Carbon nanotubes (CNTs) were incorporated into poly(styrene-block-isobutyleneblock-styrene) (SIBS) to investigate the physical characteristics of the resulting nanocomposite and its cytotoxicity to endothelial cells. CNTs were dispersed in chloroform using sonication following the addition of a SIBS solution at different ratios. We observed an uneven distribution of CNTs in the polymer matrix with sporadic bundles of interwoven nanotubes. Increasing the CNT content from 0 wt% to 8 wt% led to an increase in the tensile strength of SIBS films from 4.69 to 16.48 MPa. The engineering normal strain significantly decreased in 1 wt% SIBS–CNT films in comparison with the unmodified samples, whereas a further increase in the CNT content did not significantly affect this parameter. The incorporation of CNT into the SIBS matrix resulted in increased hydrophilicity, whereas no cytotoxicity towards endothelial cells was noted.
- nanocomposites
- carbon nanotubes
- biocompatible materials
- electrical conductivity
- triblock copolymers
1. Introduction
Carbon-based nanomaterials are currently actively tested for their potential usefulness in the modification of implantable medical devices [1][2], owing to their extraordinary high tensile strength which is attributed to the special organization of carbon atoms in a two-dimensional hexagonal grid [3]. Such cellular structure is characteristic of graphene and other allotropic modifications of carbon with sp2 hybridization such as fullerene and carbon nanotubes (CNTs).
The incorporation of carbon-based nanomaterials into polymer structure greatly enhances its mechanical properties, specifically tensile strength [4][5], which is of the upmost importance for implantable medical devices used in CVD, including vascular substitutes, chords, patches, and heart valves. On the other hand, the presence of carbon-based nanomaterials may lead to the reduced elasticity and increased rigidity of the final device, thus adversely affecting its function [6][7]. However, CNTs representing the coiled layers of graphene can be a promising material for the manufacture of implantable devices, owing to their flexibility and high tensile strength of about 50–150 GPa [8]. In addition to enhanced tensile strength, carbon-based nanomaterials may endow polymers with antimicrobial activity, electrical conductivity, or even improved hemocompatibility [9][10]. All of the above could be beneficial for implantable medical devices prone to thrombogenicity and infections.
2. Synthesis of CNT-Modified SIBS Films and Analysis of Their Structure
Triblock copolymers of isobutylene with styrene (Mn
= 50,000 g/mol, Mw
/Mn
< 1.3, central polyisobutylene block Mn
= 36,000 g/mol, Mw
/Mn
< 1.2) were synthesized by the sequential controlled cationic polymerization of isobutylene and styrene, employing the dicumyl chloride/TiCl4
/2,6-dimethylpyridine initiating system at −80 °C. The gel permeation chromatography (GPC) curve of polyisobutylene completely shifts to the high molecular weight region after the addition of the second monomer (styrene) into the polymerization mixture, thus confirming the successful formation of the desired triblock copolymer (Figure 1A). The1
H NMR spectra of SIBS (Figure 1B) exhibited well resolved signals corresponding to the phenyl protons (6.2–7.2 ppm,b
) of the polystyrene block and methyl protons (1.0–1.2 ppm,c
) of the polyisobutylene block. In addition to these signals, the spectra showed lower intensity resonances corresponding to the methyl protons of the first isobutylene unit attached to the initiator at 0.78 ppm (d
) and terminal chloromethine protons at 4.2–4.5 ppm (a
). The weight fraction of polystyrene in SIBS was calculated from the integral intensities of the corresponding signals and was 32%, which is very close to the weight fraction of styrene in the initial comonomers mixture (33%). The resultant copolymer was further used for CNT incorporation.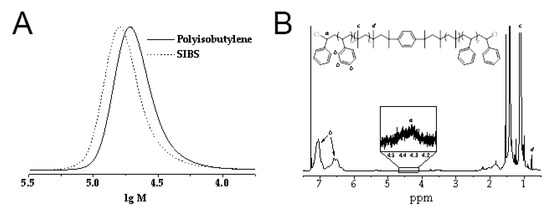
Figure 1.
A
B
1
[11]
. An absorbance peak at 2952 cm−1
corresponded to the asymmetric stretching vibrations of aliphatic –CH3
groups of isobutylene, while peaks at 2915–2847 cm−1
were determined by symmetric stretching vibrations of aliphatic –CH3
groups. The 1463 cm−1
peak was characteristic of the bending vibrations of aliphatic –CH2
and –CH3
groups. Absorbance peaks at 1373 and 1227 cm−1
correspond to the –CH3
groups of aliphatic chains and stretching vibrations of C–C bonds in the carbon backbone, respectively. The 1009 cm−1
peak reflected the stretching vibrations of C–C bonds in the carbon backbone. An increase in the CNT content led to a gradual reduction in the intensity of these absorption peaks due to the scattering of the reflected energy. The wide peak at 1600 cm−1
could be attributed to C–C vibrations in CNTs[12]
.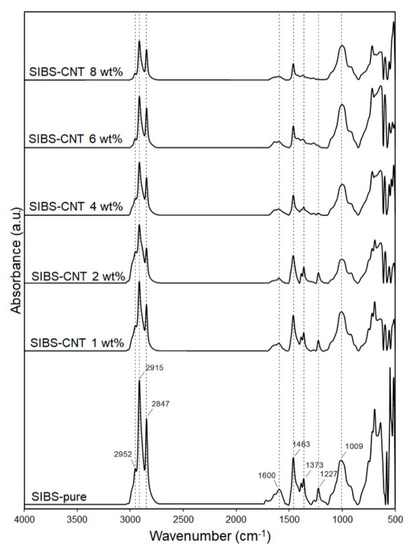
Figure 2.
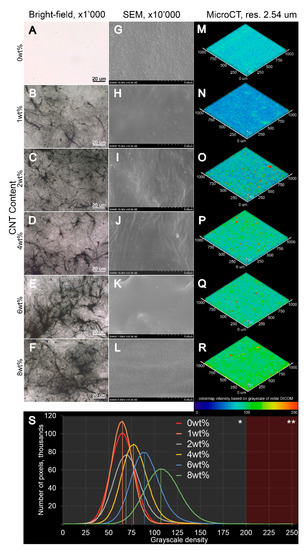
Figure 3.
A
L
A
F
G
L
M
R
S
3
(Figure 3M–R). Notably, both (i) and (ii) were found in all SIBS–CNT films regardless of their CNT concentration.3. Tensile Testing of CNT-Modified SIBS Films
All the CNT-modified SIBS films demonstrated a significantly higher ultimate tensile strength as compared to unmodified SIBS samples (p
< 0.01) (Figure 4A). Increasing the CNT content from 0 wt% to 4 wt% led to a linear increase in the Young’s modulus of SIBS films (Figure 4B). Upon reaching the CNT concentration of 4 wt%, the Young’s modulus soared to 52.7 MPa, which is 12 times higher than that of unmodified SIBS samples. In samples with a CNT concentration of 6 wt% and 8%, an even more dramatic rise in the Young’s modulus was documented (Figure 4B). The uniform elongation demonstrated a sharp drop in 1 wt% SIBS–CNT films in comparison with unmodified samples (p
< 0.0001) (Figure 4C), while a further increase in the CNT content did not significantly affect this parameter.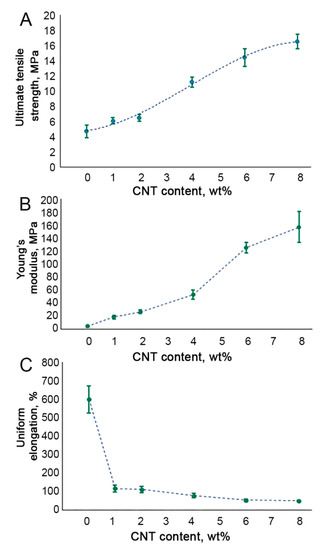
Figure 4.
A
B
C
4. Hydrophobicity Assessment of CNT-Modified SIBS Films
The incorporation of CNTs into the SIBS matrix resulted in a gradual reduction of the wetting angle, suggesting increased hydrophilicity (Figure 5). Although SIBS films with a CNT concentration of 1 wt% and 2 wt% demonstrated a similar wetting angle to that of control SIBS samples (all above 90°), a further CNT increase to 4 wt%, 6 wt% and 8 wt% led to a progressive reduction of the wetting angle to 84.4° ± 2.3°, 63.9° ± 3.2° and 66.7° ± 5.2°, respectively. Since materials with a contact angle below 90° can be considered as hydrophilic[13]
, 4 wt%, 6 wt% and 8 wt% SIBS–CNT films were characterized as hydrophilic in comparison with hydrophobic control SIBS samples.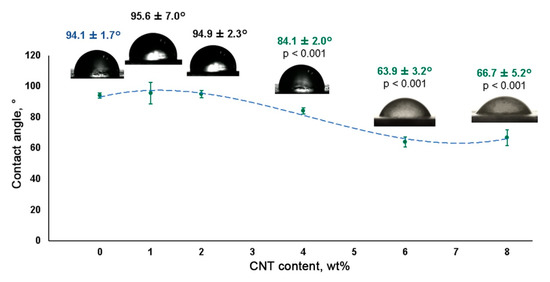
Figure 5.
5. Electrical Conductivity Measurement of CNT-Modified SIBS Films
The high electrical conductivity of a material is a requirement for the development of heart muscle patches[14]
. An increase in CNT content resulted in the elevated electrical conductivity of SIBS–CNT films from 0.07 S/cm at 1 wt% CNT to 8.55 S/cm at 8 wt% CNT (Figure 6). Considering the insulating properties of the unmodified SIBS (approximately 10−14
S/cm), it can be concluded that CNT incorporation led to a substantial (about 104
S/cm) increase in the electrical conductivity of the nanocomposite.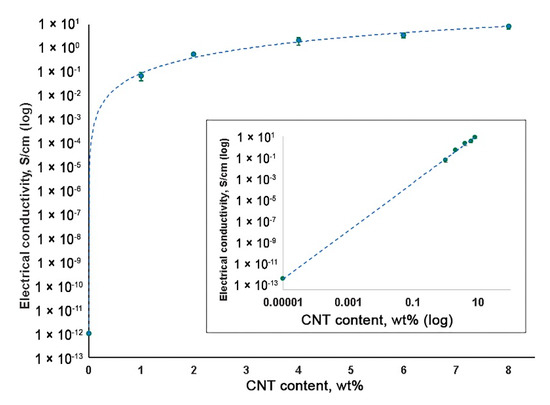
Figure 6.
6. Cytotoxicity of CNT-Modified SIBS Films
To evaluate the potential cytotoxicity of CNT-modified SIBS films, we immobilized them on cell culture plates to investigate the growth of HUVECs on their surface. Three days post seeding, the absolute number (per 1 mm2
) of HUVECs cultured on SIBS films regardless of CNT incorporation was significantly reduced in comparison with those grown in lab plastic (p
< 0.05) (Figure 7). This suggests a slower growth of HUVECs on SIBS films. Notably, the incorporation of CNTs into SIBS did not affect HUVEC growth (Figure 7A,B). The relative number of viable cells was nearly identical in all study groups except for the positive control where the cells were treated with pure CNTs (p
< 0.05) (Figure 7C). Thus, these data suggest that CNT-modified SIBS films exhibit cytotoxicity comparable to that of culture plastic and can therefore be considered biocompatible.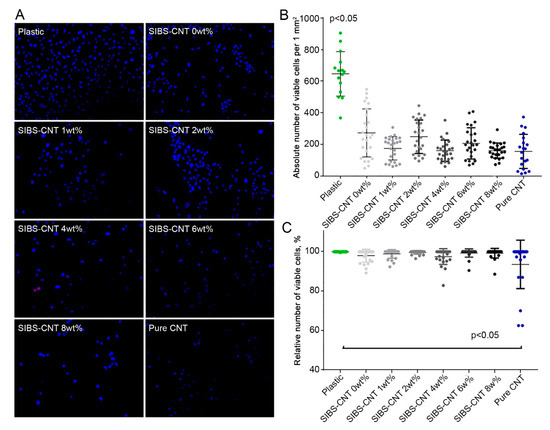
Figure 7.
A
B
2
C
7. In Vitro Oxidative Stability of CNT-Modified SIBS Films
To evaluate the stability of CNT-modified SIBS films, we performed an in vitro oxidation assay by submerging the samples in a solution of 0.1 M CoCl2 in 20% H2O2 for 14 days. We did not observe any evidence of corrosion or cracking over the course of the experiment, suggesting that both intact and CNT-modified SIBS are resistant to oxidative degradation (Figure 8A). However, the oxidative stress led in the formation of a new IR peak at 1724 cm−1, which is characteristic of stretching vibrations of C=O or –COOH bonds (Figure 8B).
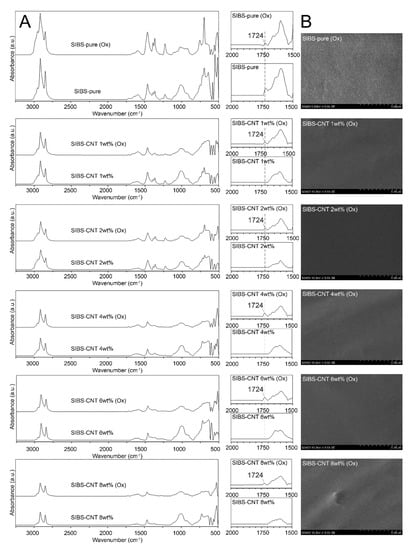
Figure 8. (A). SEM images of the surface of intact and CNT-modified SIBS films after 14 days of oxidation stress. (B). FTIR spectra of intact and CNT-modified SIBS films before and after 14 days of oxidation stress.
8. Conclusions
The incorporation of 8 wt% CNT into the SIBS matrix resulted in a significantly improved tensile strength and electrical conductivity, however, the high rigidity of this nanocomposite may limit its functionality. The most optimal results were documented for SIBS films with a CNT content of 4–6 wt%, which exhibited both high tensile strength and conductivity.
References
- Farzana Hussain; Mehdi Hojjati; Masami Okamoto; Russell E. Gorga; Review article: Polymer-matrix Nanocomposites, Processing, Manufacturing, and Application: An Overview. Journal of Composite Materials 2006, 40, 1511-1575, 10.1177/0021998306067321.
- Debabrata Maiti; Xiangmin Tong; Xiaozhou Mou; Kai Yang; Carbon-Based Nanomaterials for Biomedical Applications: A Recent Study. Frontiers in Pharmacology 2019, 9, 1401, 10.3389/fphar.2018.01401.
- Ali Eatemadi; Hadis Daraee; Hamzeh Karimkhanloo; Mohammad Kouhi; Nosratollah Zarghami; Abolfazl Akbarzadeh; Mozhgan Abasi; Younes Hanifehpour; Sang Woo Joo; Carbon nanotubes: properties, synthesis, purification, and medical applications. Nanoscale Research Letters 2014, 9, 393-393, 10.1186/1556-276x-9-393.
- Soad Z. Al Sheheri; Zahra M. Al-Amshany; Qana A. Al Sulami; Nada Y. Tashkandi; Mahmoud A. Hussein; Reda M. El-Shishtawy; The preparation of carbon nanofillers and their role on the performance of variable polymer nanocomposites. Designed Monomers and Polymers 2019, 22, 8-53, 10.1080/15685551.2019.1565664.
- Pb Kalakonda; S Banne; Enhanced mechanical properties of multiwalled carbon nanotubes/thermoplastic polyurethane nanocomposites. Nanomaterials and Nanotechnology 2019, 9, 184798041984085, 10.1177/1847980419840858.
- Alfred J. Crosby; Jong‐Young Lee; Polymer Nanocomposites: The “Nano” Effect on Mechanical Properties. Polymer Reviews 2007, 47, 217-229, 10.1080/15583720701271278.
- S.C. Tjong; Structural and mechanical properties of polymer nanocomposites. Materials Science and Engineering: R: Reports 2006, 53, 73-197, 10.1016/j.mser.2006.06.001.
- Mrinal Bhattacharya; Polymer Nanocomposites—A Comparison between Carbon Nanotubes, Graphene, and Clay as Nanofillers. Materials 2016, 9, 262, 10.3390/ma9040262.
- H.A. Khan; M.K. Sakharkar; A. Nayak; U. Kishore; A. Khan; Nanoparticles for biomedical applications: An overview. Nanobiomaterials 2018, 14, 357-384, 10.1016/b978-0-08-100716-7.00014-3.
- Ahmed Al-Jumaili; Surjith Alancherry; Kateryna Bazaka; Mohan V. Jacob; Review on the Antimicrobial Properties of Carbon Nanostructures. Materials 2017, 10, 1066, 10.3390/ma10091066.
- Shuaishuai Yuan; Jie Zhao; Shifang Luan; Shunjie Yan; Wanling Zheng; Jinghua Yin; Nuclease-Functionalized Poly(Styrene-b-isobutylene-b-styrene) Surface with Anti-Infection and Tissue Integration Bifunctions. ACS Applied Materials & Interfaces 2014, 6, 18078-18086, 10.1021/am504955g.
- Mohamed Abdel Salam; Abdullah Y. Obaid; Reda M. El-Shishtawy; Saleh A. Mohamed; Synthesis of nanocomposites of polypyrrole/carbon nanotubes/silver nano particles and their application in water disinfection. RSC Advances 2017, 7, 16878-16884, 10.1039/c7ra01033h.
- Matej Kanduč; Alexander Schlaich; Emanuel Schneck; Roland R. Netz; Water-Mediated Interactions between Hydrophilic and Hydrophobic Surfaces. Langmuir 2016, 32, 8767-8782, 10.1021/acs.langmuir.6b01727.
- Matteo Solazzo; Fergal J. O'brien; Valeria Nicolosi; Michael G. Monaghan; The rationale and emergence of electroconductive biomaterial scaffolds in cardiac tissue engineering. APL Bioengineering 2019, 3, 041501, 10.1063/1.5116579.
