Bile-duct cancers (BDC) are a group of solid tumors arising from the biliary tree. Despite their classification as rare cancers, the incidence of BDC is increasing worldwide. Poor prognosis is a common feature of this type of cancer and is mainly determined by the following factors: late diagnosis, lack of effective therapeutic approaches, and resistance to conventional treatments. In the past few years, next-generation sequencing technologies has allowed us to study the genome, exome, and transcriptome of BDC deeper, revealing a previously underestimated class of RNA: the noncoding RNA (ncRNA). MicroRNAs (miRNAs) are small ncRNAs that play an important regulatory role in gene expression. The aberrant expression of miRNAs and their pivotal role as oncogenes or tumor suppressors in biliary carcinogenesis has been widely described in BDC. Due to their ability to regulate multiple gene networks, miRNAs are involved in all cancer hallmarks, including sustaining proliferative signaling, evading growth suppressors, resisting cell death, enabling replicative immortality, inducing/accessing vasculature, activating invasion and metastasis, reprogramming cellular metabolism, and avoiding immune destruction. Their use as diagnostic, prognostic, and predictive biomarkers has been widely explored in several human cancers, including BDC. Furthermore, miRNA-based therapeutic strategies are currently the subject of numerous clinical trials that are providing evidence of their efficacy as potent anticancer agents.
- non-coding RNA
- microRNA
- bile duct cancer
- precision medicine
1. Introduction
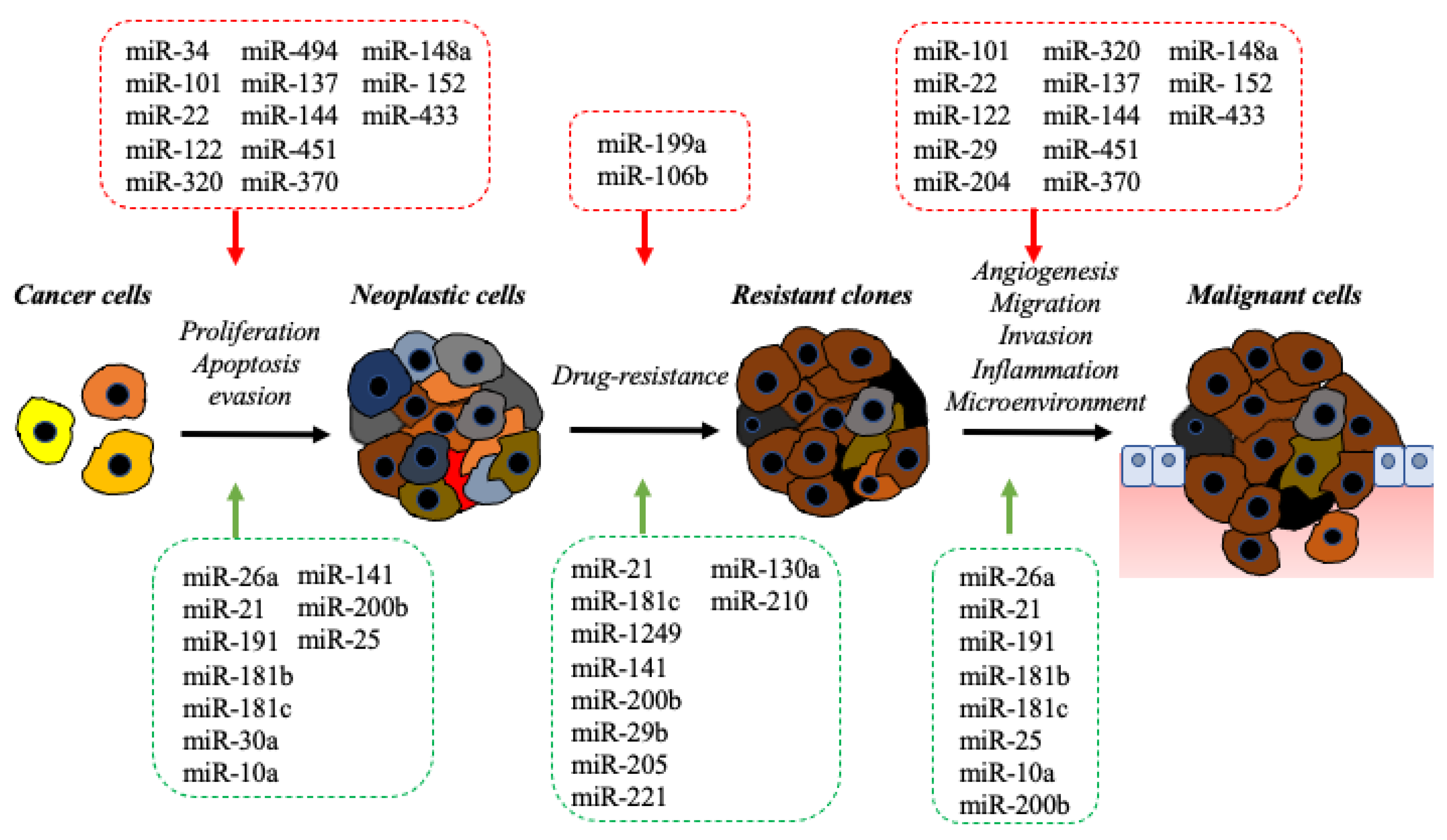
2. The Impact of the microRNA in Molecular Pathology of BDC
|
miRNA |
Target Gene |
Mechanism |
References |
||||
|---|---|---|---|---|---|---|---|
|
miR-26a |
GSK-3β; KRT19 |
Proliferation, migration, and invasion |
|||||
] |
miR-21 |
PTEN; PDCD4; TIMP3; PTPN14; 15-PGDH/HPGD |
Proliferation, apoptosis, EMT, inflammation |
[20, | |||
|
miR-101 |
EZH2, COX-2, APP, MCL-1, VEGF |
Proliferation, apoptosis; angiogenesis, inflammation; transcriptional repression |
|||||
, | , |
miR-191 |
TET |
Proliferation, invasion, and migration | |||
|
miR-22 | [23] |
||||||
SIRT1, CDK6, SP1, HDAC6 |
Proliferation, senescence, invasion, metastasis; ciliogenesis, histone modifications |
miR-181b-5p |
PARK2 |
Proliferation, migration, and invasion |
|||
|
miR-122 | [ | ||||||
ALDOA, CLIC1 |
Proliferation and invasion |
miR-181c |
NDRG2 |
Proliferation, drug-resistance, and metastasis |
[27] |
||
|
MiR-30a-5p |
SOCS3 |
Proliferation |
[28] |
||||
[ | ] | [ |
miR-25 |
DR4 |
Proliferation, invasion, and apoptosis |
||
|
miR-10a-5p |
PTEN |
Proliferation |
[31] |
|
miRNA |
Target Gene |
Mechanism |
References |
|---|---|---|---|
] | |||
|
miR-34 |
MYC, MET, CDK4/6, BCL2, CD44, NOTCH1, NOTCH2, JAGGED1 |
Proliferation, apoptosis |
|
] | |||
|
miR-29-3p |
ITGA6, ITGB1 |
Cell migration and invasion |
[43] |
|
miR-204 |
SLUG |
Cell migration, invasion, EMT |
|
|
miR-320 |
VEGF, NRP-1 |
Proliferation, invasion, EMT, tumor migration, and metastasis |
|
|
miR-494 |
CCNB1, CDK2, CDK4, CDK6, CCND1, CCNE2, HDAC1, RB1, PLK1, PTTG1, TOP2A |
Proliferation, cell cycle |
[48] |
|
miR-137 |
WNT2B |
Proliferation, migration, and invasion |
[49] |
|
miR-144-5p/miR-451 |
ST8SIA4 |
Proliferation, migration, and invasion |
[50] |
|
miR-370 |
MAP3K8 |
Proliferation, inflammation, tumor microenvironment |
|
|
miR-148a |
RASSF1 |
Proliferation, inflammation, tumor microenvironment |
|
|
miR-152 |
CDKN2A |
Proliferation, inflammation, tumor microenvironment |
3. The Impact of miRNAs in Precision Medicine of BDC
4. The Impact of miRNA-Based Therapeutic in BDC
MiRNA-based therapy is emerging as a promising novel strategy to treat cancers. miRNA-based therapeutic approaches operate by silencing overexpressed onco-miRNAs or replacing downregulated oncosuppressor-miRNAs [15]. Several miRNA-based therapeutics are under investigation in clinical trials according to www.clinicaltrials.gov (accessed on 22 January 2022) [90][58]. From the methodological point of view, the following strategies were adopted to efficiently perform onco-miRNA silencing both in vitro and in vivo: anti-miRNA oligonucleotides (AMOs), anti-miRNA locked nucleic acid (LNA), anti-miRNA sponges, and genetic knockouts based on the CRISPR/Cas9 genome-editing technologies [15]. On the other hand, miRNA replacement therapy has generally achieved restoration of onco-suppressor-miRNA by introducing synthetically modified oligonucleotides (miRNA mimics) or viral vectors [15]. However, while promising, miRNA-based therapeutics has raised the following issues to be considered: delivery, selectivity to specific target cells, degradation, and toxicity. Chemical modification of nucleotides or of the RNA backbone through methylation or LNAs, together with the development of vehicles to encapsulate the RNAs, are the main strategies put in place to protect miRNA from degradation [15]. Moreover, toxicity and side effects represent perhaps the biggest obstacle so far encountered in miRNA-based therapeutics [15]. Viral and non-viral vectors have been developed to improve delivery efficiencies to target cells, but the risk of adverse immunogenicity has restricted their use. Lipid-based and polymer-based nanoparticles (NPs) resulted as promising technical approaches as they guarantee efficient delivery and a good safety profile [15,91][15][59]. MRX34, a formulation based on miR-34 mimics in liposomal particles, was the first miRNA restoration strategy performed, subsequently entering a clinical trial recruiting patients with solid tumors, including hepatocellular carcinoma [92][60]. Preliminary results emerging from clinical investigations reported a good safety profile and significant anticancer activity for MRX34 treatment [92][60]. Recent findings showing the inhibition of BDC cell growth by the miR-34 mimic strongly suggested that miRNA-34-based therapy may be a potential efficient and safe therapeutic approach in BDC [32]. Li et al. first found that stroma-derived extracellular vesicles (EVs) containing miR-195 inhibit the proliferation, migration, and metastasis of BDC cells [91][59]. Furthermore, they observed that the systemic injection of miR-195-loaded EVs inhibited BDC tumor growth and prolonged survival in animal models [91][59]. Xie et al. explored a new therapeutic approach based on the use of nanoparticles loaded with a cholesterol-modified polymeric antagonist of CXCR4 (C-X-C receptor type 4) and anti-miR-210 [93][61]. They showed that CXCR4 antagonist- and anti-miR-210-loaded nanoparticles cooperate synergistically in inducing apoptosis and sensitizing BDC cells to Gem/Cis treatment. They presented results showing that the novel nano-therapeutic approach combining the silencing of both CXCR4 and miR-210 suppress tumor growth in BDC cell lines and animal models [93][61]. Another miRNA-based therapeutic approach is represented by the combination of chemotherapy and miRNA to establish a synergistic antitumor effect. Recently, Zhang et al. assembled NPs loaded with gemcitabine—oleic acid prodrugs (GOA) and miR-122 to form GOA/miRNAs NPs [18]. They demonstrated that GOA/miR-122-loaded NPs were efficiently delivered to the tumor area and inhibited hepatocellular carcinoma tumor growth in vivo, without significantly affecting the biosafety profile [18]. To date, miRNA-based therapeutics have been investigated by a huge number of preclinical studies, but only a small number have moved up to clinical trials. Major obstacles are still represented by the delivering of miRNA-based drugs, their stability, and safety profile. However, promising novel strategies are emerging, combining novel delivery platforms and low-toxicity profiles, providing a basis for innovative miRNA-based therapeutic approaches.References
- Rizvi, S.; Khan, S.A.; Hallemeier, C.L.; Kelley, R.K.; Gores, G.J. Cholangiocarcinoma—Evolving concepts and therapeutic strate-gies. Nat. Rev. Clin. Oncol. 2018, 15, 95–111.
- Khan, S.A.; Tavolari, S.; Brandi, G. Cholangiocarcinoma: Epidemiology and risk factors. Liver Int. 2019, 39, 19–31.
- Bertuccio, P.; Malvezzi, M.; Carioli, G.; Hashim, D.; Boffetta, P.; El-Serag, H.B.; Negri, E. Global trends in mortality from intrahepatic and extrahepatic cholangiocarcinoma. J. Hepatol. 2019, 71, 104–114.
- Kelley, R.K.; Bridgewater, J.; Gores, G.J.; Zhu, A.X. Systemic therapies for intrahepatic cholangiocarcinoma. J. Hepatol. 2020, 72, 353–363.
- Braconi, C.; Roessler, S.; Kruk, B.; Lammert, F.; Krawczyk, M.; Andersen, J.B. Molecular perturbations in cholangiocarcinoma: Is it time for precision medicine? Liver Int. 2019, 39, 32–42.
- Adeva, J.; Sangro, B.; Salati, M.; Edeline, J.; La Casta, A.; Bittoni, A.; Berardi, R.; Bruix, J.; Valle, J.W. Medical treatment for cholangiocarcinoma. Liver Int. 2019, 39, 123–142.
- De la Fouchardiere, C. Towards greater clarity in the treatment of cholangiocarcinoma. Lancet Oncol. 2020, 21, 738–739.
- Salati, M.; Caputo, F.; Baldessari, C.; Carotenuto, P.; Messina, M.; Caramaschi, S.; Dominici, M.; Bonetti, L.R. The Evolving Role of FGFR2 Inhibitors in Intrahepatic Cholangiocarcinoma: From Molecular Biology to Clinical Targeting. Cancer Manag. Res. 2021, 13, 7747–7757.
- Franco, B.; Clarke, P.; Carotenuto, P. Pemigatinib. Fibroblast growth factor receptor inhibitor, Treatment of cholangiocarci-noma. Drugs Future 2019, 44, 923.
- Guo, X.; Shen, W. Latest evidence on immunotherapy for cholangiocarcinoma (Review). Oncol. Lett. 2020, 20, 381.
- Carotenuto, P.; Hedayat, S.; Fassan, M.; Cardinale, V.; Lampis, A.; Guzzardo, V.; Braconi, C. Modulation of Biliary Cancer Chemo-Resistance Through MicroRNA-Mediated Rewiring of the Expansion of CD133+ Cells. Hepatology 2020, 72, 982–996.
- Salati, M.; Braconi, C. Noncoding RNA in Cholangiocarcinoma. Semin. Liver Dis. 2018, 39, 013–025.
- Anastasiadou, E.; Jacob, L.S.; Slack, F.J. Non-coding RNA networks in cancer. Nat. Rev. Cancer 2018, 18, 5–18.
- Gebert, L.F.R.; Macrae, I.J. Regulation of microRNA function in animals. Nat. Rev. Mol. Cell Biol. 2019, 20, 21–37.
- Rupaimoole, R.; Slack, F.J. MicroRNA therapeutics: Towards a new era for the management of cancer and other diseases. Nat. Rev. Drug Discov. 2017, 16, 203–222.
- Adrian, C.G.; Dan, D.C.; Shimizu, M.; Bichi, R.; Zupo, S.; Noch, E.; Croce, C.M. Frequent Deletions and Down-Regulation of Micro-RNA Genes miR15 and miR16 at 13q14 in Chronic Lymphocytic Leukemia . Available online: https://www.pnas.org/doi/abs/10.1073/pnas.242606799 (accessed on 22 January 2022).
- Wang, P.; Lv, L. miR-26a induced the suppression of tumor growth of cholangiocarcinoma via KRT19 approach. Oncotarget 2016, 7, 81367–81376.
- Zhang, H.-T.; Sun, J.; Yan, Y.; Cui, S.-H.; Wang, H.; Wang, C.-H.; Zhang, Q. Encapsulated microRNA by gemcitabine prodrug for cancer treatment. J. Control. Release 2019, 316, 317–330.
- Zhang, J.; Han, C.; Wu, T. MicroRNA-26a Promotes Cholangiocarcinoma Growth by Activating β-catenin. Gastroenterology 2012, 143, 246–256.e8.
- Wang, L.-J.; He, C.-C.; Sui, X.; Cai, M.-J.; Zhou, C.-Y.; Ma, J.-L.; Zhu, Q. MiR-21 Promotes Intrahepatic Cholangiocarcinoma Proliferation and Growth In Vitro and In Vivo by Targeting PTPN14 and PTEN . Volume 6. Available online: www.impactjournals.com/oncotarget (accessed on 22 January 2022).
- Liu, Z.; Jin, Z.-Y.; Liu, C.-H.; Xie, F.; Lin, X.-S.; Huang, Q. MicroRNA-21 regulates biological behavior by inducing EMT in human cholangiocarcinoma. Int. J. Clin. Exp. Pathol. 2015, 8, 4684–4694.
- Meng, F.; Henson, R.; Lang, M.; Wehbe, H.; Maheshwari, S.; Mendell, J.T.; Jiang, J.; Schmittgen, T.D.; Patel, T. Involvement of Human Micro-RNA in Growth and Response to Chemotherapy in Human Cholangiocarcinoma Cell Lines. Gastroenterology 2006, 130, 2113–2129.
- Li, H.; Zhou, Z.-Q.; Yang, Z.-R.; Tong, D.-N.; Guan, J.; Shi, B.-J.; Nie, J.; Ding, X.-T.; Li, B.; Zhou, G.-W.; et al. MicroRNA-191 acts as a tumor promoter by modulating the TET1-p53 pathway in intrahepatic cholangiocarcinoma. Hepatology 2017, 66, 136–151.
- Indrieri, A.; Carrella, S.; Carotenuto, P.; Banfi, S.; Franco, B. The Pervasive Role of the miR-181 Family in Development, Neurodegeneration, and Cancer. Int. J. Mol. Sci. 2020, 21, 2092.
- Jiang, Z.L.; Zhang, F.X.; Zhan, H.L.; Yang, H.J.; Zhang, S.Y.; Liu, Z.H.; Ke, R.S. miR-181b-5p Promotes the Progression of Cholangiocarcinoma by Targeting PARK2 via PTEN/PI3K/AKT Signaling Pathway. Biochem. Genet. 2022, 60, 223–240.
- Sun, C.; Zhu, J.; Wu, B.; Chen, J.; Zhu, Z.; Cai, P.; Guo, W.; Gu, Z.; Wang, J.; Huang, S. Diagnostic and prognostic value of microRNAs in cholangiocarcinoma: A systematic review and meta-analysis. Cancer Manag. Res. 2018, 10, 2125–2139.
- Wang, J.; Xie, C.; Pan, S.; Liang, Y.; Han, J.; Lan, Y.; Liu, L. N-myc downstream-regulated gene 2 inhibits human cholangiocarci-noma progression and is regulated by leukemia inhibitory factor/MicroRNA-181c negative feedback pathway. Hepatology 2016, 64, 1606–1622.
- Zhang, J.W.; Wang, X.; Li, G.C.; Wang, D.; Han, S.; Zhang, Y.D.; Luo, C.H.; Wang, H.W.; Jiang, W.J.; Li, C.X.; et al. MiR-30a-5p promotes cholangiocarcinoma cell proliferation through targeting SOCS3. J. Cancer 2020, 11, 3604–3614.
- Liu, H.; Ma, L.; Wang, J. Overexpression of miR-25 is associated with progression and poor prognosis of cholangiocarcinoma. Exp. Ther. Med. 2019, 18, 2687–2694.
- Razumilava, N.; Bronk, S.F.; Smoot, R.L.; Fingas, C.D.; Werneburg, N.W.; Roberts, L.; Mott, J.L. miR-25 targets TNF-related apoptosis inducing ligand (TRAIL) death receptor-4 and promotes apoptosis resistance in cholangiocarcinoma. Hepatology 2011, 55, 465–475.
- Gao, L.; Yang, X.; Zhang, H.; Yu, M.; Long, J.; Yang, T. Inhibition of miR-10a-5p suppresses cholangiocarcinoma cell growth through downregulation of Akt pathway. OncoTargets Ther. 2018, 11, 6981–6994.
- Han, Y.; Meng, F.; Venter, J.; Wu, N.; Wan, Y.; Standeford, H.; Alpini, G. miR-34a-dependent overexpression of Per1 decreases chol-angiocarcinoma growth. J. Hepatol. 2016, 64, 1295–2130.
- Slabáková, E.; Culig, Z.; Remšík, J.; Souček, K. Alternative mechanisms of miR-34a regulation in cancer. Cell Death Dis. 2017, 8, e3100.
- Au, S.L.-K.; Wong, C.C.L.; Lee, J.M.-F.; Fan, D.N.-Y.; Tsang, F.H.; Ng, I.O.-L.; Wong, C.-M. Enhancer of zeste homolog 2 epigenetically silences multiple tumor suppressor microRNAs to promote liver cancer metastasis. Hepatology 2012, 56, 622–631.
- Strillacci, A.; Griffoni, C.; Sansone, P.; Paterini, P.; Piazzi, G.; Lazzarini, G.; Tomasi, V. MiR-101 downregulation is involved in cycloox-ygenase-2 overexpression in human colon cancer cells. Exp. Cell Res. 2009, 315, 1439–1447.
- Su, H.; Yang, J.-R.; Xu, T.; Huang, J.; Xu, L.; Yuan, Y.; Zhuang, S.M. MicroRNA-101, down-regulated in hepatocellular carcinoma, pro-motes apoptosis and suppresses tumorigenicity. Cancer Res. 2009, 69, 1135–1142.
- Zhang, J.; Han, C.; Zhu, H.; Song, K.; Wu, T. miR-101 Inhibits Cholangiocarcinoma Angiogenesis through Targeting Vascular Endothelial Growth Factor (VEGF). Am. J. Pathol. 2013, 182, 1629–1639.
- Wang, J.; Li, Y.; Ding, M.; Zhang, H.; Xu, X.-M.; Tang, J. Molecular mechanisms and clinical applications of miR-22 in regulating malignant progression in human cancer (Review). Int. J. Oncol. 2016, 50, 345–355.
- Mansini, A.P.; Pisarello, M.J.L.; Thelen, K.M.; Cruz-Reyes, M.; Peixoto, E.; Jin, S.; Howard, B.N.; Trussoni, C.E.; Gajdos, G.B.; LaRusso, N.F.; et al. MicroRNA (miR)-433 and miR-22 dysregulations induce histone-deacetylase-6 overexpression and ciliary loss in cholangiocarcinoma. Hepatology 2018, 68, 561–573.
- Fu, X.; Calin, G.A. miR-122 and hepatocellular carcinoma: From molecular biology to therapeutics. EBioMedicine 2018, 37, 17–18.
- Xu, Z.; Liu, G.; Zhang, M.; Zhang, Z.; Jia, Y.; Peng, L.; Zhu, Y.; Hu, J.; Huang, R.; Sun, X. miR-122-5p Inhibits the Proliferation, Invasion and Growth of Bile Duct Carcinoma Cells by Targeting ALDOA. Cell. Physiol. Biochem. 2018, 48, 2596–2606.
- Kong, L.; Wu, Q.; Zhao, L.; Ye, J.; Li, N.; Yang, H. Upregulated lncRNA-UCA1 contributes to metastasis of bile duct carcinoma through regulation of miR-122/CLIC1 and activation of the ERK/MAPK signaling pathway. Cell Cycle 2019, 18, 1212–1228.
- Hozaka, Y.; Seki, N.; Tanaka, T.; Asai, S.; Moriya, S.; Idichi, T.; Wada, M.; Tanoue, K.; Kawasaki, Y.; Mataki, Y.; et al. Molecular Pathogenesis and Regulation of the miR-29-3p-Family: Involvement of ITGA6 and ITGB1 in Intra-Hepatic Cholangiocarcinoma. Cancers 2021, 13, 2804.
- Zhang, L.; Wang, X.; Chen, P. MiR-204 down regulates SIRT1 and reverts SIRT1-induced epithelial-mesenchymal transition, anoikis resistance and invasion in gastric cancer cells. BMC Cancer 2013, 13, 290–299.
- Chung, T.; Lau, T.; Cheung, T.; Yim, S.; Lo, K.; Siu, N.; Chan, L.; Yu, M.; Kwong, J.; Doran, G.; et al. Dysregulation of microRNA-204 mediates migration and invasion of endometrial cancer by regulating FOXC1. Int. J. Cancer 2011, 130, 1036–1045.
- Liang, Y.; Li, S.; Tang, L. MicroRNA 320, an Anti-Oncogene Target miRNA for Cancer Therapy. Biomedicines 2021, 9, 591.
- Zhu, H.; Jiang, X.; Zhou, X.; Dong, X.; Xie, K.; Yang, C.; Jiang, H.; Sun, X.; Lu, J. Neuropilin-1 regulated by miR-320 contributes to the growth and metastasis of cholangiocarcinoma cells. Liver Int. 2017, 38, 125–135.
- Olaru, A.V.; Ghiaur, G.; Yamanaka, S.; Luvsanjav, D.; An, F.; Popescu, I.; Selaru, F.M. MicroRNA down-regulated in human cholangio-carcinoma control cell cycle through multiple targets involved in the G1/S checkpoint. Hepatology 2011, 54, 2089–2098.
- Chen, T.; Lei, S.; Zeng, Z.; Pan, S.; Zhang, J.; Xue, Y.; Sun, Y.; Lan, J.; Xu, S.; Mao, D.; et al. MicroRNA-137 suppresses the proliferation, migration and invasion of cholangiocarcinoma cells by targeting WNT2B. Int. J. Mol. Med. 2020, 45, 886–896.
- Fu, W.; Yu, G.; Liang, J.; Fan, P.; Dong, K.; Zhang, B.; Chen, X.; Zhu, H.; Chu, L. miR-144-5p and miR-451a Inhibit the Growth of Cholangiocarcinoma Cells Through Decreasing the Expression of ST8SIA4. Front. Oncol. 2021, 10, 3104.
- Meng, F.; Wehbe-Janek, H.; Henson, R.; Smith, H.; Patel, T. Epigenetic regulation of microRNA-370 by interleukin-6 in malig-nant human cholangiocytes. Oncogene 2008, 27, 378–386.
- An, F.; Yamanaka, S.; Allen, S.; Roberts, L.R.; Gores, G.J.; Pawlik, T.M.; Xie, Q.; Ishida, M.; Mezey, E.; Ferguson-Smith, A.C.; et al. Silencing of miR-370 in Human Cholangiocarcinoma by Allelic Loss and Interleukin-6 Induced Maternal to Paternal Epigenotype Switch. PLoS ONE 2012, 7, e45606.
- Omar, H.A.; El-Serafi, A.T.; Hersi, F.; Arafa, E.A.; Zaher, D.M.; Madkour, M.; Arab, H.H.; Tolba, M.F. Immunomodulatory MicroRNAs in cancer: Targeting immune checkpoints and the tumor microenvironment. FEBS J. 2019, 286, 3540–3557.
- Shi, T.; Morishita, A.; Kobara, H.; Masaki, T. The Role of microRNAs in Cholangiocarcinoma. Int. J. Mol. Sci. 2021, 22, 7627.
- Hu, J.; Wang, Y.-N.; Song, D.-J.; Tan, J.-P.; Cao, Y.; Fan, J.; Zhou, J. A High-Accuracy Model Based on Plasma miRNAs Diagnoses In-trahepatic Cholangiocarcinoma: A Single Center with 1001 Samples. Diagnostics 2021, 11, 610.
- Macias, R.I.; Banales, J.; Sangro, B.; Muntané, J.; Avila, M.; Lozano, E.; Perugorria, M.J.; Padillo, F.J.; Bujanda, L.; Marin, J. The search for novel diagnostic and prognostic biomarkers in cholangiocarcinoma. Biochim. Biophys. Acta (BBA)—Mol. Basis Dis. 2018, 1864, 1468–1477.
- Lawrie, C.H.; Gal, S.; Dunlop, H.M.; Pushkaran, B.; Liggins, A.P.; Pulford, K.; Banham, A.H.; Pezzella, F.; Boultwood, J.; Wainscoat, J.S.; et al. Detection of elevated levels of tumour-associated microRNAs in serum of patients with diffuse large B-cell lymphoma. Br. J. Haematol. 2008, 141, 672–675.
- Clinical Trial Database. Available online: www.clinicaltrials.gov (accessed on 22 January 2022).
- Li, L.; Piontek, K.; Ishida, M.; Fausther, M.; Dranoff, J.A.; Fu, R.; Selaru, F.M. Extracellular vesicles carry micro RNA -195 to intrahepatic cholangiocarcinoma and improve survival in a rat model. Hepatology 2017, 65, 501–514.
- Hong, D.S.; Kang, Y.-K.; Brenner, A.J.; Sachdev, J.C.; Ejadi, S.; Borad, M.J.; Kim, T.-Y.; Lim, H.Y.; Park, K.; Becerra, C.; et al. MRX34, a liposomal miR-34 mimic, in patients with advanced solid tumors: Final dose-escalation results from a first-in-human phase I trial of microRNA therapy. J. Clin. Oncol. 2016, 34, 2508.
- Xie, Y.; Wang, Y.; Li, J.; Hang, Y.; Jaramillo, L.; Wehrkamp, C.J.; Phillippi, M.A.; Mohr, A.M.; Chen, Y.; Talmon, G.A.; et al. Cholangiocarcinoma therapy with nanoparticles that combine downregulation of MicroRNA-210 with inhibition of cancer cell invasiveness. Theranostics 2018, 8, 4305–4320.
