Bone metastasis has been considered the fatal phase of cancers, which remains incurable and to be a challenge due to the non-availability of the ideal treatment strategy. Unlike bone cancer, bone metastasis involves the spreading of the tumor cells to the bones from different origins. Bone metastasis generally originates from breast and prostate cancers. The possibility of bone metastasis is highly attributable to its physiological milieu susceptible to tumor growth. The treatment of bone-related diseases has multiple complications, including bone breakage, reduced quality of life, spinal cord or nerve compression, and pain. However, anticancer active agents have failed to maintain desired therapeutic concentrations at the target site; hence, uptake of the drug takes place at a non-target site responsible for the toxicity at the cellular level. Interestingly, lipid-based drug delivery systems have become the center of interest for researchers, thanks to their biocompatible and bio-mimetic nature. These systems possess a great potential to improve precise bone targeting without affecting healthy tissues.
- solid lipid nanoparticles
- nanostructured lipid carriers
- exosomes
1. Introduction
2. Strategies in Bone Targeting
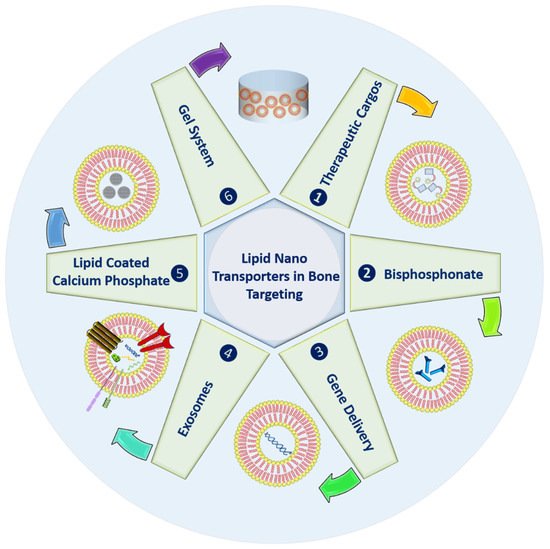
2.1. Therapeutic Cargoes
| Drug | Issue | Formulation | Outcome | Ref. |
|---|---|---|---|---|
| Metvan | Rapid oxidation, interference with blood components | Nanostructured Lipid Carriers | Quantitative encapsulation efficiency, sustained-release within 48 h, high cytotoxic effects | [25] |
| Icariin | Low water-solubility, susceptible to first-pass metabolism, and low bioavailability |
Liposomes | Amplified the mechanical strength of femoral midshaft, triggered bone turnover/remodeling | [26] |
| Simvastatin | Deterioration at a physiological pH, low water solubility, low bioavailability, high toxicity | Lipid nanoparticles | Higher encapsulation efficiency with a sustained release of 70% within 50 h, reduction in cytotoxicity | [27] |
| Doxycycline | Degradation in the anhydrous environment, poor bone penetration | Lipid- Polymer hybrid system | Zero-order release rate up to one month, eradicate bacterial bone infections | [28] |
| Edelfosine | Poor oral bioavailability, dose-dependent hemolysis | Lipid nanoparticles | Shows immediate cytotoxicity to human osteosarcoma cells, negligible tumor growth with declining of tumor volume by five-fold | [29] |
| TNF-α small interfering RNA | Short half-life, deprived extravasation from blood vessels to target cells, low cellular uptake | PEGylated solid-lipid nanoparticles | Encapsulation efficiency more than 90%, precise targeting to inflamed sites in a mouse model, declined bone loss, | [30] |
2.2. Bisphosphonate Delivery
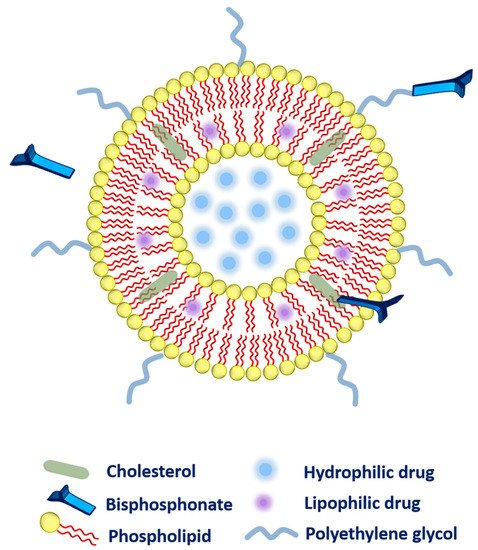
- ➢
-
Firstly, during liposomal production, bisphosphonate (alendronate sodium trihydrate, or zoledronic acid) can be added passively to the aqueous phase of the liposomes owing to high water-solubility [37][38][39][37,38,39].
- ➢
-
Secondly, the bisphosphonate can also be conjugated along with the cholesterol (as a main component of liposomal composition via a click reaction known as Cu(I)-catalyzed Huisgen 1,3-dipolar cycloaddition). The aforementioned system has exhibited a very strong affinity toward bones [40].
- ➢
-
Thirdly, bisphosphonate can be associated with the polyethylene glycol (PEG) chain to provide higher circulation time to the liposomes. More precisely, phospholipid-PEG-bisphosphonate conjugation can be employed in liposomes [41]. Polyethylene-glycol-conjugated phospholipid was used to embed zoledronic acid (as a potent inhibitor of farnesyl-pyrophosphate synthase). The fabricated liposomes were subjected to biodistribution studies that evidenced higher drug accumulation in the liver, spleen, bones, and tumor as compared to zoledronic acid in free form or entrapped in non-PEGylated liposomes. However, toxicity was the main concern as these liposomes were found very toxic to rodents [36].Moving ahead, the choice of suitable bisphosphonate is another challenge. Researchers have tried to establish a comparison between different bisphosphonates where liposomal systems composing alendronate have been investigated in T-cell immunotherapy, which delayed ovarian tumor growth in mice. Wherein, a better synergism was established in alendronate and γδ T cells as compared to zoledronic acid. In addition, concerning safety, alendronate has been found safer than zoledronic acid as concluded from cell viability studies. In this manner, the Nitrogen-containing bisphosphonates conjugation has improved the pharmacokinetic profile and enhanced passive retention at tumor sites [38].Apart from the liposomes, the solid lipid nanoparticles are also flexible enough to deliver bisphosphonates. One study reported wherein, bisphosphonate has been modified along with the surfactant of choice, which facilitated the transportation of the active molecule outstandingly. It is a well-proven fact that the surfactant is the most crucial component of lipid nanoparticles. Interestingly, Brij 78 (a non-ionic surfactant) has been conjugated with pamidronate (bisphosphonate) in order to enhance the overall bone affinity of the lipid-nanoparticles [42].
2.3. Gene Delivery
Undoubtedly, bone repair is a natural process. Nevertheless, in most cases, it is not always possible due to the inability of the body itself. In this regard, bone morphogenetic proteins (BMP) play a key role in bone regeneration via regulating cartilage and bone differentiation [43][44][45][46][43,44,45,46]. Therefore, the exogenous delivery of these types of growth factors can speed up the repairing process. Nonetheless, there are so many limitations to the delivery of BMP and liposomes can be a good candidate to encapsulate BMP either by their direct addition or transfection through gene carrying. Until now, no study has been found where these two methods have been compared with each other [47]. Protein delivery faces shortcomings as compared to gene delivery due to major obstacles including high dose, correct folding, and glycosylation of the protein [48].In this regard, gene therapy is an approach having the capacity to produce intra-cellular proteins and can express them for a longer period along with the regulation of transgene expression. Moreover, gene therapy avoids the use of a higher concentration of therapeutic moieties; hence, can be delivered only one time with a minimal dose. Henceforth, adverse effects associated with a high amount of therapeutics can be avoided. Therefore, combinational therapy of different genes in association with biomaterials could be the most favorable strategy [48][49][48,49].Comparing viral gene therapy to non-viral gene therapy is relatively toxic with unpredictable immune responses to the host. Considerably, liposomes with calcium phosphate as a non-viral siRNA vector in gene transfection have been exploited, in which dual functioning is designed firstly to avoid prompt growing of the calcium phosphate particles, secondly to prevent degradation of the entrapped siRNA of the Bcl-2 gene. This proposed system has been designed to deliver the Bcl-2 gene. To make the system pH-triggered charge-reversible, citraconic as a derivative of a maleic amide has been conjugated with cholesterol-aminocaproic acid to coat calcium phosphate particles and the further surface has been tuned by depositing the siRNA. In the last, the positively charged lipid 1,2-dioleoyl-3-trimethylammonium-propanechloridesalt (DOTAP) and negatively charged lipid dioleoylphosphatydicacid (DOPA) has been added and the charge reversal was investigated with the reversal of zeta potential results, which successfully suppress the Bcl-2 without inducing any toxicity in lung cancer A549 cells [50]. In some cases, the carrier should be negatively charged to achieve efficient transfection whilst positively charged in an acidic endolysosome environment to inhibit its uptake by disrupting the endolysosomal membrane. To tackle this difficulty, the charge reversal phenomenon has been applied [51][52][51,52].Apart from this, the conjugation between PEG-alendronate accomplished in the stem cells approach has been considered an effective tool in the treatment of various disorders related to bone degeneration due to its osteogenic potential [53][54][53,54]. In this regard, poor bone marrow homing at damaged tissues in response to injury is the main obstacle in the systemic infusion of mesenchymal stem cells (MSC), which have been addressed in a study, wherein the peptide sequence with a strong affinity to bone marrow-derived MSC has been encapsulated into liposomal nanoparticles modified with alendronate sodium (high bone mineral affinity). The aforementioned liposomes were composed of a DSPE-PEG-Alendronate complex for better bone targeting (gene delivery to osteoblastic cells). Further, the biophotonic imaging study assured the successful accumulation of genes in osseous tissue [41].2.4. Exosomes
Exosomes (30–150 nm in diameter) as bio-inspired lipid-based novel systems innately originated from biological sources such as endosomal compartments of animal and plant cells utilized as natural transporters between cells by encapsulating nucleic acids, anti-cancer agents, proteins, genetic lipids [55][56][55,56]. The use of exosomes has advantages over artificially prepared nanoparticles discussed in Figure 3.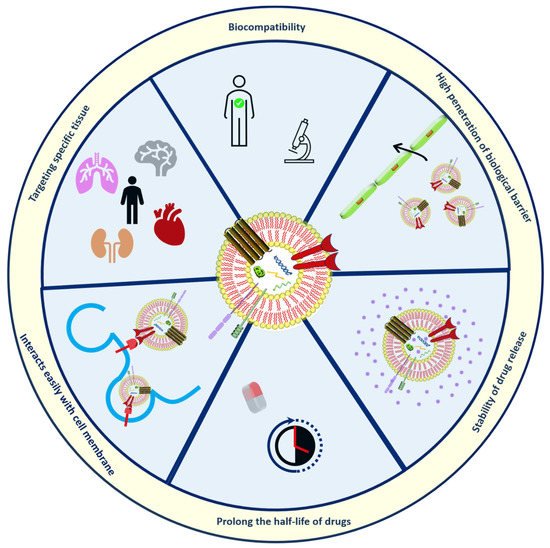 Figure 3. Structural organization and Merits of exosomes.Exosomes are more reliable because of their ability to communicate in the cells naturally [57]. Based on the diameter, extracellular vesicles can be categorized as shown in Figure 4, namely exosomes, ectosomes, and apoptotic bodies. Among the exosomes, ectosomes, and apoptotic bodies, the exosomes that are most stable can retain the content enclosed for a longer period and are also useful to enhance the bioavailability of active compounds. They can reach desired sites of action by avoiding various digestive or bio-fluid enzymes. One report has concluded that curcumin was four times more stable by loading into exosomes derived from EL-4 cells [58][59][58,59].
Figure 3. Structural organization and Merits of exosomes.Exosomes are more reliable because of their ability to communicate in the cells naturally [57]. Based on the diameter, extracellular vesicles can be categorized as shown in Figure 4, namely exosomes, ectosomes, and apoptotic bodies. Among the exosomes, ectosomes, and apoptotic bodies, the exosomes that are most stable can retain the content enclosed for a longer period and are also useful to enhance the bioavailability of active compounds. They can reach desired sites of action by avoiding various digestive or bio-fluid enzymes. One report has concluded that curcumin was four times more stable by loading into exosomes derived from EL-4 cells [58][59][58,59].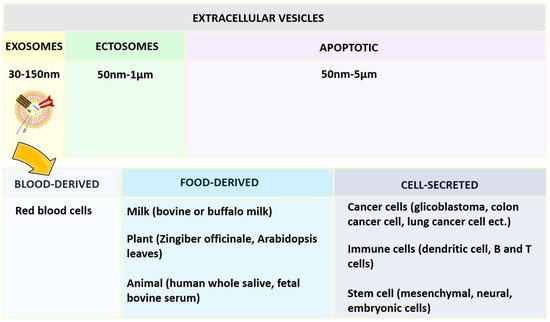 Figure 4. Classification of extracellular vesical based on the diameter and exosomes based on the source of origin.Exosomes are surrounded by the single external lamela, originating largely from blood, food, and the cell (Figure 4) and widely distributed in plasma, urine, semen, saliva, bronchial fluid, breast milk, serum, amniotic fluid, synovial fluid, tears, lymph, bile, and gastric acid [60]. In the beginning, exosomes were considered waste material from cell damage and cell homeostasis, later they were proposed to carry therapeutic moieties [61].Fascinatingly, the exosomes can disclose the biological information of the eukaryotic cells from where they have been collected practically, which is useful in curing a wide range of diseases such as chronic inflammation, specifically tumors [62][63][62,63].Exosomes are developed from the inward folding of the membrane of the early endosomes (originating from the plasma membrane of the cells), later giving rise to multivesicular bodies and taking part in various endocytic and trafficking tasks [60][64][60,64]. Further, the high cholesterol fraction containing multivesicular bodies will be guided toward fusion along with the plasma membrane to release exosomes that otherwise undergo destruction by lysosomes. Therefore, this whole event depends on the amount of cholesterol content present in multivesicular bodies [60][65][60,65]. Exosomes possess a very complex architecture containing 4400 proteins, 194 lipids, 1639 mRNAs, and 764 miRNAs, which depicts their functional diversity [66][67][66,67].Attributable to the complexity of the exosomes based on the structural organization (huge number of proteins), no robust strategy is available to provide their reproducibility. Another possible reason is the variation of exosomes derived from different matrixes. Therefore, a set of different techniques need to be applied, which increases the overall production cost. These are the frictions in the smooth production of exosomes that escalate their use at the clinical level. In general, ultracentrifugation is used for all types of exosomes collections while other methods are also available based on size, immunoaffinity capture, and precipitation of exosomes. Still, these methods are not sufficient enough for the purification of exosomal yield because the result is usually a mixture of non-differentiated content of exosomes and different extracellular vesicles [60].Moreover, in the future new strategies may be evolved involving immunoaffinity and microdevices along with good outputs and higher purified collection. Stremersch, S and co-authors have compared not only the different methods of loading to exosomes (either during biogenesis or after isolation of exosomes) but also to synthetic liposomes for the fusion capacity. No remarkable difference has been noticed in the uptake of all the above-mentioned systems. This study proposed that anionic liposomes were able to target gene knockdown under the same experimental conditions; however, exosomes cannot because of inappropriate/no release of encapsulated miRNA [68].Liposomes and exosomes are closely related in terms of composing one lipid bilayer, a diameter below 200 nm, and the capability to load both hydrophilic and lipophilic active moieties. In the case of discrepancy, exosomes offered a more complex surface with high specificity due to the existence of numerous proteins (such as tetraspanins). In a typical composition of liposomes, no proteins are associated with the surface of the external membrane. These proteins make possible the efficient targeting and uptake hence, have very good immunocompatibility [69].Although, exosomes face some disadvantages including rapid clearance, low loading capacity, and the non-availability of scale-up manufacturing techniques. In contrast, liposomes provide the possibility for PEGylation to achieve long circulation, well-defined production even at an industrial scale (many liposomal products are already on market), and the feasibility of the ligand-guided approach [69][70][69,70].Despite this fact, it has been always a topic of conflict that liposomes are better than exosomes comparatively and vice versa. To neutralize the above statement, the lipid component of liposomes can be used in exosomes to achieve higher intelligence concerning membrane fusion [71][72][71,72]. Therefore, the functioning of liposomes and exosomes merging into one system has been investigated. Herein, the surface membrane protein of exosomes hybridized not only along with the functional lipids of liposomes but also PEGylation did enhance the overall colloidal stability of the system. These two systems can be combined through processes namely freeze–thaw, incubation, and sonication [73].Bone-derived exosomes in bone-related complications can play a key role in the regulation of gene expression, migration, and proliferation. In prostate cancer, affected cells secrete both osteoblast and osteoclast stimulating factors that initiate either bone resorption or bone formation, or both. The bone metastasis disturbs the balance between the events of bone-resorbing osteoclasts and bone-forming osteoblasts [74]. In general, bone metastasis-related to prostate cells is osteoblastic in radiographs and exosomal microRNAs derived from the prostate cancerous cells are believed to promote osteoblastic bone metastasis [75][76][75,76]. Osteolytic lesions have been traced in all the subjects affected with the osteoblastic metastasis [77]. To understand this mechanism, the role of exosomes derived from osteoblastic, osteoclastic, or combination of human prostate cancer cells have been identified in one study. Wherein, investigators have found that exosomes promoted bone tumor growth via osteoclastogenesis in vitro and induced osteolysis in vivo. MicroRNA delivered through exosomes has inhibited osteoblastogenesis evidenced by inhibition of type I collagen expression in vivo. Hence, aggressive growth of prostate cancer cells inhibited, deteriorated bone matrix, and induced premetastatic niche for tumor growth, thus, playing a key role in bone homeostasis [75].
Figure 4. Classification of extracellular vesical based on the diameter and exosomes based on the source of origin.Exosomes are surrounded by the single external lamela, originating largely from blood, food, and the cell (Figure 4) and widely distributed in plasma, urine, semen, saliva, bronchial fluid, breast milk, serum, amniotic fluid, synovial fluid, tears, lymph, bile, and gastric acid [60]. In the beginning, exosomes were considered waste material from cell damage and cell homeostasis, later they were proposed to carry therapeutic moieties [61].Fascinatingly, the exosomes can disclose the biological information of the eukaryotic cells from where they have been collected practically, which is useful in curing a wide range of diseases such as chronic inflammation, specifically tumors [62][63][62,63].Exosomes are developed from the inward folding of the membrane of the early endosomes (originating from the plasma membrane of the cells), later giving rise to multivesicular bodies and taking part in various endocytic and trafficking tasks [60][64][60,64]. Further, the high cholesterol fraction containing multivesicular bodies will be guided toward fusion along with the plasma membrane to release exosomes that otherwise undergo destruction by lysosomes. Therefore, this whole event depends on the amount of cholesterol content present in multivesicular bodies [60][65][60,65]. Exosomes possess a very complex architecture containing 4400 proteins, 194 lipids, 1639 mRNAs, and 764 miRNAs, which depicts their functional diversity [66][67][66,67].Attributable to the complexity of the exosomes based on the structural organization (huge number of proteins), no robust strategy is available to provide their reproducibility. Another possible reason is the variation of exosomes derived from different matrixes. Therefore, a set of different techniques need to be applied, which increases the overall production cost. These are the frictions in the smooth production of exosomes that escalate their use at the clinical level. In general, ultracentrifugation is used for all types of exosomes collections while other methods are also available based on size, immunoaffinity capture, and precipitation of exosomes. Still, these methods are not sufficient enough for the purification of exosomal yield because the result is usually a mixture of non-differentiated content of exosomes and different extracellular vesicles [60].Moreover, in the future new strategies may be evolved involving immunoaffinity and microdevices along with good outputs and higher purified collection. Stremersch, S and co-authors have compared not only the different methods of loading to exosomes (either during biogenesis or after isolation of exosomes) but also to synthetic liposomes for the fusion capacity. No remarkable difference has been noticed in the uptake of all the above-mentioned systems. This study proposed that anionic liposomes were able to target gene knockdown under the same experimental conditions; however, exosomes cannot because of inappropriate/no release of encapsulated miRNA [68].Liposomes and exosomes are closely related in terms of composing one lipid bilayer, a diameter below 200 nm, and the capability to load both hydrophilic and lipophilic active moieties. In the case of discrepancy, exosomes offered a more complex surface with high specificity due to the existence of numerous proteins (such as tetraspanins). In a typical composition of liposomes, no proteins are associated with the surface of the external membrane. These proteins make possible the efficient targeting and uptake hence, have very good immunocompatibility [69].Although, exosomes face some disadvantages including rapid clearance, low loading capacity, and the non-availability of scale-up manufacturing techniques. In contrast, liposomes provide the possibility for PEGylation to achieve long circulation, well-defined production even at an industrial scale (many liposomal products are already on market), and the feasibility of the ligand-guided approach [69][70][69,70].Despite this fact, it has been always a topic of conflict that liposomes are better than exosomes comparatively and vice versa. To neutralize the above statement, the lipid component of liposomes can be used in exosomes to achieve higher intelligence concerning membrane fusion [71][72][71,72]. Therefore, the functioning of liposomes and exosomes merging into one system has been investigated. Herein, the surface membrane protein of exosomes hybridized not only along with the functional lipids of liposomes but also PEGylation did enhance the overall colloidal stability of the system. These two systems can be combined through processes namely freeze–thaw, incubation, and sonication [73].Bone-derived exosomes in bone-related complications can play a key role in the regulation of gene expression, migration, and proliferation. In prostate cancer, affected cells secrete both osteoblast and osteoclast stimulating factors that initiate either bone resorption or bone formation, or both. The bone metastasis disturbs the balance between the events of bone-resorbing osteoclasts and bone-forming osteoblasts [74]. In general, bone metastasis-related to prostate cells is osteoblastic in radiographs and exosomal microRNAs derived from the prostate cancerous cells are believed to promote osteoblastic bone metastasis [75][76][75,76]. Osteolytic lesions have been traced in all the subjects affected with the osteoblastic metastasis [77]. To understand this mechanism, the role of exosomes derived from osteoblastic, osteoclastic, or combination of human prostate cancer cells have been identified in one study. Wherein, investigators have found that exosomes promoted bone tumor growth via osteoclastogenesis in vitro and induced osteolysis in vivo. MicroRNA delivered through exosomes has inhibited osteoblastogenesis evidenced by inhibition of type I collagen expression in vivo. Hence, aggressive growth of prostate cancer cells inhibited, deteriorated bone matrix, and induced premetastatic niche for tumor growth, thus, playing a key role in bone homeostasis [75].
