Today, researchers are looking at new ways to treat severe infections caused by resistance to standard antibiotic therapy. This is quite challenging due to the complex and interdependent relationships involved: the cause of infection–the patient–antimicrobial agents. The sessile biofilm form is essential in research to reduce resistance to very severe infections (such as ESKAPE pathogens: Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumanni, Pseudomonas aeruginosa, and Enterobacter spp). The purpose of this study is to elucidate the mechanisms of the occurrence, maintenance, and suppression of biofilm infections. One form of biofilm suppression is the efficient action of natural antagonists of bacteria—bacteriophages. Bacteriophages effectively penetrate the biofilm’s causative cells. They infect those bacterial cells and either destroy them or prevent the infection from spreading. In this process, bacteriophages are specific, relatively easy to apply, and harmless to the patient. Antimicrobial peptides (AMPs) support the mechanisms of bacteriophages’ action. AMPs could also attack and destroy infectious agents on their own (even on biofilm). AMPs are simple, universal peptide molecules, mainly cationic peptides. Additional AMP research could help develop even more effective treatments of biofilm (bacteriophages, antibiotics, AMPs, nanoparticles). This is a review of recent unconventional agents, such as bacteriophages and AMPs, used for the eradication of biofilm, providing an overview of potentially new biofilm treatment strategies.
- antimicrobial peptides
- bacteriophage
- biofilm
- immunomodulatory action
- resistance
1. Introduction
From its beginning until the present day, the natural course of human life has been marked by transition, and it is still passing through often unknown processes of adaptation and evolution [1]. Destroying life, and even destroying the smallest carriers—microorganisms, is not easy and very often impossible [2]. Is it possible at all, and for how long, to delay an event's unfavorable and unwanted course? The ineffectiveness of antibacterial drugs is not an isolated phenomenon but an increasingly common occurrence [3][4][5][3,4,5]. Increasing bacterial resistance is connected to the patient and clinicians’ malpractice in prescribing and using antibiotics [6].
When science created the first antibiotics preparations to facilitate and raise human life quality, humanity was relieved [7]. Deadly diseases became transient conditions, and a growing selection of effective drugs guaranteed an optimistic future and extended life expectancy [8]. Simultaneously, the ever deeper delving began into the unknown principles of maintenance of life as a phenomenon.
Slowly, the growing lack of antibiotic effectiveness has led us along the path of learning about the mechanism of adaptation and even the evolution of the bacteria that carry the simplest forms of life, returning us to the very beginning and even simpler forms of life—viruses. Thus, the appearance of resistance in bacteria demonstrates one of the fundamental principles of preserving the phenomenon of life [9]. Thanks to technological advances, new ways of delivering antimicrobial peptides have been developed—one is by using nanoparticles. Those with silver are the best choice due to its antimicrobial activity [10][11][12][13][10,11,12,13].
2. Biofilm Treatment Strategies
The ability to make biofilm is an evolutionary achievement, with new “multicellular ability” traits that allow bacteria to survive, infect, multiply, and permanently infect hosts. As already mentioned, the resistance of clinically relevant bacteria (ESKAPE) and other groups of resistant bacteria is primarily the ability to infect the host despite biofilm inhibition measures taken, such as surface change and modification (medical implants or other biomaterials) using antibacterial agents, where the coating creates a barrier to bacterial adhesion [14][15][16]. Also, the use of small molecules of bacterial biofilm inhibitors creates antifilm properties that passivate the surface of implants or medical devices (such as phenols, imidazoles, indole) [17][18][19]. An alternative method in biofilm control is biologically active agents, such as predatory bacteria species [20].
Biofilm dispersion is the second strategy in treating biofilm infections. More precisely, the disruption of quorum sensing by chemical means leads to biofilm dispersion [21][22][23]. However, biofilm dispersal agents should be combined with an antimicrobial agent [17][24]. Namely, if these dispersed bacteria are not treated simultaneously with antibacterial agents, they will inevitably form new biofilms by infecting new areas [25][16]. Treatment by co-administration of drugs and dispersal agents is very complex and challenging. However, as usual, the answer to this phenomenon already exists in microbiocenosis, by “infecting” the biofilm with viruses—bacteriophages.
2.1. Bacteriophages
Bacteriophages are viruses that infect bacteria. Bacteriophages have been infectiously parasitic on bacteria from the very beginning of life. This virus–bacterium relationship is the oldest form of microbiocenosis, and perfection has been achieved in the form of a specific match between the virus –bacteriophage and the host cell-bacterium [26][27][28].
The first practical and positive experiences of using bacteriophages in controlling bacterial infections were developed during the Second World War. Bacteriophages found their application in treating war wound infections (explosive and blast injuries) before the use of antibiotics [29]. In extensive infections of such wounds, pathogen-specific bacteriophage preparations (
2.1. Bacteriophages
Pseudomonas aeruginosa
Staphylococcus aureus
Escherichia coli
Klebsiella spp.) were applied directly to the site of infection (biofilm infections) [30].
The phage action mechanism on the prokaryotic cell begins with overcoming the cell membrane’s defense mechanisms, the incorporation of the genome into the cytoplasm, and the proliferation of phages. Phages impair the bacterial cells’ normal function by their proliferation and thus inactivate or kill cells (lysogenic or lytic cycle,
Figure 2) [26][29]. The relationship between bacteriophages and bacteria in biofilm is far more complex. Bacteriophages must have the ability to encode a depolymerase that degrades the biofilm matrix, which includes degradation of polymers, capsular polysaccharides, and extracellular DNA [31][32]. Only then do they access cell membranes and receptors. In the treatment of bacterial infections, the addition of several enzymes that increases phages' activity is used, which leads to synergistic removal of bacteria. Thus, it is essential to create the conditions to bypass the bacterial biofilm matrix [33][34]. Therefore, bacteriophages can penetrate the membrane receptors, but antibiotics cannot due to the biofilm’s defense mechanisms [35][36]. The combination of phage and antibiotics seems to be the optimal combination in the fight against biofilm. In most cases, it is optimal in various combinations, even in combination with disinfectants [37][38]. However, in some situations, phage application could even lead to enhanced bacteria aggregation in the biofilm, surface adhesion, and fimbriae production. This usually occurs in Gram-negative bacteria leading to inhibition of phage penetration through the biofilm [33][39][40].
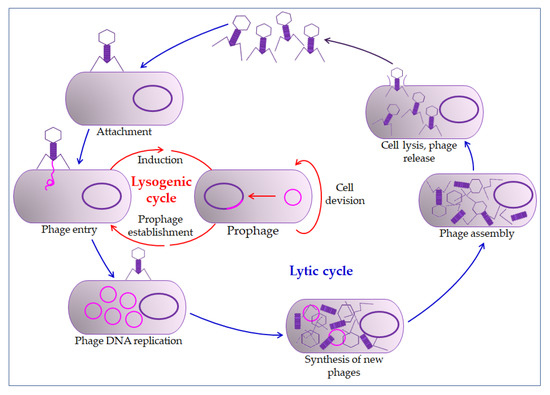
Figure 2.
Furthermore, there are circumstances in which phages stimulate biofilm formation, such as increased bacteriophage pressure in the biofilm, leading to larger aggregates, which could be considered an evolutionary adaptation [33][39]. Thus, such interaction between bacteriophage and the host could be classified as mutualistic rather than parasitic. In so doing, bacteriophages acquire some new properties which are favorable to them, such as encapsulation in the biofilm matrix, in which phages can tolerate higher concentrations of disinfectants, radiation, and other environmental factors [35][33].
All this points to the need for careful preparation before the application of bacteriophages. Due to prophage induction, extracellular DNA accumulates in the biofilm [33][41]. Although they could lead to cell death, prophages are integrated phages in the host’s lysogenic cycle. For example, they may encode virulence factors and antibiotic resistance factors (toxins, enzymes, and superantigens) in cholera [33].
Bacteriophage evolution stimulated by phage and host interaction in the biofilm potentiates and promotes mutations as common properties. It could demonstrate the real potential of bacteriophage therapy to eradicate infectious biofilms [38][42]. Thus, hundreds of years of positive practical experience in applying bacteriophages, their easy isolation, cost-effectiveness, absolute specificity to the host, self-reproduction, and non-disruption of normal microbiocenosis, without harmful side effects, make bacteriophages the choice for the future [26][43].
There are a few more potential benefits of bacteriophages. The ability to “deliver” broad-spectrum antimicrobial drugs to the infection site makes bacteriophages extremely potent in creating even more effective modular antibacterial agents [44]. Another particular interest is the concept of enhancing the phage genome to express antimicrobial peptides (AMPs) [14].
2.2. Antimicrobial Peptides (AMPs)
Biofilm eradication agents (BEAs) are the target of many modern studies, and antimicrobial peptides (AMPs) are among the most likely BEAs [17][45][46][47]. AMPs are ubiquitous compounds produced by plants, invertebrates, and animals [48] and are relatively simple molecules (from 5 to 100 amino acids) with a molecular mass of 1–5 kDa [14][49]. They are predominantly cationic, so they are also called cationic antimicrobial peptides. Their antimicrobial action mechanism is associated mainly with cytoplasmic membrane disorder and inhibition of protein accumulation or enzymatic activity. In eukaryotes, they play an essential role in innate immunity [50][51].
Plants and invertebrates lack adaptive immunity (immunity mediated by B and T cells), so AMPs play a fundamental role in protecting against bacterial and fungal infections [48]. All plant AMPs are rich in cysteine and contain many disulfide bonds. In invertebrates, AMPs are found in hemolymph, hemocytes, phagocytes, and epithelial cells [48][49]. The vertebrate immune system consists of an innate and adaptive immune system. AMPs can be isolated from leukocytes, phagocytes, epithelial macrophages, and body fluids [49][52][53][54]. The most prominent groups of mammalian AMPs are cathelicidins and defensins [50][55].
AMPs are classified according to structure, sequence, or mechanism of action. AMPs may have several activities: bactericidal, immune modulations, antiviral properties, anticarcinogenic properties, and they can prevent biofilm formation. Since AMP activity depends on their structure and sequence, it is crucial to consider both properties when categorizing them [48][49].
2.2. Antimicrobial Peptides (AMPs)
2.2.1. The Mechanism of Action of Antimicrobial Peptides
The mechanism of action of AMPs can be divided into two groups: the direct killing of microorganisms (by membrane targeting or non-membrane targeting) or immune modulation [14][51][56].
The permeabilizing membrane mechanism of action may have receptor- or non-receptor-mediated interactions. Some AMPs, such as nisin, bind with high affinity to lipid molecules in the cell membrane, producing pores in it, and act by covering the entire surface of the membrane, the so-called carpet model (detergent-like model) [22][48][57].
The direct non-membrane targeting mechanisms of action are based on AMPs targeting the bacterial cell wall to inhibit cell wall synthesis. AMPs interact with the diverse precursors needed for cell wall synthesis. For example, AMP defensins bind to the charged pyrophosphate sugar moiety of the lipid molecule [48][58]. While some AMPs can act on the cytoplasmic membrane, others accumulate in the cytoplasm and inhibit proteins and nucleic acid synthesis, thus disrupting enzyme–protein activity (
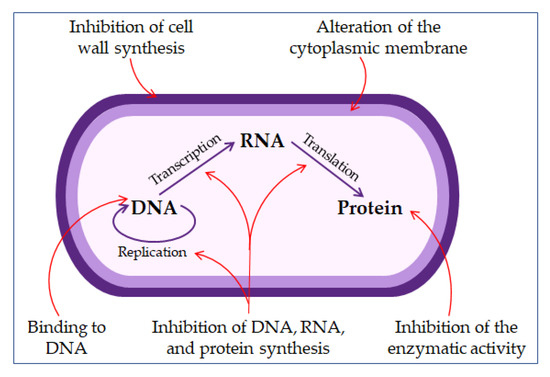
Figure 3.
The mechanisms of action of AMP in immune modulation include various immune responses. Immune cells (neutrophils, macrophages) produce AMPs, which are the first to contact the microbial invasion [60][61]. Likewise, AMPs promote a diversity of immune responses: activation, attraction, and differentiation of leukocytes. Some human AMPs (LL-37, β defensins) can attract immune cells, such as white blood cells, dendritic cells, and mast cells [48][62][63][64]. Some research suggests that AMPs might serve as vaccine adjuvants [
The properties of AMPs can be improved to enhance their delivery by optimizing their stability and toxicity. This is mainly done through polymer conjugation: conjugation with biopolymers such as chitosan and hyaluronic acid. Alternatively, it may be done by encapsulating AMPs in micelles and liposomes [48][65].
Antiviral AMPs can neutralize the virus by integrating into the viral envelope and cell membrane, causing the viral membrane's destabilization or preventing the host’s infection [62]. Antiviral AMPs defensins can bind to viral glycoprotein after which viruses (such as herpes simplex virus) cannot bind to the surface of the host cell [55]. Some AMPs (such as lactoferrin) can occupy specific mammalian cell receptors and prevent the virus from binding to its target receptor (such as heparan molecules for herpes simplex virus) and blocking viral interaction with the receptor [47][49]. Some antiviral AMPs can enter the host cell itself where they are located in the cytoplasm or the organelles and alter the host cell gene expression, thus helping the host defense mechanism [47][66][67].
Thus, AMPs, with all their properties and mechanisms of action (structural, therapeutic), are incredibly suitable molecules in the treatment, especially of infections resistant to many drugs (mainly resistant to antibiotics) [68][69][70][71]. All future research should aim at discovering the improvement of AMPs' intake and their action, and their action combined with other antimicrobial agents (antibiotics, bacteriophages) [72][73][74]. This mainly refers to their biocompatibility action in the immunomodulation system [46][64][70]. It is also necessary to avoid undesirable consequences of AMP administration such as toxicity, hemolytic activity, and changing their structure, primarily of cationic AMPs, to obtain even more efficient and safer AMPs [71].
2.2.2. The Benefit of Combined Therapy of Antimicrobial Peptides and Nanoparticles
New AMP delivery systems are being developed, which could help avoid the problems related to AMP delivery [14][75]. They improve the pharmacokinetics of AMPs [10], increase their half-life, reduce the required dose, and decrease production costs and possible toxicity [10][11][76]. All this may be achieved by encapsulating AMPs in various nanocarriers [14][75]. Nanoparticles significantly increase the penetration of AMPs into cells [77][76].
Several metal nanoparticles, such as silver and gold, have appeared as a possible choice for treating antibiotic-resistant bacterial infections [12]. Silver nanoparticles (AgNPs) are particularly interesting because they have potent antimicrobial activity [12][78]. Both AgNPs and AMPs could replace antibiotics, and the conjugation of AMPs with AgNPs has the additional advantage of the synergistic effect of their antimicrobial properties [79][80][81]. The combination of AMPs and AgNPs might produce new features, such as higher antibacterial activity, increased stability, reduced toxicity, and enhanced selectivity [13][82][83].
Combined therapy using AgNPs and AMPs represents a new approach to developing new antimicrobial drugs [84].
