Electrocatalytic CO2 reduction (ECR) is an attractive approach to convert atmospheric CO2 to value-added chemicals and fuels. However, this process is still hindered by sluggish CO2 reaction kinetics and the lack of efficient electrocatalysts. Therefore, new strategies for electrocatalyst design should be developed to solve these problems. Two-dimensional (2D) materials possess great potential in ECR because of their unique electronic and structural properties, excellent electrical conductivity, high atomic utilization and high specific surface area. In this review, we summarize the recent progress on 2D electrocatalysts applied in ECR. We first give a brief description of ECR fundamentals and then discuss in detail the development of different types of 2D electrocatalysts for ECR, including metal, graphene-based materials, transition metal dichalcogenides (TMDs), metal–organic frameworks (MOFs), metal oxide nanosheets and 2D materials incorporated with single atoms as single-atom catalysts (SACs). Metals, such as Ag, Cu, Au, Pt and Pd, graphene-based materials, metal-doped nitric carbide, TMDs and MOFs can mostly only produce CO with a Faradic efficiencies (FE) of 80~90%. [JR1] Particularly, SACs can exhibit FEs of CO higher than 90%. Metal oxides and graphene-based materials can produce HCOOH, but the FEs are generally lower than that of CO. Only Cu-based materials can produce high carbon products such as C2H4 but they have low product selectivity. It was proposed that the design and synthesis of novel 2D materials for ECR should be based on a thorough understanding of the reaction mechanism through combined theoretical prediction with experimental study, especially in situ characterization techniques. The gap between laboratory synthesis and large-scale production of 2D materials also needs to be closed for commercial applications.
- carbon dioxide
- two-dimensional materials
- electrocatalytic reduction
- graphene
- SAC
1. Introduction
2. Fundamentals of Electrochemical CO2 Reduction
2.1. CO
2
Properties and the Reduction Products
CO2 is one of the most stable molecules with a strong O=C=O double bond, and its bond energy is higher than that of C–H and C–C bonds. During electrocatalytic processes, the breaking of the O=C=O bond requires a high activation energy. In addition, since ECR consists of multiple elementary steps, these electrocatalytic processes are more demanding than, for example, water-splitting reactions.
Electrocatalysts can effectively reduce activation energy, accelerate reactions or increase desired product selectivity in ECR. It should also dissociate water in solution to promote proton–electron transfer because the proton-assisted multiple-electron transfer can be beneficial for CO2 activation. Depending on the different pathways and the number of protons and electrons transferred, a range of products can be formed, including C1 products, such as carbon monoxide (CO), methanol (CH3OH), formic acid (HCOOH), methane (CH4), or C2 products, such as ethylene (C2H4), ethanol (C2H5OH) and others [59][35]. C2 products with higher energy densities are more valuable, but the synthesis of C2 products is more difficult than that of C1 products because of larger numbers of required protons during conversion.
2.2. Single-Electron vs. Proton-Coupled Electron Reactions
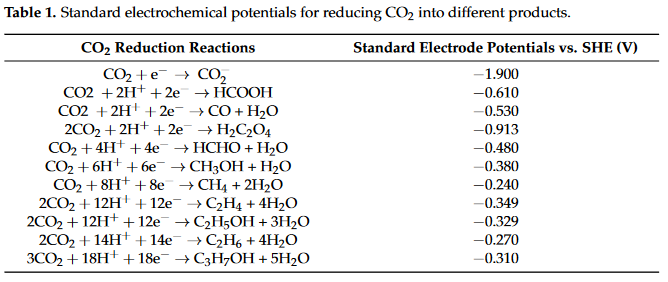
Research on ECR has been conducted ever since the early 19th century. ECR contains two half reactions occurring in the anode and the cathode, where different numbers of proton-coupled electron transfers occur. The standard electrode potentials of different reactions with reference to standard hydrogen electrons (SHEs) for different products in aqueous solutions are summarized in Table 1.
As shown in Figure 1, ECR starts by transferring a proton–electron pair to form carboxyl (*COOH) or formate (*OCHO) intermediate species [53][36]. These two intermediates can be further reduced by accepting different numbers of proton–electron pairs.
2.3. Factors Determining Product Selectivity
2.4. Key Parameters for Evaluating ECR Efficiency
In experimental ECR study, there are several key parameters for evaluating electrocatalyst performance, including overpotential, current density, Tafel slope, FE, turnover frequency (TOF) and stability of electrocatalysts. The overpotential can be regarded as the energy needed to drive the reaction. In other words, the higher the overpotential, the higher the energy required for the reaction and the more difficult the reaction to be realized. Current density reflects the rate of electrochemical reaction, and the larger the current density, the faster the electrocatalytic reaction. The Tafel slope can be obtained by fitting the linear region of the Tafel curve. With the increase in overpotential, if the current density increases dramatically, the slope will be small. FE is regarded as the fraction between charges transferred to specific products and the total charges in the electrocatalytic process, accounting for the selectivity and efficiency of the reaction. Larger FE suggests less energy loss, and smaller FE indicates more energy loss. TOF is the catalytic activity of each site under a certain overpotential, indicating the intrinsic activity of catalysts. The stability of electrocatalysts can be evaluated by long-term use under a specific voltage.3. Two-Dimensional Materials as Efficient ECR Electrocatalysts
Compared to bulk materials, 2D materials have a disordered state in their normal direction and a long-range order in the plane, showing a sheet structure. In addition, 2D materials can be exfoliated to single- or multiple-atom thickness, exhibiting interatomic covalent bonds in the plane. The physicochemical properties of 2D materials are different from that of bulk materials. The high atomic exposure rate, large specific surface area and flexible structure make them promising ECR catalysts.3.1. Two-Dimensional Metallic Materials
Bulk metal catalysts in ECR have been investigated for decades [109,110,111][39][40][41]. Metal electrocatalysts can be divided into two groups: noble metal (Au, Ag, Pt, etc.) and other earth-abundant transition metals (Co, Ni, Cu, etc.). Theoretical work has well disclosed that noble metals are better electrocatalysts compared with earth-abundant metals. Nevertheless, some intermediates, such as CO, could poison the active sites, which affects their stability. Large-scale application of precious metals is also not possible due to their high price and scarcity. It has been revealed that precious metals with 2D nanosheet structures can enhance their electrocatalytic performance effectively and improve the utilization of noble metals [72][42]. Huang et al. prepared a hexagonal Pd structure with 2.5 times higher electrocatalytic activity for the oxidation of formic acid compared to commercial Pd black [112][43]. The high performance could be attributed to nanosheet structure with more active sites, better electronic structure and larger surface area, as well as high atom utilization. The results suggested that the current density increases with the decrease in particle size. Moreover, multimetal composite nanosheets also exhibit excellent performance in electrocatalytic reactions because of the synergistic effect of different metals compared to single-metal electrocatalysts [114,115,116][44][45][46]. Nonprecious metals, including Fe, Co, Ni, etc., have also attracted much attention in electrocatalysis due to low prices, abundant resources and promising electrochemical properties as well as tunable electronic structures [118,119][47][48]. Studies have shown that Fe, Ni and Ti produce H2 as the major product in ECR due to their low HER overpotential and strong CO adsorption capability. Sn, Pb and In have poor binding energy with CO−2 intermediates and the final products are usually formate or formic acid. Moreover, Cu is the only metal catalyst that can produce C3 hydrocarbons by a C–C coupling mechanism [120][49]. Therefore, metal catalysts are effective for electrocatalysis due to their intrinsic activity and good conductivity. When the metal catalysts are synthesized with 2D nanosheets or other special nanostructures, the atom utilization, activity and selectivity could be improved, thus increasing electrocatalytic efficiency. Precious metals, such as Pd or Au, or multimetals with different ratios show excellent selectivity for CO generation. Nonprecious metals, such as Fe, Co and Ni, show poor ECR performance, while Sn, Pb and In can have good activity for the production of formate or formic acid. Particularly, Cu-based catalysts with different morphologies can exbibit different activities and selectivities for different products. Accordingly, the choice of metal, small nanosheet size and different morphology will have a positive effect on ECR activity and selectivity.3.2. Graphene and Graphene-Based Materials
Generally, graphene is a single layer of graphite with a zero-band gap. It is a promising electrocatalyst for CO2 reduction due to its high electron mobility, conductivity, unique electronic structure and large surface area. In addition, the high thermal conductivity can improve heat diffusion during the exothermic process, benefiting electrocatalysis. However, pristine graphene exhibits some drawbacks in ECR. For instance, the delocalized π bonding network of graphene negatively affects the adsorption of intermediates such as *COOH or *OCHO; therefore, graphene cannot effectively activate the CO2 molecule and presents high-energy barriers for intermediate formation, leading to low ECR activity [123][50]. Compared with nonmetal dopants, introducing single or multiple metal dopants to construct special graphene nanostructures could also exploit materials with excellent catalytic performance. Au nanoparticles embedded in graphene nanoribbon exhibit low overpotential, high FE for CO generation and excellent stability compared to amorphous carbon-supported Au nanoparticles, attributed to the change in electronic properties and the increase in active sites [127][51]. Graphene-based composite materials also possess excellent conductivities and larger surface areas compared to pristine graphene. Huang et al. synthesized N-doped graphene monolayer-coated Sn foil, showing excellent flexibility with a high FE of 92% for formate at −1.0 V vs. RHE [82][52] For graphene-like materials, graphitic carbon nitride (g–C3N4) typically exhibits good chemical and thermal stability under ambient conditions. However, poor conductivity and less active sites limit its potential application in ECR. Strategies, including doping metal and constructing composite, have been developed to increase the activity of g–C3N4. Metal atoms can effectively modulate the electronic structure of g–C3N4, thus improving the catalytic activity. The g–C3N4 nanosheet and multiwalled carbon nanotubes composite have been shown to exhibit excellent stability and good electrocatalytic performance for CO2 reduction to CO [85][53]. The electrocatalytic activity of the composite arises from the C–N bonds, and the high conductivity allows numerous electrons to transfer rapidly to the C–N sites.3.3. Two-Dimensional Transition Metal Dichalcogenides
Transition metal dichalcogenides (TMDs), such as MoS2, MoSe2 and WS2, have the general formula MX2, where M is a transition metal atom and X represents a chalcogen atom. The layers of X–M–X are stacked together by the van der Waals interaction. This is another class of 2D materials with exceptional physical and chemical properties to be applied as electrocatalysts [88][54]. The large surface area of 2D TMDs can provide more active sites for reactions such as CO2 reduction and water splitting [128,129][55][56]. DFT calculations suggested that the excellent catalytic properties for metal-terminated edges of 2D TMDs can be attributed to its metallic character and high d-electron density [87][57]. It has been confirmed that intermediates, such as COOH and CHO, prefer to adsorb on MoS2 and MoSe2 edges than the bridge S or Se atoms, and CO species adsorb selectively on the metal atoms during ECR [130,131][58][59]. Four different TMD materials, MoS2, MoSe2, WS2 and WSe2, have been investigated as ECR catalysts; of which, WSe2 nanoflakes exhibit the highest activity at an overpotential of 65 mV with an exceptional current density of 330 mA/cm2 and an FE of 85% for CO generation, surpassing other electrocatalysts [89][60]. The excellent performance can be attributed to WSe2 nanoflakes’ much lower charge transfer resistance and low work function. DFT calculations showed that the formation of CO is kinetically favorable.3.4. MOF Materials
MOFs are a new type of porous material with metal ions or clusters coordinated with organic ligands. Due to their high porosity, large specific surface area and flexible structure, MOFs have been applied in electrocatalytic research recently [43,132][34][61]. However, some disadvantages, such as poor conductivity and less active centers, hamper their electrocatalytic activity. Studies have shown that low-dimensional MOFs can achieve high electrocatalytic performance because MOF nanosheets can expose more active atoms and exhibit excellent electron transfer. Moreover, the metal atoms, ligand and the connection of metal centers with ligands also play vital roles in the electrocatalytic activity of MOFs. Kornienko et al. synthesized a thin film cobalt–porphyrin MOF on a carbon substrate. The FE of this sample was 76% at −0.70 V vs. RHE for CO2 conversion to CO. In addition, it possessed a low Tafel slope of 165 mV/decade and the electrocatalytic stability was excellent [90][62]. A Zn MOF synthesized by ionic exchange of Zn nodes with adsorbed Ni salts achieved an FE of 71.9% for CO at 10.48 mA/cm2 under the potential of −0.89V vs. RHE [91][63].3.5. Metal Oxide Nanosheets
Metal/metal oxide composites and oxide-derived metal have also been developed as ECR electrocatalysts with high performance [33,135,136][64][65][66]. Nevertheless, metal oxides still suffer problems such as instability and poor conductivity. It has been demonstrated that low-coordinated surface metal cations can be beneficial for CO2 adsorption during ECR. First principles calculation revealed that both the density of states (DOS) and the charge density around the conduction band edges can be greatly enhanced [137][67]. Therefore, faster carrier transport along the 2D ultrathin layer during ECR can be expected.3.6. Two-Dimensional Materials Incorporated Single-Atom Catalysts
Single-atom catalysts (SACs) have enormous advantages in electrocatalysis in terms of 100% atom utilization and intriguing electronic structures [138,139][68][69]. The traditionally supported nanoparticles or clusters exhibit structural irregularities on the nanoparticle surface. The surface reactivity has a close relationship with surface free energy, and the ratio between fully coordinated surface atoms and the number of vacancies distinguishes the reactivity of atoms with identical chemical compositions but different positions [140,141,142][70][71][72]. Therefore, the conventional clusters with different surface reactivities can result in poor product selectivity. SACs are different from clusters because the surface free energy is homogeneous and the number of vacancies in the nearest neighbors is the same. Thus, SACs can show good stability and special intrinsic activity [143][73]. Single-atom catalysts exhibit uniform active sites as ideal catalysts; however, one major challenge for SACs is synthesis because SACs are unstable and can agglomerate quickly due to high surface energy. Furthermore, the supported structures in the neighboring environment increase the heterogeneity of SACs’ active sites, and thus uniform activity cannot be easily achieved. However, 2D materials can be utilized as supports to improve the uniformity and performance of SACs in electrocatalytic reactions [144,145][74][75]. Studies have evidenced that 2D materials, such as MoS2, graphene and MXene, could stabilize the SACs and maintain their single-atom state [96,146,147,148,149,150,151][76][77][78][79][80][81][82].4. The Design of Electrolyzers
Numerous ECR studies are based on H-cells because they can be used conveniently to evaluate the activity of catalysts due to their easy operation and adaptability to different electrode materials and configurations [155,156][83][84]. A typical H-cell electrolyzer contains two chambers that are separated by an ion exchange membrane. The counter electrode is in the anodic chamber, whereas the working electrode and reference electrode are in the cathodic chamber. During the ECR process, CO2 is first dissolved in the liquid catholyte, such as KHCO3 and NaHCO3, and then transferred to the surface of the working electrode where ECR occurs. However, the thick diffusion layer and low solubility of CO2 in water limit CO2 mass transport to the catalyst surface, impeding its practical applications [157][85]. Commercial ECR application requires continuous operation to realize high production efficiency. A microfluidic reactor for ECR has been designed where the electrolyte can continuously flow through the electrolyzer [158,159][86][87]. The cathode and anode are separated by a very thin channel that is filled with flowing electrolytes. CO2 is supplied from the back of the cathodic gas diffusion electrode (GDE), and ECR occurs when CO2 reaches the surface of catalysts. The operation parameters, such as the composition and pH of the electrolyte, can be adjusted, which promotes ECR rates. GDEs are high-surface-area and porous electrodes. A typical GDE consists of a catalytic layer, a gas diffusion layer and a gas flow field. The structure and composition of GDE layers play a vital role in the transport of reactants and products because the transport processes influence the accumulation or depletion of intermediates on the catalysts, which determine ECR performance [160,161][88][89]. Lim et al. prepared Sn electrocatalysts with dense tips (SnDT) GDE, showing formate productivity of 65 mg h−1. It was stable for 72 h without changes in FE or current density [162][90]. A 2D nanosheet Cu-loaded GDE achieved the highest partial current density of 131 mA cm−2 for acetate production [163][91]. Although high current density was realized, the microfluidic reactor still suffered product crossover, where the reduction of oxygen diffused to the cathode and the reoxidation of cathodic products diffused to the anode, decreasing the overall energy efficiency and productivity.References
- Caldeira, K.; Wickett, M.E. Anthropogenic carbon and ocean pH. Nature 2003, 425, 365.
- Fu, Y.; Sun, D.; Chen, Y.; Huang, R.; Ding, Z.; Fu, X.; Li, Z. An amine-functionalized titanium metal-organic framework photocatalyst with visible-light-induced activity for CO2 reduction. Angew. Chem. Int. Ed. 2012, 51, 3364–3367.
- Li, J.R.; Ma, Y.; McCarthy, M.C.; Sculley, J.; Yu, J.; Jeong, H.K.; Balbuena, P.B.; Zhou, H.C. Carbon dioxide capture-related gas adsorption and separation in metal-organic frameworks. Coord. Chem. Rev. 2011, 255, 1791–1823.
- Li, X.; Yu, J.; Jaroniec, M.; Chen, X. Cocatalysts for Selective Photoreduction of CO2 into Solar Fuels. Chem. Rev. 2019, 119, 3962–4179.
- Rubin, E.S.; Davison, J.E.; Herzog, H.J. The cost of CO2 capture and storage. Int. J. Greenh. Gas Control 2015, 40, 378–400.
- Keith, D.W.; Holmes, G.; St Angelo, D.; Heidel, K. A Process for Capturing CO2 from the Atmosphere. Joule 2018, 2, 1573–1594.
- Drage, T.C.; Snape, C.E.; Stevens, L.A.; Wood, J.; Wang, J.; Cooper, A.I.; Dawson, R.; Guo, X.; Satterley, C.; Irons, R. Materials challenges for the development of solid sorbents for post-combustion carbon capture. J. Mater. Chem. 2012, 22, 2815–2823.
- Orr, J.F.M. CO2 capture and storage: Are we ready? Energy Environ. Sci. 2009, 2, 449–458.
- Maginn, E.J. What to Do with CO2. J. Phys. Chem. Lett. 2010, 1, 3478–3479.
- Ramdin, M.; de Loos, T.W.; Vlugt, T.J.H. State-of-the-Art of CO2 Capture with Ionic Liquids. Ind. Eng. Chem. Res. 2012, 51, 8149–8177.
- Wang, J.Y.; Huang, L.; Yang, R.Y.; Wu, J.W.; Gao, Y.S.; Wang, Q.; O’Hare, D.; Zhong, Z.Y. Recent advances in solid sorbents for CO2 capture and new development trends. Energy Environ. Sci. 2014, 7, 3478–3518.
- Yu, C.H.; Huang, C.H.; Tan, C.S. A Review of CO2 Capture by Absorption and Adsorption. Aerosol Air Qual. Res. 2012, 12, 745–769.
- Inoue, T.; Fujishima, A.; Konishi, S.; Honda, K. Photoelectrocatalytic reduction of carbon dioxide in aqueous suspensions of semiconductor powders. Nature 1979, 277, 637–638.
- Halmann, M. Photoelectrochemical reduction of aqueous carbon dioxide on p-type gallium phosphide in liquid junction solar cells. Nature 1978, 275, 115–116.
- Li, W. Electrocatalytic Reduction of CO2 to Small Organic Molecule Fuels on Metal Catalysts. In Advances in CO2 Conversion and Utilization; ACS Symposium Series; ACS Publisher: Washington, DC, USA, 2010; Volume 1056, pp. 55–76.
- Kortlever, R.; Peters, I.; Koper, S.; Koper, M.T.M. Electrochemical CO2 Reduction to Formic Acid at Low Overpotential and with High Faradaic Efficiency on Carbon-Supported Bimetallic Pd–Pt Nanoparticles. ACS Catal. 2015, 5, 3916–3923.
- Zafar, Q.; Mattisson, T.; Gevert, B. Integrated Hydrogen and Power Production with CO2 Capture Using Chemical-Looping ReformingsRedox Reactivity of Particles of CuO, Mn2O3, NiO, and Fe2O3 Using SiO2 as a Support. Ind. Eng. Chem. Res. 2005, 44, 3485–3496.
- Chen, X.; Wang, J.; Huang, C.; Zhang, S.; Zhang, H.; Li, Z.; Zou, Z. Barium zirconate: A new photocatalyst for converting CO2 into hydrocarbons under UV irradiation. Catal. Sci. Technol. 2015, 5, 1758–1763.
- Ding, J.; Bu, Y.; Ou, M.; Yu, Y.; Zhong, Q.; Fan, M. Facile decoration of carbon fibers with Ag nanoparticles for adsorption and photocatalytic reduction of CO2. Appl. Catal. B Environ. 2017, 202, 314–325.
- Wang, W.; Wang, S.; Ma, X.; Gong, J. Recent advances in catalytic hydrogenation of carbon dioxide. Chem. Soc. Rev. 2011, 40, 3703–3727.
- Shi, J.F.; Jiang, Y.J.; Jiang, Z.Y.; Wang, X.Y.; Wang, X.L.; Zhang, S.H.; Han, P.P.; Yang, C. Enzymatic conversion of carbon dioxide. Chem. Soc. Rev. 2015, 44, 5981–6000.
- Gong, Q.; Ding, P.; Xu, M.; Zhu, X.; Wang, M.; Deng, J.; Ma, Q.; Han, N.; Zhu, Y.; Lu, J.; et al. Structural defects on converted bismuth oxide nanotubes enable highly active electrocatalysis of carbon dioxide reduction. Nat. Commun. 2019, 10, 2807.
- Li, T.T.; Shan, B.; Xu, W.; Meyer, T.J. Electrocatalytic CO2 Reduction with a Ruthenium Catalyst in Solution and on Nanocrystalline TiO2. ChemSusChem 2019, 12, 2402–2408.
- Grodkowski, J.; Neta, P. Copper-Catalyzed Radiolytic Reduction of CO2 to CO in Aqueous Solutions. J. Phys. Chem. B 2001, 105, 4967–4972.
- Agarwal, A.S.; Zhai, Y.; Hill, D.; Sridhar, N. The electrochemical reduction of carbon dioxide to formate/formic acid: Engineering and economic feasibility. ChemSusChem 2011, 4, 1301–1310.
- Pritchard, J.; Filonenko, G.A.; van Putten, R.; Hensen, E.J.; Pidko, E.A. Heterogeneous and homogeneous catalysis for the hydrogenation of carboxylic acid derivatives: History, advances and future directions. Chem. Soc. Rev. 2015, 44, 3808–3833.
- Chernikov, A.; van der Zande, A.M.; Hill, H.M.; Rigosi, A.F.; Velauthapillai, A.; Hone, J.; Heinz, T.F. Electrical Tuning of Exciton Binding Energies in Monolayer WS2. Phys. Rev. Lett. 2015, 115, 126802.
- Wang, H.; Yuan, H.; Sae Hong, S.; Li, Y.; Cui, Y. Physical and chemical tuning of two-dimensional transition metal dichalcogenides. Chem. Soc. Rev. 2015, 44, 2664–2680.
- Wang, H.; Chen, Y.; Hou, X.; Ma, C.; Tan, T. Nitrogen-doped graphenes as efficient electrocatalysts for the selective reduction of carbon dioxide to formate in aqueous solution. Green Chem. 2016, 18, 3250–3256.
- Ao, C.; Feng, B.; Qian, S.; Wang, L.; Zhao, W.; Zhai, Y.; Zhang, L. Theoretical study of transition metals supported on g-C3N4 as electrochemical catalysts for CO2 reduction to CH3OH and CH4. J. CO2 Util. 2020, 36, 116–123.
- Zhi, X.; Jiao, Y.; Zheng, Y.; Qiao, S.Z. Impact of Interfacial Electron Transfer on Electrochemical CO2 Reduction on Graphitic Carbon Nitride/Doped Graphene. Small 2019, 15, e1804224.
- Kong, D.; Cha, J.J.; Wang, H.; Lee, H.R.; Cui, Y. First-row transition metal dichalcogenide catalysts for hydrogen evolution reaction. Energy Environ. Sci. 2013, 6, 3553–3558.
- Chhowalla, M.; Shin, H.S.; Eda, G.; Li, L.J.; Loh, K.P.; Zhang, H. The chemistry of two-dimensional layered transition metal dichalcogenide nanosheets. Nat. Chem. 2013, 5, 263–275.
- Wang, Q.; Zhang, Y.; Lin, H.; Zhu, J. Recent Advances in Metal-Organic Frameworks for Photo-/Electrocatalytic CO2 Reduction. Chem. Eur. J. 2019, 25, 14026–14035.
- Zou, X.; Liu, M.; Wu, J.; Ajayan, P.M.; Li, J.; Liu, B.; Yakobson, B.I. How Nitrogen-Doped Graphene Quantum Dots Catalyze Electroreduction of CO2 to Hydrocarbons and Oxygenates. ACS Catal. 2017, 7, 6245–6250.
- Zhu, X.; Li, Y. Review of two-dimensional materials for electrochemical CO2 reduction from a theoretical perspective. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2019, 9, e1416.
- DeCiccio, D.; Ahn, S.T.; Sen, S.; Schunk, F.; Palmore, G.T.R.; Rose-Petruck, C. Electrochemical reduction of CO2 with clathrate hydrate electrolytes and copper foam electrodes. Electrochem. Commun. 2015, 52, 13–16.
- Appel, A.M.; Bercaw, J.E.; Bocarsly, A.B.; Dobbek, H.; DuBois, D.L.; Dupuis, M.; Ferry, J.G.; Fujita, E.; Hille, R.; Kenis, P.J.; et al. Frontiers, opportunities, and challenges in biochemical and chemical catalysis of CO2 fixation. Chem. Rev. 2013, 113, 6621–6658.
- Murata, A.; Hori, Y. Product Selectivity Affected by Cationic Species in Electrochemical Reduction of CO2 and CO at a Cu Electrode. Bull. Chem. Soc. Jpn. 1991, 64, 123–127.
- Qin, B.; Li, Y.; Fu, H.; Wang, H.; Chen, S.; Liu, Z.; Peng, F. Electrochemical Reduction of CO2 into Tunable Syngas Production by Regulating the Crystal Facets of Earth-Abundant Zn Catalyst. ACS Appl. Mater. Interfaces 2018, 10, 20530–20539.
- Jiang, K.; Huang, Y.; Zeng, G.; Toma, F.M.; Goddard, W.A.; Bell, A.T. Effects of Surface Roughness on the Electrochemical Reduction of CO2 over Cu. ACS Energy Lett. 2020, 5, 1206–1214.
- Zhu, W.; Zhang, Y.J.; Zhang, H.; Lv, H.; Li, Q.; Michalsky, R.; Peterson, A.A.; Sun, S. Active and selective conversion of CO2 to CO on ultrathin Au nanowires. J. Am. Chem. Soc. 2014, 136, 16132–16135.
- Huang, X.; Tang, S.; Mu, X.; Dai, Y.; Chen, G.; Zhou, Z.; Ruan, F.; Yang, Z.; Zheng, N. Freestanding palladium nanosheets with plasmonic and catalytic properties. Nat. Nanotechnol. 2011, 6, 28–32.
- Yang, Q.; Shi, L.; Yu, B.; Xu, J.; Wei, C.; Wang, Y.; Chen, H. Facile synthesis of ultrathin Pt–Pd nanosheets for enhanced formic acid oxidation and oxygen reduction reaction. J. Mater. Chem. A 2019, 7, 18846–18851.
- Gao, F.; Zhang, Y.; Ren, F.; Shiraishi, Y.; Du, Y. Universal Surfactant-Free Strategy for Self-Standing 3D Tremella-Like Pd–M (M = Ag, Pb, and Au) Nanosheets for Superior Alcohols Electrocatalysis. Adv. Funct. Mater. 2020, 30, 2000255.
- Zhang, S.; Fan, Q.; Xia, R.; Meyer, T.J. CO2 Reduction: From Homogeneous to Heterogeneous Electrocatalysis. Acc. Chem. Res. 2020, 53, 255–264.
- Wu, Z.P.; Lu, X.F.; Zang, S.Q.; Lou, X.W. Non-Noble-Metal-Based Electrocatalysts toward the Oxygen Evolution Reaction. Adv. Funct. Mater. 2020, 30, 1910274.
- Zhu, Y.P.; Guo, C.; Zheng, Y.; Qiao, S.Z. Surface and Interface Engineering of Noble-Metal-Free Electrocatalysts for Efficient Energy Conversion Processes. Acc. Chem. Res. 2017, 50, 915–923.
- Kuhl, K.P.; Cave, E.R.; Abram, D.N.; Jaramillo, T.F. New insights into the electrochemical reduction of carbon dioxide on metallic copper surfaces. Energy Environ. Sci. 2012, 5, 7050–7059.
- Tao, H.; Gao, Y.; Talreja, N.; Guo, F.; Texter, J.; Yan, C.; Sun, Z. Two-dimensional nanosheets for electrocatalysis in energy generation and conversion. J. Mater. Chem. A 2017, 5, 7257–7284.
- Rogers, C.; Perkins, W.S.; Veber, G.; Williams, T.E.; Cloke, R.R.; Fischer, F.R. Synergistic Enhancement of Electrocatalytic CO2 Reduction with Gold Nanoparticles Embedded in Functional Graphene Nanoribbon Composite Electrodes. J. Am. Chem. Soc. 2017, 139, 4052–4061.
- Huang, J.; Guo, X.; Wei, Y.; Hu, Q.; Yu, X.; Wang, L. A renewable, flexible and robust single layer nitrogen-doped graphene coating Sn foil for boosting formate production from electrocatalytic CO2 reduction. J. CO2 Util. 2019, 33, 166–170.
- Lu, X.; Tan, T.H.; Ng, Y.H.; Amal, R. Highly Selective and Stable Reduction of CO2 to CO by a Graphitic Carbon Nitride/Carbon Nanotube Composite Electrocatalyst. Eur. J. Chem. 2016, 22, 11991–11996.
- Asadi, M.; Kumar, B.; Behranginia, A.; Rosen, B.A.; Baskin, A.; Repnin, N.; Pisasale, D.; Phillips, P.; Zhu, W.; Haasch, R.; et al. Robust carbon dioxide reduction on molybdenum disulphide edges. Nat. Commun. 2014, 5, 4470.
- Zeng, M.; Li, Y. Recent advances in heterogeneous electrocatalysts for the hydrogen evolution reaction. J. Mater. Chem. A 2015, 3, 14942–14962.
- Xie, Y.; Li, X.; Wang, Y.; Li, B.; Yang, L.; Zhao, N.; Liu, M.; Wang, X.; Yu, Y.; Liu, J.M. Reaction mechanisms for reduction of CO2 to CO on monolayer MoS2. Appl. Surf. Sci. 2020, 499, 143964.
- Abbasi, P.; Asadi, M.; Liu, C.; Sharifi-Asl, S.; Sayahpour, B.; Behranginia, A.; Zapol, P.; Shahbazian-Yassar, R.; Curtiss, L.A.; Salehi-Khojin, A. Tailoring the Edge Structure of Molybdenum Disulfide toward Electrocatalytic Reduction of Carbon Dioxide. ACS Nano 2017, 11, 453–460.
- Hong, X.; Chan, K.; Tsai, C.; Nørskov, J.K. How Doped MoS2 Breaks Transition-Metal Scaling Relations for CO2 Electrochemical Reduction. ACS Catal. 2016, 6, 4428–4437.
- Chan, K.; Tsai, C.; Hansen, H.A.; Nørskov, J.K. Molybdenum Sulfides and Selenides as Possible Electrocatalysts for CO2 Reduction. ChemCatChem 2014, 6, 1899–1905.
- Asadi, M.; Kim, K.; Liu, C.; Addepalli, A.V.; Abbasi, P.; Yasaei, P.; Phillips, P.; Behranginia, A.; Cerrato, J.M.; Haasch, R.; et al. Nanostructured transition metal dichalcogenide electrocatalysts for CO2 reduction in ionic liquid. Science 2016, 353, 467–470.
- Shao, P.; Yi, L.; Chen, S.; Zhou, T.; Zhang, J. Metal-organic frameworks for electrochemical reduction of carbon dioxide: The role of metal centers. J. Energy Chem. 2020, 40, 156–170.
- Kornienko, N.; Zhao, Y.; Kley, C.S.; Zhu, C.; Kim, D.; Lin, S.; Chang, C.J.; Yaghi, O.M.; Yang, P. Metal-organic frameworks for electrocatalytic reduction of carbon dioxide. J. Am. Chem. Soc. 2015, 137, 14129–14135.
- Zhao, C.; Dai, X.; Yao, T.; Chen, W.; Wang, X.; Wang, J.; Yang, J.; Wei, S.; Wu, Y.; Li, Y. Ionic Exchange of Metal-Organic Frameworks to Access Single Nickel Sites for Efficient Electroreduction of CO2. J. Am. Chem. Soc. 2017, 139, 8078–8081.
- Chen, Y.; Li, C.W.; Kanan, M.W. Aqueous CO2 reduction at very low overpotential on oxide-derived Au nanoparticles. J. Am. Chem. Soc. 2012, 134, 19969–19972.
- Chen, Y.; Kanan, M.W. Tin oxide dependence of the CO2 reduction efficiency on tin electrodes and enhanced activity for tin/tin oxide thin-film catalysts. J. Am. Chem. Soc. 2012, 134, 1986–1989.
- Li, C.W.; Kanan, M.W. CO2 reduction at low overpotential on Cu electrodes resulting from the reduction of thick Cu2O films. J. Am. Chem. Soc. 2012, 134, 7231–7234.
- Liu, J.; Guo, C.; Vasileff, A.; Qiao, S. Nanostructured 2D Materials: Prospective Catalysts for Electrochemical CO2 Reduction. Small Methods 2017, 1, 1600006.
- Yang, X.F.; Wang, A.Q.; Qiao, B.T.; Li, J.; Liu, J.Y.; Zhang, T. Single-Atom Catalysts: A New Frontier in Heterogeneous Catalysis. Acc. Chem. Res. 2013, 46, 1740–1748.
- Wang, A.; Li, J.; Zhang, T. Heterogeneous single-atom catalysis. Nat. Rev. Chem. 2018, 2, 65–81.
- Chorkendorff, I.; Niemantsverdriet, J.W. Concepts of Modern Catalysis and Kinetics; Wiley: Weinheim, Germany, 2003.
- Jaramillo, T.F.; Jorgensen, K.P.; Bonde, J.; Nielsen, J.H.; Horch, S.; Chorkendorff, I. Identification of Active Edge Sites for Electrochemical H2 Evolution from MoS2 Nanocatalysts. Science 2007, 317, 100–102.
- Seh, Z.W.; Kibsgaard, J.; Dickens, C.F.; Chorkendorff, I.; Norskov, J.K.; Jaramillo, T.F. Combining theory and experiment in electrocatalysis: Insights into materials design. Science 2017, 355, aad4998.
- Kyriakou, G.; Boucher, M.W.; Jewell, A.D.; Lewis, E.A.; Lawton, T.J.; Baber, A.E.; Tierney, H.L.; Flytzani-Stephanopoulos, M.; Sykes, E.C. Isolated Metal Atom Geometries as a Strategy for Selective Heterogeneous Hydrogenations. Science 2012, 335, 1209–1212.
- Zhang, N.; Zhou, T.; Chen, M.; Feng, H.; Yuan, R.; Zhong, C.a.; Yan, W.; Tian, Y.; Wu, X.; Chu, W.; et al. High-purity pyrrole-type FeN4 sites as a superior oxygen reduction electrocatalyst. Energy Environ. Sci. 2020, 13, 111–118.
- Yin, X.P.; Wang, H.J.; Tang, S.F.; Lu, X.L.; Shu, M.; Si, R.; Lu, T.B. Engineering the Coordination Environment of Single-Atom Platinum Anchored on Graphdiyne for Optimizing Electrocatalytic Hydrogen Evolution. Angew. Chem. Int. Ed. 2018, 57, 9382–9386.
- Wang, Y.; Chen, Z.; Han, P.; Du, Y.; Gu, Z.; Xu, X.; Zheng, G. Single-Atomic Cu with Multiple Oxygen Vacancies on Ceria for Electrocatalytic CO2 Reduction to CH4. ACS Catal. 2018, 8, 7113–7119.
- Xiao, M.; Gao, L.; Wang, Y.; Wang, X.; Zhu, J.; Jin, Z.; Liu, C.; Chen, H.; Li, G.; Ge, J.; et al. Engineering Energy Level of Metal Center: Ru Single-Atom Site for Efficient and Durable Oxygen Reduction Catalysis. J. Am. Chem. Soc. 2019, 141, 19800–19806.
- Zhang, H.; Yu, L.; Chen, T.; Zhou, W.; Lou, X.W.D. Surface Modulation of Hierarchical MoS2 Nanosheets by Ni Single Atoms for Enhanced Electrocatalytic Hydrogen Evolution. Adv. Funct. Mater. 2018, 28, 1807086.
- Li, H.; Wang, L.; Dai, Y.; Pu, Z.; Lao, Z.; Chen, Y.; Wang, M.; Zheng, X.; Zhu, J.; Zhang, W.; et al. Synergetic interaction between neighbouring platinum monomers in CO2 hydrogenation. Nat. Nanotechnol. 2018, 13, 411–417.
- Ye, S.; Luo, F.; Zhang, Q.; Zhang, P.; Xu, T.; Wang, Q.; He, D.; Guo, L.; Zhang, Y.; He, C.; et al. Highly stable single Pt atomic sites anchored on aniline-stacked graphene for hydrogen evolution reaction. Energy Environ. Sci. 2019, 12, 1000–1007.
- Yuan, J.; Zhang, W.; Li, X.; Yang, J. A high-performance catalyst for methane conversion to methanol: Graphene supported single atom Co. Chem. Commun. 2018, 54, 2284–2287.
- Ramalingam, V.; Varadhan, P.; Fu, H.C.; Kim, H.; Zhang, D.; Chen, S.; Song, L.; Ma, D.; Wang, Y.; Alshareef, H.N.; et al. Heteroatom-Mediated Interactions between Ruthenium Single Atoms and an MXene Support for Efficient Hydrogen Evolution. Adv. Mater. 2019, 31, e1903841.
- Choi, C.; Cai, J.; Lee, C.; Lee, H.M.; Xu, M.; Huang, Y. Intimate atomic Cu-Ag interfaces for high CO2RR selectivity towards CH4 at low over potential. Nano Res. 2021, 14, 3497–3501.
- Chen, B.; Li, B.; Tian, Z.; Liu, W.; Liu, W.; Sun, W.; Wang, K.; Chen, L.; Jiang, J. Enhancement of Mass Transfer for Facilitating Industrial-Level CO2 Electroreduction on Atomic Ni-N4 Sites. Adv. Energy Mater. 2021, 11, 2102152.
- Yang, Y.; Li, F. Reactor design for electrochemical CO2 conversion toward large-scale applications. Curr. Opin. Green Sustain. Chem. 2021, 27, 100419.
- Jayashree, R.S.; Mitchell, M.; Natarajan, D.; Markoski, L.J.; Kenis, P.J.A. Microfluidic Hydrogen Fuel Cell with a Liquid Electrolyte. Langmuir 2007, 23, 6871–6874.
- Whipple, D.T.; Finke, E.C.; Kenis, P.J.A. Microfluidic Reactor for the Electrochemical Reduction of Carbon Dioxide: The Effect of pH. Electrochem. Solid-State Lett. 2010, 13, 9.
- Higgins, D.; Hahn, C.; Xiang, C.; Jaramillo, T.F.; Weber, A.Z. Gas-Diffusion Electrodes for Carbon Dioxide Reduction: A New Paradigm. ACS Energy Lett. 2018, 4, 317–324.
- Lees, E.W.; Mowbray, B.A.W.; Parlane, F.G.L.; Berlinguette, C.P. Gas diffusion electrodes and membranes for CO2 reduction electrolysers. Nat. Rev. Mater. 2021, 7, 55–64.
- Lim, J.; Kang, P.W.; Jeon, S.S.; Lee, H. Electrochemically deposited Sn catalysts with dense tips on a gas diffusion electrode for electrochemical CO2 reduction. J. Mater. Chem. A 2020, 8, 9032–9038.
- Rabiee, H.; Ge, L.; Zhang, X.; Hu, S.; Li, M.; Yuan, Z. Gas diffusion electrodes (GDEs) for electrochemical reduction of carbon dioxide, carbon monoxide, and dinitrogen to value-added products: A review. Energy Environ. Sci. 2021, 14, 1959–2008.
