Gymnema inodorum (GI) is an indigenous medicinal plant and functional food in Thailand
that has recently helped to reduce plasma glucose levels in healthy humans. It is renowned for the
medicinal properties of gymnemic acid and its ability to suppress glucose absorption. However,
the effects of gymnemic acids on adipogenesis that contribute to the accumulation of adipose
tissues associated with obesity remain unknown. The present study aimed to determine the
effects of gymnemic acids derived from GI tea on adipogenesis. We purified and identified GiA-7
and stephanosides C and B from GI tea that inhibited adipocyte differentiation in 3T3-L1 cells.
These compounds also suppressed the expression of peroxisome proliferator-activated receptor gamma
(Pparγ)-dependent genes, indicating that they inhibit lipid accumulation and the early stage of 3T3-L1
preadipocyte differentiation. Only GiA-7 induced the expression of uncoupling protein 1 (Ucp1) and
pparγ coactivator 1 alpha (Pgc1α), suggesting that GiA-7 induces mitochondrial activity and beige-like
adipocytes. This is the first finding of stephanosides C and B in Gymnema inodorum. Our results
suggested that GiA-7 and stephanosides C and B from GI tea could help to prevent obesity.
- Gymnema inodorum
- adipogenesis
- gymnemic acid
- obesity
Note:All the information in this draft can be edited by authors. And the entry will be online only after authors edit and submit it.
Definition:Gymnema inodorum (GI) is an indigenous medicinal plant and functional food in Thailand that has recently helped to reduce plasma glucose levels in healthy humans. It is renowned for the medicinal properties of gymnemic acid and its ability to suppress glucose absorption.
1. Introduction
is a species of the genus Gymnema that is popular in India for reducing glucose levels, suppressing glucose absorption and preventing type 2 diabetes [1][2][3][4][5].
(GI) is a species of same genus that is indigenous to Thailand, particularly in the northern region, where it is widely consumed. The effects of GI on glucose absorption and blood glucose levels have recently been investigated [6][7][8]. We previously found that extracts of GI leaves decreased blood glucose in alloxan-induced diabetic rats [9] that comprise a popular model with which to study type 1 diabetes mellitus. Alloxan selectively destroys insulin production in beta cells, which consequently results in high blood glucose levels [10]. However, about 90% of patients with diabetes have type 2 diabetes mellitus (DM) which is induced by a lack of exercise and inappropriate eating habits [11]. However, obesity is the leading risk factor for type 2 DM, and it also greatly increases the risk of fatty liver disease, atherosclerosis, metabolic diseases, insulin resistance and hypertension [12][13]. Obesity is characterized at the cellular level as being differentiated from preadipocytes. White adipose tissue (WAT) is specialized to store excess energy as triglycerides composed of fatty acids. Inhibiting preadipocyte differentiation can prevent the initiation and progression of obesity [14][15].
The differentiation of 3T3-L1 fibroblast-like cells into adipocyte-like cells stimulated by insulin and synthetic glucocorticoids is a popular model of adipogenesis and lipid metabolism in vitro [16][17]. Therefore, we applied the inhibition of 3T3-L1 cell differentiation to screen gymnemic acid extracted from GI tea. Gymnemic acid is an oleanane-type triterpene glycoside [16][17] that can exist as a single entity or as a mixture of several related compounds [18][19]. The major saponin fraction in
is a gymnemic acid that comprises a complex mixture of at least nine similar glycosides and aglycone derivatives [20]. Moreover, only four gymnemic acids have been identified in GI, which renders the purification and identification of gymnemic acids difficult. Furthermore, current knowledge about these compounds purified from GI is limited. We isolated and purified GiA-7, stephanoside C and stephanoside B from GI tea that inhibited 3T3-L1 cell differentiation. We also determined the expression of the
γ
α
γ
and
genes to explain the signaling of adipogenesis inhibition in 3T3-L1 preadipocytes.
2. Results and Discussion
We measured the ability of the crude 10–100% methanol and ethanol fractions of gymnemic acid to inhibit 3T3-L1 cell differentiation. We found that the 90% methanol fraction was the most powerful inhibitor (
Figure 1). We then found that components 2, 3, 5 and 6 among the six components separated by HPLC from this fraction (
Figure 2) significantly inhibited 3T3-L1 cell differentiation (
Figure 3). Component 5 was the most powerful inhibitor. The inhibition of 3T3-L1 cell differentiation by component 5 was concentration-dependent. Component 6 also strongly inhibited 3T3-L1 cell differentiation. However, the yield of HPLC fraction 3 was very low. Therefore, HPLC fractions No. 2, 5 and 6 were further purified, and their structures were identified by NMR and mass spectrometry.
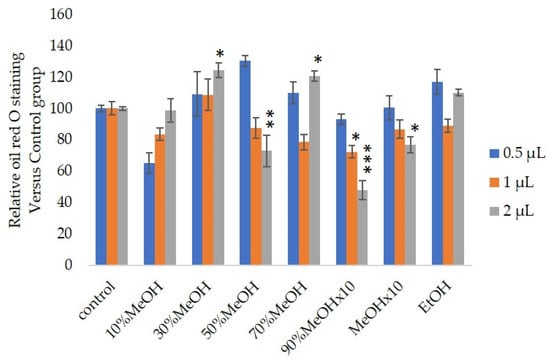
Effects of Sep-PaktC18 fractions on 3T3-L1 cell differentiation. We assessed the abilities of 10%MeOH, 30%MeOH, 50%MeOH, 70%MeOH and EtOH fractions at the concentration of 10 mg/mL in ethanol and 90%MeOH and MeOH fractions at the concentration of 1 mg/mL in ethanol to inhibit 3T3-L1 cell differentiation. Values are shown as means ± SD (
= 4). *
< 0.05, **
< 0.01 and ***
< 0.001 vs. control (ANOVA with post hoc Dunnett tests).
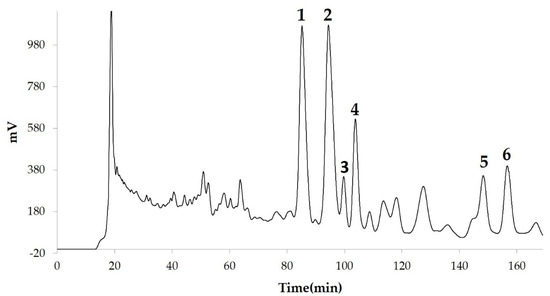
Compounds separated by high-performance liquid chromatography (HPLC) from 90% methanol fraction.
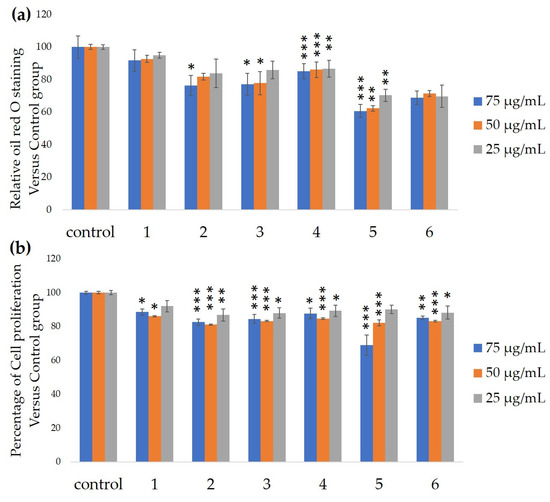
Ability of HPLC fractions to inhibit 3T3-L1 cell differentiation. (
) Inhibition of adipogenesis. (
) Cell proliferation. Values are shown as means ± SD (
= 4). *
< 0.05, **
< 0.01 and ***
< 0.001 vs. control (ANOVA and post hoc Dunnett tests).
We assessed the ability of purified 25, 50 and 100-µM GiA-7, stephanoside C and stephanoside B extracted from GI tea to inhibit 3T3-L1 cell differentiation. After 10 days, intercellular lipid accumulation and viable cells were determined. Each of GiA-7, stephanoside C and stephanoside B at 100 µM reduced intercellular lipid accumulation (
). Stephanoside C was the most effective inhibitor, which is the lowest concentration of significantly inhibited 3T3-L1 cell differentiation. Moreover, the inhibition of 3T3-L1 cell differentiation by GiA-7, stephanoside C and stephanoside B was concentration-dependent.
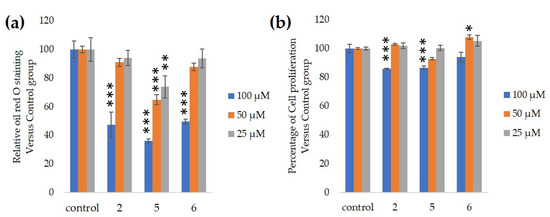
Effects of compounds 2 (GiA-7), 5 (stephanoside C) and 6 (stephanoside B) on 3T3-L1 cell differentiation. (
) Inhibition of adipogenesis. (
) Cell proliferation. Values are shown as means ± SD (
= 4). *
< 0.05, **
< 0.01 and ***
< 0.001 vs. control (ANOVA and post hoc Dunnett tests).
shows that stephanosides C, B and GiA-7 suppressed
and
gene expressions. These findings suggested that none of these compounds activated TG lipolysis. However,
directly regulates
and
gene expressions in adipocytes in vitro [21][22]. Our results suggested that these compounds downregulated
and
gene expressions by inhibiting
gene expression. Lipin-1 functions in lipid droplet biogenesis during adipocyte differentiation and generates diacylglycerol for lipid synthesis [23]. Lipin-1 is important for the process of TG accumulation during the early stage of adipogenesis. Lipin-1 is a key factor for adipocyte maturation and maintenance by regulating
and
[24].
expression is required to induce the transcription of adipogenic genes, including
and
shows that stephanosides C and B and GiA-7 significantly suppressed
expression. These findings suggest that these compounds inhibited
and
gene expressions by suppressing
gene expression. These observations confirm that GiA-7, stephanoside C and stephanoside B inhibited the early stage of adipogenesis and prevented TG accumulation. The transcriptional cofactor, Pgc1α, is important for mitochondrial biogenesis. The regulation of
expression enhances mitochondrial biogenesis through
upregulation [27][28]. The present study found that only GiA-7 induced
and
. Srebp-1c is also a key regulator of adipocytes and is involved in lipid metabolism [29][30]. These findings suggest that GiA-7 regulates mitochondrial biogenesis through the Srebp-1c-dependent upregulation of Pgc1α. GiA-7 also inhibits lipid accumulation in 3T3-L1 preadipocytes by downregulating adipogenic transcription factors and genes associated with lipid accumulation. Both
and
are brown/beige cell-specific genes. Only GiA-7 induced the expression of
and
. Beige adipocytes express low basal levels of
, whereas brown adipocytes constitutively express
. These findings suggest that GiA-7 inhibits the differentiation of white adipocytes and, also, induces beige-like adipocytes in 3T3-L1 mouse preadipocytes. Comprehensive profiles of gene expressions indicate that the characteristics of human brown and mouse beige adipocytes are compatible [29][30]. The activation of human brown adipocytes was recently examined as a possible novel therapeutic treatment for obesity [31]. Thus, GiA-7 might serve as a novel treatment for obesity in humans by inducing brown adipocytes.
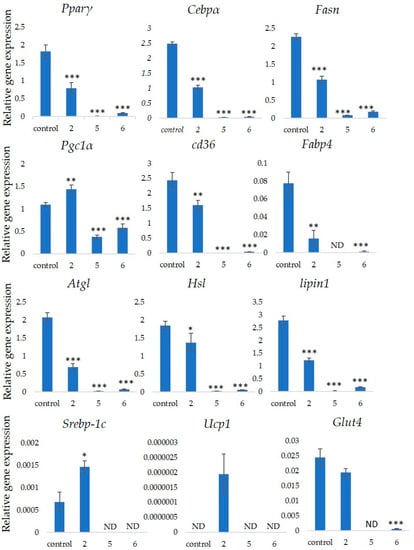
Effects of stephanosides C and B and GiA-7 extracted from
(GI) tea on gene expressions at the initial stage of 3T3-L1 cell differentiation into adipocytes. The differentiation of 3T3-L1 cells was induced, and the cells were incubated with 100 µM GiA-7, stephanoside C and stephanoside B for eight days; then, the gene expressions were measured. Values are shown as means ± SD (
= 4). *
< 0.05, **
< 0.01 and ***
< 0.001 vs. control (ANOVA with post hoc Dunnett tests. ND, not detected).
3. Discussions and Conclusions
tea has been widely applied in Thailand to control high blood glucose. Here, we screened the ability of gymnemic acids extracted from GI tea to inhibit 3T3-L1 cell differentiation into adipocytes. We isolated and purified GiA-7, stephanoside C and stephanoside B from GI tea using column chromatography and C18 HPLC, respectively, then confirmed them using NMR and mass spectrometry. All three compounds inhibited 3T3-L1 cell differentiation into adipocytes. Moreover, we determined that these compounds inhibited the early stage of adipogenesis by suppressing the
α
and
genes that are associated with adipogenesis. However, only GiA-7 induced
and
, suggesting that GiA-7 enhances mitochondrial activity and beige-like adipocytes among 3T3-L1 preadipocytes. Our findings suggest that the GiA-7, stephanoside C and stephanoside B from GI tea could help to prevent obesity.
References
- Shanmugasundaram, E.; Rajeswari, G.; Baskaran, K.; Kumar, B.R.; Shanmugasundaram, K.R.; Ahmath, B.K. Use of Gymnema sylvestre leaf extract in the control of blood glucose in insulin-dependent diabetes mellitus. J. Ethnopharmacol. 1990, 30, 281–294.
- Shimizu, K.; Iino, A.; Nakajima, J.; Tanaka, K.; Nakajyo, S.; Urakawa, N.; Atsuchi, M.; Wada, T.; Yamashita, C. Suppression of glucose absorption by some fractions extracted from Gymnema sylvestre leaves. J. Vet. Med. Sci. 1997, 59, 245–251.
- Al-Romaiyan, A.; King, A.; Persaud, S.; Jones, P. A novel extract of Gymnema sylvestre improves glucose tolerance in vivo and stimulates insulin secretion and synthesis in vitro. Phytother. Res. 2013, 27, 1006–1011.
- Paliwal, R.; Kathori, S.; Upadhyay, B. Effect of Gurmar (Gymnema sylvestre) powder intervention on the blood glucose levels among diabetics. Stud. Ethno-Med. 2009, 3, 133–135.
- Kanetkar, P.; Singhal, R.; Kamat, M. Recent advances in indian herbal drug research guest editor: Thomas Paul Asir Devasagayam Gymnema sylvestre: A memoir. J. Clin. Biochem. Nutr. 2007, 41, 77–81.
- Shimizu, K.; Ozeki, M.; Iino, A.; Nakajyo, S.; Urakawa, N.; Atsuchi, M. Structure-activity relationships of triterpenoid derivatives extracted from Gymnema inodorum leaves on glucose absorption. Jpn. J. Pharmacol. 2001, 86, 223–229.
- Chiabchalard, A.; Tencomnao, T.; Santiyanont, R. Effect of Gymnema inodorum on postprandial peak plasma glucose levels in healthy human. Afr. J. Biotechnol. 2010, 9, 1079–1085.
- Shimizu, K.; Ozeki, M.; Tanaka, K.; Itoh, K.; Nakajyo, S.; Urakawa, N.; Atsuchi, M. Suppression of glucose absorption by extracts from the leaves of Gymnema inodorum. J. Vet. Med. Sci. 1997, 59, 753–757.
- Klungsupya, P.; Muangman, T.; Theangtrong, N.; Khayungarnnawee, A.; Phatvej, W.; Thisayakorn, K.; Rerk-Am, U.; Sematong, T.; Trangvacharakul, S.; Arunpairojana, V. Antioxidant and antihyperglycemic activities of Gymnema inodorum Dence. In Proceedings of the 8th NRCT-JSPS Joint Seminar Innovative Research in Natural Products for Sustainable Development, Bangkok, Thailand, 3–5 February 2009; pp. 207–209.
- Rohilla, A.; Ali, S. Alloxan induced diabetes: Mechanisms and effects. Int. J. Res. Pharm. Biomed. Sci. 2012, 3, 819–823.
- Risérus, U.; Willett, W.C.; Hu, F.B. Dietary fats and prevention of type 2 diabetes. Prog. Lipid Res. 2009, 48, 44–51.
- Zeyda, M.; Stulnig, T.M. Obesity, inflammation, and insulin resistance—A mini-review. Gerontology 2009, 55, 379–386.
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867.
- Farmer, S.R. Molecular determinants of brown adipocyte formation and function. Genes Dev. 2008, 22, 1269–1275.
- Rosen, E.D.; MacDougald, O.A. Adipocyte differentiation from the inside out. Nat. Rev. Mol. Cell Biol. 2006, 7, 885–896.
- Green, H.; Meuth, M. An established pre-adipose cell line and its differentiation in culture. Cell 1974, 3, 127–133.
- Rubin, C.S.; Hirsch, A.; Fung, C.; Rosen, O.M. Development of hormone receptors and hormonal responsiveness in vitro. Insulin receptors and insulin sensitivity in the preadipocyte and adipocyte forms of 3T3-L1 cells. J. Biol. Chem. 1978, 253, 7570–7578.
- Taira, J.; Ogi, T. Induction of Antioxidant Protein HO-1 Through Nrf2-ARE Signaling Due to Pteryxin in Peucedanum Japonicum Thunb in RAW264. 7 Macrophage Cells. Antioxidants 2019, 8, 621.
- Oishi, K.; Ohyama, S.; Higo-Yamamoto, S. Chronic sleep disorder induced by psychophysiological stress induces glucose intolerance without adipose inflammation in mice. Biochem. Biophys. Res. Commun. 2018, 495, 2616–2621.
- Kanda, Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transpl. 2013, 48, 452–458.
- Deng, T.; Shan, S.; Li, P.P.; Shen, Z.F.; Lu, X.P.; Cheng, J.; Ning, Z.Q. Peroxisome proliferator-activated receptor-gamma transcriptionally up-regulates hormone-sensitive lipase via the involvement of specificity protein-1. Endocrinology 2006, 147, 875–884.
- Kershaw, E.E.; Schupp, M.; Guan, H.-P.; Gardner, N.P.; Lazar, M.A.; Flier, J.S. PPARγ regulates adipose triglyceride lipase in adipocytes in vitro and in vivo. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E1736–E1745.
- Sembongi, H.; Miranda, M.; Han, G.S.; Fakas, S.; Grimsey, N.; Vendrell, J.; Carman, G.M.; Siniossoglou, S. Distinct roles of the phosphatidate phosphatases lipin 1 and 2 during adipogenesis and lipid droplet biogenesis in 3T3-L1 cells. J. Biol. Chem. 2013, 288, 34502–34513.
- Kim, H.E.; Bae, E.; Jeong, D.-Y.; Kim, M.-J.; Jin, W.-J.; Park, S.-W.; Han, G.-S.; Carman, G.M.; Koh, E.; Kim, K.-S. Lipin1 regulates PPARγ transcriptional activity. Biochem. J. 2013, 453, 49–60.
- Zhang, P.; Takeuchi, K.; Csaki, L.S.; Reue, K. Lipin-1 phosphatidic phosphatase activity modulates phosphatidate levels to promote peroxisome proliferator-activated receptor gamma (PPARgamma) gene expression during adipogenesis. J. Biol. Chem. 2012, 287, 3485–3494.
- Phan, J.; Peterfy, M.; Reue, K. Lipin expression preceding peroxisome proliferator-activated receptor-gamma is critical for adipogenesis in vivo and in vitro. J. Biol. Chem. 2004, 279, 29558–29564.
- Puigserver, P.; Spiegelman, B.M. Peroxisome proliferator-activated receptor-γ coactivator 1α (PGC-1α): Transcriptional coactivator and metabolic regulator. Endocr. Rev. 2003, 24, 78–90.
- Kobayashi, M.; Fujii, N.; Narita, T.; Higami, Y. SREBP-1c-Dependent Metabolic Remodeling of White Adipose Tissue by Caloric Restriction. Int. J. Mol. Sci. 2018, 19, 3335.
- Sharp, L.Z.; Shinoda, K.; Ohno, H.; Scheel, D.W.; Tomoda, E.; Ruiz, L.; Hu, H.; Wang, L.; Pavlova, Z.; Gilsanz, V. Human BAT possesses molecular signatures that resemble beige/brite cells. PLoS ONE 2012, 7, e49452.
- Wu, J.; Bostrom, P.; Sparks, L.M.; Ye, L.; Choi, J.H.; Giang, A.H.; Khandekar, M.; Virtanen, K.A.; Nuutila, P.; Schaart, G.; et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012, 150, 366–376.
- Cannon, B.; Nedergaard, J. Neither brown nor white. Nature 2012, 488, 286–287.
