Mycotoxins are a group of natural compounds produced under a wide range of climatic conditions by filamentous fungi mainly belonging to Aspergillus, Penicillium, Fusarium, Claviceps and Alternaria species. These toxins can contaminate various agricultural commodities (cereals, dried fruits, nuts, spices and coffee being the most frequently contaminated ones) either before harvest or under postharvest conditions, thus posing a risk to human and animal health due to their toxic effects.
- mycotoxins
- food
- LC-MS methods
1. Introduction
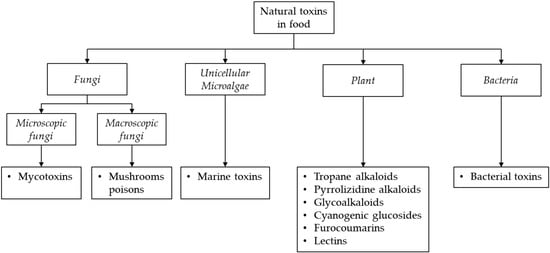
The reduction of risks related to the presence of natural toxins in food plays an essential role in protecting consumers. Indeed, WHO, together with the European Food Safety Authority (EFSA), FAO and Codex Alimentarius Commission, have established maximum residue limits (MRLs) or recommendations for many of these natural toxins to control their occurrence in food [7,8,9,10,11,12,13,14]. Mycotoxins are one of the most relevant threats to human health. They represent the most important class of chemical hazards among natural toxins recorded in the European Rapid Alert System on Feed and Food (RASFF). To ensure a correct risk assessment evaluation and compliance with the current legislation, it is important to develop sensitive, selective and robust analytical methods to determine the occurrence of these class of natural toxins in foodstuffs. However, the main challenge for the analysis of these compounds in food samples is related to their different physico-chemical properties, the inherent complexity of food matrices as well as the purpose of the analysis. These characteristics can affect the extraction efficiency of targeted toxins and then the accuracy and sensitivity of the method [4].
2. Mycotoxins
Mycotoxins are a group of natural compounds produced under a wide range of climatic conditions by filamentous fungi mainly belonging to Aspergillus, Penicillium, Fusarium, Claviceps and Alternaria species. These toxins can contaminate various agricultural commodities (cereals, dried fruits, nuts, spices and coffee being the most frequently contaminated ones) either before harvest or under postharvest conditions, thus posing a risk to human and animal health due to their toxic effects. In addition, significant losses of yields and quality of the infested commodity are observed. Among the over 300 mycotoxins that have been identified, those of major concern worldwide causing food-borne illnesses are aflatoxins, ochratoxin A, fumonisins, zearalenone, patulin, citrinin, type B trichothecenes, mainly deoxynivalenol and nivalenol, and type A trichothecenes, mainly T-2 and HT-2 toxins. Their toxic effects range from nephrotoxicity, cytotoxicity, nervous-system disturbances, gastrointestinal diseases, to immunotoxicity, mutagenicity, teratogenicity and carcinogenicity [7][8][19,20]. In particular, aflatoxins have been classified by the International Agency for Research on Cancer (IARC) as carcinogenic to humans (Group 1) [8][20]. As a legislative consequence, maximum permitted or indicative levels have been established worldwide or are under discussion [9][10][11][12][13][14][15][16][7,9,21,22,23,24,25,26]. After the infection of crop plants, mycotoxins are modified by plant enzymes and are often conjugated to more polar substances. These substances are usually not detected during routine analysis, are unregulated, and are called “bound” or alternatively “hidden”, “conjugated” or “masked” and more recently “modified” mycotoxins. Some metabolites are more toxic than the parent compound, while others are less toxic. Furthermore, depending on the type of linkage with matrix component, a part of bound mycotoxins could become bioavailable again in the digestive tract of humans and animals, thus contributing to the toxicity of parent compound [17][27]. Other unregulated mycotoxins, that are usually not detected during routine analysis, but with some relevance from a safety and economic point of view, have also been reported in raw cereals and derived products. These compounds are known as “emerging mycotoxins”. The most prominent are beauvericin, enniatins, fusaproliferin, sterigmatocystin, citrinin, ergot alkaloids, moniliformin and Alternaria toxins [18][19][20][21][22][28,29,30,31,32]. Although not all the emerging mycotoxins are of concern to human health, the European Food Standard Authority (EFSA) has recently highlighted the need for long-term studies to assess potential chronic toxic effects. Furthermore, a continuous monitoring, collection of data, expertise and tools to set food safety standards is needed to understand and manage the potential risks of emerging mycotoxins in raw materials, feeds and foods [23][33]. Several chromatographic methods, mainly based on HPLC coupled with UV/diode array and fluorescence detection, have been developed and extensively reviewed for the determination of single mycotoxin or structurally related mycotoxins in food and feed. Gas chromatography (GC) and gas chromatography coupled to mass spectrometry (GC-MS) are also available for the specific detection of trichothecenes [24][25][26][34,35,36]. The evidence of co-occurring mycotoxins in various matrices has led to the development of new multi-mycotoxin methods for their simultaneous detection in the same matrix. The absence of structural information, as well as the coelution of structurally related mycotoxins, have shifted this field towards more sophisticated detection techniques based on the use of MS detectors. As a consequence, MS coupled with HPLC and ultra-HPLC systems has turned into one of the most powerful tools for multi-mycotoxin analysis at very low concentrations in complex matrices [26][27][36,37]. Furthermore, LC tandem mass spectrometry (LC-MS/MS), multi-stage mass spectrometry (MSn) and high-resolution MS (HRMS) can provide structural and accurate mass information not only for the determination of well-known mycotoxins with remarkable sensitivity and specificity, but also for the analysis of emerging and modified mycotoxins [26][28][29][18,36,38]. A recent review providing insight into LC-MS based methods for the simultaneous determination of mycotoxins indicated that in the period 2012–2016, approximately 80% of all published LC-MS methods on mycotoxins were based on LC-MS/MS, while more than 10% were LC-HRMS methods and the remaining 10% were LC-HRMS/MS [27][37]. Specifically, HRMS are becoming a relevant trend because they offer the possibility to perform target, post target, and non-target analysis in a single run. The complexity of the matrices, mainly including those of animal origin, has led to the development and validation of specific analytical protocols allowing for quantitative extraction and detection of targeted mycotoxins. The availability of standardized methods of analysis is of paramount importance to guarantee a uniform application of the EU legislation and contribute to maintaining a high level of food and feed safety. Despite the huge number of published multimycotoxin LC-MS methods, their implementation in control laboratories has been limited in the past years due to their performance characteristics not fulfilling the acceptability criteria for mycotoxins established at the EU level [24][34]. In 2013, the European Commission (EC) invited the European Committee for Standardization (CEN) to develop and standardize 11 methods for mycotoxins determination in food within the EC standardization mandate M/520 [30][39]. Among them, six methods based on the use of LC-MS/MS have been recently adopted as CEN-EN-ISO standard methods (https://standards.cencenelec.eu/ accessed on 5 April 2022).The scientific progress in the application of LC-MS-based methods to the analysis of mycotoxins in food, considering the literature published from 2016 to 2021 is reported below. The literature search employed the Scopus online database (www.scopus.com, accessed on 31 January 2022) and keywords used for the search were “mycotoxins”, “liquid chromatography mass spectrometry” and “food. Several papers were published in the selective period with a positive trend over the years. A further refinement of selected papers was applied in order to select only those describing the development and validation of an LC-MS method (together with performance characteristics values). The novelty of the used technology, the application to new food matrices and the possibility to simultaneously analyze multiple toxins also including emerging ones were taken into account for the selection.
3. LC-MS Methods for Mycotoxin Determination in Food
A total of 28 papers were selected covering from 2 to 33 well-known detected mycotoxins in a wide range of matrices, including cereals and derived products, as well as beer, edible oils, milk, medical herbs and dried fish. The most used sample preparation approaches included SPE column clean-up or alternatively the Quick, Easy, Cheap, Effective, Rugged and Safe (QuEChERS) approach. The most frequently used MS technology was the QqQ in MRM mode. Some examples of Q-LIT, Q-TOF and Q-Orbitrap were also reported as alternative MS approaches for the analysis of these natural toxins. A multi-mycotoxin analysis method based on UPLC-ESI-MS/MS for the determination of 26 mycotoxins, including both well-known and emerging mycotoxins, in durum wheat was proposed by Juan et al. [31][40]. Accuracy and precision values were in compliance with the EU Regulation 519/2014 [32][41], while sensitivity values (i.e., limits of quantification, LOQs) were lower than the MRL established by the EC Regulation 1881/2006 [9][7] for durum wheat. The method was applied to evaluate the (co)-occurrence of targeted mycotoxins in 74 durum wheat samples harvested in central Italy. The simultaneous detection of five types of Fusarium mycotoxins in processed grains was described by Kai et al. [33][42] using an LC-MS/MS method. The multi-functional SPE procedure was used for the clean-up of sample extracts obtained using an acetonitrile–water mixture as extraction solvent. The in-house validated method applied to 55 food samples, including wheat and corn, showed the co-occurrence of targeted mycotoxins. Another paper, published by Sharmili et al. [34][43], described a simple, fast and reliable method applying a QueEChERS extraction procedure followed by dispersive SPE (d-SPE) clean-up and LC-MS/MS for the simultaneous determination of aflatoxins, ochratoxin A, deoxynivalenol and zearalenone in vegetable oil. The developed method was validated in house according to the criteria of National Public Health Laboratory’s standard operating procedure No: A03-005 for Method Validation in Chemical Analysis and then applied to the analysis of 25 commercial vegetable oil samples from Malaysia. A simple, rapid and accurate method based on QueEChERS followed by SPE C18 columns and detection with UPLC–MS/MS was optimized and validated for the simultaneous determination of 25 mycotoxins in cereals [35][44]. The optimization procedure focused on the selection of clean-up sorbents, extraction solvent, chromatographic conditions and on the matrix effect removal. Performance values were evaluated in terms of precision, accuracy and sensitivity and the method was applied to the analysis of 65 samples of cereals, including wheat, corn and rice, from different areas of China. Wang et al. [36][45] described the development and validation of a fast method based on a d-SPE clean-up, using C18 sorbent and UPLC-Q-TOFMS, for the simultaneous determination of nine mycotoxins in corn. Limits of detection and quantification were below the EU MRL [9][7]. The developed method was applied to the analysis of 130 Chinese corn samples, and fumonisins were the main class of occurring mycotoxins. Another method was developed for the simultaneous determination of 21 mycotoxins in white peony root, Radix Paeoniae Alba (RPA), by using a QuEChERS-based approach followed by d-SPE (C18 sorbent) and detection by UHPLC Q-LIT MS [37][46]. In particular, a MRM information-dependent acquisition-enhanced product ion scan mode was used, and 13 mycotoxins were detected in positive mode while the other mycotoxins were analyzed in negative mode. The developed method was applied to the analysis of 20 RPA samples, showing about 65% of contamination incidence. Zwickel et al. [38][47] reported an LC-MS/MS method for the simultaneous quantification of 12 Alternaria toxins in wine, vegetable juices and fruit juices and presented for the first time analytical data for altenuic acid, altenuisol and iso-altenuene. Furthermore, an improvement of the chromatographic performance was achieved for tenuazonic acid compared to previously reported multi-analyte methods for Alternaria targeted toxins in foodstuffs. Validation results were compliant with the requirements reported in the CEN technical report on the performance criteria for single laboratory validated methods of analysis for the determination of mycotoxins [39][48]. The validated method was applied to the analysis of 103 commercial wine and juice samples collected from Germany, with red wines and grape juices being the most frequently contaminated matrices with multiple Alternaria toxins. Al-Taher et al. [40][49] developed an LC-MS/MS method for the detection of 11 mycotoxins in infant cereals, including rice, barley, oats and mixed grains. Specifically, the method was based on the use of a single-step extraction and a stable isotope dilution assay. The use of UHPLC allowed a good separation of mycotoxins in less than 10 min, which is crucial for high throughput screenings. The method was validated in house in terms of sensitivity, linearity and accuracy, and successfully applied to the analysis of targeted mycotoxins in 64 infant cereals samples from the U.S. market. In another study, a fast, easy and cheap method was developed for the determination of eight mycotoxins in cereal-derived products (wheat flours, dry pasta, baked foods, corn meals and breakfast cereals) based on the use of QuEChERS extraction and LC-MS/MS analysis [41][50]. Performances of the validated method on wheat flour samples fulfilled the criteria set by the EU legislation [32][41]. The method was applied to the analysis of 21 cereal-derived products from the Italian market, showing the co-occurrence of some targeted mycotoxins. A method based on LC-MS/MS was developed and validated for the analysis of 11 mycotoxins in edible oils [42][51]. A simple solvent extraction approach followed by a defatting process using n-hexane was employed for a fast and high-throughput analysis. Performance of the validated method fulfilled acceptance criteria established at the EU level [43][52]. Although the method was applied to the analysis of only nine Korean edible oils, authors stated that additional studies involving larger sample sizes were needed to assess the safety of this category of samples. Another example of multiclass mycotoxin analysis was proposed also for cow milk samples by Flores-Flores and Gonzalez-Penas [44][53]. Specifically, an LC–MS/MS method was validated for the simultaneous analysis of 15 mycotoxins in cow milk, including those that have not been frequently studied in this matrix, such as fumonisins, sterigmatocystin or ochratoxin B. The sample preparation consisted of an LLE using acidified acetonitrile, and a clean-up of the extract with sodium acetate. The method was applied to the analysis of 10 Spanish cow milk samples. No mycotoxins were found, and the authors concluded that more samples should be analyzed to evaluate their presence in this matrix. Kim et al. [45][54] developed an analytical method for the simultaneous determination of 13 mycotoxins in cereal grains by LC-MS/MS after multi-mycotoxin IAC clean-up. The one-step elution provided high recovery values for all tested mycotoxins. The validated method was used in a large survey using more than 500 samples of brown rice, maize, millet, sorghum, and mixed cereals collected from local markets in South Korea. Fusarium mycotoxins were the most frequently and co-occurring detected mycotoxins in the investigated samples. An innovative approach to multi-class mycotoxin control was proposed by Arroyo-Manzanares et al. [46][55] for the analysis of 21 mycotoxins produced by Aspergillus, Fusarium, and Penicillium fungi and 12 ergot alkaloids produced by Claviceps purpurea in wheat and maize. The simple extraction procedure based on the “salting-out” LLE, minimizing the epimerization of ergot alkaloids, in combination with UPLC-MS/MS analysis, were the key factors for the rapid, robust and sensitive detection of these targeted compounds. The method was successfully applied to study the co-occurrence of targeted mycotoxins in 28 wheat and corn samples collected from six European countries. The feasibility of using an ultra-high-performance supercritical fluid chromatography (UHPSFC) and MS/MS as separation tool for 15 Fusarium mycotoxins analysis, including their modified forms, in beer has been described [47][56]. The sample preparation included an LLE and SPE C18 columns clean-up. Although they observed a limited applicability of UHPSFC to the routine multimycotoxin analysis, the authors suggested this approach as a separation technology for isomers to be used in the mycotoxin–biomarker field. In another paper, Du et al. [48][57] described the use of a rapid microwave-assisted dispersive-micro-SPE (MA-d-µ-SPE) and subsequent analysis by UHPLC-Q-TOF-MS for the determination of six mycotoxins in peach seed, milk powder, corn flour and beer. Several parameters of the d-µ-SPE were compared and optimized with nano zirconia selected as the optimal dispersant. The validated method provided limit of detection (LOD) values lower than the EU MRLs [9][7]. Li et al. [49][58] described the use of UPLC-MS/MS for the determination of 16 mycotoxins in maize. An ad hoc SPE clean-up was optimized by comparing four different approaches with Mycospin 400 column enabling acceptable recoveries for all mycotoxins including ochratoxin A and sterigmatocystin, which showed lower recoveries in the other tested protocols. The application of the developed method on 80 maize samples collected from Shandong Province in China showed that more than 70% of samples were contaminated with at least one of targeted mycotoxins. Another application of UPLC-MS/MS was described for determination of citrinin and ochratoxin A in a variety of food and feed matrices [50][59]. A QuEChERS-based extraction and 5-fold concentration were proposed as a sample preparation protocol. The developed method was validated using the criteria described by Commission Regulation No. 401/2006/EC [43][52] and Commission Decision No. 2002/657/EC [51][17] as guidance. The validated LC–MS/MS method was applied to evaluate the occurrence of target toxins in 90 Belgian foodstuffs. A comparison of different approaches commonly used for the LC-MS/MS analysis of 12 mycotoxins in cereal foods was carried out by Solfrizzo et al. [52][60]. In particular, the study compared 12 analytical methods with different extraction solvents, purification strategies (i.e., SPE, QuEChERS, and IAC), and calibration approaches (i.e., external or matrix-matched). The method providing the best results, based on water/methanol extraction followed by two consecutive IAC clean-up and external calibrations, was further validated in house on corn, rice and feed according to the EC Regulation 401/2006 [43][52]. Performance in terms of accuracy and precision made this method suitable for regulatory purposes even when using lower-performing LC-MS/MS apparatus. A modified QuEChERS method combined with nano flow LC-HRMS was proposed by Alcantara-Duran et al. [53][61] for the determination of 17 mycotoxins in peanut, almond and pistachio. After comparing two purification procedures, the d-SPE using Enhanced Matrix Removal lipid reduced the matrix effect and improved recoveries for all mycotoxins. The method, validated according to SANTE guidelines [54][62], was applied to the analysis of investigated mycotoxins in 15 samples collected in Spain. Dong et al. [55][63] described the development of a single-step SPE for the analysis of seven mycotoxins in fruit and vegetables by UHPLC-MS/MS using the dynamic MRM approach. The validated method was applied to the analysis of cherry tomato, leafy vegetables, strawberry and tomato samples. Although none of the targeted mycotoxins were detected in fresh samples, the concentration of alternariol, alternariol methyl ether and tenuazonic acid increased after storage experiments. Another comparative study was carried out by Scarpino et al. [56][64] that developed and compared two clean-up methods for the determination of 17 Fusarium and Aspergillus mycotoxins, including emerging and masked mycotoxins. Specifically, the dilute-and-shoot and SPE clean-up approaches were compared in combination with LC-MS/MS analysis. Results, in terms of precision, accuracy and reliability, indicated that both methods, thanks to the reduction of time and cost of the analysis, were promising for high throughput routine multimycotoxin analysis. It was the first time that Oasis® PRiME HLB columns were applied for the clean-up of the targeted mycotoxins considered in the study. Recently, Woo et al. [57][65] described the optimization and validation of an LC-MS/MS method after comparing three different sample preparation procedures (i.e., SPE, QuEChERS, and IAC) for the simultaneous analysis of 20 mycotoxins in doenjang, a Korean fermented soybean paste. The method based on IAC clean-up provided the best results in terms of linearity, precision, recovery, matrix effect, and measurement uncertainty. The validated method was subsequently applied to the analysis of targeted mycotoxins in 60 samples of commercial and homemade doenjang. More than 80% of doenjang samples were contaminated by at least one toxin, with the highest levels and number of co-occurring mycotoxins in homemade products compared to in commercial ones. An LC-MS/MS method for the simultaneous determination of seven major trichothecenes in wheat, wheat flour and wheat crackers was recently validated by a collaborative study involving 15 participant laboratories [58][66]. This study was carried out within the M/520 standardization mandate of the European Commission and performed according to the AOAC/IUPAC International Harmonized Protocol [59][67]. The method was based on acetonitrile–water extraction followed by SPE purification for the sample preparation and on the use of isotopically labelled mycotoxins as internal standards for the LC-MS/MS analysis. The validated method has been adopted as a CEN standard method [60][68]. The necessity to monitor mycotoxins also in dried seafood products has recently led to the development of a sensitive, selective and accurate LC-MS/MS for the quantification of aflatoxin B1, T-2 toxin, ochratoxin A and deoxynivalenol in these specific matrices [61][69]. After comparing different sample preparation approaches, the optimized method was based on an ultrasound-assisted acetonitrile/water extraction followed by a defatting clean-up with n-hexane. The analysis of 40 dried fish, dried shrimps and dried mussels samples highlighted for the first time a high occurrence of these mycotoxins in samples collected in the Zhanjiang region. Gbashi et al. [62][70] proposed for the first time the use of pressurized hot-water extraction (PHWE) methodology for the LC-MS/MS analysis of 15 mycotoxins in maize. Moreover, a chemometric approach, based on central composite design, was used for the optimization of the extraction conditions. The validated method was tested on 25 household maize meal samples from South Africa, with fumonisin B1 being the contaminant with the highest occurring frequency and contents. Although the method PHWE is a promising, suitable, cost-effective and greener alternative to traditional methods, further studies are needed to evaluate the cost-benefit of using this approach. Very recently a rapid and sensitive QuEChERS-UPLC-QTOF method, based on matrix-matched calibration, was developed and validated for the determination of 17 mycotoxins, including emerging ones, in malted barley and beer [63][71]. Validation parameters of the method were evaluated according to the International Conference of Harmonisation (ICH) [64][72] while the uncertainty associated was estimated according to EURACHEM [65][73]. For the EU-regulated mycotoxins, performance parameters fulfilled the EU acceptance criteria [32][41]. A dispersive liquid–liquid microextraction in combination with LC-MS/MS analysis was described for the simultaneous determination of 12 mycotoxins in rice bran [66][74]. The method was optimized by using the multivariate statistical techniques based on response surface methodology in combination with a Box–Behnken design. The optimized method was validated according to the EU Regulation 2002/657 EC [51][17] showing satisfactory validation characteristics. The method was applied to the analysis of 24 rice bran samples, with a rate of 42% of positive samples. Finally, a recent study has proposed a multi-mycotoxins immunoaffinity column (multi-IAC) and LC-MS/MS method to evaluate 10 mycotoxins in traditional Chinese medicinal materials (TCMMs) [67][75]. The method was validated for linearity, precision, recovery, analytical limits, and matrix effect. Furthermore, the method was successfully applied to systematically investigate the co-occurrence and contamination levels of multi-mycotoxins in 30 TCMs and functional foods that were all positive for aflatoxin B1. Despite a limited number of samples, this study showed a great multi-mycotoxins contamination rate (more than five different mycotoxins) in TCMMs. 4. ConclusionsMass spectrometry-based methods (LC-MS) have become an essential tool for the monitoring of natural toxins as well as other chemical contaminants in food in order to ensure the safety of products, preserving consumer’s health. We have reviewed the literature from 2016 to 2021 on the use of LC-MS for the detection of mycotoxins in food. Generally speaking, the most used sample preparation approaches included solid liquid extraction, followed by solid phase extraction column clean-up, or alternatively QuEChERS approaches. For the separation and detection mode, UHPLC is mainly used as an improved approach of chromatographic separation while MS/MS methodology is the current leading approach for detection, mainly for the analysis of known compounds. Specifically, the use of matrixmatched calibration curves and isotopically labelled standards allow compensating matrix effects, improving the accuracy of the methods. However, the use of LC-HRMS has become increasingly common for routine analysis. Indeed, thanks to the untargeted data acquisition, retrospective analysis can be carried out, allowing to screen and quantify also parent and unknown compounds as in the case of unexpected compounds. The LC-HRMS advance is mainly due to the capability of this technique to separate with high accuracy and sensitivity also closely related compounds, such as isomers. Hybrid instruments combining two different types of analyzers, mainly Q-TOF and Q-Orbitrap, are commonly applied to improve the selectivity and sensitivity of these analytical methods. Although the majority of available methods for the detection of natural toxins in food are in-house validated according to national or international protocols, much effort is still necessary to assess the suitability of published LC-MS methods for their standardization through interlaboratory validations according to international guidelines (such as AOAC/IUPAC, ISO, EU-DG SANTE).
5. References
- WHO Natural Toxins in food (Factsheet). Available online: www.who.int/news-room/fact-sheets/detail/natural-toxins-in-food (accessed on April 05, 2022).
- Dolan, L.C.; Matulka, R.A.; Burdock, G.A. Naturally occurring food toxins. Toxins (Basel). 2010, 2, 2289–2332, doi:10.3390/toxins2092289.
- Wong, Y.; Lewis, R.J. Analysis of Food Toxins and Toxicants; Wong, Y.C., Lewis, R.J., Eds.; John Wiley & Sons: Chichester, United Kingdom, 2017; Vol. 1–2.
- Casado, N.; Gañán, J.; Morante-Zarcero, S.; Sierra, I. New advanced materials and sorbent-based microextraction techniques as strategies in sample preparation to improve the determination of natural toxins in food samples. Molecules 2020, 25, 1–31, doi:10.3390/molecules25030702.
- Rodríguez, I.; González, J.M.; Botana, A.M.; Sainz, M.J.; Vieytes, M.R.; Alfonso, A.; Botana, L.M. Analysis of natural toxins by liquid chromatography; In Liquid Chromatography (Second Edition); Fanali, S., Haddad, P.R., Poole, C.F., Riekkola, M.L., Eds.; Elsevier: Amsterdam, Netherlands, 2017; 479-514.
- European Commission. Commission Regulation (EC) No. 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, 5–24.
- European Commission. Commission Regulation (EU) No. 2020/2040 of 11 December 2020 amending Regulation (EC) No. 1881/2006 as regards maximum levels of pyrrolizidine alkaloids in certain foodstuffs. Off. J. Eur. Union 2020, 1–5.
- European Commission. Commission Regulation (EU) No. 2016/239 of 19 February 2016 amending Regulation (EC) No 1881/2006 as regards maximum levels of tropane alkaloids in certain cereal-based foods for infants and young children. Off. J. Eur. Union 2016, 3–5.
- European Commission. Commission Regulation (EU) No. 786/2013of 16 August 2013 amending Annex III to Regulation (EC) No. 853/2004 of the European Parliament and of the Council as regards the permitted limits of yessotoxins in live bivalve mollusc. Off. J. Eur. Union 2013, 1–1.
- European Commission. Commission Regulation (EC) No. 853/2004 of the European Parliament and of the Council of 29 April 2004 laying down specific hygiene rules for on the hygiene of foodstuffs. Off. J. Eur. Union 2004, 1–151.
- EFSA. European Food Safety Authority. Risk assessment of glycoalkaloids in feed and food, in particular in potatoes and potato-derived products. EFSA J. 2020, 18, doi:10.2903/j.efsa.2020.6222.
- European Commission. Commission Regulation (EU) No. 2017/1237 of 7 July 2017 amending Regulation (EC) No. 1881/2006 as regards a maximum level of hydrocyanic acid in unprocessed whole, ground, milled, cracked, chopped apricot kernels placed on the market for the final consumer. Off. J. Eur. Union 2017, 36–38.
- European Commission. Commission Regulation (EC) No. 1334/2008 of the European Parliament and of the Council of 16 December 2008 on flavourings and certain food ingredients with flavouring properties for use in and on foods and amending Council Regulation (EEC) No 1601/91, Regulations (EC) No 2232/96 and (EC) No 110/2008 and Directive 2000/13/EC. Off. J. Eur. Union 2008, 34–50.
- Senyuva, H.Z.; Gökmen, V.; Sarikaya, E.A. Future perspectives in OrbitrapTM-high-resolution mass spectrometry in food analysis: a review. Food Addit. Contam. Part A 2015, 13, 1568–1606. doi.org/10.1080/19440049.2015.1057240
- Gavage, M.; Delahaut, P.; Gillard, N. Suitability of high-resolution mass spectrometry for routine analysis of small molecules in food, feed and water for safety and authenticity purposes: A review. Foods 2021, 10, doi:10.3390/foods10030601.
- European Commission. Commission Decision of 12 August 2002 implementing Council Directive No. 96/23/EC concerning the performance of analytical methods and the interpretation of results. Off. J. Eur. Communities 2002, L221, 8–36.
- Steiner, D.; Malachová, A.; Sulyok, M.; Krska, R. Challenges and future directions in LC-MS-based multiclass method development for the quantification of food contaminants. Anal. Bioanal. Chem. 2021, 413, 25–34, doi:10.1007/s00216-020-03015-7.
- Creppy, E.E. Update of survey, regulation and toxic effects of mycotoxins in Europe. Toxicol. Lett. 2002, 127, 19–28, doi:10.1016/S0378-4274(01)00479-9.
- IARC International Agency for Research on Cancer. Some Industrial Chemical Intermediates; International Agency for Research on Cancer: 150 cours Albert Thomas, 69372 Lyon Cedex 08, France, 2020; Vol. 125; ISBN 9789283201649.
- European Commission. Commission Regulation (EC) No. 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, 5–24.
- European Commission. Commission Regulation (EC) No. 1126/2007 of 28 September 2007 amending Regulation (EC) No. 1881/2006 setting maximum levels for certain contaminants in foodstuffs as regards Fusarium toxins in maize and maize products. Off. J. Eur. Communities 2007, 14–17.
- European Commission. Commission Recommendation of 27 March 2013 on the presence of T-2 and HT-2 toxin in cereals and cereal products (2013/165/EU). Off. J. Eur. Union 2013, 12–15.
- European Commission. Commission Regulation (EU) No 212/2014 of 6 March 2014 amending Regulation (EC) No. 1881/2006 as regards maximum levels of the contaminant citrinin in food supplements based on rice fermented with red yeast Monascus purpureus. Off. J. Eur. Union 2014, 3–4.
- EFSA. European Food Safety Authority. Evaluation of the health risks related to the presence of cyanogenic glycosides in foods other than raw apricot kernels. EFSA J. 2019, 17, doi:10.2903/j.efsa.2019.5662.
- EFSA. European Food Safety Authority. Human and animal dietary exposure to ergot alkaloids European. EFSA J. 2017, 15, 4902, doi:10.2903/j.efsa.2017.4902.
- EFSA. European Food Safety Authority. Dietary exposure assessment to Alternaria toxins in the European population. EFSA J. 2016, 14, doi:10.2903/j.efsa.2016.4654.
- Kovalsky, P.; Kos, G.; Nährer, K.; Schwab, C.; Jenkins, T.; Schatzmayr, G.; Sulyok, M.; Krska, R. Co-Occurrence of regulated, masked and emerging mycotoxins and secondary metabolites in finished feed and maize-an extensive survey. Toxins 2016, 8, 363, doi:10.3390/toxins8120363.
- EFSA. European Food Safety Authority. Alternaria toxins in feed and food. EFSA J. 2011, 9, 2407–2409, doi:10.2903/j.efsa.2011.2407.
- EFSA. European Food Safety Authority. Scientific Opinion on the risk for public and animal health related to the presence of sterigmatocystin in food and feed. EFSA J. 2013, 11, 81, doi:10.2903/j.efsa.2013.3254.
- EFSA. European Food Safety Authority. Scientific Opinion on risks for animal and public health related to the presence of nivalenol in food and feed. EFSA J. 2013, 11, 3262, doi:10.2903/j.efsa.2013.3262.
- EFSA. European Food Safety Authority. Scientific Opinion on the risks to human and animal health related to the presence of beauvericin and enniatins in food and feed. EFSA J. 2014, 12, 3802, doi:10.2903/j.efsa.2014.3802.
- Gruber-Dorninger, C.; Novak, B.; Nagl, V.; Berthiller, F. Emerging Mycotoxins: Beyond Traditionally Determined Food Contaminants. J. Agric. Food Chem. 2017, 7052–7070, doi:10.1021/acs.jafc.6b03413.
- Pascale, M.; De Girolamo, A.; Lippolis, V.; Stroka, J.; Mol, H.G.J.; Lattanzio, V.M.T. Performance evaluation of LC-MS methods for multimycotoxin determination. J. AOAC Int. 2019, 102, 1708–1720, doi: 10.5740/jaoacint.19-0068.
- Leite, M.; Freitas, A.; Silva, A.S.; Barbosa, J.; Ramos, F. Maize (Zea mays L.) and mycotoxins: A review on optimization and validation of analytical methods by liquid chromatography coupled to mass spectrometry. Trends Food Sci. Technol. 2020, 99, 542–565.
- Vargas Medina, D.A.; Bassolli Borsatto, J.V.; Maciel, E.V.S.; Lanças, F.M. Current role of modern chromatography and mass spectrometry in the analysis of mycotoxins in food. TrAC - Trends Anal. Chem. 2021, 135.
- Malachová, A.; Stránská, M.; Václavíková, M.; Elliott, C.T.; Black, C.; Meneely, J.; Hajšlová, J.; Ezekiel, C.N.; Schuhmacher, R.; Krska, R. Advanced LC–MS-based methods to study the co-occurrence and metabolization of multiple mycotoxins in cereals and cereal-based food. Anal. Bioanal. Chem. 2018, 410, 801–825, doi:10.1007/s00216-017-0750-7.
- Righetti, L.; Paglia, G.; Galaverna, G.; Dall’asta, C. Recent advances and future challenges in modified mycotoxin analysis: Why HRMS has become a key instrument in food contaminant research. Toxins (Basel). 2016, 361, doi:10.3390/toxins8120361.
- European Commission. Mandate for Standardisation Addressed to CEN for Methods of Analysis for Mycotoxins in Food 2013. Available online: https://law.resource.org/pub/eu/mandates/m52 0.pdf (accessed on 5 April 2022).
- Juan, C.; Covarelli, L.; Beccari, G.; Colasante, V.; Mañes, J. Simultaneous analysis of twenty-six mycotoxins in durum wheat grain from Italy. Food Control 2016, 62, 322–329, doi:10.1016/j.foodcont.2015.10.032.
- European Commission. Commission Regulation (EU) No 519/2014 of 16 May 2014 amending Regulation (EC) No. 401/2006 as regards methods of sampling of large lots, spices and food supplements, performance criteria for T-2, HT-2 toxin and citrinin and screening methods of analysis. Off. J. Eur. Union 2014, 29–43.
- Shigemi, K.; Kosuge, N.; Waki, M.; Miyazawa, M.; Kanazawa, H. Analysis of fusarium toxins in processed grain products using high-performance liquid chromatography/tandem mass spectrometry. Chromatography 2016, 37, 79–85, doi:10.15583/jpchrom.2016.003.
- Sharmili, K.; Jinap, S.; Sukor, R. Development, optimization and validation of QuEChERS based liquid chromatography tandem mass spectrometry method for determination of multimycotoxin in vegetable oil. Food Control 2016, 70, 152–160, doi:10.1016/j.foodcont.2016.04.035.
- Sun, J.; Li, W.; Zhang, Y.; Hu, X.; Wu, L.; Wang, B. QuEchERS purification combined with ultrahigh-performance liquid chromatography tandem mass spectrometry for simultaneous quantification of 25 mycotoxins in cereals. Toxins (Basel). 2016, 8, doi:10.3390/toxins8120375.
- Wang, Y.; Dong, Y.J.; Li, Z.M.; Deng, L.G.; Guo, C.Y.; Zhang, S.Q.; Li, D.P.; Zhao, S.C. Fast determination of multi-mycotoxins in corn by dispersive solid-phase extraction coupled with ultra-performance liquid chromatography with tandem quadrupole time-of-flight mass spectrometry. J. Integr. Agric. 2016, 15, 1656–1666, doi:10.1016/S2095-3119(15)61287-4.
- Xing, Y.; Meng, W.; Sun, W.; Li, D.; Yu, Z.; Tong, L.; Zhao, Y. Simultaneous qualitative and quantitative analysis of 21 mycotoxins in Radix Paeoniae Alba by ultra-high performance liquid chromatography quadrupole linear ion trap mass spectrometry and QuEChERS for sample preparation. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2016, 1031, 202–213, doi:10.1016/j.jchromb.2016.07.008.
- Zwickel, T.; Klaffke, H.; Richards, K.; Rychlik, M. Development of a high performance liquid chromatography tandem mass spectrometry based analysis for the simultaneous quantification of various Alternaria toxins in wine, vegetable juices and fruit juices. J. Chromatogr. A 2016, 1455, 74–85, doi:10.1016/j.chroma.2016.04.066.
- European Committee for Standardization. Technical report CEN/TR 16059:2010. Food analysis–Performance criteria for single laboratory validated methods of analysis for the determination of Mycotoxins. Technical Committee CEN/TC 275: Brussels, Belgium, 2010.
- Al-Taher, F.; Cappozzo, J.; Zweigenbaum, J.; Lee, H.J.; Jackson, L.; Ryu, D. Detection and quantitation of mycotoxins in infant cereals in the U.S. market by LC-MS/MS using a stable isotope dilution assay. Food Control 2017, 72, 27–35, doi:10.1016/j.foodcont.2016.07.027.
- Annunziata, L.; Stramenga, A.; Visciano, P.; Schirone, M.; De Colli, L.; Colagrande, M.N.; Campana, G.; Scortichini, G. Simultaneous determination of aflatoxins, T-2 and HT-2 toxins, and fumonisins in cereal-derived products by QuEChERS extraction coupled with LC-MS/MS. Anal. Bioanal. Chem. 2017, 409, 5143–5155, doi:10.1007/s00216-017-0462-z.
- Eom, T.; Cho, H.D.; Kim, J.; Park, M.; An, J.; Kim, M.; Kim, S.H.; Han, S.B. Multiclass mycotoxin analysis in edible oils using a simple solvent extraction method and liquid chromatography with tandem mass spectrometry. Food Addit. Contam. - Part A Chem. Anal. Control. Expo. Risk Assess. 2017, 34, 2011–2022, doi:10.1080/19440049.2017.1363416.
- European Commission. Commission Regulation (EC) No. 401/2006 of 23 February 2006 laying down the methods of sampling and analysis for the official control of the levels of mycotoxins in foodstuffs. 2006, 12–34.
- Flores-Flores, M.E.; González-Peñas, E. An LC–MS/MS method for multi-mycotoxin quantification in cow milk. Food Chem. 2017, 218, 378–385, doi:10.1016/j.foodchem.2016.09.101.
- Kim, D.H.; Hong, S.Y.; Kang, J.W.; Cho, S.M.; Lee, K.R.; An, T.K.; Lee, C.; Chung, S.H. Simultaneous determination of multi-mycotoxins in cereal grains collected from South Korea by LC/MS/MS. Toxins (Basel). 2017, 9, 1–6, doi:10.3390/toxins9030106.
- Arroyo-Manzanares, N.; De Ruyck, K.; Uka, V.; Gámiz-Gracia, L.; García-Campaña, A.M.; De Saeger, S.; Diana Di Mavungu, J. In-house validation of a rapid and efficient procedure for simultaneous determination of ergot alkaloids and other mycotoxins in wheat and maize. Anal. Bioanal. Chem. 2018, 410, 5567–5581, doi:10.1007/s00216-018-1018-6.
- De Boevre, M.; Van Poucke, C.; Ediage, E.N.; Vanderputten, D.; Van Landschoot, A.; De Saeger, S. Ultra-high-performance supercritical fluid chromatography as a separation tool for Fusarium mycotoxins and their modified forms. J. AOAC Int. 2018, 101, 627–632, doi:10.5740/jaoacint.17-0336.
- Du, L.J.; Chu, C.; Warner, E.; Wang, Q.Y.; Hu, Y.H.; Chai, K.J.; Cao, J.; Peng, L.Q.; Chen, Y.B.; Yang, J.; et al. Rapid microwave-assisted dispersive micro-solid phase extraction of mycotoxins in food using zirconia nanoparticles. J. Chromatogr. A 2018, 1561, 1–12, doi:10.1016/j.chroma.2018.05.031.
- Li, X.; Liu, B.; Wang, F.; Ma, X.; Li, Z.; Guo, D.; Wang, Y.; Wan, F.; Deng, L.; Zhang, S. Determination of 16 mycotoxins in maize by ultrahigh-performance liquid chromatography–tandem mass spectrometry. Anal. Lett. 2018, 51, 702–716, doi:10.1080/00032719.2017.1344246.
- Meerpoel, C.; Vidal, A.; di Mavungu, J.D.; Huybrechts, B.; Tangni, E.K.; Devreese, M.; Croubels, S.; De Saeger, S. Development and validation of an LC–MS/MS method for the simultaneous determination of citrinin and ochratoxin a in a variety of feed and foodstuffs. J. Chromatogr. A 2018, 1580, 100–109, doi:10.1016/j.chroma.2018.10.039.
- Solfrizzo, M.; Gambacorta, L.; Bibi, R.; Ciriaci, M.; Paoloni, A.; Pecorelli, I. Multimycotoxin analysis by LC-MS/MS in cereal food and feed: Comparison of different approaches for extraction, purification, and calibration. J. AOAC Int. 2018, 101, 647–657, doi:10.5740/jaoacint.17-0339.
- Alcántara-Durán, J.; Moreno-González, D.; García-Reyes, J.F.; Molina-Díaz, A. Use of a modified QuEChERS method for the determination of mycotoxin residues in edible nuts by nano flow liquid chromatography high resolution mass spectrometry. Food Chem. 2019, 279, 144–149, doi:10.1016/j.foodchem.2018.11.149.
- Dong, H.; Xian, Y.; Xiao, K.; Wu, Y.; Zhu, L.; He, J. Development and comparison of single-step solid phase extraction and QuEChERS clean-up for the analysis of 7 mycotoxins in fruits and vegetables during storage by UHPLC-MS/MS. Food Chem. 2019, 274, doi:10.1016/j.foodchem.2018.09.035.
- Scarpino, V.; Reyneri, A.; Blandino, M. Development and comparison of two multiresidue methods for the determination of 17 Aspergillus and Fusarium mycotoxins in cereals using HPLC-ESI-TQ-MS/MS. Front. Microbiol. 2019, 10, doi:10.3389/fmicb.2019.00361.
- Woo, S.Y.; Ryu, S.Y.; Tian, F.; Lee, S.Y.; Park, S.B.; Chun, H.S. Simultaneous determination of twenty mycotoxins in the Korean soybean paste doenjang by LC-MS/MS with immunoaffinity cleanup. Toxins (Basel). 2019, 11, doi:10.3390/toxins11100594.
- De Girolamo, A.; Ciasca, B.; Pascale, M.; Lattanzio, V.M.T. Determination of zearalenone and trichothecenes, including deoxynivalenol and its acetylated derivatives, nivalenol, T-2 and HT-2 toxins, in wheat and wheat products by LC-MS/MS: A collaborative study. Toxins (Basel). 2020, 12, doi:10.3390/toxins12120786.
- EN 17280:2019 Foodstuffs - Determination of zearalenone and trichothecenes including deoxynivalenol and its acetylated derivatives (3-acetyl-deoxynivalenol and 15-acetyl-deoxynivalenol), nivalenol T-2 toxin and HT-2 toxin in cereals and cereal products by LC-MS/MS. European Committee for Standardization: Brussels, Belgium, 2019.
- Deng, Y.; Wang, Y.; Deng, Q.; Sun, L.; Wang, R.; Wang, X.; Liao, J.; Gooneratne, R. Simultaneous quantification of aflatoxin B1, T-2 toxin, ochratoxin A and deoxynivalenol in dried seafood products by LC-MS/MS. Toxins (Basel). 2020, 12, doi:10.3390/toxins12080488.
- Gbashi, S.; Njobeh, P.B.; De Saeger, S.; De Boevre, M.; Madala, N.E. Development, chemometric-assisted optimization and in-house validation of a modified pressurized hot water extraction methodology for multi-mycotoxins in maize. Food Chem. 2020, 307, doi:10.1016/j.foodchem.2019.125526.
- Lago, L.O.; Nievierowski, T.H.; Mallmann, L.P.; Rodrigues, E.; Welke, J.E. QuEChERS-LC-QTOFMS for the simultaneous determination of legislated and emerging mycotoxins in malted barley and beer using matrix-matched calibration as a solution to the commercial unavailability of internal standards for some mycotoxins. Food Chem. 2021, 345, doi:10.1016/j.foodchem.2020.128744.
- Salim, S.A.; Sukor, R.; Ismail, M.N.; Selamat, J. Dispersive Liquid-Liquid Microextraction (DLLME) and LC-MS/MS analysis for multi-mycotoxin in rice bran: method development, optimization and validation. Toxins (Basel). 2021, 13, doi:10.3390/toxins13040280.
- Zhao, D.T.; Gao, Y.J.; Zhang, W.J.; Bi, T.C.; Wang, X.; Ma, C.X.; Rong, R. Development a multi-immunoaffinity column LC-MS-MS method for comprehensive investigation of mycotoxins contamination and co-occurrence in traditional Chinese medicinal materials. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2021, 1178, doi:10.1016/j.jchromb.2021.122730.
- Moreira, R.; Pereira, D.M.; Valentão, P.; Andrade, P.B. Pyrrolizidine alkaloids: Chemistry, pharmacology, toxicology and food safety. Int. J. Mol. Sci. 2018, 19, doi:10.3390/ijms19061668.
- Dusemund, B.; Nowak, N.; Sommerfeld, C.; Lindtner, O.; Schäfer, B.; Lampen, A. Risk assessment of pyrrolizidine alkaloids in food of plant and animal origin. Food Chem. Toxicol. 2018, 115, 63–72, doi:10.1016/J.FCT.2018.03.005.
- European Food Safety Authority. Scientific Opinion on Pyrrolizidine alkaloids in food and feed. EFSA J. 2011, 9, 2406.
- EFSA. European Food Safety Authority. Risks for human health related to the presence of pyrrolizidine alkaloids in honey, tea, herbal infusions and food supplements. EFSA J. 2017, 15, 4908, doi:10.2903/j.efsa.2017.4908.
- European Commission. Commission Regulation (EU) No. 2020/2040 of 11 December 2020 amending Regulation (EC) No. 1881/2006 as regards maximum levels of pyrrolizidine alkaloids in certain foodstuffs. Off. J. Eur. Union 2020, 1–5.
