COVID-19 is an infectious disease caused by the SARS-CoV-2 virus. The clinical presentations of the SARS-CoV-2 infection are widely variable and treatment strategies for COVID-19 are dependent on the infection phase. Timing the right treatment for the right phase of this disease is paramount, with correlations detected between the phase of the infection and the type of drug used to treat. The immune system activation following COVID-19 infection can further develop to a fulminant cytokine storm which can progress to acute respiratory distress syndrome. The inflammatory phase, or the hyperinflammation phase, is a later stage when patients develop acute respiratory distress syndrome (ARDS), sepsis, and kidney and other organ failure. In this stage, the virus is probably not necessary and all the damage is due to the immune system’s cytokine storm. Immunosuppressive or immunomodulatory agent administration is the major strategy in treating COVID-19 patients at this stage. On the other hand, immunodeficient patients who are treated with immunomodulator agents have attenuated immune systems that do not produce enough cytokines. Current data do not show an increased risk of severe COVID-19 in patients taking biologic therapies or targeted disease-modifying antirheumatic drugs.
- Cytokine Storm
- Delta variant
- Omicron variant
- severity
- COVID-19
- mortality
1. Introduction
2. Cytokine Storm in COVID-19: The Correlation between the Cytokine Storm, Morbidity, and Mortality
Globally, humans have suffered from many viral pandemics in the last century [21][14], with recent ones being severe acute respiratory syndrome (SARS) [22][15], the influenza virus pandemic [23][16], and the H1N1 swine influenza [23][16]. These viral infections pose risks to our health in that some of them are zoonotic infections with the threat of human-to-human transmission and excessively high mortality rates [24][17]. At the end of 2019, another strain of SARS-CoV was identified, SARS-CoV-2, which has caused the COVID-19 pandemic. The main effects of SARS-CoV-2 on the immune system are in the distribution and apoptosis of lymphocytes. There is a decrease in the total number of lymphocytes, cytotoxic and helper T cells, B, and NK cells, and almost all their subsets, especially in patients with a severe course of COVID-19 [4]. Development of fulminant cytokine storm due to the SARS-CoV-2 virus infection results in an excessive inflammatory reaction with the release of large amounts of pro-inflammatory cytokines (Figure 1). This condition mimics a dangerous situation called hemophagocytic lymphohistiocytosis, or macrophage activation syndrome, with a serious risk of death [5]. Cytokine storms are strongly correlated with severe disease and death [12,19,24][12][17][18].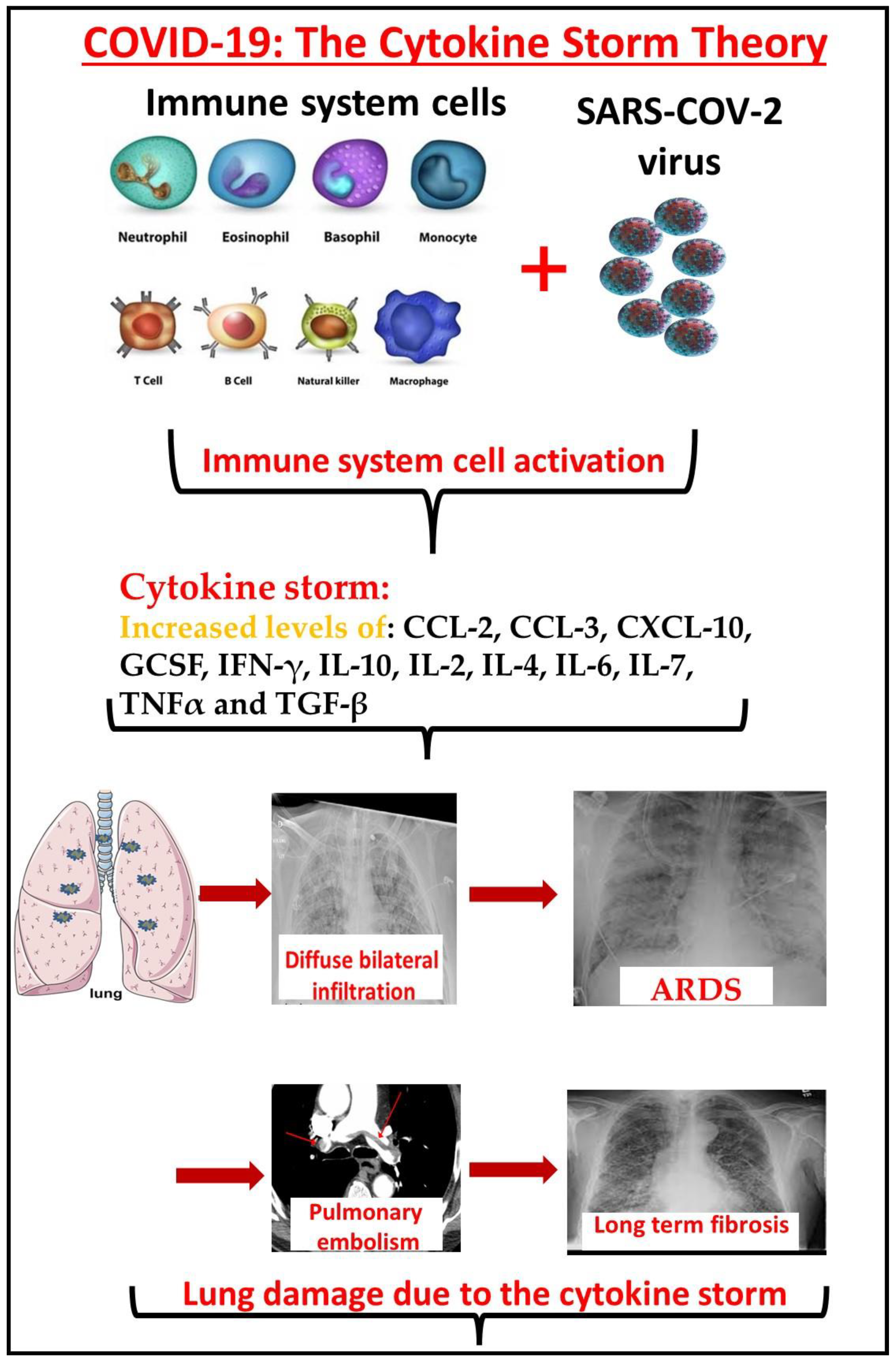
3. The Cytokine Storm as a Target Choice in the Treatment of COVID-19
The cytokine storm plays an essential role in the pathogenesis and clinical outcome of virus infections [24][17]. Blocking the cytokine storm provides greater protection than does antiviral therapy in the influenza virus [24][17]. The cytokine storm in fatal COVID-19 is represented by several pathological features, such as ARDS, coagulation, and multiorgan dysfunctions [12,19,24][12][17][18]. Patients with serious COVID-19 infections may suffer from a release of cytokines which can damage the lung tissues [12,19,24][12][17][18]. A severe COVID-19 cytokine storm is characterized by the release of proinflammatory mediators [12[12][17][18][32],19,24,67], and the amplified pulmonary inflammatory response results in enlarged alveolar–capillary gas exchange, making oxygenation difficult [67][32]. Elevated IL-6 and IL-1 concentrations correlate with intrapulmonary macrophage activation and pulmonary vascular disease [67][32]. These cytokines significantly contribute to fever, lymphopenia, coagulation, lung injury, and multiorgan failure [67,68][32][33]. IL-1β is a mediator of lung inflammation, fever, and fibrosis. Suppression of IL-1 family members and IL-6 has been shown to have a therapeutic effect in many inflammatory diseases, including viral infections such as SARS-CoV-2 [67,68,69][32][33][34]. Therefore, many drugs which target cytokines in COVID-19 were tested for their effect on mortality and morbidity [70,71,72,73,74,75,76,77,78,79,80][35][36][37][38][39][40][41][42][43][44][45]. IL-6 levels are highly correlated with the lethal complications of COVID-19, and are associated with poor prognosis and progression, as well as disease augmentation [35,69,70,71,72,73,74,75,76][30][34][35][36][37][38][39][40][41]. Agents which mediate IL-6 inhibition may improve the patients’ conditions. Tocilizumab is a recombinant, humanized, monoclonal anti-interleukin IL-6 antibody that targets the human IL-6 receptor (IL-6R). However, results using tocilizumab are mixed [69,70,71,72,73,74,75,76,77,78,79,80][34][35][36][37][38][39][40][41][42][43][44][45]. While some studies show that tocilizumab is not efficacious in improving hospitalized patients infected with severe acute respiratory syndrome coronavirus [75][40], other studies show that decreasing IL-6 levels by tocilizumab correlates with a decrease of the risk of mortality caused by COVID-19 [69,76][34][41]. Furthermore, many studies confirm that the use of tocilizumab decreases the need for mechanical ventilation [77,78,79,80,81,82,83,84,85,86,87,88,89][42][43][44][45][46][47][48][49][50][51][52][53][54]. Anti-IL-1 molecules were also tested for their efficacy on reducing morbidity and mortality in COVID-19 patients [90,91][55][56]. Anakinra is the main treatment in this group. Anakinra treatment, compared to the standard of care, shows a decrease in twenty-eight-day mortality and in hospital stay [91][56]. Anti-TNF agents are commonly used for the treatment of rheumatoid arthritis, ankylosing spondylitis, and psoriatic arthritis. While serum TNF-α levels are moderately elevated in patients with SARS [92[57][58],93], much higher serum levels are observed in COVID-19 patients, positively correlating with disease severity [92,93][57][58]. Although anti-TNF treatment was suggested as a potential treatment for COVID-19, there is insufficient data [92][57]. Some studies have demonstrated that there is a correlation between anti-TNF-α therapy with an increase in the risk of intracellular infection of SARS-CoV-2 infection via induction of the Notch-1 signaling pathway [93][58]. The efficacy of corticosteroids (CS) on inflammatory organ injury in viral pneumonia remains controversial [72,74,94][37][39][59]. Immunosuppressive glucocorticoids when administered to severely ill patients who need oxygen show the greatest benefit in preventing deterioration of these patients [36,37][31][60]. Steroids are not recommended to non-hypoxic patients [35,36,37][30][31][60]. In the absence of an oxygen requirement, patients on glucocorticoids may fare worse than those who receive standard care [35,36,37][30][31][60]. Since there is no positive evidence coming from randomized clinical trials, the WHO guidelines do not recommend routinely using systemic CS treatment for patients with COVID-19 [94][59]. Thus, the selection of patients and timing of the administration of glucocorticoids is critical for survival benefits [35][30]. Timing is paramount in this epidemic. Suppressing the host response too early during rapid viral replication is probably deleterious, whereas waiting for the requirement for respiratory support may be the appropriate time to intervene with these medications [35][30]. In clinical practice, intravenous immunoglobulin (IVIG) is used in patients with immune deficiencies for treating infectious diseases, as well as for treatment-resistant patients with autoimmune diseases as an immunomodulatory agent [95,96][61][62]. IVIG polyclonal immunoglobulin G (IgG) was also studied for its effect on morbidity and mortality in COVID-19 patients [95][61]. Previous favorable experiences from patients with SARS and MERS suggest the efficacy of a high dose of IVIG (0.3–0.5 g/kg) in patients with a serious COVID-19 infection in the early phase of the disease [95,96][61][62]. IVIG has demonstrated clinical efficacy in critically ill patients with COVID-19 [95][61]. There may be a relationship between the efficacy of IVIG and the COVID-19 disease severity [95,96][61][62]. Convalescent plasma is plasma from patients that were cured of a COVID-19 infection. Convalescent plasma may offer various beneficial actions in COVID-19 disease. In non-critical hospitalized patients, convalescent plasma therapy reduces the morbidity and mortality in moderately ill COVID-19 patients and shortens the hospitalization length [19][18]. The proposed mechanism of how convalescent plasma helps COVID-19 patients in the first stage of infection is not completely known [19][18]. Some studies show that antibodies from convalescent plasma can suppress viremia [59][63]. Administering convalescent plasma at the early stage of the disease was found to be more effective [19,59][18][63]. Mast cells may be another therapeutic target point in SARS-CoV-2 infection. This virus could activate mast cells. These cells mediate allergic and pulmonary diseases by secreting cytokines and material such as histamine, leukotrienes, and proteases [97][64]. Inhibition of mast cells could attenuate the effect of these cells on the organs, especially the lungs. The flavone luteolin could bind to the surface spike protein of SARS-CoV-2 and inhibit entry of the virus into host cells. This material also inhibits serine proteases, which is required for viral infectivity. Luteolin also inhibits mast cells and has anti-inflammatory properties, inhibiting secretion of the pro-inflammatory cytokines from human mast cells [97][64]. This drug could be effective in targeting the cytokine storm mediated by SARS-CoV-2 infection. In summary, the cytokine storm is the most dangerous factor in this disease. This stage causes permanent or long-lasting damage to the body most of the time. Some of these damages are still unknown and unclear. Targeting some of the players in the cytokine storm can possibly attenuate the storm and prove to be beneficial. More evidence is currently being accumulated about the effect of this “storm” and on possible treatment modalities.References
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W. Presenting characteristics, comorbidities and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA 2020, 323, 2052–2059.
- Oran, D.P.; Topol, E.J. The Proportion of SARS-CoV-2 Infections That Are Asymptomatic: A Systematic Review. Ann. Intern. Med. 2021, 174, 655.
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062.
- Zhang, J.J.; Dong, X.; Cao, Y.Y.; Yuan, Y.D.; Yang, Y.B.; Yan, Y.Q.; Gao, Y.D. Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy 2020, 75, 1730–1741.
- Centers for Disease Control and Prevention. SARS-CoV-2 Variant Classifications and Definitions; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2021.
- Martinez, M.A. Compounds with Therapeutic Potential against Novel Respiratory 2019 Coronavirus. Antimicrob. Agents Chemother. 2020, 64, e00399-20.
- Beigel, J.H.; Tomashek, K.M.; Dodd, L.E.; Mehta, A.K.; Zingman, B.S.; Kalil, A.C.; Lane, H.C. Remdesivir for the treatment of Covid-19: Preliminary report. N. Engl. J. Med. 2020, 383, 994.
- Cao, B.; Wang, Y.; Wen, D.; Liu, W.; Wang, J.; Fan, G.; Ruan, L.; Song, B.; Cai, Y.; Wei, M.; et al. A Trial of Lopinavir–Ritonavir in Adults Hospitalized with Severe COVID-19. N. Engl. J. Med. 2020, 382, 1787–1799.
- Bloch, E.M.; Shoham, S.; Casadevall, A.; Sachais, B.S.; Shaz, B.; Winters, J.L.; Van Buskirk, C.; Grossman, B.J.; Joyner, M.; Henderson, J.P.; et al. Deployment of convalescent plasma for the prevention and treatment of COVID-19. J. Clin. Investig. 2020, 130, 2757–2765.
- Schrezenmeier, E.; Dörner, T. Mechanisms of action of hydroxychloroquine and chloroquine: Implications for rheumatology. Nat. Rev. Rheumatol. 2020, 16, 155–166.
- Gautret, P.; Lagier, J.C.; Parola, P.; Hoang, V.T.; Meddeb, L.; Mailhe, M.; Doudier, B.; Courjon, J.; Giordanengo, V.; Vieira, V.E.; et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: Results of an open-label non-randomized clinical trial. Int. J. Antimicrob. Agents 2020, 56, 105949.
- FDA Authorizes REGEN-COV Monoclonal Antibody Therapy for Post-Exposure Prophylaxis (Prevention) for COVID-19. 2021. Available online: https://www.fda.gov/drugs/drug-safety-and-availability/fda-authorizes-regen-cov-monoclonal-antibody-therapy-post-exposure-prophylaxis-prevention-covid-19 (accessed on 8 October 2020).
- Gili, R.-Y.; Tal, G.; Mayan, G. Efficacy of a Fourth Dose of Covid-19 mRNA Vaccine against Omicron. N. Engl. J. Med. 2022, 386, 1377–1380.
- Zhang, W.P.F.; Neumann, G.; Kawaoka, Y. Orthomyxoviruses. In Fields Virology; Knipe, D.M., Howley, P.M., Griffin, D.E., Lamb, R.A., Martin, M.A., Roizman, B., Straus, S.E., Eds.; Wolters Kluwer, Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007; Chapter 48; pp. 1691–1740.
- Oldstone, M.B.A. Severe Acute Respiratory Syndrome (SARS). Viruses, Plague and History; Oxford Press: Oxford, UK, 2010; pp. 251–283.
- Dawood, F.S.; Iuliano, A.D.; Reed, C.; Meltzer, M.I.; Shay, D.K.; Cheng, P.Y. Estimated Global Mortality Associated with the First 12 Months of 2009 Pandemic Influenza A H1N1 Virus Circulation: A Modelling Study. Lancet Infect. Dis. 2012, 12, 687–695.
- Michael, B.A. Oldstone12 and Hugh Rosen13. Cytokine Storm Plays a Direct Role in the Morbidity and Mortality from Influenza Virus Infection and is Chemically Treatable with a Single Sphingosine-1-Phosphate Agonist Molecule. Sphingosine-1-Phosphate Signal. Immunol. Infect. Diseases 2014, 378, 129–147.
- Basheer, M.; Saad, E.; Hagai, R.; Assy, N. Clinical Predictors of Mortality and Critical Illness in Patients with COVID-19 Pneumonia. Metabolites 2021, 11, 679.
- Tay, M.Z.; Poh, C.M.; Rénia, L.; Macary, P.A.; Ng, L.F.P. The trinity of COVID-19: Immunity, inflammation and intervention. Nat. Rev. Immunol. 2020, 20, 363–374.
- Chen, G.; Wu, D.; Guo, W.; Cao, Y.; Huang, D.; Wang, H.; Wang, T.; Zhang, X.; Chen, H.; Yu, H.; et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J. Clin. Investig. 2020, 130, 2620–2629.
- Zhang, Y.; Li, J.; Zhan, Y.; Wu, L.; Yu, X.; Zhang, W.; Ye, L.; Xu, S.; Sun, R.; Wang, Y.; et al. Analysis of Serum Cytokines in Patients with Severe Acute Respiratory Syndrome. Infect. Immun. 2004, 72, 4410–4415.
- Coronaviridae Study Group of the International Committee on Taxonomy of V. The species severe acute respiratory syndrome-related coronavirus: Classifying 2019-nCoV and naming it SARS-CoV-2. Nat. Microbiol. 2020, 5, 536–544.
- Rabaan, A.A.; Al-Ahmed, S.H.; Muhammad, J.; Khan, A.; Sule, A.A.; Tirupathi, R.; Mutair, A.A.; Alhumaid, S.; Al-Omari, A.; Dhawan, M.; et al. Role of Inflammatory Cytokines in COVID-19 Patients: A Review on Molecular Mechanisms, Immune Functions, Immunopathology and Immunomodulatory Drugs to Counter Cytokine Storm. Vaccines 2021, 9, 436.
- Kim, J.Y.; Ko, J.H.; Kim, Y.; Kim, Y.J.; Kim, J.M.; Chung, Y.S. Viral load kinetics of SARS-CoV-2 infection in first two patients in Korea. J. Korean Med. Sci. 2020, 35, e86.
- Liu, Y.; Yang, Y.; Zhang, C.; Huang, F.; Wang, F.; Yuan, J.; Wang, Z.; Li, J.; Li, J.; Feng, C.; et al. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci. China Life Sci. 2020, 63, 364–374.
- Pan, Y.; Zhang, D.; Yang, P.; Poon, L.L.M.; Wang, Q. Viral load of SARS-CoV-2 in clinical samples. Lancet Infect. Dis. 2020, 20, 411–412.
- Phan, L.T.; Nguyen, T.V.; Luong, Q.C.; Nguyen, T.V.; Nguyen, H.T.; Le, H.Q.; Pham, Q.D. Importation and human-to-human transmission of a novel coronavirus in Vietnam. N. Engl. J. Med. 2020, 382, 872–874.
- Perreau, M.; Suffiotti, M.; Marques-Vidal, P.; Wiedemann, A.; Levy, Y.; Laouénan, C.; Ghosn, J.; Fenwick, C.; Comte, D.; Roger, T.; et al. The cytokines HGF and CXCL13 predict the severity and the mortality in COVID-19 patients. Nat. Commun. 2021, 12, 4888.
- Liu, B.; Li, M.; Zhou, Z.; Guan, X.; Xiang, Y. Can we use interleukin-6 (IL-6) blockade for coronavirus disease 2019 (COVID-19)-induced cytokine release syndrome (CRS)? J. Autoimmun. 2020, 111, 102452.
- Hojyo, S.; Uchida, M.; Tanaka, K.; Hasebe, R.; Tanaka, Y.; Murakami, M.; Hirano, T. How COVID-19 induces cytokine storm with high mortality. Inflamm. Regen. 2020, 40, 37.
- Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; Elmahi, E.; et al. Dexamethasone in Hospitalized Patients with Covid-19. N. Engl. J. Med. 2021, 384, 693–704.
- David, J.C. What Is The Role of Interleukin (IL) Inhibitors in the Treatment of Coronavirus Disease 2019 (COVID-19)? Available online: https://www.medscape.com/answers/2500114-197455 (accessed on 25 November 2020).
- Abbasifard, M.; Khorramdelazad, H. The bio-mission of interleukin-6 in the pathogenesis of COVID-19: A brief look at potential therapeutic tactics. Life Sci. 2020, 257, 118097.
- Conti, P.; Ronconi, G.; Caraffa, A.; Gallenga, C.E.; Ross, R.; Frydas, I.; Kritas, S.K. Induction of pro-inflammatory cytokines (IL-1 and IL-6) and lung inflammation by Coronavirus-19 (COVI-19 or SARS-CoV-2): Anti-inflammatory strategies. J. Biol. Regul. Homeost. Agents 2020, 34, 327–331.
- Stone, J.H.; Frigault, M.J.; Serling-Boyd, N.J.; Fernandes, A.D.; Harvey, L.; Foulkes, A.S.; Horick, N.K.; Healy, B.C.; Shah, R.; Bensaci, A.M.; et al. Efficacy of Tocilizumab in Patients Hospitalized with Covid-19. N. Engl. J. Med. 2020, 383, 2333–2344.
- Treatment of COVID-19 Patients with Anti-Interleukin Drugs (COV-AID). Available online: https://clinicaltrials.gov/ct2/show/NCT04330638 (accessed on 1 October 2021).
- Anti-IL6 and Corticosteroid Monotherapy vs. Combination in COVID-19. Available online: https://clinicaltrials.gov/ct2/show/NCT04486521 (accessed on 1 October 2021).
- Efficacy and Safety of Siltuximab vs. Corticosteroids in Hospitalized Patients with COVID-19 Pneumonia. Available online: https://clinicaltrials.gov/ct2/show/NCT04329650 (accessed on 1 October 2021).
- Castelnovo, L.; Tamburello, A.; Lurati, A.; Zaccara, E.; Marrazza, M.G.; Olivetti, M.; Mazzone, A. Anti-IL6 treatment of serious COVID-19 disease: A monocentric retrospective experience. Medicine 2021, 100, e23582.
- Rosas, I.O.; Bräu, N.; Waters, M.; Go, R.C.; Hunter, B.D.; Bhagani, S.; Skiest, D.; Aziz, M.S.; Cooper, N.; Douglas, I.S.; et al. Tocilizumab in Hospitalized Patients with Severe Covid-19 Pneumonia. N. Engl. J. Med. 2021, 384, 1503–1516.
- McGonagle, D.; Sharif, K.; O’Regan, A.; Bridgewood, C. Interleukin-6 use in COVID-19 pneumonia related macrophage activation syndrome. Autoimmun. Rev. 2020, 19, 102537.
- Luo, P.; Liu, Y.; Qiu, L.; Liu, X.; Liu, D.; Li, J. Tocilizumab treatment in COVID-19: A single center experience. J. Med. Virol. 2020, 92, 814–818.
- Hermine, O.; Mariette, X.; Tharaux, P.L.; Resche-Rigon, M.; Porcher, R.; Ravaud, P.; Korganow, A.S. Effect of tocilizumab vs usual care in adults hospitalized with COVID-19 and moderate or severe pneumonia: A randomized clinical trial. JAMA Intern. Med. 2021, 181, 32–40.
- Guaraldi, G.; Meschiari, M.; Cozzi-Lepri, A.; Milic, J.; Tonelli, R.; Menozzi, M.; Mussini, C. Tocilizumab in patients with severe COVID-19: A retrospective cohort study. Lancet Rheumatol. 2020, 2, e474–e484.
- Kaye, A.G.; Siegel, R. The efficacy of IL-6 inhibitor Tocilizumab in reducing severe COVID-19 mortality: A systematic review. PeerJ 2020, 8, e10322.
- Soin, A.S.; Kumar, K.; Choudhary, N.S.; Sharma, P.; Mehta, Y.; Kataria, S.; Govil, D.; Deswal, V.; Chaudhry, D.; Singh, P.K.; et al. Tocilizumab plus standard care versus standard care in patients in India with moderate to severe COVID-19-associated cytokine release syndrome (COVINTOC): An open-label, multicentre, randomised, controlled, phase 3 trial. Lancet Respir. Med. 2021, 9, 511–521.
- Veiga, V.C.; Prats, J.A.; Farias, D.L.; Rosa, R.G.; Dourado, L.K.; Zampieri, F.G. Effect of tocilizumab on clinical outcomes at 15 days in patients with severe or critical coronavirus disease 2019: Randomized controlled trial. BMJ 2021, 372, n84.
- Lescure, F.X.; Honda, H.; Fowler, R.A.; Lazar, J.S.; Shi, G.; Wung, P. Sarilumab in patients admitted to hospital with severe or critical COVID-19: A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Respir Med. 2021, 9, 522–532.
- Salama, C.; Han, J.; Yau, L.; Reiss, W.G.; Kramer, B.; Neidhart, J.D.; Criner, G.J.; Kaplan-Lewis, E.; Baden, R.; Pandit, L.; et al. Tocilizumab in Patients Hospitalized with Covid-19 Pneumonia. N. Engl. J. Med. 2021, 384, 20–30.
- Gordon, A.C.; Mouncey, P.R.; Al-Beidh, F.; Rowan, K.M.; Nichol, A.D.; Arabi, Y.M. Interleukin-6 receptor antagonists in critically ill patients with Covid-19. N. Engl. J. Med. 2021, 384, 1491–1502.
- Wang, D.; Fu, B.; Peng, Z.; Yang, D.; Han, M.; Li, M.; Yang, Y.; Yang, T.; Sun, L.; Li, W.; et al. Tocilizumab in patients with moderate or severe COVID-19: A randomized, controlled, open-label, multicenter trial. Front. Med. 2021, 15, 486–494.
- Salvarani, C.; Dolci, G.; Massari, M.; Merlo, D.F.; Cavuto, S.; Savoldi, L. Effect of tocilizumab vs standard care on clinical worsening in patients hospitalized with COVID-19 pneumonia: A randomized clinical trial. JAMA Intern. Med. 2021, 181, 24–31.
- Horby, P.W.; Campbell, M.; Staplin, N.; Spata, E.; Emberson, J.R.; Pessoa-Amorim, G. Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): A randomised, controlled, open-label, platform trial. Lancet 2021, 397, 1637–1645.
- Domingo, P.; Mur, I.; Mateo, G.M.; del Mar Gutierrez, M.; Pomar, V.; de Benito, N. Association between administration of IL-6 antagonists and mortality among patients hospitalized for COVID-19: A meta-analysis. JAMA 2021, 326, 499–518.
- Tharaux, P.L.; Pialoux, G.; Pavot, A.; Mariette, X.; Hermine, O.; Resche-Rigon, M. Effect of anakinra versus usual care in adults in hospital with COVID-19 and mild-to-moderate pneumonia (CORIMUNO-ANA-1): A randomised controlled trial. Lancet Respir. Med. 2021, 9, 295–304.
- Kyriazopoulou, E.; Poulakou, G.; Milionis, H.; Metallidis, S.; Adamis, G.; Tsiakos, K. Early treatment of COVID-19 with anakinra guided by soluble urokinase plasminogen receptor plasma levels: A double-blind, randomized controlled phase 3 trial. Nat. Med. 2021, 27, 1752–1760.
- Ferro, F.; Elefante, E.; Baldini, C.; Bartoloni, E.; Puxeddu, I.; Talarico, R.; Mosca, M.; Bombardieri, S. COVID-19: The new challenge for rheumatologists. Clin. Exp. Rheumatol. 2020, 38, 175–180.
- Keewan, E.; Beg, S.; Naser, S.A. Anti-TNF-α agents Modulate SARS-CoV-2 Receptors and Increase the Risk of Infection Through Notch-1 Signaling. Front. Immunol. 2021, 12, 1662.
- Shang, L.; Zhao, J.; Hu, Y.; Du, R.; Cao, B. On the use of corticosteroids for 2019-nCoV pneumonia. Lancet 2020, 395, 683–684.
- Cron, R.Q.; Caricchio, R.; Chatham, W.W. Calming the cytokine storm in COVID-19. Nat. Med. 2021, 27, 1674–1675.
- Cao, W.; Liu, X.; Bai, T.; Fan, H.; Hong, K.; Song, H.; Han, Y.; Lin, L.; Ruan, L.; Li, T. High-dose intravenous immunoglobulin as therapeutic option for deteriorating patients with coronavirus disease 2019. In Open Forum Infectious Diseases; Oxford University Press: Oxford, UK, 2020; Volume 7.
- Jawhara, S. Could Intravenous Immunoglobulin Collected from Recovered Coronavirus Patients Protect against COVID-19 and Strengthen the Immune System of New Patients? Int. J. Mol. Sci. 2020, 21, 2272.
- Maamoun, B.; Saad, E.; Laskar, O.; Schuster, O.; Rechnitzer, H.; Zisman-Rozen, S.; Azoulay, D.; Assy, N. Clearance of the SARS-CoV-2 Virus in an Immunocompromised Patient Mediated by Convalescent Plasma without B-Cell Recovery. Int. J. Mol. Sci. 2021, 22, 8902.
- Theoharides, T.C. COVID -19, pulmonary mast cells, cytokine storms, and beneficial actions of luteolin. BioFactors 2020, 46, 306–308.
