Caffeine is a natural trimethyl xanthine alkaloid in which the three methyl groups are located at positions 1, 3, and 7 (1,3,7-Trimethylxanthine). Caffeine has high oral bioavailability, with 99% of caffeine being absorbed from the gastrointestinal (GI) tract into the bloodstream 45 min after ingestion. A peak plasma concentration of 1–10 μM (0.25–2 mg/L) reached 15–120 min post oral ingestion from a cup of coffee in humans. Caffeine, a key psychoactive ingredient in coffee, is a short-acting neurostimulator with known neuromodulator effects on the brain by inhibiting phosphodiesterase, mobilizing intracellular calcium, antagonism of adenosine receptors, and modulation of GABA receptor function. Rodent studies have also reported caffeine can inhibit amylogenic-Aβ protein production and improve cognition in rodent AD models. However, results from previous clinical studies were controversial, with some reporting caffeine to be neuroprotective, while others report no effect or even detrimental effects on cognition.
- caffeine
- coffee
- cognition
- Alzheimer’s disease
- dementia
1. Introduction
2. Effect of Coffee/Caffeine
Studies by Haller et al. [14][15] showed in early cognitive decline, there is an increase in compensatory basal activity diffused through the posto-temporal region of the brain, which increases the brain’s sensitivity to the neuroprotective action of caffeine [14][15]. Furthermore, MRI studies by Ritchie et al. [16] and Haller et al. [17] showed that caffeine reduces the amount of white matter lesion/cranial volume in cognitively stable elders, contributing to cognitive decline in Dementia/AD [17][18]. Ritchie et al. [18] also showed increased cerebral perfusion in chronic coffee consumers, indicating a possible neuroprotective mechanism of coffee [18]. Moreover, Gelber et al. [19] found high caffeine levels were associated with a lower-odds of having any brain lesion types at autopsy [19]. However, an epidemiological study by Kim et al. [20] did not find any association between coffee intake and hypometabolism, atrophy of AD signature, and WMH volume; instead, it found that coffee exerted a neuroprotective effect by reducing the levels of Aβ [20].
Current literature also seems to support the notion that caffeine/coffee acts as a cognitive normalizer instead of a cognitive enhancer, and as such healthy adults or those with deteriorating cognition receive little benefit from coffee/caffeine treatment [14][15][17][21]. West et al., found that elderly participants with mild cognitive decline showed higher sensitivity to caffeine than healthy younger diabetics [21]. Furthermore, Haller et al., found no changes in neural activation among healthy adults but increased posto-temporal activation in those with MCI, further supporting the notion that caffeine does not act as a cognitive enhancer [15]. Other than that, caffeine/coffee cannot significantly enhance the cognitive function of those suffering from severe cognitive decline [14][17]. Haller et al., showed that caffeine reduces the amount of white matter lesion/cranial volume and increases cerebral perfusion in cognitively stable elders but did not extend the same benefits to elders with deteriorating cognition [17]. Furthermore, Haler et al., in another study used fMRI to study the neural activation induced by caffeine for participants with deteriorating cognition; although this research showed that caffeine reduced cognitive decline in dCON, it did not show the same level of caffeine-induced neural activation in them as seen in those with sCON [14]. The literature also shows that the neuroprotective effect of caffeine/coffee can be confounded by gender, but the evidence is not definitive for either gender, and further research is needed [18][22][23][24]. Two studies (Ritchie et al. [18][23]) only found a statistically significant neuroprotective effect of caffeine among women in the research population but not males [18][23]. Furthermore, Sugiyama et al. [22] found an overall neuroprotective effect of coffee, but this was enhanced among the female cohort and non-smokers and non-drinkers [22]. However, Iranpour et al., in a crude model, found the neuroprotective effect of caffeine extended to both genders, but after adjusting for confounding, found a weak positive correlation for the neuroprotective effect of caffeine only among the males only [24]. The exact mechanism for the difference in neuroprotective properties of caffeine/coffee between gender is unclear and warrants further research; however, it has been hypothesized that this may be due to differences in caffeine metabolism, pharmacodynamics, or hormonal influence [23][24]. Although there is evidence from the clinical studies suggesting that caffeine consumption is protective against AD cognitive decline, further clinical studies are required to prove this link. Ideally, to examine this link, there would need to be an epidemiological study with a large sample size, with multiple surveys collecting extensive data on confounding variables to be adjusted, including data on CYP genotype. The research should also use biological markers (blood tests) to assess caffeine to reduce recall bias, and caffeine data should be collected at multiple points (incl: midlife) during extended follow-up to assess changes in behavior. Furthermore, cognition should also be measured using verified methods, i.e., MMSE, CDR, CERAD, during follow-up. A sub-group should also be randomly chosen and subjected to brain imaging at set intervals during follow-up to identify changes in neural architecture before shifting in behavior. This subgroup should also be subjected to post-mortem analysis to confirm the presence and stage of AD (Figure 1).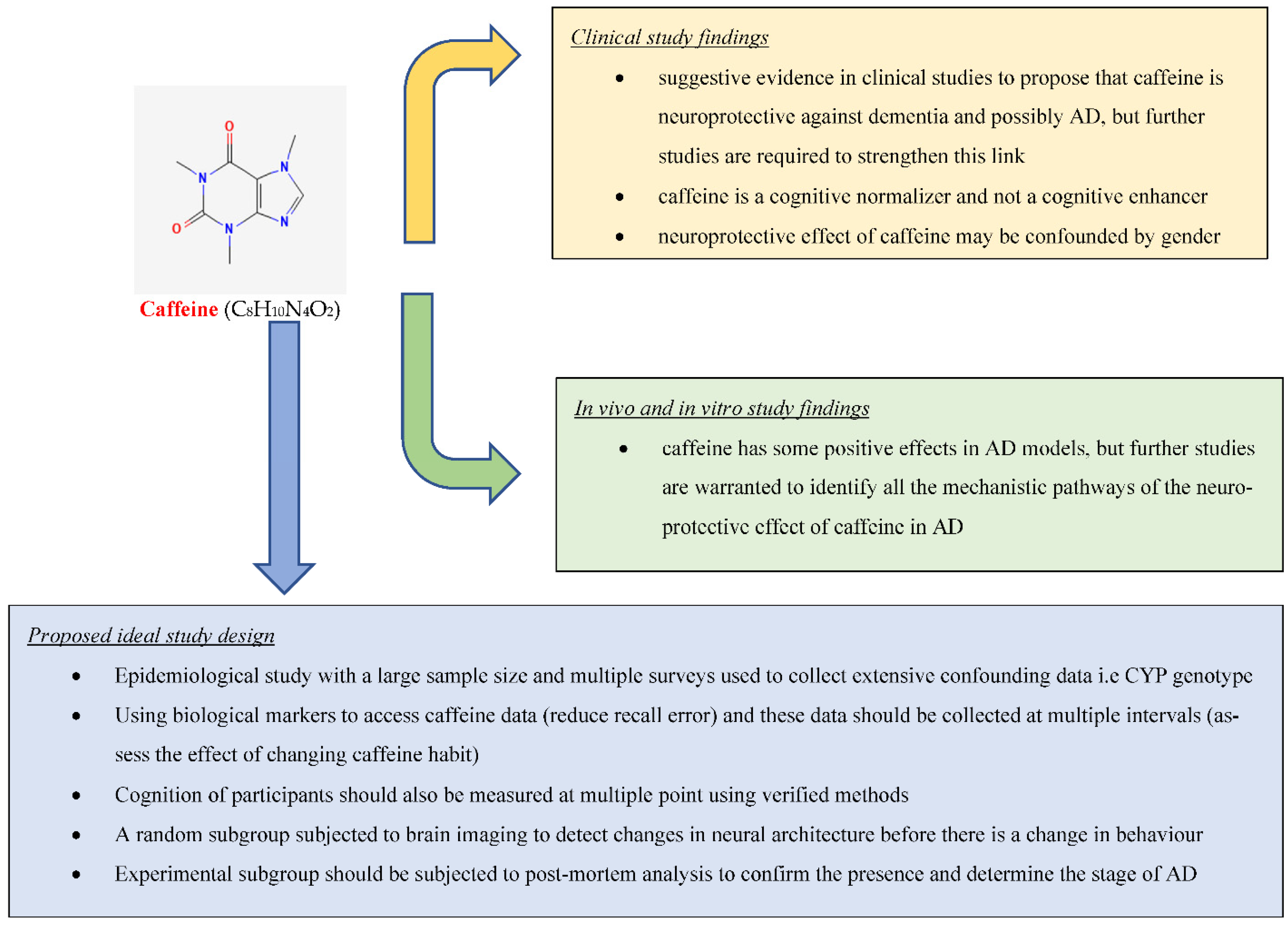
3. Conclusions
This research found suggestive evidence in clinical studies to propose that caffeine is neuroprotective against dementia and possibly AD, but further studies are required to strengthen this link (Figure 1). It also found strong evidence based on in vivo and in vitro studies that caffeine has some positive effects in AD models, but further studies are warranted to identify all the mechanistic pathways of the neuroprotective effect of caffeine in AD.References
- Scheltens, P.; Blennow, K.; Breteler, M.M.B.; de Strooper, B.; Frisoni, G.B.; Salloway, S.; Van der Flier, W.M. Alzheimer’s disease. Lancet 2016, 388, 505–517.
- Prince, M.J.; Wimo, A.; Guerchet, M.M.; Ali, G.C.; Wu, Y.-T.; Prina, M. World Alzheimer Report 2015—The Global Impact of Dementia: An Analysis of Prevalence, Incidence, Cost and Trends; Alzheimer’s Disease International: London, UK, 2015.
- Karran, E.; Mercken, M.; Strooper, B.D. The amyloid cascade hypothesis for Alzheimer’s disease: An appraisal for the development of therapeutics. Nat. Rev. Drug Discov. 2011, 10, 698–712.
- Karch, C.M.; Goate, A.M. Alzheimer’s Disease Risk Genes and Mechanisms of Disease Pathogenesis. Biol. Psychiatry 2015, 77, 43–51.
- Arnsten, A.F.T.; Datta, D.; Del Tredici, K.; Braak, H. Hypothesis: Tau pathology is an initiating factor in sporadic Alzheimer’s disease. Alzheimer’s Dement. 2021, 17, 115–124.
- Genin, E.; Hannequin, D.; Wallon, D.; Sleegers, K.; Hiltunen, M.; Combarros, O.; Bullido, M.J.; Engelborghs, S.; De Deyn, P.; Berr, C.; et al. APOE and Alzheimer disease: A major gene with semi-dominant inheritance. Mol. Psychiatry 2011, 16, 903–907.
- Norton, S.; Matthews, F.E.; Barnes, D.E.; Yaffe, K.; Brayne, C. Potential for primary prevention of Alzheimer’s disease: An analysis of population-based data. Lancet Neurol. 2014, 13, 788–794.
- De Bruijn, R.F.; Bos, M.J.; Portegies, M.L.; Hofman, A.; Franco, O.H.; Koudstaal, P.J.; Ikram, M.A. The potential for prevention of dementia across two decades: The prospective, population-based Rotterdam Study. BMC Med. 2015, 13, 132.
- Panza, F.; Solfrizzi, V.; Barulli, M.R.; Bonfiglio, C.; Guerra, V.; Osella, A.; Seripa, D.; Sabba, C.; Pilotto, A.; Logroscino, G. Coffee, tea, and caffeine consumption and prevention of late-life cognitive decline and dementia: A systematic review. J. Nutr. Health Aging 2015, 19, 313–328.
- Kim, J.; Lee, K.W. Coffee and Its Active Compounds Are Neuroprotective; Elsevier: Amsterdam, The Netherlands, 2015; pp. 423–427.
- Singh, S.S.; Rai, S.N.; Birla, H.; Zahra, W.; Kumar, G.; Gedda, M.R.; Tiwari, N.; Patnaik, R.; Singh, R.K.; Singh, S.P. Effect of Chlorogenic Acid Supplementation in MPTP-Intoxicated Mouse. Front. Pharm. 2018, 9, 757.
- Balakrishnan, R.; Azam, S.; Cho, D.-Y.; Su-Kim, I.; Choi, D.-K. Natural Phytochemicals as Novel Therapeutic Strategies to Prevent and Treat Parkinson’s Disease: Current Knowledge and Future Perspectives. Oxidative Med. Cell. Longev. 2021, 2021, 6680935.
- Fahanik-Babaei, J.; Baluchnejadmojarad, T.; Nikbakht, F.; Roghani, M. Trigonelline protects hippocampus against intracerebral Aβ(1–40) as a model of Alzheimer’s disease in the rat: Insights into underlying mechanisms. Metab. Brain Dis. 2019, 34, 191–201.
- Haller, S.; Montandon, M.-L.; Rodriguez, C.; Moser, D.; Toma, S.; Hofmeister, J.; Giannakopoulos, P. Caffeine impact on working memory-related network activation patterns in early stages of cognitive decline. Neuroradiology 2017, 59, 387–395.
- Haller, S.; Montandon, M.-L.; Rodriguez, C.; Moser, D.; Toma, S.; Hofmeister, J.; Sinanaj, I.; Lovblad, K.-O.; Giannakopoulos, P. Acute Caffeine Administration Effect on Brain Activation Patterns in Mild Cognitive Impairment. J. Alzheimers Dis. 2014, 41, 101–112.
- Ritchie, K.; Artero, S.; Portet, F.; Brickman, A.; Muraskin, J.; Beanino, E.; Ancelin, M.-L.; Carriere, I. Caffeine, Cognitive Functioning, and White Matter Lesions in the Elderly: Establishing Causality from Epidemiological Evidence. J. Alzheimers Dis. 2010, 20, S161–S166.
- Haller, S.; Montandon, M.-L.; Rodriguez, C.; Herrmann, F.R.; Giannakopoulos, P. Impact of Coffee, Wine, and Chocolate Consumption on Cognitive Outcome and MRI Parameters in Old Age. Nutrients 2018, 10, 1391.
- Ritchie, K.; Ancelin, M.L.; Amieva, H.; Rouaud, O.; Carriere, I. The association between caffeine and cognitive decline: Examining alternative causal hypotheses. Int. Psychogeriatr. 2014, 26, 581–590.
- Gelber, R.P.; Petrovitch, H.; Masaki, K.H.; Ross, G.W.; White, L.R. Coffee Intake in Midlife and Risk of Dementia and its Neuropathologic Correlates. J. Alzheimers Dis. 2011, 23, 607–615.
- Kim, J.W.; Byun, M.S.; Yi, D.; Lee, J.H.; Jeon, S.Y.; Jung, G.; Lee, H.N.; Sohn, B.K.; Lee, J.-Y.; Kim, Y.K.; et al. Coffee intake and decreased amyloid pathology in human brain. Transl. Psychiatry 2019, 9, 27.
- West, R.K.; Ravona-Springer, R.; Livny, A.; Heymann, A.; Shahar, D.; Leroith, D.; Preiss, R.; Zukran, R.; Silverman, J.M.; Schnaider, M. Age Modulates the Association of Caffeine Intake with Cognition and With Gray Matter in Elderly Diabetics. J. Gerontol. Ser. A-Biol. Sci. Med. Sci. 2019, 74, 683–688.
- Sugiyama, K.; Tomata, Y.; Kaiho, Y.; Honkura, K.; Sugawara, Y.; Tsuji, I. Association between Coffee Consumption and Incident Risk of Disabling Dementia in Elderly Japanese: The Ohsaki Cohort 2006 Study. J. Alzheimers Dis. 2016, 50, 491–500.
- Ritchie, K.; Carriere, I.; de Mendonca, A.; Portet, F.; Dartigues, J.F.; Rouaud, O.; Barberger-Gateau, P.; Ancelin, M.L. The neuroprotective effects of caffeine—A prospective population study (the Three City Study). Neurology 2007, 69, 536–545.
- Iranpour, S.; Saadati, H.M.; Koohi, F.; Sabour, S. Association between caffeine intake and cognitive function in adults; effect modification by sex: Data from National Health and Nutrition Examination Survey (NHANES) 2013–2014. Clin. Nutr. 2020, 39, 2158–2168.
- Janitschke, D.; Nelke, C.; Lauer, A.A.; Regner, L.; Winkler, J.; Thiel, A.; Grimm, H.S.; Hartmann, T.; Grimm, M.O.W. Effect of Caffeine and Other Methylxanthines on A beta-Homeostasis in SH-SY5Y Cells. Biomolecules 2019, 9, 689.
- Arendash, G.W.; Schleif, W.; Rezai-Zadeh, K.; Jackson, E.K.; Zacharia, L.C.; Cracchiolo, J.R.; Shippy, D.; Tan, J. Caffeine protects Alzheimer’s mice against cognitive impairment and reduces brain β-amyloid production. Neuroscience 2006, 142, 941–952.
- Arendash, G.W.; Mori, T.; Cao, C.; Mamcarz, M.; Runfeldt, M.; Dickson, A.; Rezai-Zadeh, K.; Tane, J.; Citron, B.A.; Lin, X.; et al. Caffeine reverses cognitive impairment and decreases brain amyloid-beta levels in aged Alzheimer’s disease mice. J. Alzheimers Dis. 2009, 17, 661–680.
- Cao, C.; Cirrito, J.R.; Lin, X.; Wang, L.; Verges, D.K.; Dickson, A.; Mamcarz, M.; Zhang, C.; Mori, T.; Arendash, G.W.; et al. Caffeine suppresses amyloid-beta levels in plasma and brain of Alzheimer’s disease transgenic mice. J. Alzheimers Dis. 2009, 17, 681–697.
- Zhao, Z.A.; Zhao, Y.; Ning, Y.L.; Yang, N.; Peng, Y.; Li, P.; Chen, X.Y.; Liu, D.; Wang, H.; Chen, X.; et al. Adenosine A(2A) receptor inactivation alleviates early-onset cognitive dysfunction after traumatic brain injury involving an inhibition of tau hyperphosphorylation. Transl. Psychiatry 2017, 7, e1123.
- Bortolotto, J.W.; de Melo, G.M.; Cognato, G.d.P.; Moreira Vianna, M.R.; Bonan, C.D. Modulation of adenosine signaling prevents scopolamine-induced cognitive impairment in zebrafish. Neurobiol. Learn. Mem. 2015, 118, 113–119.
- Li, S.; Geiger, N.H.; Soliman, M.L.; Hui, L.; Geiger, J.D.; Chen, X. Caffeine, Through Adenosine A(3) Receptor-Mediated Actions, Suppresses Amyloid-beta Protein Precursor Internalization and Amyloid-beta Generation. J. Alzheimers Dis. 2015, 47, 73–83.
- Espinosa, J.; Rocha, A.; Nunes, F.; Costa, M.S.; Schein, V.; Kazlauckas, V.; Kalinine, E.; Souza, D.O.; Cunha, R.A.; Porciuncula, L.O. Caffeine Consumption Prevents Memory Impairment, Neuronal Damage, and Adenosine A(2A) Receptors Upregulation in the Hippocampus of a Rat Model of Sporadic Dementia. J. Alzheimers Dis. 2013, 34, 509–518.
- Dall’Igna, O.P.; Fett, P.; Gomes, M.W.; Souza, D.O.; Cunha, R.A.; Lara, D.R. Caffeine and adenosine A(2a) receptor antagonists prevent beta-amyloid (25-35)-induced cognitive deficits in mice. Exp. Neurol. 2007, 203, 241–245.
- Mancini, R.S.; Wang, Y.; Weaves, D.F. Phenylindanes in Brewed Coffee Inhibit Amyloid-Beta and Tau Aggregation. Front. Neurosci. 2018, 12, 735.
- Laurent, C.; Eddarkaoui, S.; Derisbourg, M.; Leboucher, A.; Demeyer, D.; Carrier, S.; Schneider, M.; Hamdane, M.; Müller, C.E.; Buée, L.; et al. Beneficial effects of caffeine in a transgenic model of Alzheimer’s disease-like tau pathology. Neurobiol. Aging 2014, 35, 2079–2090.
- Moy, G.A.; McNay, E.C. Caffeine prevents weight gain and cognitive impairment caused by a high-fat diet while elevating hippocampal BDNF. Physiol. Behav. 2013, 109, 69–74.
- Han, K.; Jia, N.; Li, J.; Yang, L.; Min, L.-Q. Chronic caffeine treatment reverses memory impairment and the expression of brain BNDF and TrkB in the PS1/APP double transgenic mouse model of Alzheimer’s disease. Mol. Med. Rep. 2013, 8, 737–740.
- Mohamed, T.; Osman, W.; Tin, G.; Rao, P.P.N. Selective inhibition of human acetylcholinesterase by xanthine derivatives: In vitro inhibition and molecular modeling investigations. Bioorganic Med. Chem. Lett. 2013, 23, 4336–4341.
- Pohanka, M.; Dobes, P. Caffeine Inhibits Acetylcholinesterase, But Not Butyrylcholinesterase. Int. J. Mol. Sci. 2013, 14, 9873–9882.
- Gastaldo, I.P.; Himbert, S.; Ram, U.; Rheinstadter, M.C. The Effects of Resveratrol, Caffeine, beta-Carotene, and Epigallocatechin Gallate (EGCG) on Amyloid-beta(25-35) Aggregation in Synthetic Brain Membranes. Mol. Nutr. Food Res. 2020, 64, 2000632.
- Zappettini, S.; Faivre, E.; Ghestem, A.; Carrier, S.; Buee, L.; Blum, D.; Esclapez, M.; Bernard, C. Caffeine Consumption during Pregnancy Accelerates the Development of Cognitive Deficits in Offspring in a Model of Tauopathy. Front. Cell. Neurosci. 2019, 13, 438.
- Soliman, M.L.; Geiger, J.D.; Chen, X. Caffeine Blocks HIV-1 Tat-Induced Amyloid Beta Production and Tau Phosphorylation. J. Neuroimmune Pharmacol. 2017, 12, 163–170.
- Cao, C.; Wang, L.; Lin, X.; Mamcarz, M.; Zhang, C.; Bai, G.; Nong, J.; Sussman, S.; Arendash, G. Caffeine synergizes with another coffee component to increase plasmato cognitive benefits in Alzheimer’s mice. J. Alzheimers Dis. 2011, 25, 323–335.
- Qosa, H.; Abuznait, A.H.; Hill, R.A.; Kaddoumi, A. Enhanced Brain Amyloid-β Clearance by Rifampicin and Caffeine as a Possible Protective Mechanism Against Alzheimer’s Disease. J. Alzheimers Dis. 2012, 31, 151–165.
- Reznikov, L.R.; Pasumarthi, R.K.; Fadel, J.R. Caffeine elicits c-Fos expression in horizontal diagonal band cholinergic neurons. Neuroreport 2009, 20, 1609–1612.
- Laurent, C.; Burnouf, S.; Ferry, B.; Batalha, V.L.; Coelho, J.E.; Baqi, Y.; Malik, E.; Mariciniak, E.; Parrot, S.; Van der Jeugd, A.; et al. A(2A) adenosine receptor deletion is protective in a mouse model of Tauopathy. Mol. Psychiatry 2016, 21, 97–107.
