Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Sirius Huang and Version 1 by Anna-Lena Müller.
In tendinitis, as a very common and painful condition disabling musculoskeletal functionality and still lacking sustainable treatment, various plant-derived compounds have been demonstrated to affect inflammation and promote tissue healing, thus representing promising bio-active treatment agents in tendinitis therapy.
- tendinitis
- tendinopathy
- nutraceuticals
- flavones
- resveratrol
- curcuminoids
- polyphenols
1. Overview
Different natural compounds and polyphenols have been studied in the context of tendinitis, tendon tissue, and inflammation (Table 1). The chemical structures of some compounds are shown in Figure 1.
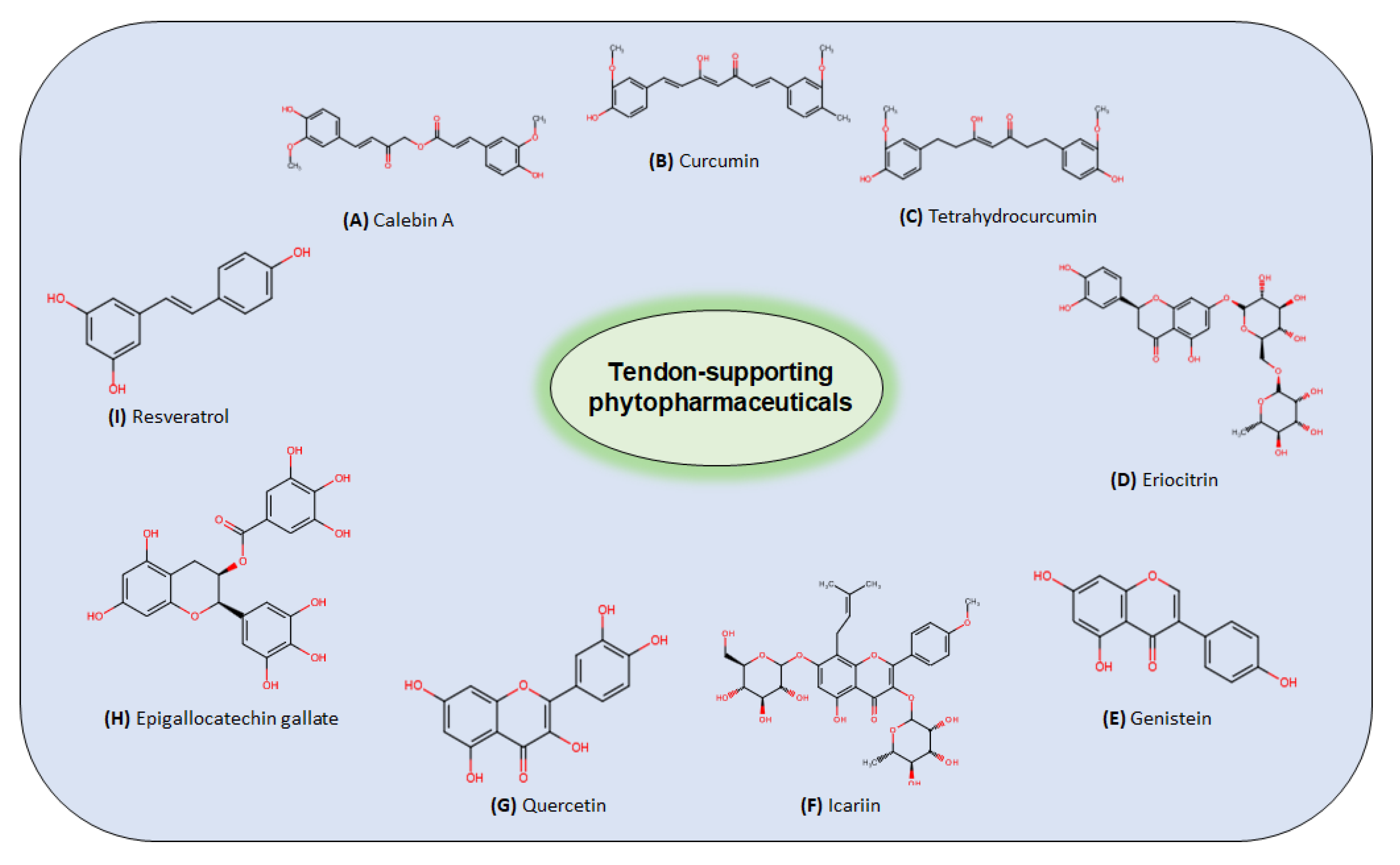
Figure 1. Chemical structures of tendon-supporting phytopharmaceuticals: curcuminoids (A–C), flavones/flavonoids (D–G), green tea extracts (H), and resveratrol (I).
Table 1. Tendon-supporting effects of phytopharmaceuticals.
| Agent | Origin | Type of Trial | Presumed Modulation | Mode of Action | Concentration/Dose Range | Reference |
|---|---|---|---|---|---|---|
| Avocado/Soybean unsaponifiables (ASU) | avocado and soybean oils | in vitro, horse tenocytes |
IL-1β COX-2 PGE2 |
Avocado/soybean unsaponifiables significantly inhibited inflammation response, such as combination therapy with glucosamine and chondroitin sulfate. | 8.3 μg/mL of ASU | [1] |
| Boswellia acid | Boswellia serrata | in vivo, Achilles tendinitis patients |
- | Boswellia acid (as Casperome®) showed pain reduction on a visual analogical scale when Casperome® was administered in addition to physical therapy in patients with Achilles tendinitis. | 250 mg of Casperome® for 15 and 30 days | [2] |
| in vivo, joint inflammation patients |
- | Boswellia acid (as Casperome®) supplementation accompanied by standard therapy reduced pain and inflammation in knee joints and tendon of rugby players. | 500 mg of Casperome® for 5 days, then 250 mg for 23 days | [3] | ||
| in vivo, supraspinatus injury patients |
- | Boswellia serrata and Curcuma longa extracts (as Tendisulfur®) reduced pain after arthroscopic supraspinatus tendon repair compared to placebo treatment. | 2 daily sachets Tendisulfur® for 15 days, then 1 daily sachet for 45 days | [4] |
25]. Besides their anti-oxidant mechanisms, green tea polyphenols are able to stimulate recovery processes during tendinitis, as shown by the promotion of ECM components and glycosaminoglycans, among other recovery elements after Achilles tendinitis in rats, in combination with a glycine diet. In addition, the synthesis of collagen I as a crucial constituent of tendon was remarkably enhanced by green tea treatment. Simultaneously, inflammatory pathways were modulated in the form of decreased MMP-9 levels, which is important for the remodeling of tissue, whereas MMP-2 and IL-1β, both involved in the active remodeling process, remained elevated during green tea treatment, indicating the potential of green tea polyphenols to accelerate tendon tissue remodeling and the regeneration after tendinitis [132,177][23][24]. Altogether, several studies indicate that green tea extracts and EGCG represent promising agents when it comes to fighting ROS and inflammation-triggering stimuli, as well as inflammation itself in tendon tissue (Table 1). However, further research and clinical trials have to be conducted in the future for validation purposes.
5. Flavonoids/Flavones
Flavonoids and flavones (Figure 1) comprise a group of secondary metabolites that are present in a variety of plants, such as celery, parsley, red pepper, chamomile, mint or gingko bilboa, and citrus fruit, and are known to exhibit anti-inflammatory and associated properties in a wide range of chronic diseases [178,179,180,181][61][62][63][64].
5.1. Anthocyanin
In an in vitro study model of rotator cuff-derived tenofibroblasts, the effect of flavonoid anthocyanin extracted from the black soybean (Glycine max (L.) MEER) as anti-oxidants was observed, thus reducing apoptosis in oxidation-stressed tenofibroblasts. Moreover, the suppression of activation of ERK1/2 and JNK accompanied by decreased ROS levels has been assumed to be an important mechanism in the effect of anthocyanins, as, in contrast, ERK1/2 and JAK were up-regulated in H2O2-induced apoptotic tenofibroblast cultures without anthocyanin treatment, indicating anthocyanins therapeutic potential in rotator cuff tendon that is exposed to oxidative stressors (Table 1) [133][27].
5.2. Eriocitrin
In previous research by Shang et al., eriocitrin, as another flavonoid derived from lemon and limes, was investigated for its potential as a bio-active reagent in tendon stem cells in vitro. Eriocitrin that has been described as the most potent anti-oxidant in citrus fruits was found to stimulate tendon stem cell proliferation and to enhance their migration activity, as an important feature of tissue healing and regeneration [147,182,183][28][65][66]. In addition, not only could pro-apoptotic caspase-3 activity be reduced in a concentration-dependent manner, but also the extent of scar formation, determined by scar-formation-related markers fibronectin and biglycan, could be markedly reduced in tenocyte stem cells when treated with eriocitrin, making it another promising plant-derived agent in tendinitis therapy (Table 1) [147][28].
5.3. Genistein
In the context of estrogen deficiency, as experienced in menopause, which consequently promotes a loss of collagen and increases the risk of tendinopathies, the flavone and phyto-oestrogen genistein was investigated on collagen synthesis and Achilles tendon in ovariectomized rats in previous research [184,185,186][29][30][67]. Ramos et al. found evidence that genistein was able to prevent collagen loss in the Achilles tendon of rats in a postmenopausal state, underlining its potential for reducing tendinitis risk in women undergoing estrogen deficiency [184][29]. Moreover, it has been recently demonstrated that genistein improved functional features of Achilles tendon in estrogen-deficient rats, mostly by modulating proliferation-related gene expression (tenomodulin, proliferating cell nuclear antigen (Pcna)), as opposed to collagen remodeling (Table 1) [185][30]. However, further studies have to be conducted in the future to verify this hypothesis.
5.4. Icariin
Icariin is the most abundant flavonoid in horny goat weed (Epimedium grandiflorum) and has been demonstrated to positively affect bone metabolism, regeneration, and density [187,188][68][69]. In a study by Ye et al., flavonoids have been shown to also support the healing and repair of tendon after rotator cuff reconstruction in rats in vivo, mainly by stimulating the synthesis of collagen type I and II. Moreover, the inhibition of bone loss and the promotion of osteogenesis and angiogenesis, as shown by vascular staining, clearly demonstrating enhanced CD31 (platelet endothelial cell adhesion molecule) and VEGF expression as a sign for intrinsic neovascularization around the tendon insertion site through icariin treatment, are assumed to be crucial components for tendon-bone healing (Table 1) [189][31]. On this background, icariin represents another flavonoid with therapeutic potential when it comes to tendinitis and tendon-associated issues, but still, cellular and molecular mechanisms have to be investigated in the future.
5.5. Quercetin
The polyphenolic flavonoid quercetin that is found in fruit and vegetables shows unique bio-active properties, such as anti-cancer, anti-inflammation, anti-oxidant, and anti-viral effects, which thus might play a role in health promotion and disease prevention, as well as in treatment [190,191,192][70][71][72]. Interestingly, previous studies have demonstrated that anti-oxidation methods reducing ROS could reduce the extent of tendon adhesion [193,194][73][74]. Furthermore, in a recent in vivo study by Liang et al., it has been demonstrated that quercetin treatment of rat tendon adhesion models can lead to increased anti-oxidant enzyme activity, as shown by enhanced levels of glutathione peroxidase and superoxide dismutase, while MDA levels were reduced in a concentration-dependent manner. In addition, histological analysis showed a lower extent of tendon adhesion in the rats treated with higher concentrations of quercetin, and no side effects or toxicity of quercetin therapy were observed [195][32].
Furthermore, Semis et al. found proof of quercetin exhibiting anti-inflammatory, anti-apoptotic, and anti-oxidant activity on rat tendon in vivo after collagenase-induced tendinitis. Moreover, inflammatory markers such as MMPs, ICAM-1, and STAT3 were activated by tendinitis induction, whereas all of them could be remarkably suppressed by quercetin administration, making it a promising agent for tendon damage protection [149][33]. In a study by Fu et al., quercetin has been investigated in combination with kaempferol and isorhamnetin as total flavones of sea buckthorn (Hippophae rhamnoides) for the healing of patellar tendon in rats. Within the study, it has been observed that the administration of the flavones not only improved the stress of healing tendons, especially at early stages, but also fiber alignment, collagen deposition, healing, and the recovery of the patellar tendon, suggesting that they could be effective for the improved recovery of tendon injuries (Table 1) [144][34].
6. Resveratrol
Resveratrol (Figure 1) is a polyphenolic compound that is found in a variety of plants, including grapes, peanuts, or mulberries. It has been reported as a health-beneficial agent with pharmacological activity positively affecting inflammation, ROS, cancer, diabetes, obesity, and other chronic diseases [117,128,196,197,198,199,200,201][75][76][77][78][79][80][81][82]. Moreover, resveratrol has been found to play a role in the modulation of a wide range of cellular mechanisms and pathways, including growth inhibition, proliferation and differentiation, apoptosis, and inflammation cascades [14,62,71,202][35][37][83][84]. In addition, resveratrol, although its mode of action is not completely understood yet, is known to be a potent activator of Sirt-1; thus, it has a modulatory impact on the Sirt-1 signaling pathway [71,203,204][83][85][86] and acts as NF-κB inhibitor [101,205,206][87][88][89]. Furthermore, in the context of tendinitis, the potential of resveratrol to inhibit the NF-κB signaling pathway, induced by IL-1β and pro-inflammatory gene end products linked with it, has been observed in human tenocytes in vitro. Similarly, IL-1β-promoted PI3K activation has also been described to be suppressed in a dose-dependent manner by resveratrol treatment in tenocytes, comparable to the effects of PI3K inhibitors, suggesting PI3K as a main target signaling pathway of resveratrol in order to suppress NF-κB [14][37]. These anti-inflammatory effects of resveratrol in tenocytes are assumed to be partly associated with the linkage of Sirt-1 and scleraxis, as well as the deacetylation of NF-κB and PI3K [14,62][35][37]. In the same context, it has been demonstrated that resveratrol not only exhibited anti-inflammatory properties, but also stimulated the synthesis of collagens, tenomodulin, and tendon-specific transcription factor scleraxis, which is necessary for tissue vitality and regeneration [14][37]. The healing capabilities of resveratrol in tendon tissue have also been revealed in diabetic rats in vivo suffering from poor wound healing, following enhanced collagen production, vascular proliferation, and higher fibroblast density, which supported the healing process of Achilles tendinitis despite diabetic conditions [61][39]. Interestingly, in another in vitro study by Busch et al., the previously mentioned target of resveratrol, Sirt-1, was down-regulated in human tenocytes, leading to the expression of apoptotic proteins (Bax, caspase-3), the acetylation of p53 tumor suppressor Akt activation, and scleraxis suppression. Resveratrol could only inhibit IL-1β-induced NF-κB activation to down-regulate inflammatory mediators, such as COX-2 and MMP-9 in tenocytes, when Sirt-1 was expressed [62][35]. The role of Sirt-1 in association with resveratrol’s anti-inflammatory effects has also been demonstrated in an in vivo study by Poulsen et al. whereby resveratrol prevented dexamethasone-induced senescence of tenocytes triggered by Sirt-1 inhibition [207][36]. As glucocorticoids are known to promote tendon senescence and collagen attenuation, the effect of resveratrol to activate Sirt-1, thus inhibiting inflammatory and tendon degrading processes, despite glucocorticoid treatment, is of great potential [29,30,208][90][91][92].
Moreover, in a recently conducted study, polydatin, a derivate of resveratrol, was found to exhibit anti-glycation effects in rat tail tendon in vitro. Glycation leads to AGE formation that causes molecular cross-linking of collagen, making it develop resistance to MMPs, leading to strongly reduced collagen turnover, in turn making collagen and tendon less flexible, and increasing the risk of tendinitis. AGE formation has been found to be accelerated by ROS; hence, resveratrol as a plant-derived anti-oxidant could be a potential natural anti-glycation agent for diabetes and could help to prevent diabetes accompanying disorders, such as tendinitis (Table 1) [209][38].
Table 1. Tendon-supporting effects of phytopharmaceuticals.
| Agent | Origin | Type of Trial | Presumed Modulation | Mode of Action | Concentration/Dose Range | Reference | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Avocado/Soybean unsaponifiables (ASU) | avocado and soybean oils | in vitro, horse tenocytes |
IL-1β COX-2 PGE2 |
Avocado/soybean unsaponifiables significantly inhibited inflammation response, such as combination therapy with glucosamine and chondroitin sulfate. | 8.3 μg/mL of ASU | [157] | |||||
| Boswellia acid | Boswellia serrata | in vivo, Achilles tendinitis patients |
- | Boswellia acid (as Casperome®) showed pain reduction on a visual analogical scale when Casperome® was administered in addition to physical therapy in patients with Achilles tendinitis. | 250 mg of Casperome® for 15 and 30 days | [210] | |||||
| in vivo, joint inflammation patients |
- | Boswellia acid (as Casperome®) supplementation accompanied by standard therapy reduced pain and inflammation in knee joints and tendon of rugby players. | 500 mg of Casperome® for 5 days, then 250 mg for 23 days | [211] | |||||||
| in vivo, supraspinatus injury patients |
- | Boswellia serrata and Curcuma longa extracts (as Tendisulfur®) reduced pain after arthroscopic supraspinatus tendon repair compared to placebo treatment. | 2 daily sachets Tendisulfur® for 15 days, then 1 daily sachet for 45 days | [212] | in vivo, tendinopathy patients |
- | Boswellia serrata and Curcuma longa extracts alleviated the symptoms (pain and functional limitation) of patients with tendon disease when applied as combinational therapy. | 2 tablets twice a day for 1 month | [5] | ||
| in vivo, tendinopathy patients |
- | Boswellia serrata and Curcuma longa extracts alleviated the symptoms (pain and functional limitation) of patients with tendon disease when applied as combinational therapy. | 2 tablets twice a day for 1 month | [65] | in vivo, rotator cuff tendinopathy, Achilles tendinopathy, and lateral epicondylitis patients |
- | Boswellia serrata, | ||||
| in vivo, rotator cuff tendinopathy, Achilles tendinopathy, and lateral epicondylitis patients | Curcuma longa and bromelain extracts (with methyl-sulfonyl-methane, hydrolysed collagen I and II, L-arginine, L-lysin, vitamin C, chondroitin sulfate, glucosamine, and myrrh as Tendisulfur | - | ® Forte), in combination with extracorporeal shock wave therapy, accelerated pain relief and remarkably reduced NSAID intake of patients. | Boswellia serrata, Curcuma longa | 2 daily tablets of Tendisulfur® Forte for 1 month, then once a day for a month | and bromelain extracts (with methyl-sulfonyl-methane, hydrolysed collagen I and II, L-arginine, L-lysin, vitamin C, chondroitin sulfate, glucosamine, and myrrh as Tendisulfur® Forte), in combination with extracorporeal shock wave therapy, accelerated pain relief and remarkably reduced NSAID intake of patients. | [6] | ||||
| 2 daily tablets of Tendisulfur | ® | Forte for 1 month, then once a day for a month | [213] | Bromelain | pineapple extracts, Ananas cosmosus | in vivo, Sprague–Dawley rats |
MDA ROS |
Pineapple flesh extract stimulated tenoblast proliferation and thus tendon healing after Achilles tendon injury. | 30 mg/kg of pineapple flesh axtract for 14 days | [7] | |
| Bromelain | pineapple extracts, Ananas cosmosus | in vivo, Sprague–Dawley rats |
MDA ROS |
Pineapple flesh extract stimulated tenoblast proliferation and thus tendon healing after Achilles tendon injury. | 30 mg/kg of pineapple flesh axtract for 14 days | [161] | in vivo, Sprague–Dawley rats |
ROS PAF |
Pineapple extract bromelain shifted the thromboxane–prostacyclin ratio towards prostacyclin and increased the tenocyte population after Achilles tendon injury. | 7 mg/kg of bromelain for 14 days | [8][9] |
| in vivo, Sprague–Dawley rats |
ROS PAF |
Pineapple extract bromelain shifted the thromboxane–prostacyclin ratio towards prostacyclin and increased the tenocyte population after Achilles tendon injury. | 7 mg/kg of bromelain for 14 days | [130,163] | in vivo, Achilles tendinopathy patients |
- | Pineapple extract bromelain (as dietary supplement Tenosan with arginine, collagen, vitamin C, methyl-sulfonyl-methane, VinitroxTM | ||||
| in vivo, Achilles tendinopathy patients | ) boosted the efficacy of extracorporeal shock wave therapy, resulting in better functional and clinical outcome, compared to placebo treatment. | 2 daily drug sachets containing 50 mg of bromelain for 60 days | [10] | ||||||||
| - | in vivo, rotator cuff tendinopathy patients |
- | Pineapple extract bromelain (as dietary supplement Tenosan with arginine L-alpha-ketoglutarate, methyl-sulfonyl-methane and hydrolysed collagen I) reduced pain and improved repair integrity of rotator cuff repair. | 2 daily drug sachets containing 50 mg of bromelain for 3 months | [11] | ||||||
| Pineapple extract bromelain (as dietary supplement Tenosan with arginine, collagen, vitamin C, methyl-sulfonyl-methane, Vinitrox | TM | ) boosted the efficacy of extracorporeal shock wave therapy, resulting in better functional and clinical outcome, compared to placebo treatment. | 2 daily drug sachets containing 50 mg of bromelain for 60 days | [214] | |||||||
| in vivo, rotator cuff tendinopathy patients |
- | Pineapple extract bromelain (as dietary supplement Tenosan with arginine L-alpha-ketoglutarate, methyl-sulfonyl-methane and hydrolysed collagen I) reduced pain and improved repair integrity of rotator cuff repair. | 2 daily drug sachets containing 50 mg of bromelain for 3 months | [215] | in vivo, rotator cuff tendinopathy, Achilles tendinopathy, and lateral epicondylitis patients |
- | |||||
| in vivo, rotator cuff tendinopathy, Achilles tendinopathy, and lateral epicondylitis patients | Pineapple extract bromelain (with methyl-sulfonyl-methane, hydrolysed collagen I and II, L-arginine, L-lysin, vitamin C, chondroitin sulfate, glucosamine, Curcuma longa, Boswellia serrata, and myrrh as Tendisulfur® Forte), in combination with extracorporeal shock wave therapy, accelerated pain relief and remarkably reduced NSAID intake of patients. | 2 daily tablets of Tendisulfur® Forte for 1 month, then once a day for an additional month | [6] | ||||||||
| - | Pineapple extract bromelain (with methyl-sulfonyl-methane, hydrolysed collagen I and II, L-arginine, L-lysin, vitamin C, chondroitin sulfate, glucosamine, Curcuma longa, Boswellia serrata, and myrrh as Tendisulfur® Forte), in combination with extracorporeal shock wave therapy, accelerated pain relief and remarkably reduced NSAID intake of patients. | 2 daily tablets of Tendisulfur® Forte for 1 month, then once a day for an additional month | [213] | Curcuminoids | turmeric, Curcuma longa | in vitro, canine tenocytes |
NF-κB scleraxis TNF-α TNF-β |
Calebin A suppressed inflammation and exhibited potential as preventive and therapeutic treatment of tendinitis by suppressing down-regulation of tenomodulin and collagen I. | |||
| Curcuminoids | turmeric, Curcuma longa | in vitro, | 1–10 µM of calebin A | canine tenocytes |
NF-κB scleraxis TNF-α | [12] | |||||
| TNF-β | Calebin A suppressed inflammation and exhibited potential as preventive and therapeutic treatment of tendinitis by suppressing down-regulation of tenomodulin and collagen I. | 1–10 µM of calebin A | [36] | in vitro, human tenocytes |
NF-κB IL-1βPI3K/p85/Akt MMPs COX-2 caspase-3 Bax/Bcl-2 |
Curcumin inhibited inflammation and apoptosis and showed potential for treatment of tendon inflammation. | 5 µM of curcumin | [13] | |||
| in vitro, human tenocytes |
NF-κB IL-1βPI3K/p85/Akt MMPs COX-2 caspase-3 Bax/Bcl-2 |
Curcumin inhibited inflammation and apoptosis and showed potential for treatment of tendon inflammation. | 5 µM of curcumin | [10] | in vivo, diabetic rats |
ROS AGE |
Curcumin reduced oxidative stress by inhibiting lipid peroxidation and prevented glycation and crosslinking of advanced glycated collagen in tail tendon and skin. | 200 mg/kg of curcumin for 8 weeks | [14] | ||
| in vivo, Sprague–Dawley rats | |||||||||||
| in vivo, diabetic rats |
ROS AGE |
Curcumin reduced oxidative stress by inhibiting lipid peroxidation and prevented glycation and crosslinking of advanced glycated collagen in tail tendon and skin. | 200 mg/kg of curcumin for 8 weeks | [168] | MDA HOPro SOD |
Curcumin improved the healing quality of tendon ruptures by promoting well-organized collagen filaments and biomechanical traits. | |||||
| in vivo, | 100 mg/kg of curcumin for 14 days | Sprague–Dawley rats |
MDA HOPro SOD | [15] | |||||||
| Curcumin improved the healing quality of tendon ruptures by promoting well-organized collagen filaments and biomechanical traits. | 100 mg/kg of curcumin for 14 days | [64] | in vivo, Sprague–Dawley rats |
adhesion of inflammatory products | Curcumin (as loaded nanoparticle) promoted the healing process of Achilles tendon rupture. | 1 injection containing 0.44 mg of curcumin/kg | |||||
| in vivo, Sprague–Dawley rats | [ | 16 | ] | ||||||||
| adhesion of inflammatory products | Curcumin (as loaded nanoparticle) promoted the healing process of Achilles tendon rupture. | 1 injection containing 0.44 mg of curcumin/kg | [169] | in vivo, Wistar albino rats |
|||||||
| in vivo, | - | Wistar albino rats | - | Curcumin showed biomechanical and histological healing (collagen I and III) promotion after surgically treated Achilles tendon ruptures. | 200 mg/kg of curcumin for 28 days | [17] | |||||
| Curcumin showed biomechanical and histological healing (collagen I and III) promotion after surgically treated Achilles tendon ruptures. | 200 mg/kg of curcumin for 28 days | [170] | in vivo, rats |
AP | Curcumin prevented tendon calcification and improved tendon regeneration by tendon stem/progenitor cells. | ||||||
| in vivo, | 3 μg of curcumin every 3 days for up to 4 weeks | rats |
AP | Curcumin prevented tendon calcification and improved tendon regeneration by tendon stem/progenitor cells. | 3 μg of curcumin every 3 days for up to 4 weeks | [18] | |||||
| [ | 131 | ] | in vivo, Sprague–Dawley rats |
||||||||
| in vivo, Sprague–Dawley rats | ROS IL-1β TNF-α MMPs |
ROS | Curcumin showed anti-oxidative and anti-inflammatory properties as part of Cur&Mg-QCS/PF hydrogel application. | 1 injection with 50 µL of hydrogel | [19] | ||||||
| IL-1β | TNF-α MMPs |
Curcumin showed anti-oxidative and anti-inflammatory properties as part of Cur&Mg-QCS/PF hydrogel application. | 1 injection with 50 µL of hydrogel | [146] | in vivo, diabetic rats |
AGE HOPro |
Curcumin’s metabolite tetrahydrocurcumin reduced accumulation and crosslinking of advanced glycated collagen. | 80 mg/kg of tetrahydrocurcumin for 45 days | [20] | ||
| in vivo, diabetic rats |
AGE HOPro |
Curcumin’s metabolite tetrahydrocurcumin reduced accumulation and crosslinking of advanced glycated collagen. | 80 mg/kg of tetrahydrocurcumin for 45 days | [172] | in vivo, tendinopathy patients |
||||||
| in vivo, tendinopathy patients | - | Curcuma longa and Boswellia serrata extracts alleviated the symptoms (pain and functional limitation) of patients with tendon disease as combinational therapy. | 2 tablets twice a day for 1 month | [5] | |||||||
| - | Curcuma longa and Boswellia serrata extracts alleviated the symptoms (pain and functional limitation) of patients with tendon disease as combinational therapy. | 2 tablets twice a day for 1 month | [65] | in vivo, rotator cuff tendinopathy, Achilles tendinopathy, and lateral epicondylitis patients |
- | ||||||
| in vivo, rotator cuff tendinopathy, Achilles tendinopathy, and lateral epicondylitis patients | Curcuma longa, Boswellia serrata and bromelain extracts (with methyl-sulfonyl-methane, hydrolysed collagen I and II, L-arginine, L-lysin, vitamin C, chondroitin sulfate, glucosamine, and myrrh as Tendisulfur® Forte), in combination with extracorporeal shock wave therapy, accelerated pain relief and remarkably reduced NSAID intake of patients. | - | 2 daily tablets of Tendisulfur | Curcuma longa, Boswellia serrata and bromelain extracts (with methyl-sulfonyl-methane, hydrolysed collagen I and II, L-arginine, L-lysin, vitamin C, chondroitin sulfate, glucosamine, and myrrh as Tendisulfur® Forte), in combination with extracorporeal shock wave therapy, accelerated pain relief and remarkably reduced NSAID intake of patients. | ® Forte for 1 month, then once a day for an additional month | 2 daily tablets of Tendisulfur® Forte for 1 month, then once a day for an additional month | [6] | ||||
| [ | 213 | ] | in vivo, supraspinatus injury patients |
- | Curcuma longa and Boswellia serrata extracts (as Tendisulfur®) reduced pain after arthroscopic supraspinatus tendon repair compared to placebo treatment. | 2 daily sachets of Tendisulfur® for 15 days and 1 daily sachet for the next 45 days | [4] | ||||
| EGCG (epigallocatechin gallate) | green tea extracts | in vitro, human tendon-derived fibroblasts |
IL-1β MMPs p54/JNK/ SAPK collagenases/ gelatinases/ aggrecanases |
Green tea’s epigallocatechin gallate targeted extracellular matrix breakdown. | 2.5–25 µM of epigallocatechin gallate | [21] | |||||
| in vivo, diabetic rats |
AGE | Green tea extract reduced collagen glycation and crosslinking in the tail tendon. | 300 mg/kg of green tea extract for 4 weeks | [22] | |||||||
| in vivo, Wistar rats |
MMPs HOPro |
Green tea promoted the synthesis of ECM components and glycosaminoglycans, and thus the recovery process after Achilles tendinitis in combination with a glycin diet. | 700 mg/kg of green tea extract for 21 days | [23] | |||||||
| in vivo, Wistar rats |
IL-1β MMPs |
Green tea modulated inflammatory action and promoted synthesis of recovery elements after Achilles tendinitis, in combination with a glycin diet. | 700 mg/kg of green tea extract for 7 days | [24] | |||||||
| in vivo, C57BL/6 mice |
ROS | Green tea extract slowed collagen aging by inhibiting crosslinking. | 21.2 mL (young mice) and 27.2 mL (adult mice) of green tea extract for 14 days | [25] | |||||||
| Echinaceae angustifolia extracts | Echinaceae angustifolia | in vivo, carpal tunnel syndrome patients |
- | Echinaceae angustifolia extract (as a dietary supplement mainly composed of alpha lipoic acid and conjugated linoleic acid) showed significant improvement in pain, symptoms, and functionality. | 2 capsules containing 250 mg of echinacea extract for 40 days, then 1 capsule for 80 days | [26] | |||||
| Flavones/Flavonoids | celery, parsley, red peppers, chamomile, mint and gingko bilboa, citrus fruit |
in vitro, rat tenofibroblast |
ROS ERK1/2 JNK |
Flavonoid anthocyanin acted as an anti-apoptotic and showed the therapeutic potential of rotator cuff tendon. | 10–200 µg/mL of anthocyanins | [27] | |||||
| in vitro, tendon stem cells |
caspase-3 | Flavonoid eriocitrin inhibited apoptosis and scar formation (biglycan, fibronectin, COMP) and improved woundhealing by stimulating proliferation and migration of tendon stem cells. | 25–75 of µM eriocitrin | [28] | |||||||
| in vivo, Sprague–Dawley rats |
- | Flavone genistein protected ovariectomy-induced collagen reduction in Achilles tendon. | 300 mg/kgof genistein for 6 weeks | [29] | |||||||
| in vivo, Sprague–Dawley rats | |||||||||||
| in vivo, supraspinatus injury patients |
- | Curcuma longa and Boswellia serrata extracts (as Tendisulfur®) reduced pain after arthroscopic supraspinatus tendon repair compared to placebo treatment. | 2 daily sachets of Tendisulfur® for 15 days and 1 daily sachet for the next 45 days | [212] | |||||||
| EGCG (epigallocatechin gallate) | green tea extracts | in vitro, human tendon-derived fibroblasts |
IL-1β MMPs p54/JNK/ SAPK collagenases/ gelatinases/ aggrecanases |
Green tea’s epigallocatechin gallate targeted extracellular matrix breakdown. | 2.5–25 µM of epigallocatechin gallate | [148] | |||||
| in vivo, diabetic rats |
AGE | Green tea extract reduced collagen glycation and crosslinking in the tail tendon. | 300 mg/kg of green tea extract for 4 weeks | [145] | |||||||
| in vivo, Wistar rats |
MMPs HOPro |
Green tea promoted the synthesis of ECM components and glycosaminoglycans, and thus the recovery process after Achilles tendinitis in combination with a glycin diet. | 700 mg/kg of green tea extract for 21 days | [177] | |||||||
| in vivo, Wistar rats |
IL-1β MMPs |
Green tea modulated inflammatory action and promoted synthesis of recovery elements after Achilles tendinitis, in combination with a glycin diet. | 700 mg/kg of green tea extract for 7 days | [132] | |||||||
| in vivo, C57BL/6 mice |
ROS | Green tea extract slowed collagen aging by inhibiting crosslinking. | 21.2 mL (young mice) and 27.2 mL (adult mice) of green tea extract for 14 days | [176] | |||||||
| Echinaceae angustifolia extracts | Echinaceae angustifolia | in vivo, carpal tunnel syndrome patients |
- | Echinaceae angustifolia extract (as a dietary supplement mainly composed of alpha lipoic acid and conjugated linoleic acid) showed significant improvement in pain, symptoms, and functionality. | 2 capsules containing 250 mg of echinacea extract for 40 days, then 1 capsule for 80 days | [216] | |||||
| Flavones/Flavonoids | celery, parsley, red peppers, chamomile, mint and gingko bilboa, citrus fruit |
in vitro, rat tenofibroblast |
ROS ERK1/2 JNK |
Flavonoid anthocyanin acted as an anti-apoptotic and showed the therapeutic potential of rotator cuff tendon. | 10–200 µg/mL of anthocyanins | [133] | |||||
| in vitro, tendon stem cells |
caspase-3 | Flavonoid eriocitrin inhibited apoptosis and scar formation (biglycan, fibronectin, COMP) and improved woundhealing by stimulating proliferation and migration of tendon stem cells. | 25–75 of µM eriocitrin | [147] | |||||||
| in vivo, Sprague–Dawley rats |
- | Flavone genistein protected ovariectomy-induced collagen reduction in Achilles tendon. | 300 mg/kgof genistein for 6 weeks | [184] | Pcna Timp1 |
Flavone genistein enhanced tendon function at an estrogen-deficit through the modulation of tenomodulin. | 6 mg/kg of genistein for 6 weeks | ||||
| in vivo, Sprague–Dawley rats |
Pcna Timp1 | [30] | |||||||||
| Flavone genistein enhanced tendon function at an estrogen-deficit through the modulation of tenomodulin. | 6 mg/kg of genistein for 6 weeks | [185] | in vivo, Sprague–Dawley rats |
AP CD31 VEGF |
Flavonoid icariin supported healing and angiogenesis after rotator cuff reconstruction through promoting collagen I/II. | ||||||
| in vivo, | 0.125 mg/g of icariin for 2 and 4 weeks | Sprague–Dawley rats |
AP CD31 VEGF | [31] | |||||||
| Flavonoid icariin supported healing and angiogenesis after rotator cuff reconstruction through promoting collagen I/II. | 0.125 mg/g of icariin for 2 and 4 weeks | [189] | in vivo, Wistar rats |
ROS | |||||||
| in vivo, Wistar rats | MDA SOD GPX |
ROS MDA SOD GPX | Flavonoid quercetin prevented the adhesion of tendon tissue. | 50–100 mg/kg for 4 weeks | [32] | ||||||
| Flavonoid quercetin prevented the adhesion of tendon tissue. | 50–100 mg/kg for 4 weeks | [195] | in vivo, Sprague–Dawley rats |
MMPs ICAM-1 |
Flavonoid quercetin prevented collagenase-induced tendon damage at Achilles tendinopathy. | 25–50 mg/kg for 7 days | [33] | ||||
| in vivo, rats |
- | Flavonoid quercetin, kaempferol, and isorhamnetin (Hippophae rhamnoides’ flavones) improved fibre alignment, collagen deposition, healing, and recovery after patellar tendon injury. | 1 injection with 0.1 mg of Hippophae rhamnoides’ flavones | [34] | |||||||
| Resveratrol | red grapes, Vitis vinifera |
in vitro, human tenocytes |
NF-κB p53 Sirt-1 IL-1β COX-2 MMPs Akt/scleraxis Bax/caspase-3 |
Resveratrol regulated tenocytes homeostatic and inhibited inflammation of cascades and apoptosis. | 5 µM of resveratrol | [35] | |||||
| in vitro, human tenocytes |
Sirt-1 | Resveratrol averted dexamethasone-induced senescence despite glucocorticoid treatment. | 30 µM of resveratrol | [36] | |||||||
| in vitro, human tenocytes |
NF-κB PI3K IL-1β scleraxis |
Resveratrol inhibited inflammation cascades; prevented apoptosis; and promoted collagen I, collagen III, and tenomodulin expression. | 0.1–20 µM of resveratrol | [37] | |||||||
| in vitro, Wistar rat tail tendon |
ROS | Resveratrol’s derivate polydatin protected from advanced glycation as an anti-oxidant property. | 50–500 µg of polydatin | [38] | |||||||
| in vivo, Sprague–Dawley rats |
- | Resveratrol promoted the collagens and the healing process of Achilles tendinopathy, despite diabetic condition. | 10 mg/kg of resveratrol for 14 days | [39] |
12. Avocado/Soybean Unsaponifiables
Avocado and soybean unsaponifiables (ASUs) are made of oils extracted from fruit and seeds of avocado and soybean that exhibit active anti-inflammatory properties; thus, they are recommended as supplementary treatment in inflammation-linked chronic diseases, such as osteoarthritis (OA) [153,154,155,156][40][41][42][43]. In a recent study by Grzanna et al., the effects of ASU have been examined in IL-1β-induced inflammation horse tenocytes. ASUs were concomitantly administered with glucosamine (GLU) and chondroitin sulfate (CS), because this mixture has been used in osteoarthritis joint-inflammation therapy before. As a result of the study, it has been demonstrated that ASU was able to significantly suppress IL-1β-induced inflammation and to reduce COX-2 and prostaglandin E2 expression, indicating that ASU can act as a potential treatment agent in tendon-associated inflammation for combination therapy with GLU and CS (Table 1) [157][1]. However, because of the small number of studies conducted with ASU in the context of tendinitis, these results must be considered as an only vague hint of ASU’s potential in tendinitis treatment and further research has to be carried out.
2. Bromelain
Bromelain is a natural complex of proteolytic enzymes that are derived from fruit or stem of the Ananas cosmosus (pineapple). It has been used as another phytopharmaceutical in folk medicine for centuries due to its anti-inflammatory, anti-cancer, immune-modulating, as well as anti-thrombotic properties, to name just a few, as well as its safety for administration [158,159,160][44][45][46]. In musculoskeletal injuries or disorders, it is also known to reduce acute pain and swelling [161][7]. In a study by Aiyegbusi et al., the effects of aqueous extracts of different parts of the pineapple plant were investigated on tenocyte proliferation in rats in vivo after crush injury of the Achilles tendon. In addition, the tendon malondialdehyde (MDA) level, which is a marker of oxidative stress, was analyzed [161,162][7][47]. Finally, the extracts of pineapple flesh and bark were found to promote the proliferation of tenoblasts, comparable to untreated tendon, whereas leaves and core extracts negatively affected the proliferation of tenocytes. Using the pineapple flesh extract, even the MDA level in tendon could be alleviated, suggesting that the anti-oxidant properties of the pineapple are located in its flesh, while both flesh and bark showed the potential to promote tendon regeneration and injury healing by promoting tenoblast proliferation [161][7]. The proliferation-stimulating properties of bromelain in tendon were supported by further studies in rats, arguably due to the role for reactive oxygen species (ROS), or, on the other hand, the increased expression of platelet activating factor (PAF) by modulating the cytokine system, leading to the proliferation of tenoblasts [130,163][8][9].
3. Curcuminoids
Curcuminoids (Figure 1) describe a group of bio-active nutraceutical compounds found in the rhizome of turmeric (Curcuma longa) that exhibit considerable inflammation-protective and anti-oxidant properties and have a long history in traditional Ayurvedic and Chinese medicine [34,35,164,165,166][48][49][50][51][52].
3.1. Curcumin
Curcumin is the most abundant polyphenolic compound found in the group of curcuminoids that gives turmeric its characteristic intensive yellow colour [167][53]. Due to its multiple health-beneficial effects in a wide range of chronic diseases, including rheumatoid arthritis (RA), OA, cancer, neurological, cardiovascular or respiratory disorders, it has reached a lot of attention as an anti-inflammatory agent in therapy [35,54,111,127,167][49][53][54][55][56]. As a well-known curcuminoid, curcumin has also been widely investigated in the context of tendon and tendinitis in several studies, where it was proven to stimulate tendon vitality and regeneration [10,64,131,146,168,169,170,171][13][14][15][16][17][18][19][57]. For instance, in previous in vitro research using human tenocyte cultures, it was found that curcumin was able to suppress NF-κB activation triggered by IL-1β through inhibition of IκBα and NF-κB -dependent pro-inflammatory COX-2 and matrix metalloproteinases (MMPs) as well as Bcl-2, Bcl-xL and TRFA-1 that are involved in apoptosis. Moreover, it was demonstrated that these effects were at least partly executed by down-regulation of PI3K/Akt signaling promoted by curcumin, suggesting the polyphenol as an effective treatment option for tendinitis as well as for prophylaxis by its modulation of pro-inflammatory NF-κB signaling [10][13]. The strong anti-inflammatory effect of curcumin was also found in a recent in vivo study by Chen et al., demonstrating the down-regulation of pro-inflammatory ROS, TNF-α, IL-1β and MMPs in rats during curcumin treatment in the form of curcumin/Mg2+ hydrogels as well [146][19].
In addition, in a study by Sajithlal et al. investigating the preventive and therapeutic potential of curcumin in tail tendon of diabetic rats, it has been demonstrated that curcumin treatment significantly reduced oxidative stress by the suppression of lipid peroxidation and even more, prevented increased accumulation of advanced glycation end products and collagen crosslinking in tendon as an issue coming along with diabetes [168][14]. In another study, of diabetic rats similar observations were made using tetrahydro-curcumin (a metabolite of curcumin) for treatment [172][20], showing curcumins potential in tendon-associated therapy as an effective anti-oxidant [168,172][14][20]. Besides its anti-inflammatory effects, curcumin has also been proven in vitro (human tenocytes) and in vivo (rats) to help in tendon regeneration and healing by promoting collagen I and II synthesis [10,170][13][17] and their organization as filaments enhancing tendons biomechanical traits [64][15] as well as by suppressing the peritendinous adhesion of inflammatory products [169][16]. Moreover, curcumin has been shown to prevent tendon calcification in rats as a common issue of late-stage tendinitis due to aberrant tendon stem and progenitor cells osteogenic differentiation triggered by inflammation, by promoting tenogenesis while suppressing osteogenesis at the respective pathological sites [131][18], as summarized in Table 1.
3.2. Calebin A
Besides curcumin, another bio-active compound of turmeric, namely calebin A, has been recently investigated in in vitro tendinitis study models by our group. Within ourne study, calebin A was demonstrated with the ability to suppress inflammatory conditions in tenocytes by inhibiting the NF-κB signaling pathway and its associated gene end products, such as COX-2, MMP-9, and caspase-3, which are responsible for matrix degrading and apoptotic processes, leading to tissue inflammation and degeneration. By suppressing these catabolic events, calebin A helps in inflammation protection and tendon regeneration, which was shown by an increased expression of tendon-specific transcription factor scleraxis, tenomodulin, as well as collagen I. The down-regulation of NF-κB with the simultaneous up-regulation of tenogenic scleraxis indicates a multi-modulatory effect of calebin A in tendinitis by targeting the NF-κB–scleraxis axis (Table 1) [36][12].
Therefore, calebin A represents another powerful nutraceutical with great potential in tendinitis therapy, as well as in its prevention. However, further studies are needed to examine the full potential of the natural compound calebin A in fighting inflammatory and degrading processes in tendinitis [36][12].
4. Green Tea Extracts (Epigallocatechin Gallate)
Epigallocatechin gallate (Figure 1), as the most abundant polyphenol component of green tea, has been shown to be beneficial in many different diseases, including cancer, the metabolic syndrome, and neurodegenerative disorders, because of its anti-oxidant and anti-inflammatory characteristics [173,174,175][58][59][60]. These effects were also found in a study investigating human tendon-derived fibroblasts in vitro, in which EGCG administration suppressed IL-1β-stimulated collagenase and stromelysin, as well as the expression of MMPs, and simultaneously reduced the stimulation of p54/JNK/SAPK phosphorylation. Altogether, these outcomes suggest ECM breakdown as an important target for EGCG and other green tea polyphenols, thus proposing a potential target for EGCG therapy in tendon injury [148][21]. In addition to that, green tea extract has also been shown to remarkably reduce glycation, i.e., the resulting formation of advanced glycation end products (AGE), as well as crosslink collagen in the tail tendon of diabetic rats. These outcomes demonstrate the therapeutic potential of green tea extracts in treating diabetes-related tendon glycation, since the formation of glycation and AGE in tendon finally leads to impaired tendon turnover, making it even more vulnerable to tendinitis [145][22]. Furthermore, in an in vivo study by Rutter et al., it has been demonstrated that green tea polyphenols could prevent collagen aging markers from rising and to even delay collagen crosslinking by anti-oxidant mechanisms in rats, further supporting green tea extract as a promising anti-glycation agent. These effects could be even enhanced by the concomitant supplementation of vitamin C and E [176][
| in vivo, | ||||||
| Sprague–Dawley rats | ||||||
| MMPs | ||||||
| ICAM-1 | ||||||
| Flavonoid quercetin prevented collagenase-induced tendon damage at Achilles tendinopathy. | ||||||
| 25–50 mg/kg for 7 days | ||||||
| [ | ||||||
| 149 | ||||||
| ] | ||||||
| in vivo, | ||||||
| rats | ||||||
| - | ||||||
| Flavonoid quercetin, kaempferol, and isorhamnetin ( | ||||||
| Hippophae rhamnoides’ | ||||||
| flavones) improved fibre alignment, collagen deposition, healing, and recovery after patellar tendon injury. | ||||||
| 1 injection with 0.1 mg of | ||||||
| Hippophae rhamnoides’ | ||||||
| flavones | ||||||
| [ | ||||||
| 144 | ||||||
| ] | ||||||
| Resveratrol | red grapes, Vitis vinifera |
in vitro, human tenocytes |
NF-κB p53 Sirt-1 IL-1β COX-2 MMPs Akt/scleraxis Bax/caspase-3 |
Resveratrol regulated tenocytes homeostatic and inhibited inflammation of cascades and apoptosis. | 5 µM of resveratrol | [62] |
| in vitro, human tenocytes |
Sirt-1 | Resveratrol averted dexamethasone-induced senescence despite glucocorticoid treatment. | 30 µM of resveratrol | [207] | ||
| in vitro, human tenocytes |
NF-κB PI3K IL-1β scleraxis |
Resveratrol inhibited inflammation cascades; prevented apoptosis; and promoted collagen I, collagen III, and tenomodulin expression. | 0.1–20 µM of resveratrol | [14] | ||
| in vitro, Wistar rat tail tendon |
ROS | Resveratrol’s derivate polydatin protected from advanced glycation as an anti-oxidant property. | 50–500 µg of polydatin | [209] | ||
| in vivo, Sprague–Dawley rats |
- | Resveratrol promoted the collagens and the healing process of Achilles tendinopathy, despite diabetic condition. | 10 mg/kg of resveratrol for 14 days | [61] |
References
- Grzanna, M.W.; Au, R.Y.; Au, A.Y.; Rashmir, A.M.; Frondoza, C.G. Avocado/Soybean Unsaponifiables, Glucosamine and Chondroitin Sulfate Combination Inhibits Proinflammatory COX-2 Expression and Prostaglandin E2 Production in Tendon-Derived Cells. J. Med. Food 2020, 23, 139–146.
- Riva, A.; Allegrini, P.; Franceschi, F.; Togni, S.; Giacomelli, L.; Eggenhoffner, R. A novel boswellic acids delivery form (Casperome®) in the management of musculoskeletal disorders: A review. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 5258–5263.
- Franceschi, F.; Togni, S.; Belcaro, G.; Dugall, M.; Luzzi, R.; Ledda, A.; Pellegrini, L.; Eggenhoffner, R.; Giacomelli, L. A novel lecithin based delivery form of Boswellic acids (Casperome®) for the management of osteo-muscular pain: A registry study in young rugby players. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 4156–4161.
- Merolla, G.; Dellabiancia, F.; Ingardia, A.; Paladini, P.; Porcellini, G. Co-analgesic therapy for arthroscopic supraspinatus tendon repair pain using a dietary supplement containing Boswellia serrata and Curcuma longa: A prospective randomized placebo-controlled study. Musculoskelet. Surg. 2015, 99, S43–S52.
- Henrotin, Y.; Dierckxsens, Y.; Delisse, G.; Seidel, L.; Albert, A. Curcuminoids and Boswellia serrata extracts combination decreases tendinopathy symptoms: Findings from an open-label post-observational study. Curr. Med. Res. Opin. 2021, 37, 423–430.
- Vitali, M.; Naim Rodriguez, N.; Pironti, P.; Drossinos, A.; Di Carlo, G.; Chawla, A.; Gianfranco, F. ESWT and nutraceutical supplementation (Tendisulfur Forte) vs. ESWT-only in the treatment of lateral epicondylitis, Achilles tendinopathy, and rotator cuff tendinopathy: A comparative study. J. Drug Assess. 2019, 8, 77–86.
- Aiyegbusi, A.I.; Duru, F.I.; Awelimobor, D.; Noronha, C.C.; Okanlawon, A.O. The role of aqueous extract of pineapple fruit parts on the healing of acute crush tendon injury. Niger. Q. J. Hosp. Med. 2010, 20, 223–227.
- Aiyegbusi, A.I.; Duru, F.I.; Anunobi, C.C.; Noronha, C.C.; Okanlawon, A.O. Bromelain in the early phase of healing in acute crush Achilles tendon injury. Phytother. Res. PTR 2011, 25, 49–52.
- Aiyegbusi, A.I.; Olabiyi, O.O.; Duru, F.I.; Noronha, C.C.; Okanlawon, A.O. A comparative study of the effects of bromelain and fresh pineapple juice on the early phase of healing in acute crush achilles tendon injury. J. Med. Food 2011, 14, 348–352.
- Notarnicola, A.; Pesce, V.; Vicenti, G.; Tafuri, S.; Forcignanò, M.; Moretti, B. SWAAT study: Extracorporeal shock wave therapy and arginine supplementation and other nutraceuticals for insertional Achilles tendinopathy. Adv. Ther. 2012, 29, 799–814.
- Gumina, S.; Passaretti, D.; Gurzì, M.D.; Candela, V. Arginine L-alpha-ketoglutarate, methylsulfonylmethane, hydrolyzed type I collagen and bromelain in rotator cuff tear repair: A prospective randomized study. Curr. Med. Res. Opin. 2012, 28, 1767–1774.
- Mueller, A.L.; Brockmueller, A.; Kunnumakkara, A.B.; Shakibaei, M. Calebin A, a Compound of Turmeric, Down-Regulates Inflammation in Tenocytes by NF-κB/Scleraxis Signaling. Int. J. Mol. Sci. 2022, 23, 1695.
- Buhrmann, C.; Mobasheri, A.; Busch, F.; Aldinger, C.; Stahlmann, R.; Montaseri, A.; Shakibaei, M. Curcumin modulates nuclear factor kappaB (NF-kappaB)-mediated inflammation in human tenocytes in vitro: Role of the phosphatidylinositol 3-kinase/Akt pathway. J. Biol. Chem. 2011, 286, 28556–28566.
- Sajithlal, G.B.; Chithra, P.; Chandrakasan, G. Effect of curcumin on the advanced glycation and cross-linking of collagen in diabetic rats. Biochem. Pharmacol. 1998, 56, 1607–1614.
- Jiang, D.; Gao, P.; Lin, H.; Geng, H. Curcumin improves tendon healing in rats: A histological, biochemical, and functional evaluation. Connect. Tissue Res. 2016, 57, 20–27.
- Zhang, W.; Li, X.; Comes Franchini, M.; Xu, K.; Locatelli, E.; Martin, R.C.; Monaco, I.; Li, Y.; Cui, S. Controlled release of curcumin from curcumin-loaded nanomicelles to prevent peritendinous adhesion during Achilles tendon healing in rats. Int. J. Nanomed. 2016, 11, 2873–2881.
- Güleç, A.; Türk, Y.; Aydin, B.K.; Erkoçak, Ö.F.; Safalı, S.; Ugurluoglu, C. Effect of curcumin on tendon healing: An experimental study in a rat model of Achilles tendon injury. Int. Orthop. 2018, 42, 1905–1910.
- Chen, Y.; Xie, Y.; Liu, M.; Hu, J.; Tang, C.; Huang, J.; Qin, T.; Chen, X.; Chen, W.; Shen, W.; et al. Controlled-release curcumin attenuates progression of tendon ectopic calcification by regulating the differentiation of tendon stem/progenitor cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 103, 109711.
- Chen, B.; Liang, Y.; Zhang, J.; Bai, L.; Xu, M.; Han, Q.; Han, X.; Xiu, J.; Li, M.; Zhou, X.; et al. Synergistic enhancement of tendon-to-bone healing via anti-inflammatory and pro-differentiation effects caused by sustained release of Mg2+/curcumin from injectable self-healing hydrogels. Theranostics 2021, 11, 5911–5925.
- Pari, L.; Murugan, P. Influence of tetrahydrocurcumin on tail tendon collagen contents and its properties in rats with streptozotocin-nicotinamide-induced type 2 diabetes. Fundam. Clin. Pharmacol. 2007, 21, 665–671.
- Corps, A.N.; Curry, V.A.; Buttle, D.J.; Hazleman, B.L.; Riley, G.P. Inhibition of interleukin-1beta-stimulated collagenase and stromelysin expression in human tendon fibroblasts by epigallocatechin gallate ester. Matrix Biol. J. Int. Soc. Matrix Biol. 2004, 23, 163–169.
- Babu, P.V.; Sabitha, K.E.; Shyamaladevi, C.S. Effect of green tea extract on advanced glycation and cross-linking of tail tendon collagen in streptozotocin induced diabetic rats. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2008, 46, 280–285.
- Vieira, C.P.; Guerra Fda, R.; de Oliveira, L.P.; Almeida, M.S.; Marcondes, M.C.; Pimentell, E.R. Green tea and glycine aid in the recovery of tendinitis of the Achilles tendon of rats. Connect. Tissue Res. 2015, 56, 50–58.
- Vieira, C.P.; De Oliveira, L.P.; Da Ré Guerra, F.; Marcondes, M.C.; Pimentel, E.R. Green Tea and Glycine Modulate the Activity of Metalloproteinases and Collagen in the Tendinitis of the Myotendinous Junction of the Achilles Tendon. Anat. Rec. 2016, 299, 918–928.
- Rutter, K.; Sell, D.R.; Fraser, N.; Obrenovich, M.; Zito, M.; Starke-Reed, P.; Monnier, V.M. Green tea extract suppresses the age-related increase in collagen crosslinking and fluorescent products in C57BL/6 mice. Int. J. Vitam. Nutr. Res. 2003, 73, 453–460.
- Notarnicola, A.; Maccagnano, G.; Tafuri, S.; Fiore, A.; Pesce, V.; Moretti, B. Comparison of shock wave therapy and nutraceutical composed of Echinacea angustifolia, alpha lipoic acid, conjugated linoleic acid and quercetin (perinerv) in patients with carpal tunnel syndrome. Int. J. Immunopathol. Pharmacol. 2015, 28, 256–262.
- Park, H.B.; Hah, Y.S.; Yang, J.W.; Nam, J.B.; Cho, S.H.; Jeong, S.T. Antiapoptotic effects of anthocyanins on rotator cuff tenofibroblasts. J. Orthop. Res. 2010, 28, 1162–1169.
- Shang, C.; Tian, Z.; Li, G.; Liu, G.; Zhang, H. Effect of Eriocitrin on Cell Proliferation, Apoptosis, Migration, and Scar Formation-Related Genes Expression in Tendon Stem Cells. Doklady. Biochem. Biophys. 2021, 500, 402–407.
- Ramos, J.E.; Al-Nakkash, L.; Peterson, A.; Gump, B.S.; Janjulia, T.; Moore, M.S.; Broderick, T.L.; Carroll, C.C. The soy isoflavone genistein inhibits the reduction in Achilles tendon collagen content induced by ovariectomy in rats. Scand. J. Med. Sci. Sports 2012, 22, e108–e114.
- Carroll, C.C.; Patel, S.H.; Simmons, J.; Gordon, B.D.; Olson, J.F.; Chemelewski, K.; Saw, S.; Hale, T.M.; Howden, R.; Sabbaghi, A. The Impact of Genistein Supplementation on Tendon Functional Properties and Gene Expression in Estrogen-Deficient Rats. J. Med. Food 2020, 23, 1266–1274.
- Ye, C.; Zhang, W.; Wang, S.; Jiang, S.; Yu, Y.; Chen, E.; Xue, D.; Chen, J.; He, R. Icariin Promotes Tendon-Bone Healing during Repair of Rotator Cuff Tears: A Biomechanical and Histological Study. Int. J. Mol. Sci. 2016, 17, 1780.
- Liang, Y.; Xu, K.; Zhang, P.; Zhang, J.; Chen, P.; He, J.; Fang, Y.; Zhou, Y.; Wang, J.; Bai, J. Quercetin reduces tendon adhesion in rat through suppression of oxidative stress. BMC Musculoskelet. Disord. 2020, 21, 608.
- Semis, H.S.; Gur, C.; Ileriturk, M.; Kandemir, F.M.; Kaynar, O. Evaluation of Therapeutic Effects of Quercetin Against Achilles Tendinopathy in Rats via Oxidative Stress, Inflammation, Apoptosis, Autophagy, and Metalloproteinases. Am. J. Sports Med. 2022, 50, 486–498.
- Fu, S.C.; Hui, C.W.; Li, L.C.; Cheuk, Y.C.; Qin, L.; Gao, J.; Chan, K.M. Total flavones of Hippophae rhamnoides promotes early restoration of ultimate stress of healing patellar tendon in a rat model. Med. Eng. Phys. 2005, 27, 313–321.
- Busch, F.; Mobasheri, A.; Shayan, P.; Stahlmann, R.; Shakibaei, M. Sirt-1 is required for the inhibition of apoptosis and inflammatory responses in human tenocytes. J. Biol. Chem. 2012, 287, 25770–25781.
- Poulsen, R.C.; Watts, A.C.; Murphy, R.J.; Snelling, S.J.; Carr, A.J.; Hulley, P.A. Glucocorticoids induce senescence in primary human tenocytes by inhibition of sirtuin 1 and activation of the p53/p21 pathway: In vivo and in vitro evidence. Ann. Rheum. Dis. 2014, 73, 1405–1413.
- Busch, F.; Mobasheri, A.; Shayan, P.; Lueders, C.; Stahlmann, R.; Shakibaei, M. Resveratrol modulates interleukin-1β-induced phosphatidylinositol 3-kinase and nuclear factor κB signaling pathways in human tenocytes. J. Biol. Chem. 2012, 287, 38050–38063.
- Selvakumar, G.; Venu, D.; Kuttalam, I.; Lonchin, S. Inhibition of Advanced Glycation End Product Formation in Rat Tail Tendons by Polydatin and p-Coumaric acid: An In Vitro Study. Appl. Biochem. Biotechnol. 2022, 194, 339–353.
- Zeytin, K.; Ciloğlu, N.S.; Ateş, F.; Vardar Aker, F.; Ercan, F. The effects of resveratrol on tendon healing of diabetic rats. Acta Orthop. Et Traumatol. Turc. 2014, 48, 355–362.
- Salehi, B.; Rescigno, A.; Dettori, T.; Calina, D.; Docea, A.O.; Singh, L.; Cebeci, F.; Özçelik, B.; Bhia, M.; Dowlati Beirami, A.; et al. Avocado-Soybean Unsaponifiables: A Panoply of Potentialities to Be Exploited. Biomolecules 2020, 10, 130.
- Ernst, E. Avocado-soybean unsaponifiables (ASU) for osteoarthritis—A systematic review. Clin. Rheumatol. 2003, 22, 285–288.
- Angermann, P. Avocado/soybean unsaponifiables in the treatment of knee and hip osteoarthritis. Ugeskr. Laeger 2005, 167, 3023–3025.
- Al-Afify, A.S.A.; El-Akabawy, G.; El-Sherif, N.M.; El-Safty, F.E.A.; El-Habiby, M.M. Avocado soybean unsaponifiables ameliorates cartilage and subchondral bone degeneration in mono-iodoacetate-induced knee osteoarthritis in rats. Tissue Cell 2018, 52, 108–115.
- Rathnavelu, V.; Alitheen, N.B.; Sohila, S.; Kanagesan, S.; Ramesh, R. Potential role of bromelain in clinical and therapeutic applications. Biomed. Rep. 2016, 5, 283–288.
- Orsini, R.A. Bromelain. Plast. Reconstr. Surg. 2006, 118, 1640–1644.
- Fitzhugh, D.J.; Shan, S.; Dewhirst, M.W.; Hale, L.P. Bromelain treatment decreases neutrophil migration to sites of inflammation. Clin. Immunol. 2008, 128, 66–74.
- Del Rio, D.; Stewart, A.J.; Pellegrini, N. A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr. Metab. Cardiovasc. Dis. NMCD 2005, 15, 316–328.
- Aggarwal, B.B.; Yuan, W.; Li, S.; Gupta, S.C. Curcumin-free turmeric exhibits anti-inflammatory and anticancer activities: Identification of novel components of turmeric. Mol. Nutr. Food Res. 2013, 57, 1529–1542.
- He, Y.; Yue, Y.; Zheng, X.; Zhang, K.; Chen, S.; Du, Z. Curcumin, inflammation, and chronic diseases: How are they linked? Molecules 2015, 20, 9183–9213.
- Silvestro, S.; Sindona, C.; Bramanti, P.; Mazzon, E. A State of the Art of Antioxidant Properties of Curcuminoids in Neurodegenerative Diseases. Int. J. Mol. Sci. 2021, 22, 3168.
- Gupta, S.C.; Sung, B.; Kim, J.H.; Prasad, S.; Li, S.; Aggarwal, B.B. Multitargeting by turmeric, the golden spice: From kitchen to clinic. Mol. Nutr. Food Res. 2013, 57, 1510–1528.
- Tyagi, A.K.; Prasad, S.; Majeed, M.; Aggarwal, B.B. Calebin A, a novel component of turmeric, suppresses NF-κB regulated cell survival and inflammatory gene products leading to inhibition of cell growth and chemosensitization. Phytomed. Int. J. Phytother. Phytopharm. 2017, 34, 171–181.
- Pulido-Moran, M.; Moreno-Fernandez, J.; Ramirez-Tortosa, C.; Ramirez-Tortosa, M. Curcumin and Health. Molecules 2016, 21, 264.
- Buhrmann, C.; Brockmueller, A.; Mueller, A.L.; Shayan, P.; Shakibaei, M. Curcumin Attenuates Environment-Derived Osteoarthritis by Sox9/NF-kB Signaling Axis. Int. J. Mol. Sci. 2021, 22, 7645.
- Wang, Y.; Tang, Q.; Duan, P.; Yang, L. Curcumin as a therapeutic agent for blocking NF-κB activation in ulcerative colitis. Immunopharmacol. Immunotoxicol. 2018, 40, 476–482.
- Wang, P.; Ye, Y.; Yuan, W.; Tan, Y.; Zhang, S.; Meng, Q. Curcumin exerts a protective effect on murine knee chondrocytes treated with IL-1β through blocking the NF-κB/HIF-2α signaling pathway. Ann. Transl. Med. 2021, 9, 940.
- Fusini, F.; Bisicchia, S.; Bottegoni, C.; Gigante, A.; Zanchini, F.; Busilacchi, A. Nutraceutical supplement in the management of tendinopathies: A systematic review. Muscles Ligaments Tendons J. 2016, 6, 48–57.
- Ohishi, T.; Goto, S.; Monira, P.; Isemura, M.; Nakamura, Y. Anti-inflammatory Action of Green Tea. Anti-Inflamm. Anti-Allergy Agents Med. Chem. 2016, 15, 74–90.
- Legeay, S.; Rodier, M.; Fillon, L.; Faure, S.; Clere, N. Epigallocatechin Gallate: A Review of Its Beneficial Properties to Prevent Metabolic Syndrome. Nutrients 2015, 7, 5443–5468.
- Chu, C.; Deng, J.; Man, Y.; Qu, Y. Green Tea Extracts Epigallocatechin-3-gallate for Different Treatments. BioMed Res. Int. 2017, 2017, 5615647.
- Serafini, M.; Peluso, I.; Raguzzini, A. Flavonoids as anti-inflammatory agents. Proc. Nutr. Soc. 2010, 69, 273–278.
- Wang, K.; Lv, Q.; Miao, Y.M.; Qiao, S.M.; Dai, Y.; Wei, Z.F. Cardamonin, a natural flavone, alleviates inflammatory bowel disease by the inhibition of NLRP3 inflammasome activation via an AhR/Nrf2/NQO1 pathway. Biochem. Pharmacol. 2018, 155, 494–509.
- Kempuraj, D.; Thangavel, R.; Kempuraj, D.D.; Ahmed, M.E.; Selvakumar, G.P.; Raikwar, S.P.; Zaheer, S.A.; Iyer, S.S.; Govindarajan, R.; Chandrasekaran, P.N.; et al. Neuroprotective effects of flavone luteolin in neuroinflammation and neurotrauma. BioFactors 2021, 47, 190–197.
- He, B.; Nohara, K.; Park, N.; Park, Y.S.; Guillory, B.; Zhao, Z.; Garcia, J.M.; Koike, N.; Lee, C.C.; Takahashi, J.S.; et al. The Small Molecule Nobiletin Targets the Molecular Oscillator to Enhance Circadian Rhythms and Protect against Metabolic Syndrome. Cell Metab. 2016, 23, 610–621.
- Ferreira, P.S.; Manthey, J.A.; Nery, M.S.; Cesar, T.B. Pharmacokinetics and Biodistribution of Eriocitrin in Rats. J. Agric. Food Chem. 2021, 69, 1796–1805.
- Xu, P.; Deng, B.; Zhang, B.; Luo, Q.; Song, G. Stretch-Induced Tenomodulin Expression Promotes Tenocyte Migration via F-Actin and Chromatin Remodeling. Int. J. Mol. Sci. 2021, 22, 4928.
- Sureda, A.; Sanches Silva, A.; Sánchez-Machado, D.I.; López-Cervantes, J.; Daglia, M.; Nabavi, S.F.; Nabavi, S.M. Hypotensive effects of genistein: From chemistry to medicine. Chem. Biol. Interact. 2017, 268, 37–46.
- Wang, Z.; Wang, D.; Yang, D.; Zhen, W.; Zhang, J.; Peng, S. The effect of icariin on bone metabolism and its potential clinical application. Osteoporos. Int. 2018, 29, 535–544.
- Zhang, X.; Liu, T.; Huang, Y.; Wismeijer, D.; Liu, Y. Icariin: Does it have an osteoinductive potential for bone tissue engineering? Phytother. Res. PTR 2014, 28, 498–509.
- Li, Y.; Yao, J.; Han, C.; Yang, J.; Chaudhry, M.T.; Wang, S.; Liu, H.; Yin, Y. Quercetin, Inflammation and Immunity. Nutrients 2016, 8, 167.
- Chen, S.; Jiang, H.; Wu, X.; Fang, J. Therapeutic Effects of Quercetin on Inflammation, Obesity, and Type 2 Diabetes. Mediat. Inflamm. 2016, 2016, 9340637.
- Wong, S.K.; Chin, K.Y.; Ima-Nirwana, S. Quercetin as an Agent for Protecting the Bone: A Review of the Current Evidence. Int. J. Mol. Sci. 2020, 21, 6448.
- Hung, L.K.; Fu, S.C.; Lee, Y.W.; Mok, T.Y.; Chan, K.M. Local vitamin-C injection reduced tendon adhesion in a chicken model of flexor digitorum profundus tendon injury. J. Bone Jt. Surgery. Am. Vol. 2013, 95, e41.
- Friedman, S.L. Mechanisms of hepatic fibrogenesis. Gastroenterology 2008, 134, 1655–1669.
- Yang, G.; Chang, C.C.; Yang, Y.; Yuan, L.; Xu, L.; Ho, C.T.; Li, S. Resveratrol Alleviates Rheumatoid Arthritis via Reducing ROS and Inflammation, Inhibiting MAPK Signaling Pathways, and Suppressing Angiogenesis. J. Agric. Food Chem. 2018, 66, 12953–12960.
- Meng, T.; Xiao, D.; Muhammed, A.; Deng, J.; Chen, L.; He, J. Anti-Inflammatory Action and Mechanisms of Resveratrol. Molecules 2021, 26, 229.
- Truong, V.L.; Jun, M.; Jeong, W.S. Role of resveratrol in regulation of cellular defense systems against oxidative stress. BioFactors 2018, 44, 36–49.
- Li, Y.R.; Li, S.; Lin, C.C. Effect of resveratrol and pterostilbene on aging and longevity. BioFactors 2018, 44, 69–82.
- Nunes, S.; Danesi, F.; Del Rio, D.; Silva, P. Resveratrol and inflammatory bowel disease: The evidence so far. Nutr. Res. Rev. 2018, 31, 85–97.
- Poulsen, M.M.; Fjeldborg, K.; Ornstrup, M.J.; Kjær, T.N.; Nøhr, M.K.; Pedersen, S.B. Resveratrol and inflammation: Challenges in translating pre-clinical findings to improved patient outcomes. Biochim. Biophys. Acta 2015, 1852, 1124–1136.
- De Ligt, M.; Timmers, S.; Schrauwen, P. Resveratrol and obesity: Can resveratrol relieve metabolic disturbances? Biochim. Biophys. Acta 2015, 1852, 1137–1144.
- Szkudelski, T.; Szkudelska, K. Resveratrol and diabetes: From animal to human studies. Biochim. Biophys. Acta 2015, 1852, 1145–1154.
- Shakibaei, M.; Buhrmann, C.; Mobasheri, A. Resveratrol-mediated SIRT-1 interactions with p300 modulate receptor activator of NF-kappaB ligand (RANKL) activation of NF-kappaB signaling and inhibit osteoclastogenesis in bone-derived cells. J. Biol. Chem. 2011, 286, 11492–11505.
- Joe, A.K.; Liu, H.; Suzui, M.; Vural, M.E.; Xiao, D.; Weinstein, I.B. Resveratrol induces growth inhibition, S-phase arrest, apoptosis, and changes in biomarker expression in several human cancer cell lines. Clin. Cancer Res. 2002, 8, 893–903.
- Howitz, K.T.; Bitterman, K.J.; Cohen, H.Y.; Lamming, D.W.; Lavu, S.; Wood, J.G.; Zipkin, R.E.; Chung, P.; Kisielewski, A.; Zhang, L.L.; et al. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature 2003, 425, 191–196.
- Kaeberlein, M.; McDonagh, T.; Heltweg, B.; Hixon, J.; Westman, E.A.; Caldwell, S.D.; Napper, A.; Curtis, R.; DiStefano, P.S.; Fields, S.; et al. Substrate-specific activation of sirtuins by resveratrol. J. Biol. Chem. 2005, 280, 17038–17045.
- Ren, Z.; Wang, L.; Cui, J.; Huoc, Z.; Xue, J.; Cui, H.; Mao, Q.; Yang, R. Resveratrol inhibits NF-kB signaling through suppression of p65 and IkappaB kinase activities. Die Pharm. 2013, 68, 689–694.
- Shang, X.; Lin, K.; Yu, R.; Zhu, P.; Zhang, Y.; Wang, L.; Xu, J.; Chen, K. Resveratrol Protects the Myocardium in Sepsis by Activating the Phosphatidylinositol 3-Kinases (PI3K)/AKT/Mammalian Target of Rapamycin (mTOR) Pathway and Inhibiting the Nuclear Factor-κB (NF-κB) Signaling Pathway. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2019, 25, 9290–9298.
- Constanze, B.; Popper, B.; Aggarwal, B.B.; Shakibaei, M. Evidence that TNF-β suppresses osteoblast differentiation of mesenchymal stem cells and resveratrol reverses it through modulation of NF-κB, Sirt1 and Runx2. Cell Tissue Res. 2020, 381, 83–98.
- Tillander, B.; Franzén, L.E.; Karlsson, M.H.; Norlin, R. Effect of steroid injections on the rotator cuff: An experimental study in rats. J. Shoulder Elb. Surg. 1999, 8, 271–274.
- Akpinar, S.; Hersekli, M.A.; Demirors, H.; Tandogan, R.N.; Kayaselcuk, F. Effects of methylprednisolone and betamethasone injections on the rotator cuff: An experimental study in rats. Adv. Ther. 2002, 19, 194–201.
- Mikolyzk, D.K.; Wei, A.S.; Tonino, P.; Marra, G.; Williams, D.A.; Himes, R.D.; Wezeman, F.H.; Callaci, J.J. Effect of corticosteroids on the biomechanical strength of rat rotator cuff tendon. J. Bone Jt. Surgery. Am. Vol. 2009, 91, 1172–1180.
More
