ZnSnO3 semiconductor nanostructures have several applications as photocatalysis, gas sensors, and energy harvesting. However, due to its multicomponent nature, the synthesis is far more complex than its binary counter parts. The complexity increases even more when aiming for low-cost and low-temperature processes as in hydrothermal methods. Knowing in detail the influence of all the parameters involved in these processes is imperative, in order to properly control the synthesis to achieve the desired final product. Thus, this paper presents a study of the influence of the physical parameters involved in the hydrothermal synthesis of ZnSnO3 nanowires, namely volume, reaction time, and process temperature. Based on this study a growth mechanism for the complex Zn:Sn:O system is proposed. Two zinc precursors, zinc chloride and zinc acetate, were studied, showing that although the growth mechanism is inherent to the material itself, the chemical reactions for different conditions need to be considered.
- ZnSnO3
- Nanowires
- Hydrothermal synthesis
- ZTO
1. Introduction
As a result of its impressive multifunctionality, ZnO-based thin-films and nanostructures have received a lot of attention in the last decade [1][2][3]. While ZnO on its own captured a large interest, doping or mixing with other binary compounds brings a new level of possibilities, as the material properties can be improved/tailored to different applications depending on the cationic ratio. This has been widely explored in oxide thin-films, for example with zinc-tin oxide (ZTO) [4][5]. ZTO structures crystalize by solid-state reaction in the stable inverse spinel ortho-stannate Zn2SnO4 phase or in the metastable ZnSnO3 phase, either in perovskite (orthorhombic, orth, or face centered, fcc) or rhombohedral forms [6][7][8][9]. ZnSnO3 is a ferro/piezoelectric material with a high polarization along the z-axis (~59 μC/cm2) which is much higher than that of ZnO (~5 μC/cm2) [10][11][12][13][14]. Its band-gap of 3.9 eV is higher than that of Zn2SnO4 [15][16].
While vapor phase processes can be used to synthesize high-quality ZTO nanostructures [17][18], these are expensive, cumbersome and require high temperatures (>700 °C). The demand for low-cost processes compatible with flexible substrates requires solution processes that allow for the synthesis of nanostructures at low-cost, using simple and easy methods, ideally upscalable to industrial-scale quantities [19].
2. Seed-lLayer Free Hydrothermal Synthesis of ZnSnO3 Nanowires
The multicomponent nature of ZTO makes the synthesis process quite challenging, given the different ionic sizes and diffusivity of the cations. Furthermore, each ZTO structure has different nucleation and growth times and requires a specific range of synthesis temperature [20]. In literature it has been shown the possibility to control the shape and type of the nanostructures and consequently the electrical, optical, and mechanical properties, by controlling the chemical and physical parameters of the synthesis [7][21][22].
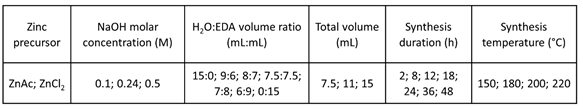
In this study, ZnSnO3 nanowires were synthesized via hydrothermal method in a conventional oven [23][24]. Briefly, the ZTO hydrothermal synthesis was performed by dissolving zinc chloride, ZnCl2, (or zinc acetate, ZnAc) and tin chloride (SnCl4·5H2O) separately in deionized water and then mixing these solutions. The surfactant ethylenediamine (EDA) was then added and stirred for 30 min, after which the mineralizer sodium hydroxide (NaOH) was added. The solution was then transferred into an autoclave and kept in an electric oven varying the temperature and the reaction time. The final product comprising the nanostructures was alternately washed with deionized water and isopropyl alcohol and centrifuged. After washed, the nanostructures were dried at 60 °C, in vacuum. Aiming to study the influence of the chemico-physical parameters in the ZTO nanostructures growth, the conditions showed in Table 1 were tested. This work was conducted without employing any seed-layer in the synthesis of ZTO nanostructures; hence, the obtained structures depend exclusively on the chemico-physical parameters of the synthesis. Moreover, the obtained nanostructures are in powder form, which through a variety of transfer methods allow for a higher degree of freedom for integration on different substrates, without contamination from the seed-layer material [25][26].
Table 1 – Conditions studied to optimize the hydrothermal synthesis of ZnSnO3 nanowires.
3. Influence of Chemico-physical Parameters of the Synthesis in the ZnSnO3 Nanowires Growth
Two zinc precursors were used in this study and their solubility in the surfactant (EDA) showed to be one of the critical parameters to optimize the synthesis of the ZnSnO3 nanowires. Comparing the tin precursor solubility in EDA with the zinc precursors’ solubility it was possible to conclude that the lower solubility of ZnAc favors tin-based structures while the higher solubility of ZnCl2 promotes zinc-based structures. This results in different optimal values of Zn:Sn molar ratios, for each of these two precursors. Furthermore, using ZnCl2 as zinc precursor results in larger and significantly more homogeneous nanostructures (less mixture of phases/structures) and more reproducible reactions, which is due to its higher solubility, promoting a faster evolution of the species in synthesis. As directing growth agent, the surfactant has crucial role to achieve the nanowire form. Similarly to what is observed for the NaOH concentration, the EDA concentration influences the solution’s pH which favors the formation of either tin-based structures (lower concentrations) or zinc-based structures (higher concentrations). The growth of ZnSnO3 nanowires was achieved for intermediate values only, proving that both EDA and NaOH optimal concentrations are a compromise between the role they play in the synthesis and their influence on the pH [23].
ZnSnO3 is achievable for more energetic syntheses, i.e., under higher pressure (higher volume), while for lower energetic processes the more energetically stable phases (Zn2SnO4 and SnO2) are obtained. It was observed that at least 12 h of synthesis at 200 °C were necessary to predominantly obtain nanowires, while very long synthesis (36 h), or at higher temperatures (220 °C) result in the decomposition of the ZnSnO3 phase into Zn2SnO4 and SnO2 [24].
The optimal conditions to achieve the ZnSnO3 nanowires were a Zn:Sn molar ratio of 2:1 (1:1) using ZnCl2 (ZnAc) as zinc precursor, a NaOH concentration of 0.24 M with a H2O:EDA volume ratio of 7.5:7.5 mL:mL for syntheses at 200 °C for 24 h (Figure 1). This shows the viability of the hydrothermal process to achieve a metastable ZTO phase at low temperatures, provided that proper control of the synthesis parameters exists.
Figure 1 – ZnSnO3 nanowires produced using as zinc precursor (a) ZnCl2 and (b) ZnAc. From reference [24].
4. Properties and Applications
The ZnSnO3 nanowires achieved were characterized concerning their optical, electrical and piezoelectrical properties, resulting in electrical resistivity (measured in vacuum inside SEM) of 1.4 kΩ·cm, optical band-gap of 3.6 eV and piezoelectric constant of 23 pm/V [23][27]. These properties allow to envisage application on numerous next-generation nanoscale devices such as nanogenerators (reported in [27]), sensors, photocatalysis, solar cells, resistive switching memories, and transistors [18][28][29][30][31][32][33][34].
References
- L. Schmidt-Mende, J.L. MacManus-Driscoll; ZnO - nanostructures, defects, and devices. Mater. Today. 2007, 10, 40–48, 10.1016/S1369-7021(07)70078-0.
- Kołodziejczak-Radzimska A., Jesionowski T.; Zinc Oxide—From Synthesis to Application: A Review. Materials 2014, 7, 2833-2881, 10.3390/ma7042833.
- Pimentel, A.; Rodrigues, J.; Duarte, P.; Nunes, D.; Costa, F.M.; Monteiro, T.; Martins, R.; Fortunato, E.; Effect of solvents on ZnO nanostructures synthesized by solvothermal method assisted by microwave radiation: a photocatalytic study. J. Mater. Sci. 2015, 50, 5777–5787, 10.1007/s10853-015-9125-7.
- Fernandes, C.; Santa, A.; Santos, Â.; Bahubalindruni, P.; Deuermeier, J.; Martins, R.; Fortunato, E.; Barquinha, P.; A Sustainable Approach to Flexible Electronics with Zinc-Tin Oxide Thin-Film Transistors. Adv. Electron. Mater. 2018, 4 , 1800032, 10.1002/aelm.201800032.
- Salgueiro, D.; Kiazadeh, A.; Branquinho, R.; Santos, L.; Barquinha, P.; Martins, R.; Fortunato, E.; Solution based zinc tin oxide TFTs: the dual role of the organic solvent. J. Phys. D. Appl. Phys. 2017, 50 , 065106, 10.1088/1361-6463/50/6/065106.
- Sun, S.; Liang, S.; Morphological zinc stannate: synthesis, fundamental properties and applications. J. Mater. Chem. A. 2017, 5, 20534–20560, 10.1039/C7TA06221D.
- Baruah, S.; Dutta, J.; Zinc stannate nanostructures: hydrothermal synthesis. Sci. Technol. Adv. Mater. 2011, 12, 013004, 10.1088/1468-6996/12/1/013004.
- Guo, R.; Guo, Y.; Duan, H.; Li, H.; Liu, H.; Synthesis of Orthorhombic Perovskite-Type ZnSnO3 Single-Crystal Nanoplates and Their Application in Energy Harvesting. ACS Appl. Mater. Interfaces. 2017 , 9, 8271–8279, 10.1021/acsami.6b16629.
- Wu, J.M.; Chen, C.-Y.; Zhang, Y.; Chen, K.-H.; Yang, Y.; Hu, Y.; He, J.-H.; Wang, Z.L.; Ultrahigh Sensitive Piezotronic Strain Sensors Based on a ZnSnO3 Nanowire/Microwire, . ACS Nano 2012, 6, 4369–4374, 10.1021/nn3010558..
- Lo, M.-K.; Lee, S.-Y.; Chang, K.-S.; Study of ZnSnO3-Nanowire Piezophotocatalyst Using Two-Step Hydrothermal Synthesis. J. Phys. Chem. C. 2015, 119, 5218–5224, 10.1021/acs.jpcc.5b00282.
- Nakayama, M.; Nogami, M.; Yoshida, M.; Katsumata, T.; Inaguma, Y.; First-Principles Studies on Novel Polar Oxide ZnSnO3; Pressure-Induced Phase Transition and Electric Properties. Adv. Mater. 2010, 22, 2579–2582, 10.1002/adma.200903432.
- Dal Corso, A.; Posternak, M.; Resta, R.; Baldereschi, A.; Ab initio study of piezoelectricity and spontaneous polarization in ZnO. Phys. Rev. B. 1994, 50 , 10715–10721, 10.1103/PhysRevB.50.10715.
- Yuanbing Mao; Tae-Jin Park; Stanislaus S. Wong; Synthesis of classes of ternary metal oxide nanostructures. Chemical Communications 2005, n.a., 5721-5735, 10.1039/b509960a.
- Anuja Datta; Devajyoti Mukherjee; Corisa Kons; Sarath Witanachchi; Pritish Mukherjee; Evidence of Superior Ferroelectricity in Structurally Welded ZnSnO3Nanowire Arrays. Small 2014, 10, n.a., 10.1002/smll.201401249.
- Masahiro Miyauchi; Zhifu Liu; Zhi-Gang Zhao; Srinivasan Anandan; Kohjiro Hara; Single crystalline zinc stannate nanoparticles for efficient photo-electrochemical devices. Chemical Communications 2010, 46, 1529-1531, 10.1039/b921010e.
- Ming Lei; Yue Sheng; Long Wan; K. Bi; Kai Huang; RenXu Jia; Jun Liu; Yonggang Wang; A novel self-catalytic route to zinc stannate nanowires and cathodoluminescence and electrical transport properties of a single nanowire. Journal of Alloys and Compounds 2016, 657, 394-399, 10.1016/j.jallcom.2015.10.085.
- Christina Pang; Bin Yan; Lei Liao; Bo Liu; Zhe Zheng; Tom Wu; Handong Sun; Ting Yu; Synthesis, characterization and opto-electrical properties of ternary Zn2SnO4nanowires. Nanotechnology 2010, 21, n.a., 10.1088/0957-4484/21/46/465706.
- Taekyung Lim; Hwansoo Kim; M. Meyyappan; Sanghyun Ju; Photostable Zn2SnO4 Nanowire Transistors for Transparent Displays. ACS Nano 2012, 6, 4912-4920, 10.1021/nn300401w.
- Di Chen; Zhe Liu; Bo Liang; Xianfu Wang; Guozhen Shen; Transparent metal oxide nanowire transistors. Nanoscale 2012, 4, 3001, 10.1039/c2nr30445g.
- K. Shantha Shankar; A.K. Raychaudhuri; Fabrication of nanowires of multicomponent oxides: Review of recent advances. Materials Science and Engineering: C 2005, 25, 738-751, 10.1016/j.msec.2005.06.054.
- Xiaoxu Ji; Xin-Tang Huang; Jinping Liu; Jian Jiang; Xin Li; Ruimin Ding; Yingying Hu; Fei Wu; Qiang Li; Hydrothermal synthesis of novel Zn2SnO4 octahedron microstructures assembled with hexagon nanoplates. Journal of Alloys and Compounds 2010, 503, L21-L25, 10.1016/j.jallcom.2009.12.038.
- Jia Zeng; MuDi Xin; Li; Hao Wang; Hui Yan; Wenjun Zhang; Transformation Process and Photocatalytic Activities of Hydrothermally Synthesized Zn2SnO4 Nanocrystals. The Journal of Physical Chemistry C 2008, 112, 4159-4167, 10.1021/jp7113797.
- Ana Rovisco; Rita Branquinho; Jorge Martins; Maria João Oliveira; Daniela Nunes; Elvira Fortunato; R. Martins; Pedro Barquinha; Seed-Layer Free Zinc Tin Oxide Tailored Nanostructures for Nanoelectronic Applications: Effect of Chemical Parameters. ACS Applied Nano Materials 2018, 1, 3986-3997, 10.1021/acsanm.8b00743.
- Ana Rovisco; Rita Albuquerque; Jorge Martins; Elvira Fortunato; Rodrigo Martins; P. Barquinha; Growth Mechanism of Seed-Layer Free ZnSnO3 Nanowires: Effect of Physical Parameters.. Nanomaterials 2019, 9, 1002, 10.3390/nano9071002.
- Bin Su; Yuchen Wu; Lei Jiang; The art of aligning one-dimensional (1D) nanostructures. Chemical Society Reviews 2012, 41, 7832-7856, 10.1039/c2cs35187k.
- T. Tharsika; A.S.M.A. Haseeb; Sheikh A. Akbar; Mohd Faizul Mohd Sabri; Y.H. Wong; Gas sensing properties of zinc stannate (Zn2SnO4) nanowires prepared by carbon assisted thermal evaporation process. Journal of Alloys and Compounds 2015, 618, 455-462, 10.1016/j.jallcom.2014.08.192.
- Ana Rovisco; Andreia Dos Santos; Tobias Cramer; Jorge Martins; Rita Albuquerque; Hugo Águas; Beatrice Fraboni; Elvira Fortunato; Rodrigo Martins; Rui Igreja; et al.P. Barquinha Piezoelectricity Enhancement of Nanogenerators Based on PDMS and ZnSnO3 Nanowires through Microstructuration. ACS Applied Materials & Interfaces 2020, 12, 18421-18430, 10.1021/acsami.9b21636.
- Jyh Ming Wu; Chen Xu; Yan Zhang; Zhong Lin Wang; Lead-Free Nanogenerator Made from Single ZnSnO3 Microbelt. ACS Nano 2012, 6, 4335-4340, 10.1021/nn300951d.
- Jyh Ming Wu; Chen Xu; Yan Zhang; Ya Yang; Yusheng Zhou; Zhong Lin Wang; Flexible and Transparent Nanogenerators Based on a Composite of Lead-Free ZnSnO3Triangular-Belts. Advanced Materials 2012, 24, 6094-6099, 10.1002/adma.201202445.
- Yongming Fu; Yuxin Nie; Yayu Zhao; Penglei Wang; Lili Xing; Yan Zhang; Xinyu Xue; Detecting Liquefied Petroleum Gas (LPG) at Room Temperature Using ZnSnO3/ZnO Nanowire Piezo-Nanogenerator as Self-Powered Gas Sensor. ACS Applied Materials & Interfaces 2015, 7, 10482-10490, 10.1021/acsami.5b01822.
- X. Y. Xue; Y. J. Chen; Y. G. Wang; T. H. Wang; Synthesis and ethanol sensing properties of ZnSnO3 nanowires. Applied Physics Letters 2005, 86, 233101, 10.1063/1.1944204.
- Lili Wang; Tingting Zhou; Rui Zhang; Zheng Lou; Jianan Deng; Sen Liu; Comparison of toluene sensing performances of zinc stannate with different morphology-based gas sensors. Sensors and Actuators B: Chemical 2016, 227, 448-455, 10.1016/j.snb.2015.12.097.
- Muhammad Najam Khan; Muna H. Al-Hinai; Ashraf Al-Hinai; Joydeep Dutta; Visible light photocatalysis of mixed phase zinc stannate/zinc oxide nanostructures precipitated at room temperature in aqueous media. Ceramics International 2014, 40, 8743-8752, 10.1016/j.ceramint.2014.01.094.
- Sawanta S. Mali; Chang Su Shim; Chang Kook Hong; Highly porous Zinc Stannate (Zn2SnO4) nanofibers scaffold photoelectrodes for efficient methyl ammonium halide perovskite solar cells. Scientific Reports 2015, 5, 11424, 10.1038/srep11424.