Glucagon-like peptide 1 receptor agonists (GLP-1 RAs) are a relatively new class of anti-diabetic medications that have exhibited very promising results in the treatment of type 2 diabetes mellitus (T2DM). According to the 2021 American Diabetes Associ drugs that act by mimicking the function of endogenous glucagon-like peptide 1. They constitute valuable agents for the management of T2DM as, in addition to exerting a strong hypoglycemic action, they present cardiorenal protective properties, promote weight loss, and have a good safety profile, particularly with respect to the risk of hypoglycemia. Knowledge of the potential sex-specific effects of these medications is extremely useful for the implementation guidelines, they constitute one ofof individualized therapeutic plans in the treatment of T2DM. Due to the preferred add-on agents when metformin monotherapy and lifestyle modifications have failed to achieve adequate glycemic controlcedent of studies having identified sexual dimorphic elements regarding the action of other antidiabetic agents, ongoing research has attempted to examine whether this is also the case for GLP-1 RAs. This narrative review aims to present the available data regarding the sex-specific action of GLP-1 RAs as well as to discuss the potential pathophysiologic mechanisms explaining these dissimilarities.
- GLP-1 receptor agonists
- sex/gender differences
- diabetes
- weight loss
1. Introduction
2. GLP-1 RAs: An Overview of the Class
| Exenatide | Liraglutide | Albiglutide | Lixisenatide | Dulaglutide | Semaglutide | |
|---|---|---|---|---|---|---|
| Molecular weight (Dalton) | 4187 [26] | 3751 [27] | 3283.6 [28] | 4858 [29] | 59,669 [30] | 4114 [31] |
| Molecular formation | C184H282N50O60S [26] | C172H265N43O51 [27] | C148H224N40O45 [28] | C215H347N61O65S [29] | C2646H4044N704O836S18 [32] | C187H291N45O59 [31] |
| Structure | Natural peptide (exendin-4) from the saliva of the lizard Heloderma suspectum (53% homology) [33] | Slightly modified GLP-1 (97% homology) with free fatty acid side chain attached [ |
4. Sex as a Determinant of Different Adverse Outcomes of GLP-1 RA Treatment
In general, GLP-1 RAs possess a good safety profile with minor adverse events. The most commonly reported side effects of these drugs are GI adverse events (GI AE) such as nausea and vomiting, while symptoms that are less frequently encountered are diarrhea and constipation. Additionally, there have been a few associations with incidents of major adverse outcomes such as episodes of acute pancreatitis and thyroid malignancy. However, these have been observed mainly in animal studies and a cause–effect relationship among GLP-1 Ras, and these outcomes have not been established in human clinical trials [71].
In an analysis of data derived from two randomized control trials, testing monotherapy with dulaglutide versus liraglutide for the treatment of T2DM, more women reported overall treatment-emergent adverse events compared to men in both arms of the study (86.5% of women vs. 61.4% of men for dulaglutide and 83.3% vs. 65.5% for liraglutide, respectively). GI AE in particular also occurred more frequently in women. Interestingly, patients with higher percentages of GI AE were more likely to achieve greater weight loss. Thus, sex-related differences in GI AE occurrence may be indirectly associated with increased GLP-1 RA efficacy among women.
The apparent sex-specific differences in the frequency of GI AE and in the efficacy of GLP-1 RA treatment were partially attributed by the authors of this analysis to higher exposures (defined by the authors as “higher drug plasma concentrations”) of dulaglutide and liraglutide in females, possibly due to their lower average body weight [57]. The association between higher levels of GLP-1 RA exposure and a higher frequency of adverse GI events has already been highlighted in several studies. For instance an exposure-focused study of liraglutide identified nausea and vomiting as established exposure-dependent adverse events of liraglutide treatment [72]. Nonetheless, the findings of an exposure–response analysis of semaglutide provided more insight into the matter by concluding that these particular GI AE, although being exposure dependent in general, were more frequent in females across similar levels of exposure [73]. This could potentially suggest that exposure does not comprise the main determinant of the sex-differences observed in the incidence of GI AE after treatment with GLP-1 RAs. Table 2 provides a more detailed view on the relationship between exposure/dose, effectiveness and AE of GLP-1 RAs.
Table 2 Relationship between exposure/dose, effectiveness and AE of GLP-1 RAs.| Exposure–Response Analyses of Semaglutide (Kristin C.C. Petri et al.) [73] | Exposure–Response Analyses of Liraglutide (J.P.H.Wilding et al.) [72] |
Dose-Finding Study of Semaglutide (Michael A. Nauck et al.) [74] | ||||
|---|---|---|---|---|---|---|
| HbA1c reduction | Exposure dependent | Exposure dependent | Dose dependent | |||
| 33 | ] | Two modified GLP-1 molecules amino-terminally attached to the linear structure of albumin | [33] | Exenatide plus poly-lysine tail [33] | Two modified GLP-1 molecules attached to an immunoglobulin (Fc) fragment [33] | Slightly modified GLP-1 (94% homology) with free fatty acid side chain attached |
| Hypoglycemia | ||||||
| Data not provided | ||||||
| Exposure independent ( | p-value for slope = 0.83) | Dose independent | ||||
| Adverse effects of the gallbladder, malignant neoplasms, malignant breast neoplasms or benign colorectal neoplasms | Data not provided | Exposure independent | Data not provided | |||
| Acute pancreatitis | Data not provided | Exposure indepndent | No effect |
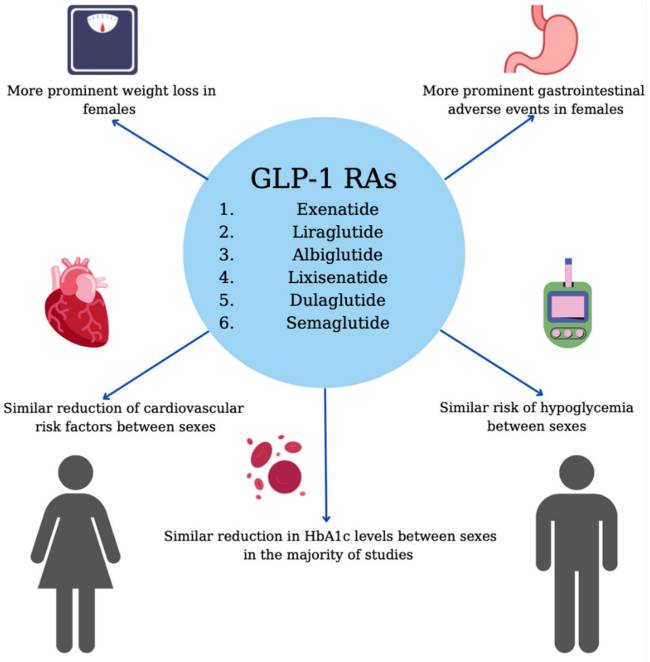
Further exploring this subject, a study analyzing data derived from two national Korean databases concluded that females were approximately twice as likely than males to report any adverse events linked to GLP-1 RA use, with a reporting ratio of 2.34 (reporting ratio: reporting rate of women/reporting rate of men). Regarding GI AE, the higher percentage reported by women could be partially attributed to the greater prevalence of functional GI disorders among the latter. Another interesting finding of this analysis was that women had a higher probability of experiencing headaches after treatment with GLP-1 RAs compared to men (risk ratio of 7.97) [76]. In contrast to the above, the percentage of patients having experienced episodes of nocturnal or total hypoglycemia did not differ significantly between sexes [57]. Figure 1 describes the sex-specific effects of GLP-1 RAs.
Sex-specific effects of GLP-1 RAs.
5. Discussion
It is beyond doubt that GLP-1 RAs constitute an effective tool in the management of T2DM and obesity. However, the hypothesis of whether sex could pose as a significant factor determining the efficacy and tolerability of GLP-1 RA treatment has been postulated and remains to be verified. In more detail, although several studies have indicated that sex may comprise a predictor of HbA1c reduction after the initiation of GLP-1 RA treatment, other studies have not found such a correlation, and therefore a definitive conclusion has not yet been drawn on the matter. In contrast, female sex appears to be a well-recognized independent factor linked to greater weight loss achievement after treatment with GLP-1 RAs. This is also the case for adverse events resulting from the use of these medications, which appear to manifest in higher percentages in women, mainly affecting the GI tract. According to existing studies, no conclusive result has emerged on whether sex may influence other parameters, such as WC, BP, lipid profile, and incidence of cardiovascular events. Table 3 summarizes the above-mentioned sex differences with respect to treatment with GLP-1 RAs.
| Differences in Response | ||||||
|---|---|---|---|---|---|---|
| Hypoglycemic efficacy | No sex differences noted in the majority of studies (ref. [21,50,51]) Female superiority noted in a few studies (ref. [5,6,52,53,54]) Male superiority noted in one study (ref. [22]) |
Body weight loss | Exposure dependent | Exposure dependent | Dose dependent | |
| Weight loss | Female superiority noted in the majority of studies (ref. [6,22,50,56,57]) | Increase in pulse rate | ||||
| MACE CVD risk factors i. WC | Exposure independent | ii. BP iii. Lipid profile |
No sex differences noted (ref. [68,69]) Higher risk in females (ref. [66] Lower risk in females (ref. [70]) Similar reduction between sexes (ref. [5]) Similar reduction between sexes (ref. [5,21]) Similar alterations (except for no reduction of LDL-C in females ref. [ | Exposure independent (p-value for slope = ∼0.18) | Dose dependent | [33] |
| Time to peak (h/days) | 2.1–2.2 h [34] | 11.0–13.75 h [35] | 3–5 days [36] | ≈2 h [37] | 48 h [38] | 24 h (subcutaneous injection) [39] |
| Elimination half-life (t 1/2) | 3.3–4 h [34] | 12.6–14.3 h [35] | 5.7–6.8 days [36] | 2.6 h [37] | 4.7–5.5 days (0.75 mg); 4.7 days (1.5 mg) [40] |
7.6 days [39] |
| Drug-drug interactions | Drug-drug interactions with digoxin, lovastatin, lisinopril, and acetaminophen [41] | Very low potential for pharmacokinetic drug–drug interactions related to cytochrome P450. No clinically relevant interactions between steady-state liraglutide and insulin detemir, atorvastatin, griseofulvin, paracetamol, digoxin, lisinopril or oral contraceptives [42] | Coadministration with chloroquine, hydroxychloroquine, lanreotide, octreotide, pasireotide, thioctic acid is not recommended [43] | Delays gastric emptying and can reduce the rate of absorption of oral medications such as acetaminophen, ethinyl estradiol, and warfarin. Does not affect the activity of cytochrome P450 isoenzymes [44] | Delays gastric emptying and can reduce the rate of absorption of oral medications. Concomitant use with an insulin secretagogue (e.g., sulfonylurea) or with insulin may increase the risk of hypoglycemia [38] | Minor delay of gastric emptying. No clinically relevant effect on the exposure of metformin, warfarin, atorvastatin or digoxin [45] |
| Adverse effects | Nausea, vomiting, diarrhea, dyspepsia, dizziness, headache [41] | Nausea, vomiting, diarrhea, dyspepsia, constipation, injection site reactions, low incidence of hypoglycemia [42] | Nausea, vomiting, diarrhea, constipation, gastroesophageal reflux disease, abdominal pain [46] | Nausea, vomiting, diarrhea. Concomitant use with an additional medication known to cause hypoglycemia can increase the risk of the latter [44] | Nausea, vomiting, diarrhea, abdominal pain, decreased appetite, hypoglycemia [47] | Nausea, vomiting and diarrhea, increased risk of cholelithiasis [48] |
3. Sex-Specific Efficacy of GLP-1 RAs
3.1. Hypoglycemic Efficacy
3.2. Weight Loss
3.3. Cardiovascular Risk and Major Adverse Cardiovascular Events (MACE)
| Episodes of nausea | |||||
i. GI AE | Exposure dependent | ii. Headaches iii. Hypoglycemia |
More frequent in females (ref. [57,73,76]) | Exposure dependent for episodes of any severity (p-value for slope = 0.004) | Dose dependent |
| 21 | ]) | More frequent in females (ref. [76]) |
Exposure independent for moderate/severe episodes (p-value for slope = 0.90) | ||
| No sex differences noted (ref. [ | 57 | Episodes of vomiting | Exposure dependent | Exposure dependent for episodes of any severity with doses up to 1.8 mg | Dose dependent |
| Exposure independent for moderate/severe episodes (p | |||||
| Adverse events | -value for slope = 0.85) | ||||
| ]) | Diarrhoea | Exposure independent | Data not provided | Dose dependent | |
| Constipation | Exposure independent | Data not provided | Data not provided | ||
| Elevated calcitonin levels (biomarker of C-cell activity and mass [75]) | Exposure independent | Exposure independent (p-value for slope = ∼0.49) | No effect |
Approaching the matter of sexual dimorphism of T2DM from a different angle, current research has shed light on several anatomical and functional dissimilarities between the female and male pancreas. More specifically, according to a study conducted on biopsies of human pancreases, the percentage of β-cells in the female pancreatic islets appeared to exceed that of the male islets (6% more β-cells on average in females) [81]. In contrast, another study of human pancreatic islets from 87 human pancreases showed no statistically significant difference in β-cell number between sexes. However, this same study highlighted the presence of different patterns of methylation between male and female islets, mainly with regard to the X chromosome and especially involving various genes that control insulin secretion and function. One of these genes is the Dual Specificity Phosphatase 9 (DUSP9) gene, which is protective against insulin resistance and which exhibited a higher percentage of methylation in female pancreatic islets. Increased methylation of a gene’s promoter is known to induce gene silencing [82], and thus increased methylation of the DUSP9 gene could act as a risk factor for insulin resistance and T2DM [83]. According to recent data stemming from the use of single-cell RNA sequencing, several both sex-specific and sex-independent differentially expressed genes (DEGs) related to insulin secretion and pathophysiology of diabetes have been identified in healthy and diabetic mice. With regard to sex-specific DEGs, the insulin II gene may serve as an example since it appeared to have a more pronounced expression in male compared to female β-cells of healthy mice. In the same study, other T2D sex-specific DEGs were identified in diabetic mice in comparison with healthy mice, some showing female superiority, while others being expressed to a greater extent in male mice. In females, some of these DEGs were associated with impaired endoplasmic reticulum stress responses. In male mice, specific upregulated DEGs were associated with mitochondrial function, which is known to affect the process of insulin secretion. All of the above indicate a high level of sexual dimorphism regarding the transcriptome of β-cells in healthy and diabetic mice. These data therefore support the notion that sex plays a pivotal role in the pathophysiology of T2DM and thus may be an important factor to consider when opting for personalized T2DM management. However, the two sexes share some aspects of the disease pathogenesis since sex-independent DEGs involving several disease-specific processes were also demonstrated [84].
Taking into consideration all of the above, it is evident that some aspects of GLP-1 RA pathophysiology and mechanism of action need to be further evaluated for better guidance regarding their use in the clinical setting. More specifically, since there seems to be a more prominent effect of GLP-1 RAs regarding weight reduction in women, it would be beneficial to shed light on the exact root of such differentiation. Therefore, more research needs to be done focusing not only on the role of increased drug exposure in women, but also on the impact of the female hormonal profile on the efficacy of GLP-1 RAs. Animal studies examining the impact of sex on the anorexigenic effects of GLP-1 RAs have demonstrated that, at least in part, sex hormones are responsible for the differential responses between males and females. More specifically, it has been shown that estrogen signaling is a crucial component via which GLP-1 RAs modify the food-reward aspect of food seeking. In this sense, higher estrogen concentrations in females, especially during their premenopausal years, could potentially serve as an explanation for why women may respond better to GLP-1 RAs [85]. This hypothesis is further supported by studies exploring the fluctuations in food intake of women throughout their menstrual cycle. These studies have observed that food intake tends to be lower during the follicular phase and the periovulatory period, albeit higher during the luteal phase of the cycle, thus exhibiting an inverse correlation to estrogen levels [86]. Prompted by these findings, some researchers have suggested the combined administration of GLP-1 RA and estrogen by developing a new conjugate molecule. The goal of this newly developed drug would be to hypothetically maximize weight loss results and achieve better control of metabolic syndrome, while simultaneously limiting the potential adverse gynecological events and tumorigenesis potential of estrogen, by ensuring targeted estrogen delivery specifically to GLP-1 receptor expressing cells [87]. A very interesting element of this strategy is that when administered as a conjugate molecule, GLP-1 RAs and estrogen act in a synergistic manner, activating a specific brain area which controls food intake, the supramammillary nucleus. This activation results in a greater modification of the food-reward behavior and therefore can potentially lead to a greater reduction in body weight [88]. Nonetheless, this approach has been primarily tested in animal studies and requires further research to be introduced in the clinical setting. Should substantial evidence supporting this correlation arise, the potential of co-administering estrogen with GLP-1 RAs as single-molecule peptides should be further evaluated.
Additionally, it is of utmost importance to clarify whether GLP-1 RA treatment exhibits sex-specific properties regarding the reduction in cardiovascular risk, by modifying several parameters such as WC, BP and lipids, as well as the decrease of MACE frequency. Should such a sexual dimorphism be demonstrated, physicians might have more opportunities to develop a tailored approach to diabetes and obesity, seeing that certain patients may benefit from earlier initiation of specific treatment.
Another aspect that should not be overlooked when attempting to examine the sexual dimorphic properties of GLP-1 RA treatment is the potential sex-specific differences encountered with regard to treatment adherence. In further detail, a study focusing on a variety of factors influencing adherence rates to noninsulin antidiabetic agents indicated that male sex was independently associated with greater treatment adherence (OR 1.14, p = 0.0001) [89]. These results were in accordance with the conclusions drawn by a retrospective study measuring the adherence and discontinuation rates of injectable GLP-1 RAs. At the 12- and 24-month time points after the initiation of GLP-1 RA, the percentage of men who qualified as adherent exceeded that of women (adherence rate: 54.2% for men versus 48.4% for women at 12 months and 51.1% versus 44.6% at 24 months, respectively, p < 0.01). With regard to the same time intervals, males also presented with lower rates of discontinuation of treatment (p < 0.01) [90]. These data suggest that adherence to prescribed treatment could be considered a factor that differentially affects the efficacy of GLP-1 RAs, possibly not allowing women to obtain the full benefit of their course of treatment. As a result, a sex-specific pattern in adherence rates may possibly obscure further primary differences in treatment outcomes between sexes. Hence, this subject could constitute a potential research topic in future studies, which could further clarify the impact of differences in adherence rates on the sexual dimorphism of GLP-1 RA treatment, whereas at the same time, achieve a better understanding regarding the sex-specific mechanism of action of these medications.
Our review of the literature has a number of limitations. Firstly, the majority of referenced studies are characterized by extremely heterogeneous patient populations. More specifically, the patient population in most studies differs with regard to duration of diabetes, current pharmacologic regimen, and/or previous medication history, among other factors. This lack of uniformity may confound some additional parameters that may influence the final results of each study. Furthermore, most of the available studies that examine the impact of sex on the outcome of GLP-1 RA treatment are post hoc analyses and, therefore, their conclusions constitute secondary measure outcomes. It is also important to note that although in several cases the association of GLP-1 RA with a certain variable had been evaluated, sex stratification was not conducted. Hence, the lack of sex-disaggregated data seems to be a fundamental limitation of our report and a major exclusion criterion for many other relevant studies currently available in the literature.
6. Conclusions
In conclusion, it is evident that the male and female responses to GLP-1 RAs differ at some level with respect to the achievement of weight loss and the incidence of GI AE. Regarding other parameters, namely HbA1c levels and cardiovascular risk, it remains to be seen whether there is a sex difference. This difference could be partly attributed to the sex specific pharmacodynamics of GLP-1 RAs which lead to dissimilar drug exposure levels. Alternatively, this dimorphism could be attributed to specific hormonal profiles of the sex. However, clinical trials do not always provide sufficient sex-specific data, which is of paramount importance to shed more light on the sex-specificity of the GLP-1 RA action. Thus, henceforward, sex-disaggregated data should stand in the epicenter of the research field, in order to enable the drawing of more accurate scientific conclusions and optimize the management of T2DM.
Author Contributions
E.R. and S.P. reviewed the literature and drafted the first version of the manuscript. T.K., M.G. and K.K. reviewed the literature and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
T.K. has received honoraria as a speaker from AstraZeneca, Boehringer Ingelheim, Pharmaserve Lilly and Novo Nordisk and has participated in sponsored studies by Eli-Lilly. M.G. has participated in sponsored studies by Eli-Lilly. K.K. has received honoraria for lectures/advisory boards and research support from Astra Zeneca, Boehringer Ingelheim, Pharmaserve Lilly, Sanofi-Aventis, ELPEN, MSD, and Novo Nordisk. E.R. and S.P. declare no conflict of interest.
References
- Castellana M., Cignarelli A., Brescia F., Perrini S., Natalicchio A., Laviola L., Giorgino F. Efficacy and safety of GLP-1 receptor agonists as add-on to SGLT2 inhibitors in type 2 diabetes mellitus: A meta-analysis. Sci. Rep. 2019;9:1–8. doi: 10.1038/s41598-019-55524-w.Castellana M., Cignarelli A., Brescia F., Perrini S., Natalicchio A., Laviola L., Giorgino F. Efficacy and safety of GLP-1 receptor agonists as add-on to SGLT2 inhibitors in type 2 diabetes mellitus: A meta-analysis. Sci. Rep. 2019;9:1–8. doi: 10.1038/s41598-019-55524-w. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- American Diabetes Association 6 Glycemic Targets: Standards of Medical Care in Diabetes—2021. Diabetes Care. 2020;44:S73–S84. doi: 10.2337/dc21-s006.American Diabetes Association 6 Glycemic Targets: Standards of Medical Care in Diabetes—2021. Diabetes Care. 2020;44:S73–S84. doi: 10.2337/dc21-s006. [PubMed] [CrossRef] [Google Scholar]
- Koufakis T., Papanas N., Zebekakis P., Kotsa K. Treatment options following metformin in primary prevention populations with type 2 diabetes: Which is the right road to take? Expert Rev. Clin. Pharmacol. 2021;14:1189–1192. doi: 10.1080/17512433.2021.1942843.Koufakis T., Papanas N., Zebekakis P., Kotsa K. Treatment options following metformin in primary prevention populations with type 2 diabetes: Which is the right road to take? Expert Rev. Clin. Pharmacol. 2021;14:1189–1192. doi: 10.1080/17512433.2021.1942843. [PubMed] [CrossRef] [Google Scholar]
- American Diabetes Association Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes—2021. Diabetes Care. 2021;44:S111–S124. doi: 10.2337/dc21-S009.American Diabetes Association Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes—2021. Diabetes Care. 2021;44:S111–S124. doi: 10.2337/dc21-S009. [PubMed] [CrossRef] [Google Scholar]
- Chiefari E., Capula C., Vero A., Oliverio R., Puccio L., Liguori R., Pullano V., Greco M., Foti D., Tirinato D., et al. Add-On Treatment with Liraglutide Improves Glycemic Control in Patients with Type 2 Diabetes on Metformin Therapy. Diabetes Technol. Ther. 2015;17:468–474. doi: 10.1089/dia.2014.0412. Chiefari E., Capula C., Vero A., Oliverio R., Puccio L., Liguori R., Pullano V., Greco M., Foti D., Tirinato D., et al. Add-On Treatment with Liraglutide Improves Glycemic Control in Patients with Type 2 Diabetes on Metformin Therapy. Diabetes Technol. Ther. 2015;17:468–474. doi: 10.1089/dia.2014.0412. [PubMed] [CrossRef] [Google Scholar]
- Quan H., Zhang H., Wei W., Fang T. Gender-related different effects of a combined therapy of Exenatide and Metformin on overweight or obesity patients with type 2 diabetes mellitus. J. Diabetes Its Complicat. 2016;30:686–692. doi: 10.1016/j.jdiacomp.2016.01.013. Quan H., Zhang H., Wei W., Fang T. Gender-related different effects of a combined therapy of Exenatide and Metformin on overweight or obesity patients with type 2 diabetes mellitus. J. Diabetes Its Complicat. 2016;30:686–692. doi: 10.1016/j.jdiacomp.2016.01.013. [PubMed] [CrossRef] [Google Scholar]
- Ryan P.M., Seltzer S., Hayward N.E., Rodriguez D.A., Sless R.T., Hawkes C.P. Safety and Efficacy of Glucagon-Like Peptide-1 Receptor Agonists in Children and Adolescents with Obesity: A Meta-Analysis. J. Pediatr. 2021;236:137–147. doi: 10.1016/j.jpeds.2021.05.009.Ryan P.M., Seltzer S., Hayward N.E., Rodriguez D.A., Sless R.T., Hawkes C.P. Safety and Efficacy of Glucagon-Like Peptide-1 Receptor Agonists in Children and Adolescents with Obesity: A Meta-Analysis. J. Pediatr. 2021;236:137–147. doi: 10.1016/j.jpeds.2021.05.009. [PubMed] [CrossRef] [Google Scholar]
- Chadda K.R., Cheng T.S., Ong K.K. GLP-1 agonists for obesity and type 2 diabetes in children: Systematic review and meta-analysis. Obes. Rev. 2021;22:e13177. doi: 10.1111/obr.13177.Chadda K.R., Cheng T.S., Ong K.K. GLP-1 agonists for obesity and type 2 diabetes in children: Systematic review and meta-analysis. Obes. Rev. 2021;22:e13177. doi: 10.1111/obr.13177. [PubMed] [CrossRef] [Google Scholar]
- Mayes J.S., Watson G.H. Direct effects of sex steroid hormones on adipose tissues and obesity. Obes. Rev. 2004;5:197–216. doi: 10.1111/j.1467-789X.2004.00152.x.Mayes J.S., Watson G.H. Direct effects of sex steroid hormones on adipose tissues and obesity. Obes. Rev. 2004;5:197–216. doi: 10.1111/j.1467-789X.2004.00152.x. [PubMed] [CrossRef] [Google Scholar]
- Kalish G.M., Barrett-Connor E., Laughlin G.A., Gulanski B.I. Association of Endogenous Sex Hormones and Insulin Resistance among Postmenopausal Women: Results from the Postmenopausal Estrogen/Progestin Intervention Trial. J. Clin. Endocrinol. Metab. 2003;88:1646–1652. doi: 10.1210/jc.2002-021375.Kalish G.M., Barrett-Connor E., Laughlin G.A., Gulanski B.I. Association of Endogenous Sex Hormones and Insulin Resistance among Postmenopausal Women: Results from the Postmenopausal Estrogen/Progestin Intervention Trial. J. Clin. Endocrinol. Metab. 2003;88:1646–1652. doi: 10.1210/jc.2002-021375. [PubMed] [CrossRef] [Google Scholar]
- Kautzky-Willer A., Harreiter J., Pacini G. Sex and Gender Differences in Risk, Pathophysiology and Complications of Type 2 Diabetes Mellitus. Endocr. Rev. 2016;37:278–316. doi: 10.1210/er.2015-1137.Kautzky-Willer A., Harreiter J., Pacini G. Sex and Gender Differences in Risk, Pathophysiology and Complications of Type 2 Diabetes Mellitus. Endocr. Rev. 2016;37:278–316. doi: 10.1210/er.2015-1137. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Pasquali R., Vicennati V., Gambineri A., Pagotto U. Sex-dependent role of glucocorticoids and androgens in the pathophysiology of human obesity. Int. J. Obes. 2008;32:1764–1779. doi: 10.1038/ijo.2008.129.Pasquali R., Vicennati V., Gambineri A., Pagotto U. Sex-dependent role of glucocorticoids and androgens in the pathophysiology of human obesity. Int. J. Obes. 2008;32:1764–1779. doi: 10.1038/ijo.2008.129. [PubMed] [CrossRef] [Google Scholar]
- Arnetz L., Ekberg N.R., Alvarsson M. Sex differences in type 2 diabetes: Focus on disease course and outcomes. Diabetes Metab. Syndr. Obes. Targets Ther. 2014;7:409–420. doi: 10.2147/DMSO.S51301.Arnetz L., Ekberg N.R., Alvarsson M. Sex differences in type 2 diabetes: Focus on disease course and outcomes. Diabetes Metab. Syndr. Obes. Targets Ther. 2014;7:409–420. doi: 10.2147/DMSO.S51301. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Mauvais-Jarvis F. Gender differences in glucose homeostasis and diabetes. Physiol. Behav. 2018;187:20–23. doi: 10.1016/j.physbeh.2017.08.016.Mauvais-Jarvis F. Gender differences in glucose homeostasis and diabetes. Physiol. Behav. 2018;187:20–23. doi: 10.1016/j.physbeh.2017.08.016. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Chiu C.-J., Wray L.A. Gender Differences in Functional Limitations in Adults Living with Type 2 Diabetes: Biobehavioral and Psychosocial Mediators. Ann. Behav. Med. 2010;41:71–82. doi: 10.1007/s12160-010-9226-0. Chiu C.-J., Wray L.A. Gender Differences in Functional Limitations in Adults Living with Type 2 Diabetes: Biobehavioral and Psychosocial Mediators. Ann. Behav. Med. 2010;41:71–82. doi: 10.1007/s12160-010-9226-0. [PubMed] [CrossRef] [Google Scholar]
- Roche M.M., Wang P.P. Sex Differences in All-Cause and Cardiovascular Mortality, Hospitalization for Individuals with and Without Diabetes, and Patients with Diabetes Diagnosed Early and Late. Diabetes Care. 2013;36:2582–2590. doi: 10.2337/dc12-1272.Roche M.M., Wang P.P. Sex Differences in All-Cause and Cardiovascular Mortality, Hospitalization for Individuals with and Without Diabetes, and Patients with Diabetes Diagnosed Early and Late. Diabetes Care. 2013;36:2582–2590. doi: 10.2337/dc12-1272. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Mauvais-Jarvis F., Clegg D.J., Hevener A.L. The Role of Estrogens in Control of Energy Balance and Glucose Homeostasis. Endocr. Rev. 2013;34:309–338. doi: 10.1210/er.2012-1055. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Root-Bernstein R., Podufaly A., Dillon P.F. Estradiol Binds to Insulin and Insulin Receptor Decreasing Insulin Binding in vitro. Front. Endocrinol. 2014;5:5. doi: 10.3389/fendo.2014.00118. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Plows J.F., Stanley J.L., Baker P.N., Reynolds C.M., Vickers M.H. The Pathophysiology of Gestational Diabetes Mellitus. Int. J. Mol. Sci. 2018;19:3342. doi: 10.3390/ijms19113342. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Koning S.H., Lutgers H.L., Hoogenberg K., Trompert C.A., Berg P.P.V.D., Wolffenbuttel B.H.R. Postpartum glucose follow-up and lifestyle management after gestational diabetes mellitus: General practitioner and patient perspectives. J. Diabetes Metab. Disord. 2016;15:1–9. doi: 10.1186/s40200-016-0282-2. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Pencek R., Blickensderfer A., Li Y., Brunell S.C., Anderson P.W. Exenatide Twice Daily: Analysis of Effectiveness and Safety Data Stratified by Age, Sex, Race, Duration of Diabetes, and Body Mass Index. Postgrad. Med. 2012;124:21–32. doi: 10.3810/pgm.2012.07.2567. [PubMed] [CrossRef] [Google Scholar]
- Anichini R., Cosimi S., Di Carlo A., Orsini P., De Bellis A., Seghieri G., Franconi F., Baccetti F. Gender difference in response predictors after 1-year exenatide therapy twice daily in type 2 diabetic patients: A real world experience. Diabetes Metab. Syndr. Obes. 2013;6:123–129. doi: 10.2147/DMSO.S42729. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Shah M., Vella A. Effects of GLP-1 on appetite and weight. Rev. Endocr. Metab. Disord. 2014;15:181–187. doi: 10.1007/s11154-014-9289-5. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Garber A.J. Long-Acting Glucagon-Like Peptide 1 Receptor Agonists. Diabetes Care. 2011;34:S279–S284. doi: 10.2337/dc11-s231. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Trujillo J.M., Nuffer W., Smith B.A. GLP-1 receptor agonists: An updated review of head-to-head clinical studies. Ther. Adv. Endocrinol. Metab. 2021;12:204201882199732. doi: 10.1177/2042018821997320. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- PubChem Exenatide. [(accessed on 28 February 2022)]; Available online: https://pubchem.ncbi.nlm.nih.gov/compound/45588096.
- PubChem Liraglutide. [(accessed on 28 February 2022)]; Available online: https://pubchem.ncbi.nlm.nih.gov/compound/16134956.
- PubChem Albiglutide. [(accessed on 28 February 2022)]; Available online: https://pubchem.ncbi.nlm.nih.gov/compound/145994868.
- PubChem Lixisenatide. [(accessed on 28 February 2022)]; Available online: https://pubchem.ncbi.nlm.nih.gov/compound/90472060.
- Drugs and Lactation Database (LactMed) National Library of Medicine (US); Bethesda, MD, USA: 2006. Dulaglutide. [PubMed] [Google Scholar]
- PubChem Semaglutide. [(accessed on 28 February 2022)]; Available online: https://pubchem.ncbi.nlm.nih.gov/compound/56843331.
- Dulaglutide. [(accessed on 28 February 2022)]. Available online: https://go.drugbank.com/drugs/DB09045.
- Nauck M.A., Meier J.J. Management of endocrine disease: Are all GLP-1 agonists equal in the treatment of type 2 diabetes? Eur. J. Endocrinol. 2019;181:R211–R234. doi: 10.1530/EJE-19-0566. [PubMed] [CrossRef] [Google Scholar]
- Kolterman O.G., Kim D.D., Shen L., Ruggles J.A., Nielsen L.L., Fineman M.S., Baron A.D. Pharmacokinetics, pharmacodynamics, and safety of exenatide in patients with type 2 diabetes mellitus. Am. J. Health Pharm. 2005;62:173–181. doi: 10.1093/ajhp/62.2.173. [PubMed] [CrossRef] [Google Scholar]
- Damholt B., Golor G., Wierich W., Pedersen P., Ekblom M., Zdravkovic M. An Open-Label, Parallel Group Study Investigating the Effects of Age and Gender on the Pharmacokinetics of the Once-Daily Glucagon-Like Peptide-1 Analogue Liraglutide. J. Clin. Pharmacol. 2006;46:635–641. doi: 10.1177/0091270006288215. [PubMed] [CrossRef] [Google Scholar]
- Matthews J.E., Stewart M.W., De Boever E.H., Dobbins R.L., Hodge R.J., Walker S.E., Holland M.C., Bush M.A. Albiglutide Study Group: Pharmacodynamics, Pharmacokinetics, Safety, and Tolerability of Albiglutide, a Long-Acting Glucagon-Like Peptide-1 Mimetic, in Patients with Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2008;93:4810–4817. doi: 10.1210/jc.2008-1518. [PubMed] [CrossRef] [Google Scholar]
- Becker R.H.A., Stechl J., Steinstraesser A., Golor G., Pellissier F. Lixisenatide reduces postprandial hyperglycaemia via gastrostatic and insulinotropic effects. Diabetes/Metab. Res. Rev. 2015;31:610–618. doi: 10.1002/dmrr.2647. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Smith L.L., Mosley J.F., Parke C., Brown J., Barris L.S., Phan L.D. Dulaglutide (Trulicity): The Third Once-Weekly GLP-1 Agonist. Pharm. Ther. 2016;41:357–360. [PMC free article] [PubMed] [Google Scholar]
- Marbury T.C., Flint A., Jacobsen J.B., Karsbøl J.D., Lasseter K. Pharmacokinetics and Tolerability of a Single Dose of Semaglutide, a Human Glucagon-Like Peptide-1 Analog, in Subjects with and Without Renal Impairment. Clin. Pharmacokinet. 2017;56:1381–1390. doi: 10.1007/s40262-017-0528-2. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Geiser J.S., Heathman M.A., Cui X., A Martin J., Loghin C., Chien J.Y., De La Peña A. Clinical Pharmacokinetics of Dulaglutide in Patients with Type 2 Diabetes: Analyses of Data from Clinical Trials. Clin. Pharmacokinet. 2015;55:625–634. doi: 10.1007/s40262-015-0338-3. [PubMed] [CrossRef] [Google Scholar]
- Bray G.M. Exenatide. Am. J. Health Pharm. 2006;63:411–418. doi: 10.2146/ajhp050459. [PubMed] [CrossRef] [Google Scholar]
- Scott L.J. Liraglutide: A Review of Its Use in Adult Patients with Type 2 Diabetes Mellitus. Drugs. 2014;74:2161–2174. doi: 10.1007/s40265-014-0321-6. [PubMed] [CrossRef] [Google Scholar]
- Albiglutide (Subcutaneous Route) Side Effects-Mayo Clinic. [(accessed on 28 February 2022)]. Available online: https://www.mayoclinic.org/drugs-supple-ments/albiglutide-subcutaneous-route/side-effects/drg-20095263?p=1.
- Leon N., LaCoursiere R., Yarosh D., Patel R.S. Lixisenatide (Adlyxin): A Once-Daily Incretin Mimetic Injection for Type-2 Diabetes. Pharm. Ther. 2017;42:676–711. [PMC free article] [PubMed] [Google Scholar]
- Hausner H., Karsbøl J.D., Holst A.G., Jacobsen J.B., Wagner F.-D., Golor G., Anderson T.W. Effect of Semaglutide on the Pharmacokinetics of Metformin, Warfarin, Atorvastatin and Digoxin in Healthy Subjects. Clin. Pharmacokinet. 2017;56:1391–1401. doi: 10.1007/s40262-017-0532-6. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Leiter L.A., Mallory J.M., Wilson T.H., Reinhardt R.R. Gastrointestinal safety across the albiglutide development programme. Diabetes Obes. Metab. 2016;18:930–935. doi: 10.1111/dom.12679. [PubMed] [CrossRef] [Google Scholar]
- Fala L. Trulicity (Dulaglutide): A New GLP-1 Receptor Agonist Once-Weekly Subcutaneous Injection Approved for the Treatment of Patients with Type 2 Diabetes. Am. Health Drug Benefits. 2015;8:131–134. [PMC free article] [PubMed] [Google Scholar]
- Smits M.M., Van Raalte D.H. Safety of Semaglutide. Front. Endocrinol. 2021;12:645563. doi: 10.3389/fendo.2021.645563. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Kaiafa G., Veneti S., Polychronopoulos G., Pilalas D., Daios S., Kanellos I., Didangelos T., Pagoni S., Savopoulos C. Is HbA1c an ideal biomarker of well-controlled diabetes? Postgrad. Med. J. 2021;97:380–383. doi: 10.1136/postgradmedj-2020-138756. [PubMed] [CrossRef] [Google Scholar]
- Gallwitz B., Dagogo-Jack S., Thieu V., Garcia-Perez L.E., Pavo I., Yu M., Robertson K.E., Zhang N., Giorgino F. Effect of once-weekly dulaglutide on glycated haemoglobin (HbA1c) and fasting blood glucose in patient subpopulations by gender, duration of diabetes and baseline HbA1c. Diabetes Obes. Metab. 2018;20:409–418. doi: 10.1111/dom.13086. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Onishi Y., Oura T., Nishiyama H., Ohyama S., Takeuchi M., Iwamoto N. Subgroup analysis of phase 3 studies of dulaglutide in Japanese patients with type 2 diabetes. Endocr. J. 2016;63:263–273. doi: 10.1507/endocrj.EJ15-0518. [PubMed] [CrossRef] [Google Scholar]
- Aleidi S., Issa A., Bustanji H., Khalil M., Bustanji Y. Adiponectin serum levels correlate with insulin resistance in type 2 diabetic patients. Saudi Pharm. J. 2015;23:250–256. doi: 10.1016/j.jsps.2014.11.011. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Hemmer A., Maiter D., Buysschaert M., Preumont V. Long-term effects of GLP-1 receptor agonists in type 2 diabetic patients: A retrospective real-life study in 131 patients. Diabetes Metab. Syndr. Clin. Res. Rev. 2019;13:332–336. doi: 10.1016/j.dsx.2018.09.007. [PubMed] [CrossRef] [Google Scholar]
- Durden E., Lenhart G., Lopez-Gonzalez L., Hammer M., Langer J. Predictors of glycemic control and diabetes-related costs among type 2 diabetes patients initiating therapy with liraglutide in the United States. J. Med. Econ. 2016;19:403–413. doi: 10.3111/13696998.2015.1131703. [PubMed] [CrossRef] [Google Scholar]
- Takahashi T.A., Johnson K.M. Menopause. Med. Clin. N. Am. 2015;99:521–534. doi: 10.1016/j.mcna.2015.01.006. [PubMed] [CrossRef] [Google Scholar]
- Buysschaert M., Preumont V., Oriot P., Paris I., Ponchon M., Scarnière D., Selvais P. One-year metabolic outcomes in patients with type 2 diabetes treated with exenatide in routine practice. Diabetes Metab. 2010;36:381–388. doi: 10.1016/j.diabet.2010.03.009. [PubMed] [CrossRef] [Google Scholar]
- Onishi Y., Oura T., Matsui A., Matsuura J., Iwamoto N. Analysis of efficacy and safety of dulaglutide 0.75 mg stratified by sex in patients with type 2 diabetes in 2 randomized, controlled phase 3 studies in Japan. Endocr. J. 2017;64:553–560. doi: 10.1507/endocrj.EJ16-0552. [PubMed] [CrossRef] [Google Scholar]
- Mirabelli M., Chiefari E., Caroleo P., Arcidiacono B., Corigliano D.M., Giuliano S., Brunetti F.S., Tanyolaç S., Foti D.P., Puccio L., et al. Long-Term Effectiveness of Liraglutide for Weight Management and Glycemic Control in Type 2 Diabetes. Int. J. Environ. Res. Public Health. 2020;17:207. doi: 10.3390/ijerph17010207. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Overgaard R.V., Petri K.C.C., Jacobsen L.V., Jensen C.B. Liraglutide 3.0 mg for Weight Management: A Population Pharmacokinetic Analysis. Clin. Pharmacokinet. 2016;55:1413–1422. doi: 10.1007/s40262-016-0410-7. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Sun F., Wu S., Guo S., Yu K., Yang Z., Li L., Zhang Y., Ji L., Zhan S. Effect of GLP-1 receptor agonists on waist circumference among type 2 diabetes patients: A systematic review and network meta-analysis. Endocrine. 2014;48:794–803. doi: 10.1007/s12020-014-0373-0. [PubMed] [CrossRef] [Google Scholar]
- Wang B., Zhong J., Lin H., Zhao Z., Yan Z., He H., Ni Y., Liu D., Zhu Z. Blood pressure-lowering effects of GLP-1 receptor agonists exenatide and liraglutide: A meta-analysis of clinical trials. Diabetes Obes. Metab. 2013;15:737–749. doi: 10.1111/dom.12085. [PubMed] [CrossRef] [Google Scholar]
- Sun F., Wu S., Wang J., Guo S., Chai S., Yang Z., Li L., Zhang Y., Ji L., Zhan S. Effect of Glucagon-like Peptide-1 Receptor Agonists on Lipid Profiles Among Type 2 Diabetes: A Systematic Review and Network Meta-analysis. Clin. Ther. 2015;37:225–241. doi: 10.1016/j.clinthera.2014.11.008. [PubMed] [CrossRef] [Google Scholar]
- Leon B.M., Maddox T.M. Diabetes and cardiovascular disease: Epidemiology, biological mechanisms, treatment recommendations and future research. World J. Diabetes. 2015;6:1246–1258. doi: 10.4239/wjd.v6.i13.1246. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Poudel I., Tejpal C., Rashid H., Jahan N. Major Adverse Cardiovascular Events: An Inevitable Outcome of ST-elevation myocardial infarction? A Literature Review. Cureus. 2019;11:e5280. doi: 10.7759/cureus.5280. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Hupfeld C., Mudaliar S. Navigating the “MACE” in Cardiovascular Outcomes Trials and decoding the relevance of Atherosclerotic Cardiovascular Disease benefits versus Heart Failure benefits. Diabetes Obes. Metab. 2019;21:1780–1789. doi: 10.1111/dom.13740. [PubMed] [CrossRef] [Google Scholar]
- Maric C. Risk factors for cardiovascular disease in women with diabetes. Gend. Med. 2010;7:551–556. doi: 10.1016/j.genm.2010.11.007. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Lin D.S.-H., Lee J.-K., Chen W.-J. Major adverse cardiovascular and limb events in patients with diabetes treated with GLP-1 receptor agonists vs DPP-4 inhibitors. Diabetologia. 2021;64:1949–1962. doi: 10.1007/s00125-021-05497-1. [PubMed] [CrossRef] [Google Scholar]
- Singh A., Singh R. Gender difference in cardiovascular outcomes with SGLT-2 inhibitors and GLP-1 receptor agonist in type 2 diabetes: A systematic review and meta-analysis of cardio-vascular outcome trials. Diabetes Metab. Syndr. Clin. Res. Rev. 2020;14:181–187. doi: 10.1016/j.dsx.2020.02.012. [PubMed] [CrossRef] [Google Scholar]
- D’Andrea E., Kesselheim A.S., Franklin J.M., Jung E.H., Hey S.P., Patorno E. Heterogeneity of antidiabetic treatment effect on the risk of major adverse cardiovascular events in type 2 diabetes: A systematic review and meta-analysis. Cardiovasc. Diabetol. 2020;19:1–11. doi: 10.1186/s12933-020-01133-1. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Raparelli V., Elharram M., Moura C.S., Abrahamowicz M., Bernatsky S., Behlouli H., Pilote L. Sex Differences in Cardiovascular Effectiveness of Newer Glucose-Lowering Drugs Added to Metformin in Type 2 Diabetes Mellitus. J. Am. Hear. Assoc. 2020;9:e012940. doi: 10.1161/JAHA.119.012940. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Nathan B.M., Rudser K.D., Abuzzahab M.J., Fox C.K., Coombes B., Bomberg E.M., Kelly A.S. Predictors of weight-loss response with glucagon-like peptide-1 receptor agonist treatment among adolescents with severe obesity. Clin. Obes. 2015;6:73–78. doi: 10.1111/cob.12128. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Wilding J.P.H., Overgaard R.V., Jacobsen L.V., Jensen C.B., Le Roux C.W. Exposure-response analyses of liraglutide 3.0 mg for weight management. Diabetes Obes. Metab. 2016;18:491–499. doi: 10.1111/dom.12639. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Petri K.C.C., Ingwersen S.H., Flint A., Zacho J., Overgaard R.V. Exposure-response analysis for evaluation of semaglutide dose levels in type 2 diabetes. Diabetes Obes. Metab. 2018;20:2238–2245. doi: 10.1111/dom.13358. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Nauck M.A., Petrie J.R., Sesti G., Mannucci E., Courrèges J.P., Lindegaard M.L., Jensen C.B., Atkin S.L. A Phase 2, Randomized, Dose-Finding Study of the Novel Once-Weekly Human GLP-1 Analog, Semaglutide, Compared with Placebo and Open-Label Liraglutide in Patients with Type 2 Diabetes. Diabetes Care. 2016;39:231–241. doi: 10.2337/dc15-0165. [PubMed] [CrossRef] [Google Scholar]
- Costante G., Meringolo D. Calcitonin as a biomarker of C cell disease: Recent achievements and current challenges. Endocrine. 2020;67:273–280. doi: 10.1007/s12020-019-02183-6. [PubMed] [CrossRef] [Google Scholar]
- Joung K.-I., Jung G.-W., Park H.-H., Lee H., Park S.-H., Shin J.-Y. Gender differences in adverse event reports associated with antidiabetic drugs. Sci. Rep. 2020;10:1–10. doi: 10.1038/s41598-020-74000-4. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- McGill J., Vlajnic A., Knutsen P., Recklein C., Rimler M., Fisher S. Effect of gender on treatment outcomes in type 2 diabetes mellitus. Diabetes Res. Clin. Pr. 2013;102:167–174. doi: 10.1016/j.diabres.2013.10.001. [PubMed] [CrossRef] [Google Scholar]
- Han E., Kim A., Lee S.J., Kim J.-Y., Kim J.H., Lee W.J., Lee B.-W. Characteristics of Dapagliflozin Responders: A Longitudinal, Prospective, Nationwide Dapagliflozin Surveillance Study in Korea. Diabetes Ther. 2018;9:1689–1701. doi: 10.1007/s13300-018-0470-9. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Li J., Li J., Shan Z., Yang W., Liu J., Tian H., Zhou Z., Ji Q., Weng J., Jia W., et al. Gender-differential effects on blood glucose levels between acarbose and metformin in Chinese patients with newly diagnosed type 2 diabetes: A sub-analysis of the MARCH trial. Endocr. J. 2021;68:69–79. doi: 10.1507/endocrj.EJ20-0006. [PubMed] [CrossRef] [Google Scholar]
- Campesi I., Seghieri G., Franconi F. Type 2 diabetic women are not small type 2 diabetic men: Sex-and-gender differences in antidiabetic drugs. Curr. Opin. Pharmacol. 2021;60:40–45. doi: 10.1016/j.coph.2021.06.007. [PubMed] [CrossRef] [Google Scholar]
- Marchese E., Rodeghier C., Monson R.S., McCracken B., Shi T., Schrock W., Martellotto J., Oberholzer J., Danielson K.K. Enumerating β-Cells in Whole Human Islets: Sex Differences and Associations with Clinical Outcomes After Islet Transplantation. Diabetes Care. 2015;38:e176–e177. doi: 10.2337/dc15-0723. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Hall E., Volkov P., Dayeh T., Esguerra J.L.S., Salö S., Eliasson L., Rönn T., Bacos K., Ling C. Sex differences in the genome-wide DNA methylation pattern and impact on gene expression, microRNA levels and insulin secretion in human pancreatic islets. Genome Biol. 2014;15:1–22. doi: 10.1186/s13059-014-0522-z. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Gannon M., Kulkarni R.N., Tse H.M., Mauvais-Jarvis F. Sex differences underlying pancreatic islet biology and its dysfunction. Mol. Metab. 2018;15:82–91. doi: 10.1016/j.molmet.2018.05.017. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Liu G., Li Y., Zhang T., Li M., Li S., He Q., Liu S., Xu M., Xiao T., Shao Z., et al. Single-cell RNA Sequencing Reveals Sexually Dimorphic Transcriptome and Type 2 Diabetes Genes in Mouse Islet β Cells. Genom. Proteom. Bioinform. 2021;19:408–422. doi: 10.1016/j.gpb.2021.07.004. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Richard J., Anderberg R.H., López-Ferreras L., Olandersson K., Skibicka K.P. Sex and estrogens alter the action of glucagon-like peptide-1 on reward. Biol. Sex Differ. 2016;7:6. doi: 10.1186/s13293-016-0059-9. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Gong E.J., Garrel D., Calloway D.H. Menstrual cycle and voluntary food intake. Am. J. Clin. Nutr. 1989;49:252–258. doi: 10.1093/ajcn/49.2.252. [PubMed] [CrossRef] [Google Scholar]
- Finan B., Yang B., Ottaway N., Stemmer K., Müller T.D., Yi C.-X., Habegger K., Schriever S.C., García-Cáceres C., Kabra D.G., et al. Targeted estrogen delivery reverses the metabolic syndrome. Nat. Med. 2012;18:1847–1856. doi: 10.1038/nm.3009. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Vogel H., Wolf S., Rabasa C., Rodriguez-Pacheco F., Babaei C.S., Stöber F., Goldschmidt J., DiMarchi R.D., Finan B., Tschöp M.H., et al. GLP-1 and estrogen conjugate acts in the supramammillary nucleus to reduce food-reward and body weight. Neuropharmacology. 2016;110:396–406. doi: 10.1016/j.neuropharm.2016.07.039. [PubMed] [CrossRef] [Google Scholar]
- Kirkman M.S., Rowan-Martin M.T., Levin R., Fonseca V.A., Schmittdiel J.A., Herman W.H., Aubert R.E. Determinants of Adherence to Diabetes Medications: Findings from a Large Pharmacy Claims Database. Diabetes Care. 2015;38:604–609. doi: 10.2337/dc14-2098. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Weiss T., Carr R.D., Pal S., Yang L., Sawhney B., Boggs R., Rajpathak S., Iglay K. Real-World Adherence and Discontinuation of Glucagon-Like Peptide-1 Receptor Agonists Therapy in Type 2 Diabetes Mellitus Patients in the United States. Patient Prefer. Adherence. 2020;14:2337–2345. doi: 10.2147/PPA.S277676. [PMC free article] [PubMed] [CrossRef] [Google Scholar]