Earthworms and leeches are sentinel animals that represent the annelid phylum within terrestrial and freshwater ecosystems, respectively. One early stress signal in these organisms is related to innate immunity, but how nanomaterials affect it is poorly characterized. In this survey, we compare the latest literature on earthworm and leeches with examples of their molecular/cellular responses to inorganic (silver nanoparticles) and organic (carbon nanotubes) nanomaterials. A special focus is placed on the role of annelid immunocytes in the evolutionarily conserved antioxidant and immune mechanisms and protein corona formation and probable endocytosis pathways involved in nanomaterial uptake. Our summary helps to realize why these environmental sentinels are beneficial to study the potential detrimental effects of nanomaterials.
- nanoparticles
- innate immunity
- earthworms
- leeches
1. Introduction
[4]
[2]
[7]
2. Risk Assessment of Silver Nanoparticles to Soil Invertebrates
+
+
[12]
+
3
[14]
3. Routes of AgNP Uptake in Invertebrates
The interaction of NPs and immune cells, be it through the protein corona or not, in general leads to uptake and/or activation of signaling cascades downstream of receptor ligation. Here we focus on the routes of NP uptake in invertebrates, for which some insights are available in the literature that may also be relevant in understanding the very process in annelids. According to the general classification for eukaryotes, there are two major endocytic pathways, such as phagocytosis and pinocytosis that can be distinguished based on the cell type-specificity and target entities that are taken up[30]
. Phagocytosis is involved in the immune function against large non-self elements of biological (bacteria and viruses) and chemical (including microparticles) origins and is performed mostly by professional immunocytes, such as macrophages, monocytes, dendritic cells, and neutrophils[31]
. Classically, the process starts with the recognition of opsonized particles, and proceeds to actin-driven ingestion and the forming of the phagolysosome[30]
. Macropinocytosis is another type of actin-dependent bulk engulfment, but it differs from phagocytosis in that it captures solutes in a non-specific manner. During macropinocytosis, membrane protrusions with the help of actin fuse around the solutes, forming 1 µm or bigger macropinosomes, which then merge with lysosomes. Practically, all types of cells are capable of endocytosis, which takes place by at least four different pathways, including micropinocytosis, clathrin-mediated endocytosis (CME), caveolin-mediated endocytosis (CvME), and clathrin/caveolin-independent endocytosis . Briefly, internalization by CME occurs with the help of clathrin, which forms a basket-like structure at the cell membrane, typically around 150 nm in size[30]
. This process, just like phagocytosis, leads to the fusion with lysosomes forming endolysosomes . CvME is characterized by the formation of 50–80 nm flask-shaped membrane invaginations, the main component of which is caveolin, a cholesterol-binding protein. In contrast to abovementioned pathways, a formed caveosome does not contain enzymatic proteins, and the vesicle does not go through lysosomal degradation . Among the clathrin, caveolin-independent endocytic pathways, particularly well described is a mechanism involving 40–50 nm sized cholesterol-rich lipid “rafts”, which participate in sorting of intracellular molecules[34]
; however, these pathways are not reported to be extensively involved in NP uptake ()[35]
.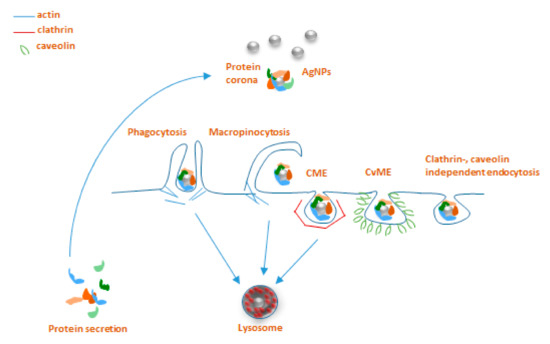
Figure 1. Main endocytosis pathways possibly involved in internalization of AgNPs: phagocytosis, macropinocytosis, clathrin-mediated endocytosis (CME), caveolin-mediated endocytosis (CvME), and clathrin/caveolin-independent endocytosis.
Very limited data are available about the involvement of particular endocytosis pathways for AgNP uptake in invertebrates, with practically no information on these mechanisms for earthworms. On the other hand, somewhat better unravelled AgNPs endocytosis mechanisms have been reported for other invertebrate species of nematodes, snails, and mussels. These studies applied various in vivo and in vitro approaches using pharmacological inhibitors originally developed for use in vertebrates. Endocytic mechanisms of the nematode
Caenorhabditis elegans
[36]
[37]
Peringia ulvae
+
Mytilus galloprovincialis
E. fetida [40]
[41], which might indicate the involvement of this mechanism in earthworms as well. To conclude, the summarized studies give an understanding of evolutionarily conserved processes potentially participating in AgNP uptake in annelids. Nevertheless, the area needs further investigations to obtain the complete picture of the NP entry to the cell and the immune responses in invertebrates.
4. Carbon Nanotubes and Related Toxicological Risks
[47]
[50]
[51]
[5]
4.1. Carbon Nanotubes and Acquatic Invertebrates
[52]
[53]
Artemia salina
[54]
Diopatra neapolitana
D. neapolitana
Hediste diversicolor
[55]
Ficopomatus enigmaticus
[56]
H. diversicolor
[55]
[59]
Crenomytilus grayanus, Swiftopecten swifti, Modiolus modiolus
[62]
S. swifti
[61].
References
- Hansen, S.F.; Michelson, E.S.; Kamper, A.; Borling, P.; Stuer‐Lauridsen, F.; Baun, A. Categorization framework to aid exposure assessment of nanomaterials in consumer products. Ecotoxicology 2008, 17, 438–447, doi:10.1007/s10646‐008‐0210‐4.
- Ray, P.C.; Yu, H.; Fu, P.P. Toxicity and environmental risks of nanomaterials: Challenges and future needs. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2009, 27, 1–35, doi:10.1080/10590500802708267.
- Bayda, S.; Adeel, M.; Tuccinardi, T.; Cordani, M.; Rizzolio, F. The history of nanoscience and nanotechnology: From chemical‐physical applications to nanomedicine. Molecules 2019, 25, 112, doi:10.3390/molecules25010112.
- Prajitha, N.; Athira, S.S.; Mohanan, P.V. Bio‐interactions and risks of engineered nanoparticles. Environ. Res. 2019, 172, 98–108, doi:10.1016/j.envres.2019.02.003.
- Oberdörster, G.; Oberdörster, E.; Oberdörster, J. Nanotoxicology: An emerging discipline evolving from studies of ultrafine particles. Environ. Health Perspect. 2005, 113, 823–839, doi:10.1289/ehp.7339.
- Soni, D.; Naoghare, P.K.; Saravanadevi, S.; Pandey, R.A. Release, transport and toxicity of engineered nanoparticles. Rev. Environ. Contam. Toxicol. 2015, 234, 1–47, doi:10.1007/978‐3‐319‐10638‐0_1.
- Carlson, C.; Hussain, S.M.; Schrand, A.M.; Braydich‐Stolle, L.K.; Hess, K.L.; Jones, R.L.; Schlager, J.J. Unique cellular interaction of silver nanoparticles: Size‐dependent generation of reactive oxygen species. J. Phys. Chem. B. 2008, 112, 13608–13619, doi:10.1021/jp712087m.
- Vance, M.E.; Kuiken, T.; Vejerano, E.P.; McGinnis, S.P.; Hochella, M.F.; Rejesk, D.; Hull, M.S. Nanotechnology in the real world: Redeveloping the nanomaterial consumer products inventory. Beilstein, J. Nanotechnol. 2015, 6, 1769–1780, doi:10.3762/bjnano.6.181.
- Ahmed, K.B.; Nagy, A.M.; Brown, R.P.; Zhang, Q.; Malghan, S.G.; Goering, P.L. Silver nanoparticles: Significance of physicochemical properties and assay interference on the interpretation of in vitro cytotoxicity studies. Toxicol. In Vitro 2017, 38, 179–192, doi:10.1016/j.tiv.2016.10.012.
- Du, J.; Tang, J.; Xu, S.; Ge, J.; Dong, Y.; Li, H.; Jin, M. A review on silver nanoparticles‐induced ecotoxicity and the underlying toxicity mechanisms. Regul. Toxicol. Pharmacol. 2018, 98, 231–239, doi:10.1016/j.yrtph.2018.08.003.
- Shoults‐Wilson, W.A.; Reinsch, B.C.; Tsyusko, O.V.; Bertsch, P.M.; Lowry, G.V.; Unrine, J.M. Effect of silver nanoparticle surface coating on bioaccumulation and reproductive toxicity in earthworms (Eisenia fetida). Nanotoxicology 2011, 5, 432–444, doi:10.3109/17435390.2010.537382.
- Hayashi, Y.; Engelmann, P.; Foldbjerg, R.; Szabó, M.; Somogyi, I.; Pollák, E.; Molnár, L.; Autrup, H.; Sutherland, D.S.; Scott‐Fordsmand, J.; et al. Earthworms and humans in vitro: Characterizing evolutionarily conserved stress and immune responses to silver nanoparticles. Environ. Sci. Technol. 2012, 46, 4166–4173, doi:10.1021/es3000905.
- Zhang, C.; Hu, Z.; Deng, B. Silver nanoparticles in aquatic environments: Physiochemical behavior and antimicrobial mechanisms. Water Res. 2016, 88, 403–427, doi:10.1016/j.watres.2015.10.025.
- Rajala, J.E.; Vehniäinen, E.R.; Väisänen, A.; Kukkonen, J.V.K. Toxicity of silver nanoparticles to Lumbriculus variegatus is a function of dissolved silver and promoted by low sediment pH. Environ. Toxicol. Chem. 2018,37, 1889–1897, doi:10.1002/etc.4136.5624.
- Tang, S.; Zheng, J. Antibacterial activity of silver nanoparticles: Structural effects. Adv. Healthc. Mater. 2018,7, e1701503, doi:10.1002/adhm.201701503.
- Shoults‐Wilson, W.A.; Zhurbich, O.I.; McNear, D.H.; Tsyusko, O.V.; Bertsch, P.M.; Unrine, J.M. Evidence for avoidance of Ag nanoparticles by earthworms (Eisenia fetida). Ecotoxicology 2011, 20, 385–396, doi:10.1007/s10646‐010‐0590‐0.
- Makama, S.; Piella, J.; Undas, A.; Dimmers, W.J.; Peters, R.; Puntes, V.F.; van den Brink, N.W. Properties of silver nanoparticles influencing their uptake in and toxicity to the earthworm Lumbricus rubellus following exposure in soil. Environ. Pollut. 2016, 218, 870–878, doi:10.1016/j.envpol.2016.08.016.
- Exbrayat, J.M.; Moudilou, E.N.; Lapied, E. Harmful effects of nanoparticles on animals. J. Nanobiotechnol. 2015, 2015, 1–10.
- Kim, S.; Ryu, D.Y. Silver nanoparticle‐induced oxidative stress, genotoxicity and apoptosis in cultured cells and animal tissues. J. Appl. Toxicol. 2013, 33, 78–89, doi:10.1002/jat.2792.
- Tao, C. Antimicrobial activity and toxicity of gold nanoparticles: Research progress, challenges and prospects. Lett. Appl. Microbiol. 2018, 67, 537–543, doi:10.1111/lam.13082.
- Crane, J.K. Metal nanoparticles in infection and immunity. Immunol. Invest. 2020, 1–14, doi:10.1080/08820139.2020.1776724.
- Scott‐Fordsmand, J.J.; Krogh, P.H.; Schaefer, M.; Johansen, A. The toxicity testing of double‐walled nanotubes‐contaminated food to Eisenia veneta earthworms. Ecotoxicol. Environ. Saf. 2008, 71, 616–619, doi:10.1016/j.ecoenv.2008.04.011.
- Lapied, E.; Moudilou, E.; Exbrayat, J.M.; Oughton, D.H.; Joner, E.J. Silver nanoparticle exposure causes apoptotic response in the earthworm Lumbricus terrestris (Oligochaeta). Nanomedicine 2010, 5, 975-984, doi:10.2217/nnm.10.58.
- Demuynck, S.; Grumiaux, F.; Mottier, V.; Schikorski, D.; Lemière, S.; Leprêtre, A. Cd/Zn exposure interactions on metallothionein response in Eisenia fetida (Annelida, Oligochaeta). Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2007, 145, 658–668, doi:10.1016/j.cbpc.2007.03.001.
- Irizar, A.; Rodríguez, M.P.; Izquierdo, A.; Cancio, I.; Marigómez, I.; Soto, M. Effects of soil organic matter content on cadmium toxicity in Eisenia fetida: Implications for the use of biomarkers and standard toxicity tests. Arch. Environ. Contam. Toxicol. 2015, 68, 181–192, doi:10.1007/s00244‐014‐0060‐4.
- Calisi, A.; Lionetto, M.G.; Schettino, T. Pollutant‐induced alterations of granulocyte morphology in the earthworm Eisenia foetida. Ecotoxicol. Environ. Saf. 2009, 72, 1369–1377, doi:10.1016/j.ecoenv.2009.03.010.
- Bodó, K.; Hayashi, Y.; Gerencsér, G.; László, Z.; Kéri, A.; Galbács, G.; Telek, E.; Mészáros, M.; Deli, A.M.; Kokhanyuk, B.; et al.; Engelmann, P. Species‐specific sensitivity of Eisenia earthworms towards nobel metal nanoparticles: A multiparametric in vitro study. Environ. Sci. Nano. 2020, doi:10.1039/C9EN0105E.
- Gomes, S.I.L.; Soares, A.M.V.M.; Scott‐Fordsmand, J.J.; Amorim, M.J.B. Mechanisms of response to silver nanoparticles on Enchytraeus albidus (Oligochaeta): Survival, reproduction and gene expression profile. J. Hazard. Mater. 2013, 254‐255, 336–344, doi:10.1016/j.jhazmat.2013.04.005.
- Velicogna, J.R.; Ritchie, E.E.; Scroggins, R.P.; Princz, J.I. A comparison of the effects of silver nanoparticles and silver nitrate on a suite of soil dwelling organisms in two field soils. Nanotoxicology 2016, 10, 1144–1151, doi:10.1080/17435390.2016.1181807.
- Conner, S.D.; Schmid, S.L. Regulated portals of entry into the cell. Nature 2003, 422, 37–44, doi:10/1038/nature01451.
- Aderem, A.; Underhill, D.M. Mechanisms of phagocytosis in macrophages. Annu. Rev. Immunol. 1999, 17, 593–623, doi:10.1146/annurev.immunol.17.1.593.
- Hillaireau, H.; Couvreur, P. Nanocarriers’ entry into the cell: Relevance to drug delivery. Cell. Mol. Life Sci. 2009, 66, 2873–2896, doi:10.1007/s00018‐009‐0053‐z.
- Bareford, L.M.; Swaan, P.W. Endocytic mechanisms for targeted drug delivery. Adv. Drug Deliv. Rev. 2007, 59, 748–758, doi:10.1016/j.addr.2007.06.008
- Edidin, M. Shrinking patches and slippery rafts: Scales of domains in the plasma membrane. Trends Cell Biol. 2001, 11, 492–496, doi:10.1016/s0962‐89.
- Manzanares, D.; Ceña, V. Endocytosis: The nanoparticle and submicron nanocompounds gateway into the cell. Pharmaceutics 2020, 12, 371, doi:10.3390/pharmaceutics12040371
- Maurer, L.L.; Yang, X.; Schindler, A.J.; Taggart, R.K.; Jiang, C.; Hsu‐Kim, H.; Sherwood, D.R.; Meyer, J.N. Intracellular trafficking pathways in silver nanoparticle uptake and toxicity in Caenorhabditis elegans. Nanotoxicology 2016, 10, 831–835, doi:10.3109/17435390.2015.1110759
- Khan, F.R.; Misra, S.K.; Bury, N.R.; Smith, B.D.; Rainbow, P.S.; Luoma, S.N.; Valsami‐Jones, E. Inhibition of potential uptake pathways for silver nanoparticles in the estuarine snail Peringia ulvae. Nanotoxicology 2015, 9, 493–501, doi:10.3109/17435390.2014.948519
- Bouallegui, Y.; Younes, R.B.; Turki, F.; Oueslati, R. Impact of exposure time, particle size and uptake pathway on silver nanoparticle effects on circulating immune cells in Mytilus galloprovincialis. J. Immunotoxicol. 2017, 14, 116–124, doi:10.1080/1547691X.2017.1335810.
- Bouallegui, Y.; Ben Younes, R.; Turki, F.; Mezni, A.; Oueslati, R. Effect of exposure time, particle size and uptake pathways in immune cell lysosomal cytotoxicity of mussels exposed to silver nanoparticles. Drug Chem. Toxicol. 2018, 41, 169–174, doi:10.1080/01480545.2017.1329317
- Novo, M.; Lahive, E.; Diez‐Ortiz, M.; Matzke, M.; Morgan, A.J.; Spurgeon, D.J.; Svendsen, C.; Kille, P. Different routes, same pathways: Molecular mechanisms under silver ion and nanoparticle exposures in the soil sentinel Eisenia fetida. Environ. Pollut. 2015, 205, 385–393, doi:10.1016/j.envpol.2015.07.010
- Benmerah, A. The ciliary pocket. Curr. Opin. Cell Biol. 2013, 25, 78–84, doi:10.1016/j.ceb.2012.10.011
- Mauter, M.S.; Eimelech, M.; Osuji, C.O. Nanocomposites of vertically aligned single‐walled carbon nanotubes by magnetic alignment and polymerization of a lyotropic precursor. ACS Nano 2010, 4, 6651-6658, doi:10.1021/nn102047j.
- Serpell, C.J.; Kostarelos, K.; Davis, B.G. Can carbon nanotubes deliver on their promise in biology; Harnessing unique properties for unparalleled applications. ACS Cent. Sci. 2016, 2, 190–200, doi:10.1021/acscentralsci.6b00005
- Saria, R.; Mouchet, F.; Perrault, A.; Flahaut, E.; Laplanche, C.; Boutonnet, C.; Pinelli, E.; Gauthier, L. Short term exposure to multi‐walled carbon nanotubes induce oxidative stress and DNA damage in Xenopus laevis tadpoles. Ecotox. Environ. Saf. 2014, 107, 22–29, doi:10.1016/j.ecoenv.2014.05.010.
- Pereira, M.M.; Mouton, L.; Yéprémian, C.; Couté, A.; Lo, J.; Marconcini, J.M.; Ladeira, L.O.; Raposo, N.R.; Brandão, H.M.; Brayner, R. Ecotoxicological effects of carbon nanotubes and cellulose nanofibers in Chlorella vulgaris. J. Nanobiotechnol. 2014, 12, 15, doi:10.1186/1477‐3155‐12‐15.
- Velseboer, I.; Peeters, E.T.; Koelmans, A.A. Multiwalled carbon nanotubes at environmentally relevant concentrations affect the composition of benthic communities. Environ. Sci. Technol. 2013, 47, 7475 7482, doi:10.1021/es400777j.
- Funahashi, S.; Okazaki, Y.; Ito, D.; Asakawa, A.; Nagai, H.; Tajima, M.; Toyokuni, S. Asbestos and multiwalled carbon nanotubes generate distinct oxidative responses in inflammatory cells. J. Clin. Biochem. Nutr. 2015, 56, 111–117, doi:10.3164/jcbn.14‐92.
- Poland, C.A.; Duffin, R.; Kinloch, I.; Maynard, A.; Wallace, W.A.; Seaton, A.; Stone, V.; Brown, S.; Macnee, W.; Donaldson, K. Carbon nanotubes introduced into the abdominal cavity of mice show asbestos‐like pathogenicity in a pilot study. Nat. Nanotechnol. 2008, 3, 423–428, doi:10.1038/nnano.2008.111.
- Murphy, F.A.; Schinwald, A.; Poland, C.A.; Donaldson, K. The mechanism of pleural inflammation by long carbon nanotubes: Interaction of long fibres with macrophages stimulates them to amplify proinflammatory responses in mesothelial cells. Part. Fibre Toxicol. 2012, 9, 8, doi:10.1186/1743‐8977‐9‐8
- Dörger, M.; Münzing, S.; Allmeling, A.M.; Messmer, K.; Krombach, F. Differential responses of rat alveolar and peritoneal macrophages to man‐made vitreous fibers in vitro. Environ. Res. 2001, 85, 207–214, doi:10.1006/enrs.2001.4234
- Boyles, M.S.; Young, L.; Brown, D.M.; MacCalman, L.; Cowie, H.; Moisala, A.; Smail, F.; Smith, P.J.; Proudfoot, L.; Windle, A.H.; et al. Multi‐walled carbon nanotube induced frustrated phagocytosis, cytotoxicity and pro-inflammatory conditions in macrophages are length dependent and greater than that of asbestos. Toxicol. Vitro 2015, 29, 1513–1528, doi:10.1016/j.tiv.2015.06.012
- Petersen, E.J.; Zhang, L.; Mattison, N.T.; OʹCarroll, D.M.; Whelton, A.J.; Uddin, N.; Nguyen, T.; Huang, Q.; Henry, T.B.; Holbrook, R.D.; et al. Potential release pathways, environmental fate, and ecological risks of carbon nanotubes. Environ. Sci. Technol. 2011, 45, 9837–9856, doi:10.1021/es201579y.
- Zhang, P.; Selck, H.; Tanga, S.R.; Pang, C.; Zhao, B. Bioaccumulation and effects of sediment‐associated gold‐ and graphene oxide nanoparticles on Tubifex tubifex. J. Environ. Sci. 2017, 51, 138–145, doi:10.1016/j.jes.2016.08.015
- Mesarič, T.; Gambardella, C.; Milivojević, T.; Faimali, M.; Drobne, D.; Falugi, C.; Makovec, D.; Jemec, A.; Sepčić, K. High surface adsorption properties of carbon‐based nanomaterials are responsible for mortality, swimming inhibition, and biochemical responses in Artemia salina larvae. Aquat. Toxicol. 2015, 163, 121–129, doi:10.1016/j.aquatox.2015.03.014
- De Marchi, L.; Neto, V.; Pretti, C.; Figueira, E.; Chiellini, F.; Morelli, A.; Soares, A.; Freitas, R. The influence of salinity on the effects of Multi‐walled carbon nanotubes on polychaetes. Sci. Rep. 2018, 8, 8571, doi:10.1038/s41598‐018‐26729‐2
- Oliva, M.; De Marchi, L.; Vieira S.M.; Pires, A.; Cuccaro, A.; Baratti, M.; Chiellini, F.; Morelli, A.; Freitas, R.; Pretti, C. Atlantic and Mediterranean populations of the widespread serpulid Ficopomatus enigmaticus: Developmental responses to carbon nanotubes. Mar. Pollut. Bull. 2020, 156, 111265, doi:10.1016/j.marpolbul.2020.111265
- Saleh, N.B.; Pfefferle, L.D.; Elimelech, M. Aggregation kinetics of multi‐walled carbon nanotubes in aquatic systems: Measurements and environmental implications. Environ. Sci. Technol. 2008, 42, 7963 7969, doi:10.1021/es801251c.
- Babakhani, P. The impact of nanoparticle aggregation on their size exclusion during transport in porous media: One‐and three‐dimensional modelling investigations. Sci. Rep. 2019, 9, 1–12, doi:10.1038/s41598 019‐50493‐6.
- Ward, J.E.; Kach, D.J. Marine aggregates facilitate ingestion of nanoparticles by suspension feeding bivalves. Mar. Environ. Res. 2009, 68, 137–142, doi:10.1016/j.marenvres.2009.05.002
- Anisimova, A.A.; Chaika, V.V.; Kuznetsov, V.L.; Golokhvast, K.S. Study of the influence of multiwalled carbon nanotubes (12–14 nm) on the main target tissues of the bivalve Modiolus modiolus. Nanotechnol. Russia. 2015, 10, 278–287, doi:10.1134/S1995078015020020.
- Anisimova, A.; Lukyanova, O.; Chaika, V.; Kalitnik, A.; Danilenko, S.; Kuznetsov, V.; Golokhvast, K. S. Short‐time effect of multi‐walled carbon nanotubes on some histological and biochemical parameters in marine bivalves Crenomytilus grayanus (Dunker, 1853) and Swiftopecten swifti (Bernardi, 1858). Nano Hybr. Comp. 2016, 13, 225–231, doi:10.4028/www.scientific.net/NHC.13.225
- Ringwood, A.H.; Levi Polyachenko, N.; Carroll, D.L. Fullerene exposures with oysters: Embryonic, adult, and cellular responses. Environ. Sci. Technol. 2009, 43, 7136–7141, doi:10.1021/es900621j.
