MicroRNAs (miRNAs) are small, noncoding RNA molecules containing approximately 22 nucleotides. miRNAs are found in plants, animals, and some viruses that function in RNA silencing and posttranscriptional regulation of gene expression
[56][76]. Many microRNAs, such as let-7, miR-223, miR-29, miR-103, and miR-107, are known to regulate metabolic disorders (including diabetes) through various molecular pathways, such as modulation of lipid or glucose metabolism, liver gluconeogenesis, insulin secretion, and autophagy
[57][58][59][60][61][77,78,79,80,81]. MiRNA-carrying exosomes have advantages that work at distant tissues more efficiently. Thus, some exosomal miRNAs are pathological factors in diabetes by targeting key proteins and acting as crucial modulators of insulin sensitivity. In cells, insulin combined with its substrate, insulin receptor substrate-1 (IRS-1), stimulates a cascade signal, including phosphorylation of protein kinase B (PKB/AKT), at serine 473, leading to phosphorylated AKT promoting the translocation of glucose transporter-4 (Glut4) from the cytosol to the membrane for glucose uptake
[62][82]. Katayama and colleagues examined the miRNA expression profile of serum-derived exosome-enriched extracellular vesicles in healthy controls and type 2 diabetic patients. They found that exosome-derived extracellular miR-20b-5p is highly abundant in type 2 diabetic patients, and further in vitro studies showed that exosomal miR-20b-5p targeted AKT-interacting protein (AKTIP), which interacts directly with AKT and modulates AKT activity by enhancing the phosphorylation of AKT regulatory sites and reducing glycogen accumulation in primary human skeletal muscle, resulting in insulin resistance
[63][83]. Consistent with the above findings, another study demonstrated that pancreatic cancer-derived exosomes enter C2C12 myotubes and inhibit insulin and PI3K/Akt signaling, thereby preserving insulin-induced FoxO1 nuclear exclusion and impairing Glut4 trafficking.
In this Mstudy, microarray methods revealed that certain exosomal microRNAs (miR-450b-3p and miR-151-3p) likely play key roles in this process (
Figure 1)
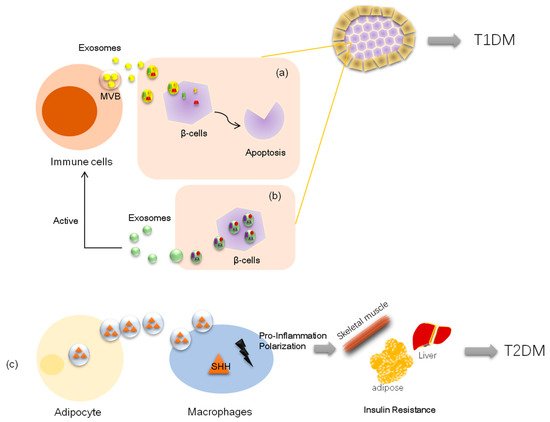
[64][84]. Further, exosome miR-155 derived from adipose tissue macrophages in obesity-induced diabetic mice causes glucose intolerance and insulin resistance by targeting PPARγ, a transcription factor and key regulator of lipid metabolism, when administered to lean mice
[65][85]. Similarly, another elegant experiment on obese mice showed that exosomes from obese mice could induce glucose intolerance in lean mice;
in th
e researcheis study, the authors transfected exosomes with the selected miRNA (miR-192, miR-122, miR-27a-3p, and miR-27b-3p; all were demonstrated to be increased in obese mice) and then injected the reconstituted exosomes into lean mice. Surprisingly, the lean mice developed glucose intolerance and insulin resistance as well. The data further showed that exosomal miRNAs induced diabetes in white adipose tissues in lean mice by targeting PPARα
[66][86], which is also a transcription factor and a major regulator of lipid metabolism.
Inadequate pancreatic β-cell numbers is a pathological manifestation of type 1 diabetes. Researchers have found that lymphocyte-derived exosomes miR-142-3p and miR-142-5p from prediabetic mice transferred to pancreatic β-cells trigger the expression of genes involved in chemokine signaling, including Ccl2, Ccl7, and Cxcl10. The induction of these genes may promote the recruitment of immune cells and exacerbate β-cell death during an autoimmune attack, which also contributes to diabetes development
[31][49]. Nonetheless, a recent study demonstrated that bone marrow mesenchymal stem cells derived exosome miR-29b-3p from aged mice mediated insulin resistance in young mice
[47][65], which indicated that exosomal miRNAs could exert their role independently. Thus, exosome cargo with miRNAs is also considered a novel therapeutic agent. Exosome miR-222 was reduced in rabbits with type 1 diabetes; however, MSC-derived exosomes containing miR-222 injected subconjunctivally and intraocularly to these diabetic rabbits could attenuate retinal degeneration
[67][87]. Similarly, intravenous administration of exosome miR-106b and miR-222 derived from bone narrow promoted post-injury β-cell proliferation through Cip/Kip family downregulation in insulin-deficient mice
[68][88]. In addition, MSC-derived exosome miR-146a has anti-inflammatory effects on damaged astrocytes and prevents diabetes-induced cognitive impairment
[69][89].
Consequently, many exosomal miRNAs have been found to be increased in the plasma or the urine of diabetic patients, making them promising circulating biomarkers associated with type 2 diabetes, such as exosomal miR-21-5p, miR-375-3p, miR-133b, miR-342, and miR-30, which are all upregulated in the serum of diabetic subjects
[70][71][72][90,91,92], and miR-451-5p, let-7c-5p, miR-362-3p, miR-877-3p, miR-150-5p, and miR-15a-5p, which are upregulated in the urine of diabetic subjects
[73][74][75][93,94,95]. It should be noted that, for certain miRNAs, total circulating miRNA levels are distinct from circulating extracellular vesicle miRNA content. For example, a clinical study including 19 children with type 1 diabetes along with 16 healthy controls showed that miR-21-5p derived from serum extracellular vesicles was increased threefold compared with that in nondiabetic individuals, while total serum miR-21-5p was reduced in diabetic participants
[72][92], suggesting that exosomal RNA may be a distinct biomarker from total serum RNA levels.