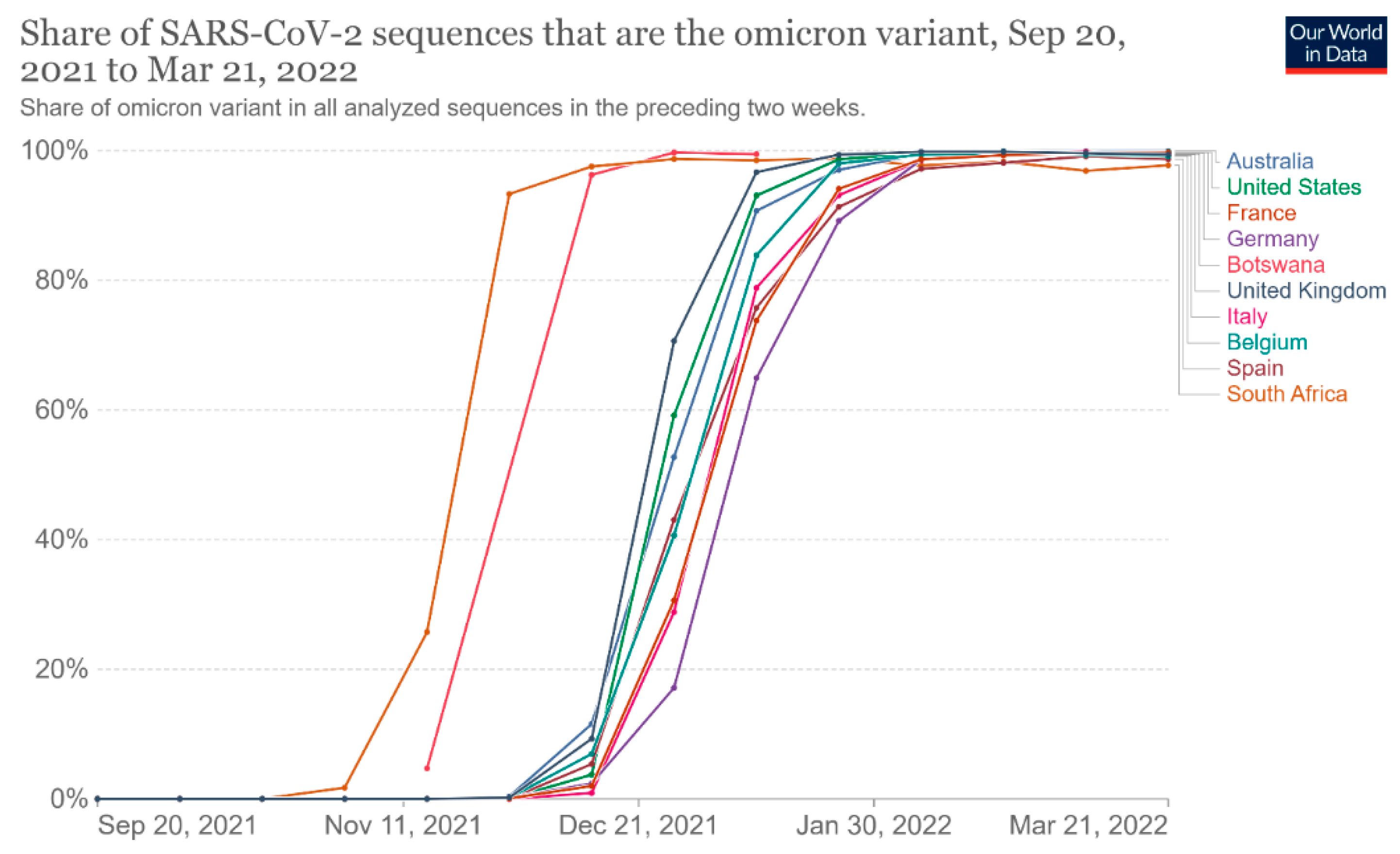
Figure 4. Share of omicron variant in all analyzed sequences. This dashboard is continually updated and can be accessed at https://ourworldindata.org/grapher/covid-cases-omicron?tab=chart&country=GBR~FRA~BEL~DEU~ITA~ESP~USA~ZAF~BWA~AUS (accessed on 17 June 2022).
The time from the infection to the onset of symptoms can vary from 2 to 14 days, and the median incubation period reported by the WHO was 5–6 days [9,10][9][10].
Older age and other risk factors, such as a history of underlying diseases and/or co-infections, play an important role in determining the severity of symptoms, leading to a higher risk to develop severe illness and death [11].
The clinical initial manifestations of COVID-19 can be aspecific. First of all, a large number of patients show symptoms of common cold, such as dry cough, sore throat, low-grade fever, or myalgia [11]. Less frequent manifestations of the infection may be nausea, diarrhea, dysgeusia, and persistent olfactory dysfunction (hyposmia) due to mucosal edema and nasal inflammation [12,13][12][13]. Some findings underlined the potential transmission of SARS-CoV-2 mediated by asymptomatic subjects, who may represent potential reservoirs for the spreading and re-emergence of the infection since the viral load in such patients was comparable to that of symptomatic patients [14].
Even if the mechanism of SARS-CoV-2 infection is not yet completely known, the high transmissibility of the virus can be partially explained by the higher affinity of the virus for cells located in the lower airways, where the virus binds using the host receptor for the conversion enzyme of angiotensin 2 (ACE2) and replicates, causing pneumonia [15].
The radiologic signs of bilateral pneumonia are abnormal ground-glass opacities found in chest X-ray and computed tomographic (CT) scans. The worst clinical picture patients can present is multiorgan dysfunction, acute renal failure, and acute respiratory distress syndrome (ARDS) [16].
People older than 60 years old and patients with chronic comorbidities (especially diabetes, high blood pressure, and cardiovascular diseases) were found to be more prone to develop severe clinical disease and fatal outcomes, which occurred only in critical cases. Fortunately, children often seem to show mild symptoms of COVID-19 [15].
SARS-CoV-2 has a human-to-human typical transmission by means of respiratory droplets and indirect contact, whereas the conjunctival and mother-to-fetus modes still need to be confirmed [17].
Some evidence suggested that infected individuals can spread infection through bodily secretions, such as the saliva and nasal fluid produced by talking, sneezing, and coughing. The high presence of the virus in saliva may be explained by the binding of SARS-CoV-2 with ACE2 receptors, which are highly concentrated in salivary glands [18,19,20][18][19][20].
Moreover, the contact of contaminated hands with the mucous membranes of the mouth and/or nose may lead to the onset of COVID-19 infection. The fecal–oral mode also may be another important route for nosocomial spread. Consequently, the disinfection of objects, handwashing, and social distancing (beyond six feet) are strongly recommended in order to control the community outbreak of the disease [17,18,19,20][17][18][19][20].
In particular, dental offices could be easily contaminated since the use of high-speed handpieces or ultrasonic instruments could cause the aerosolization of patients’ secretions, such as saliva or blood. Thus, dental professionals are exposed to a high risk of contracting SARS-CoV-2 because social distancing is unachievable during dental procedures [21].
On the other hand, the increased susceptibility of patients in dental offices is another important concern: elderly age, diabetes, chemotherapy, pregnancy, or conditions of an impaired immune defense system may easily lead to a worsening clinical picture or fatal outcome in the case of SARS-CoV-2 infection [11,22][11][22].
Among the branches of dentistry, prosthodontics is the part of restorative dentistry concerned with the design, manufacture, and fitting of artificial replacements for missing teeth and the associated soft tissues. Many aspects and devices used during prosthetic procedures may offer the opportunity for cross-contamination, requiring careful attention and rigorous protocols for the prevention of infection spreading.
To date, there are scarce data in the scientific literature about the management of COVID-19 infection during prosthodontics procedures in order to prevent COVID-19 cross-infections.
2. Dental Impression
Dental impression and wax or silicone interocclusal records could be contaminated with patients’ saliva and, just as frequently, blood (Figure 65).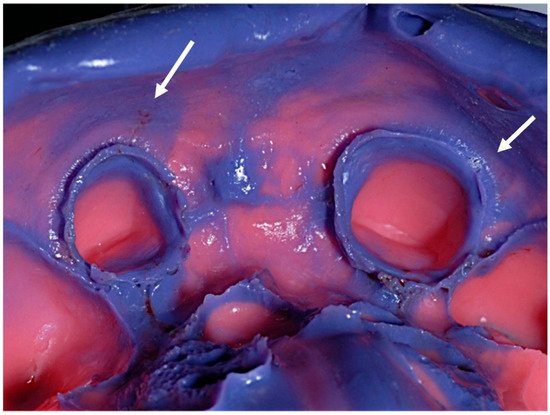
Figure 65.
Debris of saliva and blood on a conventional elastomeric dental impression (arrows indicated).
3. Prosthodontics Aids
In order to reduce the probability of contagion, maximum attention has to be paid to the disinfection and sterilization of the instruments and aids that can be used during prosthetic clinical practice. For this purpose, it is useful to dwell on the following indications reported in the literature. The disinfection of some aids, such as occlusal rims or casts, could be achieved by spraying with 5.25% sodium hypochlorite solution and allowing to set for 10 min [35,36,37][33][34][35]. Moist heat sterilization is performed using a steam autoclave at 121 °C for 15–20 min at 15 lb pressure/square inch, and it is suggested for handpieces, stainless steel instruments, tissue retraction pluggers, dapen dishes, and glass slabs in combination with ethylene oxide at a concentration of 450–800 mg/L [38,39][36][37]. Moreover, ethylene oxide might be used for the disinfection of mouth and face mirrors, carbon steel hand instruments, three-way syringes, saliva ejectors, and evacuators [40][38]. Regarding impression trays, metallic ones should be heat-sterilized via autoclave, chemical vapor, or dry heat and ethylene oxide sterilization. Custom acrylic resin should be disinfected with tuberculocidal hospital disinfectant for reuse during the same patient’s next visit, while it is suggested to avoid using plastic trays because they are more difficult to sterilize efficiently [41][39]. Acrylic dentures must be soaked in glutaraldehyde solution for 12 h after being rinsed with running water and stored in an ultrasonic cleaner. Subsequently, they must be cleaned with running water, scraped with chlorhexidine, and then exposed to chlorine dioxide for 3 min. Then, ethylene oxide is used to sterilize [40][38]. Regarding dentures made of metal, it is suggested to spray with 2% glutaraldehyde solution and place the prostheses in a plastic bag for 10 min [32][30].4. Metal–Ceramic Materials
Immersion in glutaraldehydes for the duration indicated by the disinfectant manufacturer can be used to disinfect fixed metal/porcelain prostheses. In addition, fixed prostheses may be disinfected by immersing them in diluted hypochlorite for a brief period of time without causing harm. The higher the noble metal concentration, the lower the risk of harmful effects on the metal core [42][40]. Care should be taken to limit the amount of time in which metals are exposed to potentially corrosive chemicals. Iodophors might be utilized as well, but there is no evidence to back this up. Fixed metal prostheses can be disinfected with ethylene oxide or even autoclaving if needed. Conversely, unglazed porcelain should not be exposed to any disinfectant because porcelain firing/glazing might be sufficient [42][40]. Before delivering to the patient, any aid that was treated with a disinfectant must be properly cleaned.5. All-Ceramic Materials
All-ceramic materials require surface treatments aimed at favoring the adhesion of the material to the dental surfaces. This surface treatment is called “etching” and mainly concerns the glass ceramics, such as feldspathic or leucite-based ceramics, lithium silicates, and disilicates, in order to cement these materials to the prepared tooth adhesively, increasing the resistance to fracture of the final restoration [43,44,45][41][42][43]. Regarding zirconia ceramics, they are characterized by the absence of glass in their composition, and the surface treatment takes place through a tribochemical silica coating or low-pressure air abrasion (Al2O3, 50 µm at 2 bars) for both conventional and adhesive cementations [46,47,48][44][45][46]. The surface treatment procedures of all-ceramic materials require some rinsing and drying steps, both in the laboratory and in the dental office. Unfortunately, the rinsing and drying phases lead to nebulization and the formation of bio-aerosols. Bio-aerosols are liquid/solid particles generated by different sources and are responsible for the transmission of airborne microorganisms, using droplet nuclei (1–5 μm) or droplets (>5 μm). High-speed handpieces, ultrasonic scalers, air turbines, air polishing, and air–water syringes are all sources of bio-aerosols during dental practice. These particles can stay suspended in the air for a variable time and fall, contaminating all the surfaces of dental offices [49][47]. Tooth preparation procedures or chairside prosthesis modifications require the use of dental handpieces, and the production of aerosols is unavoidable, resulting in a high risk of indirect infections for patients and dental professionals, which is difficult to contain [50][48]. SARS-CoV-2 spread may occur via respiratory droplets and contact transmission. Hence, it is advisable to meticulously manage the patients to minimize the risk of COVID-19 infections.References
- Pan, A.; Liu, L.; Wang, C.; Guo, H.; Hao, X.; Wang, Q.; Huang, J.; He, N.; Yu, H.; Lin, X.; et al. Association of Public Health Interventions with the Epidemiology of the COVID-19 Outbreak in Wuhan, China. JAMA 2020, 323, 1915–1923.
- European Centre for Disease Prevention and Control. An agency of the European Union. Available online: https://www.ecdc.europa.eu/en/geographical-distribution-2019-ncov-cases (accessed on 5 January 2022).
- Kakodkar, P.; Kaka, N.; Baig, M. A comprehensive literature review on the clinical presentation, and management of the pandemic coronavirus disease 2019 (COVID-19). Cureus 2020, 12, e7560.
- Ye, Z.W.; Yuan, S.; Yuen, K.S.; Fung, S.Y.; Chan, C.P.; Jin, D.Y. Zoonotic origins of human coronaviruses. Int. J. Biol. Sci. 2020, 16, 1686–1697.
- Araf, Y.; Akter, F.; Tang, Y.D.; Fatemi, R.; Parvez, M.S.A.; Zheng, C.; Hossain, M.G. Omicron variant of SARS-CoV-2: Genomics, transmissibility, and responses to current COVID-19 vaccines. J. Med. Virol. 2022, 94, 1825–1832.
- Aleem, A.; Akbar Samad, A.B.; Slenker, A.K. Emerging Variants of SARS-CoV-2 And Novel Therapeutics against Coronavirus (COVID-19). In StatPearls; StatPearls Publishing LLC: Treasure Island, FL, USA, 2021.
- World Health Organization. Classification of Omicron (B.1.1.529): SARS-CoV-2 Variant of Concern. Available online: https://www.who.int/news/item/26-11-2021-classification-of-omicron-(b.1.1.529)-sars-cov-2-variant-of-concern (accessed on 20 June 2022).
- Torjesen, I. COVID-19: Omicron may be more transmissible than other variants and partly resistant to existing vaccines, scientists fear. BMJ 2021, 375, n2943.
- Coronavirus Disease 2019 (COVID-19) Situation Report–30. 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200219-sitrep-30-covid-19.pdf?sfvrsn=3346b04f_2 (accessed on 31 December 2021).
- Backer, J.A.; Klinkenberg, D.; Wallinga, J. Incubation period of 2019 novel coronavirus (2019-nCoV) infections among travellers from Wuhan, China, 20–28 January 2020. Euro Surveill 2020, 25, 2000062.
- Kolifarhood, G.; Aghaali, M.; Mozafar Saadati, H.; Taherpour, N.; Rahimi, S.; Izadi, N.; Hashemi Nazari, S.S. Epidemiological and clinical aspects of COVID-19; a narrative review. Arch. Acad. Emerg. Med. 2020, 8, e41.
- Soler, Z.M.; Patel, Z.M.; Turner, J.H.; Holbrook, E.H. A primer on viral-associated olfactory loss in the era of COVID-19. Int. Forum Allergy Rhinol. 2020, 10, 814–820.
- Giacomelli, A.; Pezzati, L.; Conti, F.; Bernacchia, D.; Siano, M.; Oreni, L.; Rusconi, S.; Gervasoni, C.; Ridolfo, A.L.; Rizzardini, G.; et al. Self-reported olfactory and taste disorders in SARS-CoV-2 patients: A cross-sectional study. Clin. Infect. Dis. 2020, 71, 889–890.
- Zou, L.; Ruan, F.; Huang, M.; Liang, L.; Huang, H.; Hong, Z.; Yu, J.; Kang, M.; Song, Y.; Xia, J.; et al. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N. Engl. J. Med. 2020, 382, 1177–1179.
- Gabutti, G.; D’Anchera, E.; Sandri, F.; Savio, M.; Stefanati, A. Coronavirus: Update Related to the Current Outbreak of COVID-19. Infect. Dis. Ther. 2020, 9, 241–253.
- Chih-Cheng, L.; Tzu-Ping, S.; Wen-Chien, K.; Hung-Jen, T.; Po-Ren, H. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges. Int. J. Antimicrob. Agents 2020, 55, 105924.
- Yang, C.; Ma, Q.Y.; Zheng, Y.H.; Yang, Y.X. Transmission routes of 2019-novel coronavirus (2019-nCoV). Zhonghua Yu Fang Yi Xue Za Zhi 2020, 54, 374–377.
- Dietz, L.; Horve, P.; Coil, D.A.; Fretz, M.; Eisen, J.A.; Van Den Wymelenberg, K. 2019 Novel Coronavirus (COVID-19) pandemic: Built environment considerations to reduce transmission. mSystems 2020, 5, e00245-20.
- To, K.K.; Tsang, O.T.; Yip, C.C.; Chan, K.H.; Wu, T.C.; Chan, J.M.; Leung, W.S.; Chik, T.S.; Choi, C.Y.; Kandamby, D.H.; et al. Consistent Detection of 2019 Novel Coronavirus in Saliva. Clin. Infect. Dis. 2020, 71, 841–843.
- Ong, S.W.X.; Tan, Y.K.; Chia, P.Y.; Lee, T.H.; Ng, O.T.; Wong, M.S.Y.; Marimuthu, K. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient. JAMA 2020, 323, 1610–1612.
- Meng, L.; Hua, F.; Bian, Z. Coronavirus Disease 2019 (COVID-19): Emerging and future challenges for dental and oral medicine. J. Dent. Res. 2020, 99, 481–487.
- Dropulic, L.K.; Lederman, H.M. Overview of infections in the immunocompromised host. Microbiol. Spectr. 2016, 4, 1–50.
- Barenghi, L.; Barenghi, A.; Cadeo, C.; Di Blasio, A. Innovation by Computer-Aided Design/Computer-Aided Manufacturing technology: A look at infection prevention in dental settings. Biomed. Res. Int. 2019, 2019, 6092018.
- Sofou, A.; Larsen, T.; Fiehn, N.E.; Owall, B. Contamination level of alginate impressions arriving at a dental laboratory. Clin. Oral Investig. 2002, 6, 161–165.
- Al Shikh, A.; Milosevic, A. Effectiveness of alcohol and aldehyde spray disinfectants on dental impressions. Clin. Cosmet. Investig. Dent. 2020, 12, 25–30.
- British Dental Association. Health Technical Memorandum 01-05: Decontamination in Primary Care Dental Practices. London: Department of Health. Available online: https://www.england.nhs.uk/wp-content/uploads/2021/05/HTM_01-05_2013.pdf (accessed on 31 December 2021).
- Kotsiomiti, E.; Tzialla, A.; Hatjivasiliou, K. Accuracy and stability of impression materials subjected to chemical disinfection—A literature review. J. Oral Rehabil. 2008, 35, 291–299.
- Fathizadeh, H.; Maroufi, P.; Momen-Heravi, M.; Dao, S.; Köse, Ş.; Ganbarov, K.; Pagliano, P.; Esposito, S.; Kafil, H.S. Protection and disinfection policies against SARS-CoV-2 (COVID-19). Infez. Med. 2020, 28, 185–191.
- Kampf, G.; Todt, D.; Pfaender, S.; Steinmannb, E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J. Hosp. Infect. 2020, 104, 246–251.
- Prabu, P.; Kattak, S.A.K.; Prabu, N.; Beenu, V.; Nair, S.J.; Sreeleksmi, M.S. Asepsis in prosthodontics. J. Prosthet. Implant Dent. 2021, 4, 67–71.
- Punj, A.; Bompolaki, D.; Garaicoa, J. Dental impression materials and techniques. Dent. Clin. North Am. 2017, 61, 779–796.
- Markarian, R.A.; da Silva, R.L.B.; Burgoa, S.; Pinhata-Baptista, O.H.; No-Cortes, J.; Cortes, A.R.G. Clinical relevance of Digital Dentistry during COVID-19 outbreak: A scoped review. Braz. J. Oral Sci. 2020, 19, e200201.
- Naylor, W.P. Prosthodontic items of interest. Int. J. Prosthodont. 1992, 5, 188–189.
- Rampal, N.; Pawah, S.; Kaushik, P. Infection Control in Prosthodontics. J. Oral Health Comm. Dent. 2010, 4, 7–11.
- ADA Council on Scientific Affairs; ADA Council on Dental Practice. Infection control recommendations for the dental and the dental laboratory. J. Am. Dent. Assoc. 1996, 127, 672–680.
- Sumanth, K.S.; Poovani, S.; Shetty, G.; Kumar, N.S.B.; Sudhakar, S.K. Infection Control Protocol in Prosthodontic—A Review. Int. J. Scien. Res. 2019, 8, 75–78.
- Council on dental materials. Infection control recommendation for a dental office and dental laboratory by the council of dental materials. J. Am. Dent. Assoc. 1988, 116, 241–248.
- Sarangi, D. Infection Control in Prosthodontics: A Review. Indian J. Forensic Med. Toxicol. 2020, 14, 8180–8183.
- Bhat, V.S.; Shetty, M.S.; Shenoy, K.K. Infection control in the prosthodontic laboratory. J. Indian Prosthodont. Soc. 2007, 7, 62–65.
- Jain, S.; Yadav, B.; Phogat, S.; Madan, R. Disinfection in prosthodontics. Int. J. Dent. Health Sci. 2014, 1, 779–787.
- Zidan, O.; Ferguson, G.C. The retention of complete crowns prepared with three different tapers and luted with four different cements. J. Prosthet. Dent. 2003, 89, 565–571.
- Gönüldaş, F.; Öztürk, C.; Atalay, P.; Öztaş, D. Influence of different surface finishing techniques on machinable feldspathic and leucite-reinforced ceramics. Dent. Mater. J. 2019, 38, 317–322.
- Maroulakos, G.; Thompson, G.A.; Kontogiorgos, E.D. Effect of cement type on the clinical performance and complications of zirconia and lithium disilicate tooth-supported crowns: A systematic review. Report of the Committee on Research in Fixed Prosthodontics of the American Academy of Fixed Prosthodontics. J. Prosthet. Dent. 2019, 121, 754–765.
- Kim, M.J.; Kim, K.H.; Kim, Y.K.; Kwon, T.Y. Degree of conversion of two dual-cured resin cements light-irradiated through zirconia ceramic disks. J. Adv. Prosthodont. 2013, 5, 464–470.
- Kern, M. Bonding to oxide ceramics-laboratory testing versus clinical outcome. Dent. Mater. 2015, 31, 8–14.
- Özcan, M.; Bernasconi, M. Adhesion to zirconia used for dental restorations: A systematic review and meta-analysis. J. Adhes. Dent. 2015, 17, 7–26.
- Zemouri, C.; de Soet, H.; Crielaard, W.; Laheij, A. A scoping review on bio-aerosols in healthcare and the dental environment. PLoS ONE 2017, 12, e0178007.
- Akin, H.; Karabay, O.; Toptan, H.; Furuncuoglu, H.; Kaya, G.; Akin, E.G.; Koroglu, M. Investigation of the Presence of SARS-CoV-2 in Aerosol After Dental Treatment. Int. Dent. J. 2022, 72, 211–215.
More
