Chitosan is obtained from chitin that in turn is recovered from marine crustacean wastes. The recovery methods and their varying types and the advantages of the recovery methods are briefly discussed. TChe bioactive properties of chitosan, which emphasize the unequivocal deliverables contained by this biopolymer, have bitin is the major component of cuticles of insects (cockroach, beetle, true fly, and worm), fungal cell walls (Aspergillus niger, Mucor rouxii, Penicillum notatum, yeenast) concisely presentedand green algae. The variations of chitosan and its derivatives and their unique properties are discussed. The antioxidant propertirecovery methods and their varying types and the advantages of chitosan have been presented and the need for more work targeted towards harnessing the antioxidant property of chitosan has been emphasizthe recovery methods are briefly discussed.
- chitosan
- chitin
- crustacean shells
- waste
- antioxidant
- derivatives
- applications
- nanotechnology
1. Introduction
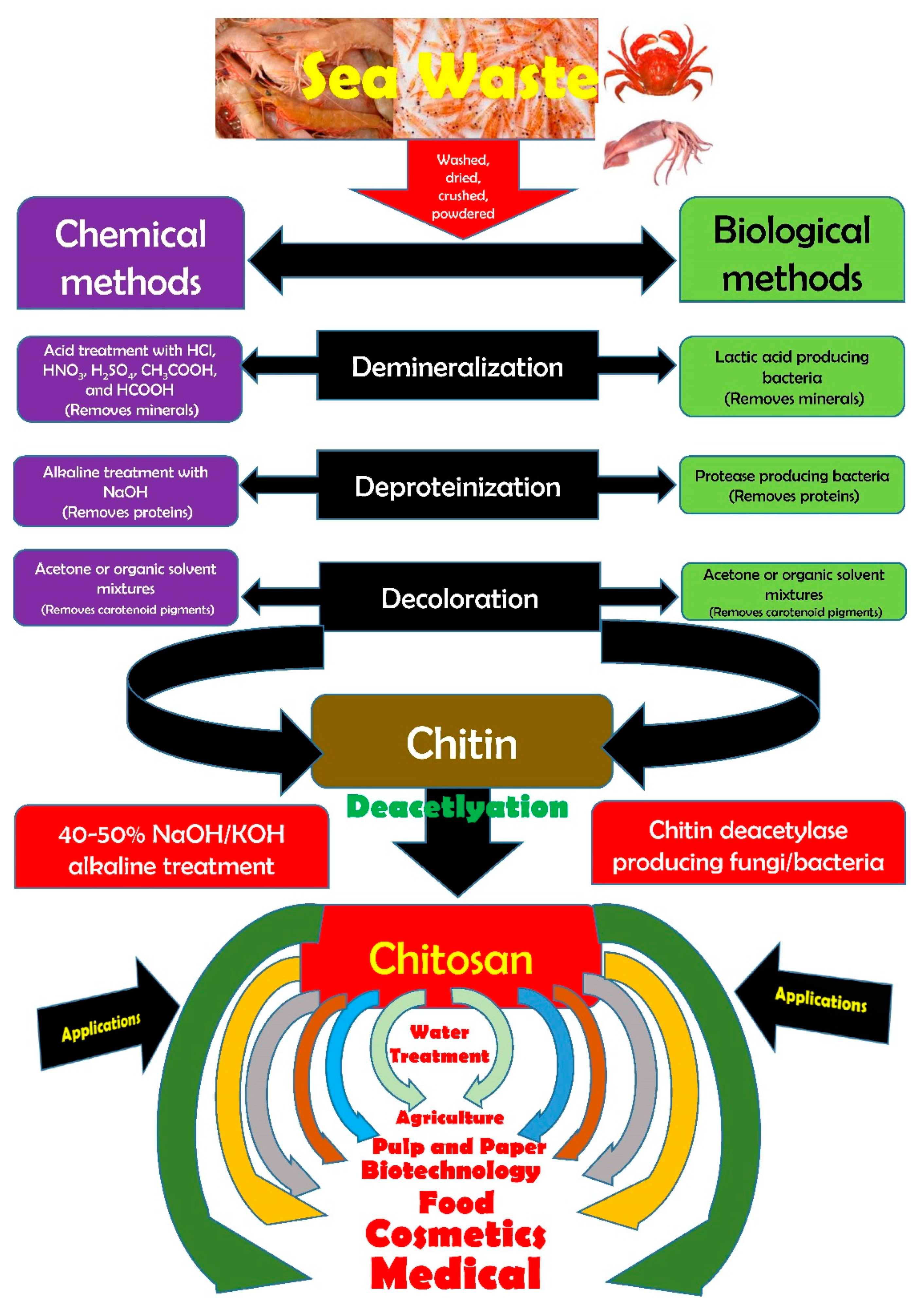
2. Chitin–Chitosan Metamorphosis
The seafood processing industry produces large quantities of byproducts and discards such as heads, tails, skins, scales, viscera, backbones, and shells of marine organisms. Although these are waste residues, they still are an excellent source of lipids, proteins, pigments, and small molecules, and moreso a source of chitinous materials. One of the limitations in the use of chitin on a large-scale is its water insolubility, this is why water-soluble derivatives have been sought after. Chitosan is the most important of these. Chitosan is obtained from chitin by a process called deacetylation, whereby acetyl groups (CH3-CO) are removed resulting in a molecule that is soluble in most diluted acids [27]. The deacetylation process releases amine groups (-NH2) rendering chitosan a cationic nature. Chitosan, a linear polysaccharide is made up of β-(1–4)-linked d-glucosamine and N-acetyl-d-glucosamine moieties [28,29,30,31,32,33,34][28][29][30][31][32][33][34]. Chitosan is derived from chitin by chemical or enzymatic deacetylations. Although chemical deacetylation is preferentially cheaper and warrants mass production, the major disadvantage is the energy consumption and high environmental pollution risks. Alternatives in the form of enzymatic methods that utilize chitin deacetylases have been explored via enzymatic deacetylation of chitin. Research has identified that selected fungal, viral, and bacterial chitin deacetylases could produce partially acetylated chitosan tetramers with a defined degree of acetylation and a pattern of acetylation [35]. With the recent progress in extraction methodologies and instrumentation sophistication, chitosan extraction from marine crustaceans has also been achieved outside of chemical extraction through autoclave-based methods [36[36][37][38][39][40][41][42],37,38,39,40,41,42], microwave-based methods [39,43[39][43][44][45][46],44,45,46], ultrasonication-based methods [47,48[47][48][49],49], and Graviola extract combined with magnetic stirring on hot plate [50]. Chitin and chitosan exhibit several biological properties such as anti-cancer [109][51], antioxidant [110][52], antimicrobial [111][53], and anti-coagulant [112][54] properties. In addition, they are used as biomaterials in a wide range of applications: for biomedical purposes such as for artificial skin, bones, and cartilage regeneration [113[55][56],114], for food preservation such as for edible films [115][57], and for pharmaceutical purposes such as for drug delivery [116][58]. Chitin is a versatile, environmentally friendly, modern material [117][59]. Chitin and chitin derivatives have been used in virtually every significant segment of the economy (e.g., water treatment, pulp and paper industry, biomedical devices and therapies, cosmetics, biotechnology, agriculture, food science, and membrane technology) [118][60]. Chitin and chitosan are important bioactive materials, with many highly potent activities such as immune function, hemostasis and wound healing, antioxidant action, antimicrobial activity, and heavy metal and other pollutant removal [119][61]. Therefore, as renewable resources, chitin and its derivatives have a wide range of applications in food and nutrition [120][62], pharmaceutical [121][63], biotechnological [122][64], cosmetic [123][65], packaging [124][66], textile, wastewater treatment [125][67], and agricultural [126][68] industries.3. Snap Shot of the Bioactive Properties of Chitosan
Chitosan has three reactive groups, the primary amine group and the primary and secondary hydroxyl groups at C-2, C-3, and C-6 positions, respectively [127][69]. Among the three groups, the primary amine at the C-2 position is reported to be responsible for the bioactivity of chitosan [128][70]. The chemical modification of chitosan adds unique functional properties for use towards biological and biomedical applications [129,130,131,132,133,134,135,136,137,138,139,140,141,142,143,144,145,146,147,148,149,150,151,152,153,154,155,156,157,158,159,160,161][71][72][73][74][75][76][77][78][79][80][81][82][83][84][85][86][87][88][89][90][91][92][93][94][95][96][97][98][99][100][101][102][103]. The biodegradability, biocompatibility, mucoadhesion, hemostatic, analgesic, adsorption enhancer, antimicrobial, anticholesterolemic, and antioxidant attributes of chitosan are those which make it suitable for biomedical applications. Chitosan has been well established as an alternative to antibiotics, undertaking antimicrobial and antifungal roles. Because of its cationic properties, chitosan is able to lead to membrane-disrupting effects [162,163,164[104][105][106][107],165], which are higher against Gram-positives than Gram-negatives [165][107]. The antibacterial activity of chitosan is influenced by the molecular weight of chitosan and allied physicochemical properties. A number of chitosan derivatives with different modifications have shown improved antibacterial activity; in this way, chitosan micro/nanoparticles display unique physical and chemical features [166][108]. The chitosan nanoparticles penetrate inside the cell, interacting with phosphorus- and sulfur-containing compounds such as DNA and protein, eventually causing damage to the cells [163,164][105][106]. Successful experiments were performed using chitosan and reticulated chitosan microparticles against aquaculture related trouble-makers: Lactococcus garvieae (Gram +), Vibrio parahaemolyticus, and Vibrio alginolyticus (Gram −). These microorganisms are the most predominant bacterial pathogens of mariculture industry and are responsible for crucial economic losses in cultured fish and seafood worldwide [167][109]. The antimicrobial activity of chitin, chitosan, and their derivatives against different groups of microorganisms, such as bacteria, yeast, and fungi, has received considerable attention in recent years [120,168,169][62][110][111]. Traditional chemotherapeutic agents kill actively dividing cells, characteristic of most cancer cells. Cytotoxic drugs continue to play a crucial role in cancer therapy, although side effects such as the destruction of lymphoid and bone marrow cells is inevitable. In this direction, constant efforts to improve cancer therapy-based side effects are sought after. This is why biocompatible anticancer drugs are needed for cancer therapy. The introduction of several groups into chitosan modifies its structure significantly, thereby increasing the biological activity of chitosan. The introduction of sulfates and phenyl groups in carboxymethyl benzylamide dextrans into chitosan, lead to enhanced anticancer activity in breast cancer cells. Sulfated chitosan (SCS) and sulfated benzaldehyde chitosan (SBCS) significantly inhibited cell proliferation, induced apoptosis, and blocked the fibroblast growth factors (FGF)-2-induced phosphorylation of extracellular signal-regulated kinase (ERK) in Middle cranial fossa (MCF)-7 cells [170][112]. Dialkylaminoalkylation and reductive amination followed by quaternization of chitosan could elicit inhibitory effects on the proliferation of tumor cell lines [171][113].4. Antioxidant Activity of Chitosan
Being extracted from crustacean’s exoskeleton and fungi cell walls, chitosan products are biocompatible and biodegradable, and their range of applications include food, wastewater treatment, cell culture, cosmetics, textiles, agrochemicals, and medical devices [30]. Additionally, chitosan also exhibits antioxidant activity [184,185][114][115] and can be used as a replacement for synthetic antioxidants such as butylated hydroxytoluene (BHT), butylated hydroxy-anisole (BHA), propyl gallate, and tert-butylhydroquinone (TBHQ) [186][116]. Reactive oxygen species (ROS) such as H2O2, hydroxyl radicals, and superoxides lead to oxidative stress which is the key behind a wide range of pathologies: cancer [187][117], cardiovascular disease [188[118][119],189], premature aging [190][120], rheumatoid arthritis, and inflammation [191][121]. Chitin, similar to vitamin C, exhibits antioxidant effects [192][122] and can be used as an ingredient for the production of functional foods in order to circumvent age-related and diet-related diseases [193][123]. Due to oxidation of lipids in food, off-flavors and rancidity manifests, this is why BHT and BHA (synthetic antioxidants) are used. BHT and BHA are well known for their potential health hazards [194][124], and hence safe and natural alternatives are being sought [195][125]. The addition of 1% chitosan resulted in 70% decrease in the 2-thiobarbituric acid reactive substance (TBARS) values of frozen meat. Chitosan addition is reported to have lead to chelation of the free iron in heme proteins of meat that are released during processing [196][126]. In seafoods, oxidation of high concentrations of prooxidants such as hemoglobin and metal ions in fish muscles is also reported [197][127]. Antioxidant effect of chitosan is reported [198][128] to be directly proportional to its molecular weight, concentration, and viscosity. Chitosans from crab shell wastes were tested on herring flesh and chitosan with different viscosity were also tested on fish samples. The highest activity was observed with low viscosity chitosan (14 cP) and its action was similar to that of BHA, BHT, and TBHQ. Chitosans are speculated to slowdown lipid oxidation by chelating ferrous ions in fish. This eliminates the prooxidant activity of ferrous ions by preventing their conversion to ferric ion [197][127]. Kim and Thomas [199][129] have also reported identical inferences in Atlantic salmon (Salmo salar). Free radical reaction is connected with several specific human diseases and has gained paramount attention. In the human body, reactive oxygen species (ROS) produced during metabolic process oxidize lipids, proteins, carbohydrates and nucleic material, resulting in oxidative stress [192][122]. The ROS generated, may activate enzymes that eventually manifest as life-threatening disorders such as cancer, aging, cardiovascular diseases wrinkle formation, rheumatoid arthritis, inflammation, hypertension, dyslipidemia, atherosclerosis, myocardial infraction, angina pectoris, heart failure, and neurodegenerative diseases such as Alzheimer’s, Parkinson’s, and amyotrophic lateral sclerosis [200,201,202,203,204][130][131][132][133][134]. The term ROS refers to oxygen-derived free radicals like superoxide, hydroxyl radical, and nitric oxide, and is extended to nonradical oxygen derivatives of high reactivity like singlet oxygen, hydrogen peroxide, peroxynitrite, and hypochlorite [205,206][135][136]. Mitochondria in biological cells are responsible for ROS generation [207][137]. The cellular defense includes enzymes such as catalase, superoxide dismutase, and glutathione peroxidase [200][130]. When excessive ROS are generated, the defense mechanism is unable to respond appropriately and thus oxidative stress manifests. The antioxidant activity of chitosan has gained paramount importance, with chitosan exhibiting confirmed scavenging activity against various radical species. The degree of deactylation (DDA) and molecular weight (MW) of chitosan determine the scavenging capacity of chitosan [208][138] and the chitosan NH2 groups are responsible for free radical scavenging effect. Mahdy Samar et al. confirmed that a high rate of DDA and low MW of chitosan exhibits higher antioxidant activity [44]. Hajji et al. studied chitosan obtained from Tunisian marine shrimp (Penaeus kerathurus) waste (DDA: 88%), crab (Carcinus mediterraneus) shells (DDA: 83%), and cuttlefish (Sepia officinalis) bones (DDA: 95%) [74][139] and tested their antioxidant activities. Cuttlefish with 95% DDA exhibited the highest antioxidant activity. Sun et al. [209][140], reported that chitosan oligomers exhibited stronger scavenging activity with lower MW. Chang et al. [210][141] demonstrated that lower MW chitosan (~2.2 kDa) greatly impacts the scavenging ability. Although the antioxidant activity of chitosan has been proven, yet it is limited by the lack of an H-atom donor, to serve as a good chain-breaking antioxidant [211][142]. The scavenging capacity of free radicals is related to the bond dissociation energy of O–H or N–H and the stability of the formed radicals. Due to the presence of strong intramolecular and intermolecular hydrogen bonds in chitosan molecules, the OH and NH2 groups find it difficult to dissociate and react with hydroxyl radicals [212][143]. This is why various modifications of chitosan molecules by grafting functional groups into the molecular structure have evolved. Modification of chitosan by grafting polyphenols, has been observed to enhance the antioxidant activity. This has resulted owing to the synergetic effects obtained from both chitosan and polyphenols [213][144]. Chito-oligosaccharides (COS) are known to be highly promising compounds for use as natural antioxidants in biological systems [62,214][145][146]. Li et al. 2018 [215][147] have recently investigated the preparation and potential free radical scavenging activity of chitosan derivatives with 1,2,3-triazoles and 1,2,3-triazoliums. Their results indicated that all the chitosan derivatives exhibited higher radical scavenging activity than chitosan and the scavenging ability was further enhanced following the N-methylation of 1,2,3-triazole moieties. Other researchers [216,217][148][149] have also reported the antioxidant activity of quaternary ammonium groups in chitosan derivatives. Antioxidant agents like chitosan play a role in scavenging the free radicals and by inhibiting the oxidative damage caused by free radicals (Figure 2). Antioxidant mechanism of chitosan functions by protecting the host against oxidative stress induced damages via interfering with the oxidation chain reaction. The exact mechanism of free radical scavenging activity of chitosan has still not been established. However, Riaz et al., 2019 [218][150] put forth a plausible theory that the unsteady free radicals may react with the OH group and the amino group at C-2, C-3, and C-6 positions of the pyranose ring to produce a stable macromolecule. This entreviewy calls to attention that this (elucidating the mechanism of antioxidant activity of chitosan) is one of the gray areas worth working on with respect to the antioxidant activity of chitosan.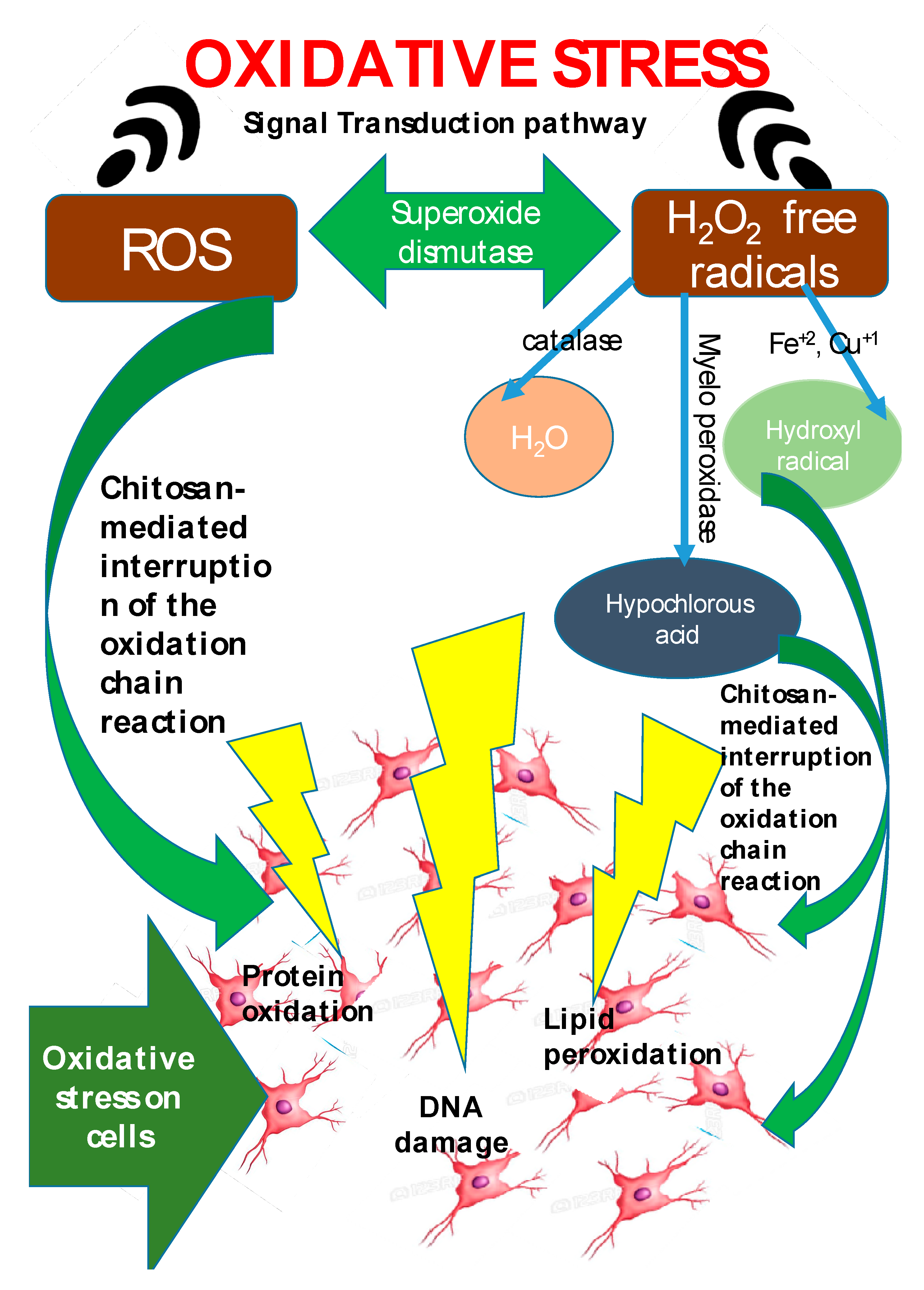
References
- Einbu, A.; Vårum, K.M. Characterization of chitin and its hydrolysis to GlcNAc and GlcN. Biomacromolecules 2008, 9, 1870–1875.
- Sato, H.; Mitzutani, S.; Tsuge, S. Determination of the degree of acetylation of chitin/chitosan by pyrolysis-gas chromatography in the presence of oxalic acid. Anal. Chem. 1998, 70, 7–12.
- Kikkawa, Y.; Tokuhisa, H.; Shingai, H.; Hiraishi, T.; Houjou, H.; Kanesato, M.; Imanaka, T.; Tanaka, T. Interaction force of chitin-binding domains onto chitin surface. Biomacromolecules 2008, 9, 2126–2131.
- Shahidi, F.; Abuzaytoun, R. Chitin, chitosan, and co-products: Chemistry, production, applications, and health effects. Adv. Food Nutr. Res. 2005, 49, 93–135.
- Wang, X.; Xing, B. Importance of structural makeup of biopolymers for organic contaminant sorption. Environ. Sci. Technol. 2007, 41, 3559–3565.
- Zargar, V.; Asghari, M.; Dashti, A. A review on chitin and chitosan polymers: Structure, chemistry, solubility, derivatives, and applications. Chembioeng. Rev. 2015, 2, 204–226.
- Yan, N.; Chen, X. Sustainability: Don’t waste seafood waste. Nat. News 2015, 524, 155.
- Kjartansson, G.T.; Zivanovic, S.; Kristbergsson, K.; Weiss, J. Sonication-assisted extraction of chitin from North Atlantic shrimps (Pandalus borealis). J. Agric. Food Chem. 2006, 54, 5894–5902.
- Chang, K.L.B.; Tsai, G. Response surface optimization and kinetics of isolating chitin from pink shrimp (Solenocera melantho) shell waste. J. Agric. Food Chem. 1997, 45, 1900–1904.
- Mojarrad, J.S.; Nemati, M.; Valizadeh, H.; Ansarin, M.; Bourbour, S. Preparation of glucosamine from exoskeleton of shrimp and predicting production yield by response surface. J. Agric. Food Chem. 2007, 55, 2246–2250.
- Xu, Y.; Gallert, C.; Winter, J. Chitin purification from shrimp wastes by microbial deproteination and decalcification. Appl. Microbiol. Biotechnol. 2008, 79, 687–697.
- Zhai, X.; Hawkins, S.J. Interactions of aquaculture and waste disposal in the coastal zone. J. Ocean. Univ. Qingdao 2002, 1, 8–12.
- Gimeno, M.; Ramirez-Hernandez, J.Y.; Mártinez-Ibarra, C.; Pacheco, N.; Garcia-Arrazola, R.; Barzana, E.; Shirai, K. One-solvent extraction of astaxanthin from lactic acid fermented shrimp wastes. J. Agric. Food Chem. 2007, 55, 10345–10350.
- Wang, S.L.; Chang, T.J.; Liang, T.W. Conversion and degradation of shellfish wastes by Serratia sp. TKU016 fermentation for the production of enzymes and bioactive materials. Biodegradation 2010, 21, 321–333.
- Arbia, W.; Arbia, L.; Adour, L.; Amrane, A. Chitin extraction from crustacean shells using biological methods—A review. Food Technol. Biotech. 2013, 51, 12–25.
- Yadav, M.; Goswami, P.; Paritosh, K.; Kumar, M.; Pareek, N.; Vivekanand, V. Seafood waste: A source for preparation of commercially employable chitin/chitosan materials. Bioresour. Bioprocess. 2019, 6, 8.
- Casadidio, C.; Peregrina, D.V.; Gigliobianco, M.R.; Deng, S.; Censi, R.; Di Martino, P. Chitin and chitosans: Characteristics, eco-friendly processes, and applications in cosmetic science. Mar. Drugs 2019, 17, 369.
- Synowiecki, J.; Al-Khateeb, N.A. Production, properties, and some new applications of chitin and its derivatives. Crit. Rev. Food Sci. Nutr. 2003, 43, 145–171.
- Tolaimate, A.; Desbrieres, J.; Rhazi, M.; Alagui, A. Contribution to the preparation of chitins and chitosans with controlled physico-chemical properties. Polymer 2003, 44, 7939–7952.
- Das, S.; Ganesh, E.A. Extraction of chitin from trash crabs (Podophthalmus vigil) by an eccentric method. Curr. Res. J. Biol. Sci. 2010, 2, 72–75.
- Bajaj, M.; Winter, J.; Gallert, C. Effect of deproteination and deacetylation conditions on viscosity of chitin and chitosan extracted from Crangon crangon shrimp waste. Biochem. Eng. J. 2011, 56, 51–62.
- Haddar, A.; Hmidet, N.; Ghorbel-Bellaaj, O.; Fakhfakh-Zouari, N.; Sellami-Kamoun, A.; Nasri, M. Alkaline proteases produced by Bacillus licheniformis rp1 grown on shrimp wastes: Application in chitin extraction, chicken feather-degradation and as a dehairing agent. Biotechnol. Bioprocess. Eng. 2011, 16, 669.
- Sila, A.; Nasri, R.; Bougatef, A.; Nasri, M. Digestive alkaline proteases from the goby (Zosterisessor ophiocephalus): Characterization and potential application as detergent additive and in the deproteinization of shrimp wastes. J. Aquat. Food Prod. Technol. 2012, 21, 118–133.
- Mhamdi, S.; Ktari, N.; Hajji, S.; Nasri, M.; Kamoun, A.S. Alkaline proteases from a newly isolated Micromonospora chaiyaphumensis s103: Characterization and application as a detergent additive and for chitin extraction from shrimp shell waste. Int. J. Biol. Macromol. 2017, 94, 415–422.
- Hamdi, M.; Hammami, A.; Hajji, S.; Jridi, M.; Nasri, M.; Nasri, R. Chitin extraction from blue crab (Portunus segnis) and shrimp (Penaeus kerathurus) shells using digestive alkaline proteases from P. segnis viscera. Int. J. Biol. Macromol. 2017, 101, 455–463.
- Castro, R.; Guerrero-Legarreta, I.; Bórquez, R. Chitin extraction from Allopetrolisthes punctatus crab using lactic fermentation. Biotechnol. Rep. 2018, 20, e00287.
- Du, J.; Tan, E.; Kim, H.J.; Zhang, A.; Bhattacharya, R.; Yarema, K.J. Comparative evaluation of chitosan, cellulose acetate, and polyethersulfone nanofiber scaffolds for neural differentiation. Carbohydr. Polym. 2014, 99, 483–490.
- Peter, M.G. Applications and Environmental Aspects of Chitin and Chitosan. J. Macromol. Sci. Part A 1995, 32, 629–640.
- Kurita, K. Chemistry and application of chitin and chitosan. Polym. Degrad. Stab. 1998, 59, 117–120.
- Kumar, R.M.N. A review of chitin and chitosan applications. React. Funct. Polym. 2000, 46, 1–27.
- Dutta, P.K.; Dutta, J.; Tripathi, V.S. Chitin and chitosan: Chemistry, properties and applications. J. Sci. Ind. Res. India 2004, 63, 20–31.
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632.
- Pillai, C.K.S.; Paul, W.; Sharma, C.P. Chitin and chitosan polymers: Chemistry, solubility and fiber formation. Prog. Polym. Sci. 2009, 34, 641–678.
- Venkatesan, J.; Kim, S.K. Chitosan composites for bone tissue engineering—An overview. Mar. Drugs 2010, 8, 2252–2266.
- Hembach, L.; Cord-Landwehr, S.; Moerschbacher, B.M. Enzymatic production of all fourteen partially acetylated chitosan tetramers using different chitin deacetylases acting in forward or reverse mode. Sci. Rep. 2017, 7, 17692.
- Abdou, E.S.; Nagy, K.S.A.; Elsabee, M.Z. Extraction and characterization of chitin and chitosan from local sources. Bioresour. Technol. 2008, 99, 1359–1367.
- Al-Hassan, A.A. Utilization of waste: Extraction and characterization of chitosan from shrimp byproducts. Civil. Environ. Res. 2016, 8, 117–123.
- Vilar Junior, J.C.; Ribeaux, D.R.; Alves da Silva, C.A.; Campos-Takaki, G.M. Physicochemical and antibacterial properties of chitosan extracted from waste shrimp shells. Int. J. Microbiol. 2016, 5127515, 1–7.
- Sedaghat, F.; Yousefzadi, M.; Toiserkani, H.; Najafipour, S. Bioconversion of shrimp waste Penaeus merguiensis using lactic acid fermentation: An alternative procedure for chemical extraction of chitin and chitosan. Int. J. Biol. Macromol. 2017, 104, 883–888.
- Varun, T.K.; Senani, S.; Jayapal, N.; Chikkerur, J.; Roy, S.; Tekulapally, V.B.; Gautam, M.; Kumar, N. Extraction of chitosan and its oligomers from shrimp shell waste, their characterization and antimicrobial effect. Vet. World 2017, 10, 170–175.
- Aboudamia, F.Z.; Kharroubi, M.; Neffa, M.; Aatab, F.; Hanoune, S.; Bouchdoug, M.; Jaouad, A. Potential of discarded sardine scales (Sardina pilchardus) as chitosan sources. J. Air Waste Manag. Assoc. 2020, 70, 1186–1197.
- Said Al Hoqani, H.A.; AL-Shaqsi, N.; Hossain, M.A.; Al Sibani, M.A. Isolation and optimization of the method for industrial production of chitin and chitosan from Omani shrimp shell. Carbohydr. Res. 2020, 492, 108001.
- Sagheer, F.A.A.; Al-Sughayer, M.A.; Muslim, S.; Elsabee, M.Z. Extraction and characterization of chitin and chitosan from marine sources in Arabian Gulf. Carbohydr. Polym. 2009, 77, 410–419.
- Samar, M.M.; El-Kalyoubi, M.; Khalaf, M.; El-Razik, M.A. Physicochemical, functional, antioxidant and antibacterial properties of chitosan extracted from shrimp wastes by microwave technique. Ann. Agric. Sci. 2013, 58, 33–41.
- EL Knidri, H.; Dahmani, J.; Addaou, A.; Laajeb, A.; Lahsini, A. Rapid and efficient extraction of chitin and chitosan for scale-up production: Effect of process parameters on deacetylation degree and molecular weight. Int. J. Biol. Macromol. 2019, 139, 1092–1102.
- Santos, V.P.; Maia, P.; Alencar, N.S.; Farias, L.; Andrade, R.F.S.; Souza, D.; Ribeaux, D.R.; Franco, L.O.; Campos-Takaki, G.M. Recovery of chitin and chitosan from shrimp waste with microwave technique and versatile application. Arq. Inst. Biol. 2019, 86, e0982018.
- Hafsa, J.; Smach, M.A.; Charfeddine, B.; Limem, K.; Majdoub, H.; Rouatbi, S. Antioxidant and antimicrobial proprieties of chitin and chitosan extracted from Parapenaeus Longirostris shrimp shell waste. Ann. Pharm. Françaises 2016, 74, 27–33.
- Borja-Urzola, A.D.C.; García-Gómez, R.S.; Flores, R.; Durán-Domínguez-de-Bazúa, M.D.C. Chitosan from shrimp residues with a saturated solution of calcium chloride in methanol and water. Carbohydr. Res. 2020, 497, 108116.
- Huang, L.; Bi, S.; Pang, J.; Sun, M.; Feng, C.; Chen, X. Preparation and characterization of chitosan from crab shell (Portunus trituberculatus) by NaOH/urea solution freeze-thaw pretreatment procedure. Int. J. Biol. Macromol. 2020, 147, 931–936.
- Gopal, J.; Muthu, M.; Dhakshanamurthy, T.; Kim, K.J.; Hasan, N.; Kwon, S.J.; Chun, S. Sustainable ecofriendly phytoextract mediated one pot green recovery of chitosan. Sci. Rep. 2019, 9, 13832.
- Salah, R.; Michaud, P.; Mati, F.; Harrat, Z.; Lounici, H.; Abdi, N.; Drouiche, N.; Mameri, N. Anticancer activity of chemically prepared shrimp low molecular weight chitin evaluation with the human monocyte leukaemia cell line, THP-1. Int. J. Biol. Macromol. 2013, 52, 333–339.
- Yen, M.T.; Yang, J.H.; Mau, J.L. Antioxidant properties of chitosan from crab shells. Carbohydr. Polym. 2008, 74, 840–844.
- Goy, R.C.; de Britto, D.; Assis, O.B.G. A review of the antimicrobial activity of chitosan. Polim. Cienc. E Tecnol. 2009, 19, 241–247.
- Vongchan, P.; Sajomsang, W.; Kasinrerk, W.; Subyen, D.; Kongtawelert, P. Anticoagulant activities of the chitosan polysulfate synthesized from marine crab shell by semi-heterogeneous conditions. Sci. Asia 2003, 29, 115–120.
- Dash, M.; Chiellini, F.; Ottenbrite, R.M.; Chiellini, E. Chitosan—A versatile semi-synthetic polymer in biomedical applications. Prog. Polym. Sci. 2011, 36, 981–1014.
- Parvez, S.; Rahman, M.M.; Khan, M.A.; Khan, M.A.H.; Islam, J.M.M.; Ahmed, M.; Rahman, M.F.; Ahmed, B. Preparation and characterization of artificial skin using chitosan and gelatin composites for potential biomedical application. Polym. Bull. 2012, 69, 715–731.
- Muzzarelli, R.A.; Muzzarelli, C. Chitosan chemistry: Relevance to the biomedical sciences. Adv. Polym. Sci. 2005, 186, 151–209.
- Riva, R.; Ragelle, H.; des Rieux, A.; Duhem, N.; Jérôme, C.; Préat, V. Chitosan and chitosan derivatives in drug delivery and tissue engineering. Adv. Polym. Sci. 2011, 244, 19–44.
- Mahmoud, N.S.; Ghaly, A.E.; Arab, F. Unconventional approach for demineralization of deproteinized crustacean shells for chitin production. Am. J. Biochem. Biotechnol. 2007, 3, 1–9.
- Li, Q.; Dunn, E.T.; Grandmaison, E.W.; Goosen, M.F.A. Applications and properties of chitin and chitosan. In Applications of Chitin and Chitosan; Mattheus, F.A., Goosen, M.F.A., Eds.; Chitin Recovery; Technomic Publishing Company: Lancaster, PA, USA, 1997; Volume 37, pp. 1359–1366.
- Khoushab, F.; Yamabhai, M. Chitin research revisited. Mar. Drugs 2010, 8, 1988–2012.
- Limam, Z.; Selmi, S.; Sadok, S.; El Abed, A. Extraction and characterization of chitin and chitosan from crustacean byproducts: Biological and physicochemical properties. Afr. J. Biotechnol. 2011, 10, 640–647.
- Kato, Y.; Onishi, H.; Machida, Y. Application of chitin and chitosan derivatives in the pharmaceutical field. Curr. Pharm. Biotechnol. 2003, 4, 303–309.
- Kim, S.K.; Mendis, E. Bioactive compounds from marine processing byproducts—A review. Food Res. Int. 2006, 39, 383–393.
- Muzzarelli, R.A.A.; Boudrant, J.; Meyer, D.; Manno, N.; Demarchis, M.; Paoletti, M.G. Current views on fungal chitin/chitosan, human chitinases, food preservation, glucans, pectins and inulin: A tribute to Henri Braconnot, precursor of the carbohydrate polymers science, on the chitin bicentennial. Carbohydr. Polym. 2012, 87, 995–1012.
- Leceta, I.; Guerrero, P.; Cabezudo, S.; Caba, K.D.L. Environmental assessment of chitosan-based films. J. Clean. Prod. 2013, 41, 312–318.
- Bhatnagar, A.; Sillanpää, M. Applications of chitin-and chitosan-derivatives for the detoxification of water and wastewater-a short review. Adv. Colloid Interface 2009, 152, 26–38.
- Jin, R.D.; Suh, J.W.; Park, R.D.; Kim, Y.W.; Krishnan, H.B.; Kim, K.Y. Effect of chitin compost and broth on biological control of Meloidogyne incognita on tomato (Lycopersicon esculentum Mill.). Nematology 2005, 7, 125–132.
- Venkatesan, J.; Lowe, B.; Anil, S.; Manivasagan, P.; Kheraif, A.A.A.; Kang, K.H.; Kim, S.K. Seaweed polysaccharides and their potential biomedical applications. Starch 2015, 67, 381–390.
- Aranaz, I.; Harris, R.; Heras, A. Chitosan amphiphilic derivatives. Chemistry and applications. Curr. Org. Chem. 2010, 14, 308–330.
- Cárdenas, G.; Cabrera, G.; Taboada, E.; Rinaudo, M. Synthesis and characterization of chitosan alkyl phosphates. J. Chil. Chem. Soc. 2006, 51, 815–820.
- Jayakumar, R.; Nagahama, H.; Furuike, T.; Tamura, H. Synthesis of phosphorylated chitosan by novel method and its characterization. Int. J. Biol. Macromol. 2008, 42, 335–339.
- Jayakumar, R.; Nwe, N.; Tokura, S.; Tamura, H. Sulfated chitin and chitosan as novel biomaterials. Int. J. Biol. Macromol. 2007, 40, 175–181.
- Jayakumar, R.; Reis, R.; Mano, J. Chemistry and applications of phosphorylated chitin and chitosan. e-Polymers 2006, 35, 1–16.
- Jayakumar, R.; Selvamurugan, N.; Nair, S.; Tokura, S.; Tamura, H. Preparative methods of phosphorylated chitin and chitosan—An overview. Int. J. Biol. Macromol. 2008, 43, 221–225.
- Li, B.; Huang, L.; Wang, X.; Ma, J.; Xie, F. Biodegradation and compressive strength of phosphorylated chitosan/chitosan/hydroxyapatite bio-composites. Mater. Des. 2011, 32, 4543–4547.
- Li, B.; Huang, L.; Wang, X.; Ma, J.; Xie, F.; Xia, L. Effect of micropores and citric acid on the bioactivity of phosphorylated chitosan/chitosan/hydroxyapatite composites. Ceram. Int. 2013, 39, 3423–3427.
- Li, Q.L.; Chen, Z.Q.; Darvell, B.W.; Liu, L.K.; Jiang, H.B.; Zen, Q.; Peng, Q.; Ou, G.M. Chitosan-phosphorylated chitosan polyelectrolyte complex hydrogel as an osteoblast carrier. J. Biomed. Mater. Res. 2007, 82, 481–486.
- Li, Q.; Chen, Z.; Darvell, B.; Zeng, Q.; Li, G.; Ou, G.; Wu, M. Biomimetic synthesis of the composites of hydroxyapatite and chitosan–phosphorylated chitosan polyelectrolyte complex. Mater. Lett. 2006, 60, 3533–3536.
- Li, Q.; Huang, L.N.; Chen, Z.; Tang, X. Biomimetic synthesis of the nanocomposite of phosphorylatd chitosan and hydroxyapatite and its bioactivity in vitro. Key Eng. Mater. 2007, 330–332, 721–724.
- Li, Z.; Ramay, H.R.; Hauch, K.D.; Xiao, D.; Zhang, M. Chitosan-alginate hybrid scaffolds for bone tissue engineering. Biomaterials 2005, 26, 3919–3928.
- Chesnutt, B.M.; Yuan, Y.; Brahmandam, N.; Yang, Y.; Ong, J.L.; Haggard, W.O.; Bumgardner, J.D. Characterization of biomimetic calcium phosphate on phosphorylated chitosan films. J. Biomed. Mater. Res. Part A 2007, 82, 343–353.
- Cao, Z.; Xu, H.; Tang, R.; Wei, W.; Xu, W. Synthesis, characterization and molluscicidal activity of triphenyltin phosphorylated chitosan. Chem. Bull./Huaxue Tongbao 2008, 71, 528–532.
- Datta, P.; Dhara, S.; Chatterjee, J. Hydrogels and electrospun nanofibrous scaffolds of N-methylene phosphonic chitosan as bioinspired osteoconductive materials for bone grafting. Carbohydr. Polym. 2012, 87, 1354–1362.
- Liu, J.; Zheng, Y.; Wang, W.; Wang, A. Preparation and swelling properties of semi-IPN hydrogels based on chitosan-g-poly(acrylic acid) and phosphorylated polyvinyl alcohol. J. Appl. Polym. Sci. 2009, 114, 643–652.
- Srakaew, V.; Ruangsri, P.; Suthin, K.; Thunyakitpisal, P.; Tachaboonyakiat, W. Sodium-phosphorylated chitosan/zinc oxide complexes and evaluation of their cytocompatibility: An approach for periodontal dressing. J. Biomater. Appl. 2012, 27, 403–412.
- Yeh, H.Y.; Lin, J.C. Surface phosphorylation for polyelectrolyte complex of chitosan and its sulfonated derivative: Surface analysis, blood compatibility and adipose derived stem cell contact properties. J. Biomater. Sci. Polym. Ed. 2012, 23, 233–250.
- Zhao, D.; Xu, J.; Wang, L.; Du, J.; Dong, K.; Wang, C.; Liu, X. Study of two chitosan derivatives phosphorylated at hydroxyl or amino groups for application as flocculants. J. Appl. Polym. Sci. 2012, 125, E299–E305.
- Saiki, I.; Murata, J.; Nakajima, M.; Tokura, S.; Azuma, I. Inhibition by sulfated chitin derivatives of invasion through extracellular matrix and enzymatic degradation by metastatic melanoma cells. Cancer Res. 1990, 50, 3631–3637.
- Ryzhenkov, V.E.; Solovyeva, M.A.; Remesova, O.V.; Okunevich, I.V. Hypolipidemic action of sulfated polysaccharides. Voprosy Meditsinskoj Khimii 1996, 42, 118–119.
- Kornilaeva, G.V.; Makarova, T.V.; Gamzazade, A.I.; Sklyar, A.M.; Nasibov, S.M.; Karamov, E.V. Sulphated chitosan derivatives as HIV-infection inhibitors. Immunologiya 1995, 1, 13–16.
- Hirano, S.; Tanaka, Y.; Hasegawa, M.; Tobetto, K.; Nishioka, A. Effect of sulfated derivatives of chitosan on some blood coagulant factors. Carbohydr. Res. 1985, 137, 205–215.
- Gamzazade, A.; Sklyar, A.; Nasibov, S.; Sushkov, I.; Shashkov, A.; Knirel, Y. Structural features of sulfated chitosans. Carbohydr. Polym. 1997, 34, 113–116.
- Mariappan, M.R.; Alas, E.A.; Williams, J.G.; Prager, M.D. Chitosan and chitosan sulfate have opposing effects on collagen-fibroblast interactions. Wound Repair Regen. 1999, 7, 400–406.
- Ma, B.; Huang, W.; Kang, W.; Yan, J. Studies on preparation of sulfated derivatives of chitosan from Mucor rouxianus. Ion Exch. Adsorpt. 2007, 23, 451–458.
- Vongchan, P.; Sajomasang, W.; Subyen, D.; Kongtawelert, P. Anticoagulant activity of a sulfated chitosan. Carbohydr. Res. 2002, 337, 1239–1242.
- Ou, Y.; Wu, Y.; Li, H.; Wang, K.; Zhang, D. Synthesis, characterization and antibacterial activity of hybrid materials of rare earth and chitosan. J. Chin. Rare Earth Soc. 2013, 31, 211–216.
- Xing, R.; Liu, S.; Yu, H.; Zhang, Q.; Li, Z.; Li, P. Preparation of low-molecular-weight and highsulfate-content chitosans under microwave radiation and their potential antioxidant activity in vitro. Carbohyd. Res. 2004, 339, 2515–2519.
- Xing, R.; Yu, H.; Liu, S.; Zhang, W.; Zhang, Q.; Li, Z. Antioxidative activity of differently regioselective chitosan sulfates in vitro. Bioorg. Med. Chem. 2005, 13, 1387–1392.
- Karadeniz, F.; Karagozlu, M.Z.; Pyun, S.Y.; Kim, S.K. Sulfation of chitosan oligomers enhances their anti-adipogenic effect in 3T3-L1 adipocytes. Carbohydr. Polym. 2011, 86, 666–671.
- Fan, L.; Wu, P.; Zhang, J.; Gao, S.; Wang, L.; Li, M.; Sha, M.; Xie, W.; Nie, M. Synthesis and anticoagulant activity of the quaternary ammonium chitosan sulfates. Int. J. Biol. Macromol. 2012, 50, 31–37.
- Pires, N.R.; Cunha, P.L.; Maciel, J.S.; Angelim, A.L.; Melo, V.M.; De Paula, R.C.; Feitosa, J.P. Sulfated chitosan as tear substitute with no antimicrobial activity. Carbohydr. Polym. 2013, 91, 92–99.
- Shelma, R.; Sharma, C.P. Development of lauroyl sulfated chitosan for enhancing hemocompatibility of chitosan. Colloid. Surf. B 2011, 84, 561–570.
- Fang, S.W.; Li, C.F.; Shih, D.Y.C. Antifungal activity of chitosan and its preservative effect on low-sugar candied kumwuat. J. Food Prot. 1994, 56, 136–140.
- Chung, Y.-C.; Chen, C.-Y. Antibacterial characteristics and activity of acid-soluble chitosan. Bioresour. Technol. 2008, 99, 2806–2814.
- Eaton, P.; Fernandes, J.C.; Pereira, E.; Pintado, M.E.; Malcata, F.X. Atomic force microscopy study of the antibacterial effects of chitosans on Escherichia coli and Staphylococcus aureus. Ultramicroscopy 2008, 108, 1128–1134.
- Helander, I.M.; Nurmiaho-Lassila, E.L.; Ahvenainen, R.; Rhoades, J.; Roller, S. Chitosan disrupts the barrier properties of the outer membrane of gram-negative bacteria. Int. J. Food Microbiol. 2001, 71, 235–244.
- Assis, O.B.G.; Pessoa, J.D.C. Scientific note: Preparation of thin films of chitosan for use as edible coatings to inhibit fungal growth on sliced fruits. Braz. J. Food Sci. Tech. 2004, 7, 17–22.
- Másson, M.; Holappa, J.; Hjálmarsdóttir, M.; Rúnarsson, Ö.; Nevalainen, T.; Järvinen, T. Antimicrobial activity of piperazine derivatives of chitosan. Carbohydr. Polym. 2008, 74, 566–571.
- Khanafari, A.; Marandi, R.; Sanatei, S. Recovery of chitin and chitosan from shrimp waste by chemical and microbal methods. Iran. J. Environ. Health Sci. Eng. 2008, 5, 19–24.
- Li, Q.; Dunn, E.T.; Grandmaison, E.W.; Goosen, M.F.A. Applications and properties of chitosan. J. Bioact. Compat. Polym. 1992, 7, 370–397.
- Farley, J.; Clear, N.; Leroy, B.; Davis, T.; Mcpherson, G. Age, growth and preliminary estimates of maturity of bigeye tuna, Thunnus obesus, in the Australian region. Mar. Freshwater Res. 2006, 57, 713–724.
- Lee, J.K.; Lim, H.S.; Kim, J.H. Cytotoxic activity of aminoderivatized cationic chitosan derivatives. J. Bioorgan. Med. Chem. Lett. 2002, 12, 2949–2951.
- Cerdá, C.; Sánchez, C.; Climent, B.; Vázquez, A.; Iradi, A.; El Amrani, F.; Bediaga, A.; Sáez, G.T. Oxidative stress and DNA damage in obesity-related tumorigenesis. Adv. Exp. Med. Biol. 2014, 824, 5–17.
- Wölfle, U.; Seelinger, G.; Bauer, G.; Meinke, M.C.; Lademann, J.; Schempp, C.M. Reactive molecule species and antioxidative mechanisms in normal skin and skin aging. Ski. Pharmacol. Physiol. 2014, 27, 316–332.
- Ito, N.; Hirose, M.; Fukushima, H.; Tsuda, T.; Shirai, T.; Tatenatsu, M. Studies on antioxidants: Their carcinogenic and modifying effects on chemical carcinogens. Food Chem. Toxicol. 1986, 24, 1071–1092.
- Manda, G.; Nechifor, M.T.; Neagu, T.M. Reactive oxygen species, cancer and anticancer therapies. Curr. Chem. Biol. 2009, 3, 342–366.
- Zhang, N.; Bradley, T.A.; Zhang, C. Inflammation and reactive oxygen species in cardiovascular Disease. World J. Cardiol. 2010, 2, 408–410.
- Zhang, J.; Xia, W.; Liu, P.; Cheng, Q.; Tahirou, T.; Gu, W.; Li, B. Chitosan modification and pharmaceutical/biomedical applications. Mar. Drugs 2010, 8, 1962–1987.
- Cui, H.; Kong, Y.; Zhang, H. Oxidative stress, mitochondrial dysfunction, and aging. J. Signal. Transduct 2012, 2012, 646354.
- Filippin, L.I.; Vercelino, R.; Marroni, N.P.; Xavier, R.M. Redox signalling and the inflammatory response in rheumatoid arthritis. Clin. Exp. Immunol. 2008, 152, 415–422.
- Park, B.K.; Kim, M.-M. Applications of chitin and its derivatives in biological medicine. Int. J. Mol. Sci. 2010, 11, 5152–5164.
- Kerch, G. The potential of chitosan and its derivatives in prevention and treatment of age-related diseases. Mar. Drugs 2015, 13, 2158–2182.
- Younes, I.; Rinaudo, M. Chitin and chitosan preparation from marine sources. Structure, properties and applications. Mar. Drugs 2015, 13, 1133–1174.
- Harish Prashanth, K.V.; Tharanathan, R.N. Chitin/chitosan: Modifications and their unlimited application potential—An overview. Trends Food Sci. Technol. 2007, 18, 117–131.
- Tharanathan, R.N.; Kittur, F.S. Chitin—The undisputed biomolecule of great potential. Crit. Rev. Food Sci. Nutr. 2003, 43, 61–87.
- No, H.K.; Meyers, S.P.; Prinyawiwatkul, W.; Xu, Z. Applications of chitosan for improvement of quality and shelf life of foods: A review. J. Food Sci. 2007, 72, 87–100.
- Kamil, J.Y.V.A.; Jeon, Y.J.; Shahidi, F. Antioxidative activity of chitosans of different viscosity in cooked comminuted flesh of herring (Clupea harengus). Food Chem. 2002, 79, 69–77.
- Kim, K.W.; Thomas, R.L. Antioxidative activity of chitosans with varying molecular weights. Food Chem. 2007, 101, 308–313.
- Pittayapruek, P.; Meephansan, J.; Prapapan, O.; Komine, M.; Ohtsuki, M.; Maki, M. Role of matrix metalloproteinases in photoaging and photocarcinogenesis. Int. J. Mol. Sci. 2016, 17, 868.
- Yoo, S.-J.; Go, E.; Kim, Y.-E.; Lee, S.; Kwon, J. Roles of reactive oxygen species in rheumatoid arthritis pathogenesis. J. Rheum. Dis. 2016, 23, 340–347.
- Mirshafiey, A.; Mohsenzadegan, M. The role of reactive oxygen species in immunopathogenesis of rheumatoid arthritis. Iran. J. Allergy Asthma Immunol. 2008, 7, 195–202.
- Rahman, T.; Hosen, I.; Islam, M.M.T.; Shekhar, H.U. Oxidative stress and human health. Adv. Biosci. Biotechnol. 2012, 3, 997–1019.
- Aranaz, I.; Mengíbar, M.; Harris, R.; Paños, I.; Miralles, B.; Acosta, N.; Galed, G.; Heras, A. Functional characterization of chitin and chitosan. Curr. Chem. Biol. 2009, 3, 203–230.
- Bhattacharya, S. Reactive oxygen species and cellular defense system. In Free Radicals in Human Health and Disease; Rani, V., Yadav, U.C.S., Eds.; Springer: New Delhi, India, 2015; pp. 17–29.
- Lushchak, V.I. Free radicals, reactive oxygen species, oxidative stress and its classification. Chem. Biol. Interact. 2014, 224, 164–175.
- Patlevic, P.; Vascova, J.; Svork, P., Jr.; Vasko, L.; Svork, P. Reactive oxygen species and antioxidant defense in human gastrointestinal diseases. Integr. Med. Res. 2016, 5, 250–258.
- Bhattacharyya, A.; Chattopadhyay, R.; Mitra, S.; Crowe, S.E. Oxidative stress: An essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol. Rev. 2014, 94, 329–354.
- Hajji, S.; Younes, I.; Rinaudo, M.; Jellouli, K.; Nasri, M. Characterization and in vitro evaluation of cytotoxicity, antimicrobial and antioxidant activities of chitosans extracted from three different marine sources. Appl. Biochem. Biotech. 2015, 177, 18–35.
- Sun, T.; Zhou, D.; Xie, J.; Mao, F. Preparation of chitosan oligomers and their antioxidant activity. Eur. Food Res. Technol. 2007, 225, 451–456.
- Chang, S.; Wu, C.; Tsai, G.J. Effects of chitosan molecular weight on its antioxidant and antimutagenic properties. Carbohydr. Polym. 2018, 181, 1026–1032.
- Božič, M.; Gorgieva, S.; Kokol, V. Laccase-mediated functionalization of chitosan by caffeic and galic acids for modulating antioxidant and antimicrobial properties. Carbohydr. Polym. 2012, 87, 2388–2398.
- Xie, W.; Xu, P.; Liu, Q. Antioxidant activity of water-soluble chitosan derivatives. Bioorg. Med. Chem. Lett. 2001, 11, 1699–1701.
- Sousa, F.; Guebitz, G.M.; Kokol, V. Antimicrobial and antioxidant properties of chitosan enzymatically functionalized with flavonoids. Process. Biochem. 2009, 44, 749–756.
- Benhabiles, M.S.; Salah, R.; Lounici, H.; Drouiche, N.; Goosen, M.F.A.; Mameri, N. Antibacterial activity of chitin, chitosan and its oligomers prepared from shrimp shell waste. Food Hydrocoll. 2012, 29, 48–56.
- Fernandes, J.C.; Eaton, P.; Nascimento, H.; Gião, M.S.; Ramos, Ó.S.; Belo, L.; Santos-Silva, A.; Pintado, M.E.; Malcata, F.X. Antioxidant activity of chitooligosaccharides upon two biological systems: Erythrocytes and bacteriophages. Carbohydr. Polym. 2010, 79, 1101–1106.
- Li, Q.; Sun, X.; Gu, G.; Guo, Z. Novel water soluble chitosan derivatives with 1,2,3-triazolium and their free radical-scavenging activity. Mar. Drugs 2018, 16, 107.
- Liu, J.; Sun, H.; Dong, F.; Xue, Q.; Wang, G.; Qin, S.; Guo, Z. The influence of the cation of quaternized chitosans on antioxidant activity. Carbohydr. Polym. 2009, 78, 439–443.
- Wan, A.; Xu, Q.; Sun, Y.; Li, H. Antioxidant activity of high molecular weight chitosan and N,O-quaternized chitosans. J. Agric. Food Chem. 2013, 61, 6921–6928.
- Rajoka, M.S.R.; Zhao, L.; Mehwish, H.M.; Wu, Y.; Mahmood, S. Chitosan and its derivatives: Synthesis, biotechnological applications, and future challenges. Appl. Microbiol. Biotechnol. 2019, 103, 1557–1571.
