Pharmacological treatment of presbyopia may be an alternative for those who want a spectacle-free scenario and an easy-to-use method with lower risk of irreversible ocular adverse events. There are two main agents, miotics and lens softeners, investigated as agents for the pharmacological treatment. Miotic agents treat presbyopia by creating a pinhole effect which may increase the depth of focus at all working distances. Lens softeners increase the elasticity of the lens, which is targeted at one of the etiologic mechanisms of presbyopia.
- presbyopia
- pharmacological
- medical
- correction
- treatment
1. Introduction
2. Pharmacological Treatment in Presbyopia
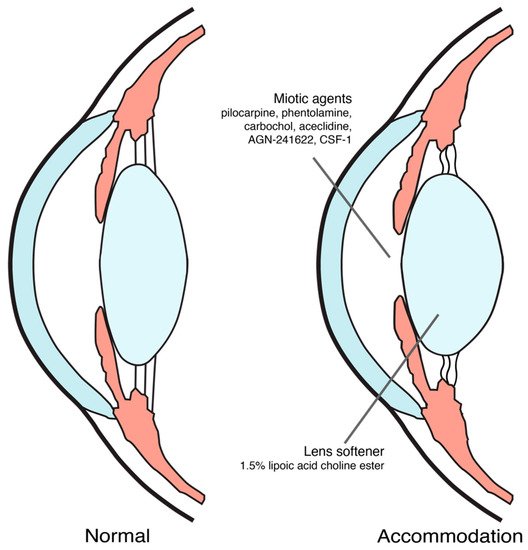
Until now, proposed mechanisms of action for pharmacological treatment of presbyopia were inducing miosis and softening the lens [9][18][19], as shown in Figure 1. The ongoing trials on pharmacological treatment of presbyopia were summarized in Table 1.
| Drugs | N | Study Design | Instillation Method | Primary Outcome | NCT ID | Phase |
|---|---|---|---|---|---|---|
| Miotic agents | ||||||
| 1.25% pilocarpine [20] | 230 | Multi-center, double-masked, randomized, vehicle-controlled, parallel-group study | Twice daily binocularly for 14 days | Percentage of participants gaining 3 lines or more in mesopic, high contrast, binocular DCNVA at day 14 | NCT04983589 | 3 |
| 1.25% pilocarpine [21] | 54 | Randomized, double-masked, crossover study | Twice daily binocularly for 14 days | Overall Composite Driving Z score approximately 1 h after study intervention instillation | NCT04837482 | 3 |
| AGN-241622 [22] | 144 | Phase 1/2, dose-ascending, multi-center, randomized, double-masked, vehicle-controlled study | Single drop binocularly | Number of patients experiencing a treatment emergent adverse event after single administration of AGN-241622 at day 2 and day 14 | NCT04403763 | 1/2 |
| CSF-1 [23] | 300 | 4-visit, multi-center, randomized, double-masked, vehicle-controlled study | Twice daily binocularly for 2 weeks | Percentage of subjects with a ≥ 3-line gain in BDCVA at 40 cm and no loss in BDCVA ≥ 5 letters at 4 m at day 8 | NCT04599933 | 3 |
| CSF-1 [24] | 300 | 4-visit, multi-center, randomized, double-masked, vehicle-controlled study | Twice daily binocularly for 2 weeks | Percentage of subjects with a ≥ 3-line gain in BDCVA at 40 cm and no loss in BDCVA ≥ 5 letters at 4 m at day 8 | NCT04599972 | 3 |
| 1% phentolamine [25] | 150 | Randomized, quadruple-masked, parallel-group study | Single drop binocularly | Percentage of subjects with ≥15 letters of improvement in photopic binocular DCNVA after 6 h | NCT04675151 | 2 |
| Carbachol and brimonidine [26] | 450 | Multi-center, randomized, double-masked study | Single drop binocularly | Percentage of subjects with 3-line gains in near VA with the loss of at least 1 line in DVA | NCT05135286 | 3 |
| PBOHB compound [27] | 11 | Single group study | Single drop binocularly | Jaeger near uncorrected visual acuity improvement after 1 h | NCT05006911 | 1 |
| Pilocarpine cream [28] | 120 | Multi-center, randomized, double-masked, placebo-controlled, parallel group study | Once daily binocularly for 28 days | Binocular DCNVA after 28 days | NCT05124275 | 2 |
| Pilocarpine Spray [29] | 139 | Randomized, triple-masked, crossover, placebo-controlled study | Single drop binocularly | Proportion of subjects gaining ≥ 15 letters in mesopic, high contrast, binocular DCNVA at 120 min post-treatment | NCT05114486 | 3 |
| Lens softeners | ||||||
| 1.5% lipoic acid choline ester [30] | 225 | Multi-center, randomized, placebo-controlled, double-masked, dose-ranging study | Twice daily binocularly | Change in Binocular DNCVA From Baseline at Month 3 | NCT04806503 | 2 |
2.1. Miotic Agents
Most of the current presbyopia pharmacological treatment options aim at inducing temporary miosis causing a pinhole effect to increase the depth of focus through parasympathetic pathway. Miotic agents were used as either a monotherapy or in combination therapy with another miotic agent or other agents for treatment of presbyopia. Whereas most of them were used in combination therapy, the only two agents studied for monotherapy were pilocarpine and phentolamine.2.1.1. Pilocarpine HCl Ophthalmic Solution 1.25%—The U.S. FDA Approved Agent for Presbyopia
Pilocarpine is a miotic agent that has been used and studied in different concentrations, different forms and also as a combination with other drugs for presbyopia. It can induce miosis and ciliary body contraction which would also help in lens accommodation [31]. The daily use of pilocarpine hydrochloride ophthalmic solution 1.25% monotherapy in both eyes is the regimen which has been approved by the U.S. FDA. Pilocarpine ophthalmic solution was stored at a pH between 3.5 to 5.5 for its stability, the agent would have low bioavailability at this acidity. However, it was claimed that pilocarpine HCl ophthalmic solution 1.25% for the treatment of presbyopia could adapt to the pH of ocular surface within 1 min [32]. In phase 3 studies of this agent (GEMINI 1 and GEMINI 2), which were submitted for this approval, the eye drop was applied in each eye daily for 30 days and compared with placebo [33][34]. The key inclusion criteria of these studies were patients with presbyopia aged 40–55 years old, distance-corrected near visual acuity (DCNVA) between 20/40 to 20/100 and best distance correction between −4.00 to +1.00 D sphere and between −2.00 to +2.00 D cylinder. Participants were randomized into the treatment group (n = 375) and placebo group (n = 375). The mean age of participants was 49.6 ± 3.75 and 49.8 ± 3.38 in the treatment group and placebo group, respectively, and the majority of participants (85%) was Caucasian [35]. Since presbyopia affects both near and intermediate vision, outcome measures of both studies included four outcomes for near vision and one for intermediate vision. All of these outcomes were determined at different hours post-instillation on the 30th day of continuous use, therefore in the following sentences, ‘hours’ means hour post-instillation on the 30th day of continuous use. The outcomes for near vision were (1) the primary outcome, a percentage of participants with at least 3-line improvement in mesopic, high-contrast, binocular DCNVA without losing more than 5 letters of mesopic, high contrast, binocular corrected-distance visual acuity (CDVA) at 3 h, (2) a percentage of participants with at least 3-line improvement in mesopic, high-contrast, binocular DCNVA from 0.25 to 8 h, (3) changes from baseline in mesopic DCNVA from 0.25 to 10 h, and (4) a percentage of participants achieving photopic, binocular DCNVA of 20/40 or better at 1 h and 3 h. The outcome for intermediate vision was changes from baseline in photopic, high-contrast, binocular distance-corrected intermediate visual acuity (DCIVA) at 1 h and 3 h [33][34][35].2.1.2. Other Miotic Agents
Another form of pilocarpine topical drug as ophthalmic cream monotherapy for presbyopia has also been studied. There is an ongoing phase 2 study on various concentrations of this pilocarpine ophthalmic topical cream on their safety and efficacy after using the medication twice a day for 28 days and assessing DCNVA as the primary outcome [28]. A phase 3 study on a 2% pilocarpine ophthalmic solution spray, another alternative mode of delivery, has been conducted with the primary outcome measures as DCNVA at 120 min after instillation [29]. Phentolamine is a nonselective alpha-adrenergic antagonist that has been studied for monotherapy. An ophthalmic solution of 1% phentolamine mesylate could decrease pupil diameter and create pinhole effect. The results from a phase 2b study by Pepose et al., using this drug once daily in the evening for 14 days, compared with placebo, showed that patients in the treatment group had at least 1-line improvement in DCNVA at day 15. There was no difference in conjunctival hyperemia between both treatment and placebo groups [36]. For the combination of a miotic agent with another miotic agent or other agents, the aim is either to gain addition effect between each drug or to reduce the side effects from the miotic agent. There are several studies evaluating pilocarpine in combination with anti-inflammatory agents or vasoconstrictive agents to minimize adverse events of conjunctival injection or ocular inflammation that may be induced by pilocarpine. An ongoing randomized controlled study has been initiated to compare between monotherapy of miotic agents and the combination of two miotic agents. There were four treatment groups for the comparison in this study: phentolamine and pilocarpine, phentolamine alone, pilocarpine alone and placebo, DCNVA in these treatment groups were compared at baseline and at 6 h [25].2.2. Lens Softeners
Loss of lens elasticity is associated with advanced age and presbyopia. This may be related to an increase in disulfide bonds formation in the collagen of aging lens, possibly due to oxidative stress [37]. Inducing miosis might be an effective mechanism for treatment of presbyopia by creating a pinhole effect but it might not address the etiology of this condition. Lens softening, on the other hand, might be an agent that could make a change at its pathophysiology. There was in vitro evidence showing that lipoic acid, an antioxidant, could reduce disulfide bonds in the lens protein, and thus increase lens elasticity [37]. However, lipoic acid alone as an eye drop had limited ocular penetration because of its lipid solubility [38]. Bonding lipoic acid with choline allows the drug to better penetrate into the aqueous humor [39]. In a study by Korenfeld et al., 1.5% lipoic acid choline ester ophthalmic solution (UNR844) was compared with placebo for DCNVA and adverse events. The inclusion criteria of this study were patients with presbyopia aged 45–55 years, monocular DCNVA worse than 20/40 in each eye, best-corrected distance visual acuity of 20/20 or better, and manifested spherical equivalent between −4.00 to +4.00 D. From a total of 75 patients enrolled, they were randomized into the treatment group and the placebo group with 2:1 ratio, resulting in 50 patients in the former and 25 patients in the latter. The mean age of the treatment group and the placebo group was 50.1 ± 3.2 and 51.4 ± 3.0, respectively; 70.67% of patients were Caucasians [39]. The treatment group were given 1.5% lipoic acid choline ester ophthalmic solution unilaterally in their nondominant eye twice daily on day 1–7, and then given bilaterally twice daily on day 8–91. There was significantly improved in DCNVA from baseline in the treatment group compared with the placebo group over the course of 91 days (0.198 vs. 0.099 LogMAR VA units). Moreover, about a third of the patients with 1.5% lipoic acid had a sustained bilateral improvement of at least 1-line DCNVA at day 301. In terms of adverse events, the most common was eye irritation at 6%. Patients also reported asthenopia, eye pruritus, and foreign body sensation. For ocular safety, there were no clinical changes in non-dilated pupil diameter, ocular comfort, intraocular pressure (IOP), distance vision, or other ocular findings in the treated eyes [39].3. Conclusions
With the recent U.S. FDA approval of 1.25% pilocarpine for treatment of presbyopia, this agent is becoming an alternative option for those in the US who may not want to wear spectacles for near tasks. It is possible that this agent may be approved in other countries and more patients may be able to access this agent in the future. This would give an opportunity to investigate on safety and efficacy of this agent in the real world, particularly for long-term use, which is essential. Pharmacological treatment of presbyopia is, without a doubt, one of the promising fields in research in ophthalmology since all people who are more than 45 years old will have this condition eventually.References
- Wolffsohn, J.S.; Davies, L.N. Presbyopia: Effectiveness of correction strategies. Prog. Retin. Eye Res. 2019, 68, 124–143.
- Holden, B.A.; Fricke, T.R.; Wilson, D.A.; Jong, M.; Naidoo, K.S.; Sankaridurg, P.; Wong, T.Y.; Naduvilath, T.J.; Resnikoff, S. Global Prevalence of Myopia and High Myopia and Temporal Trends from 2000 through 2050. Ophthalmology 2016, 123, 1036–1042.
- Strenk, S.A.; Strenk, L.M.; Koretz, J.F. The mechanism of presbyopia. Prog. Retin. Eye Res. 2005, 24, 379–393.
- Michael-Titus, A.; Revest, P.; Shortland, P. The Visual System. In The Nervous System; Michael-Titus, A., Revest, P., Shortland, P., Eds.; Elsevier: Amsterdam, The Netherlands, 2010; pp. 121–140. ISBN 978-0-7020-3373-5.
- Weikert, M.P. Update on bimanual microincisional cataract surgery. Curr. Opin. Ophthalmol. 2006, 17, 62–67.
- Vargas, V.; Radner, W.; Allan, B.D.; Reinstein, D.Z.; Burkhard Dick, H.; Alió, J.L. Methods for the study of near, intermediate vision, and accommodation: An overview of subjective and objective approaches. Surv. Ophthalmol. 2019, 64, 90–100.
- Socea, S.; Mimouni, M.; Andreja, V.; Blumenthal, E.Z. Drops for Presbyopia: Results of CSF-1, a multicenter randomized double-masked placebo-controlled crossover study. Investig. Ophthalmol. Vis. Sci. 2019, 60, 1385.
- Montés-Micó, R.; Charman, W.N. Pharmacological Strategies for Presbyopia Correction. J. Refract. Surg. 2019, 35, 803–814.
- Orman, B.; Benozzi, G. Pharmacological strategies for treating presbyopia. Curr. Opin. Ophthalmol. 2021, 32, 319–323.
- Smith, C.E.; Allison, R.S.; Wilkinson, F.; Wilcox, L.M. Monovision: Consequences for depth perception from large disparities. Exp. Eye Res. 2019, 183, 62–67.
- Hayashi, K.; Yoshida, M.; Manabe, S.; Hayashi, H. Optimal Amount of Anisometropia for Pseudophakic Monovision. J. Refract. Surg. 2011, 27, 332–338.
- Rueff, E.M.; Varghese, R.J.; Brack, T.M.; Downard, D.E.; Bailey, M.D. A Survey of Presbyopic Contact Lens Wearers in a University Setting. Optom. Vis. Sci. 2016, 93, 848–854.
- Lim, C.H.L.; Stapleton, F.; Mehta, J.S. Review of Contact Lens–Related Complications. Eye Contact Lens Sci. Clin. Pract. 2018, 44, S1–S10.
- Gil-Cazorla, R.; Shah, S.; Naroo, S.A. A review of the surgical options for the correction of presbyopia. Br. J. Ophthalmol. 2016, 100, 62–70.
- Kelava, L.; Barić, H.; Bušić, M.; Čima, I.; Trkulja, V. Monovision Versus Multifocality for Presbyopia: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Adv. Ther. 2017, 34, 1815–1839.
- Labiris, G.; Toli, A.; Perente, A.; Ntonti, P.; Kozobolis, V.P. A systematic review of pseudophakic monovision for presbyopia correction. Int. J. Ophthalmol. 2017, 10, 992.
- Harrison, L. FDA Approves Eye Drops for Presbyopia. Available online: https://www.medscape.com/viewarticle/961999#vp_2 (accessed on 11 November 2021).
- Katz, J.A.; Karpecki, P.M.; Dorca, A.; Chiva-Razavi, S.; Floyd, H.; Barnes, E.; Wuttke, M.; Donnenfeld, E. Presbyopia—A Review of Current Treatment Options and Emerging Therapies. Clin. Ophthalmol. 2021, 15, 2167–2178.
- Chang, D.H.; Waring, G.O., 4th; Hom, M.; Barnett, M. Presbyopia Treatments by Mechanism of Action: A New Classification System Based on a Review of the Literature. Clin. Ophthalmol. 2021, 15, 3733–3745.
- Clinicaltrials.gov. A Study to Assess Safety and Efficacy in Participants Age 40 to 55 With Presbyopia (Old Eye) Who Receive AGN-190584 in Both Eyes Twice Daily. Available online: https://clinicaltrials.gov/ct2/show/NCT04983589 (accessed on 11 November 2021).
- Clinicaltrials.gov. A Study to Assess the Impact and Adverse Events of Topical Eyedrops of AGN-190584 on Night-Driving Performance in Participants, 40 to 55 Years of Age. Available online: https://clinicaltrials.gov/ct2/show/NCT04837482?term=agn-190584&cond=Presbyopia&draw=2&rank=6 (accessed on 12 November 2021).
- Clinicaltrials.gov. Phase 1/2 Study of AGN-241622 in Healthy Participants and Participants with Presbyopia. Available online: https://clinicaltrials.gov/ct2/show/NCT04403763 (accessed on 21 November 2021).
- Clinicaltrials.gov. An Evaluation of the Efficacy and Safety of CSF-1 in the Temporary Correction of Presbyopia (NEAR-2). Available online: https://clinicaltrials.gov/ct2/show/NCT04599972?term=medication&cond=Presbyopia&draw=2&rank=8 (accessed on 5 December 2021).
- Clinicaltrials.gov. An Evaluation of the Efficacy and Safety of CSF-1 in the Temporary Correction of Presbyopia (NEAR-1). Available online: https://clinicaltrials.gov/ct2/show/NCT04599933?term=medication&cond=Presbyopia&draw=2&rank=9 (accessed on 5 December 2021).
- Clinicaltrials.gov. Safety and Efficacy of Nyxol with Pilocarpine Eye Drops in Subjects with Presbyopia. Available online: https://clinicaltrials.gov/ct2/show/NCT04675151 (accessed on 1 December 2021).
- Clinicaltrials.gov. Safety and Efficacy Study of BRIMOCHOLTM in Subjects with Emmetropic Phakic and Pseudophakic Presbyopia. Available online: https://clinicaltrials.gov/ct2/show/NCT05135286?term=medication&cond=Presbyopia&draw=4&rank=29 (accessed on 5 December 2021).
- Clinicaltrials.gov. Pilocarpine, Brimonidine, Oxymetazoline (PBO) Compound to Control Presbyopia Symptoms. Available online: https://clinicaltrials.gov/ct2/show/NCT05006911 (accessed on 5 December 2021).
- Clinicaltrials.gov. Safety and Efficacy of Pilocarpine Ophthalmic Topical Cream (4%, 6%, 8%) for the Treatment of Presbyopia. Available online: https://clinicaltrials.gov/ct2/show/NCT05124275 (accessed on 5 December 2021).
- Clinicaltrials.gov. Safety & Efficacy of Pilocarpine Ophthalmic Spray for Temporary Improvement of Near Vision in Presbyopic Adults. Available online: https://clinicaltrials.gov/ct2/show/NCT05114486 (accessed on 5 December 2021).
- Clinicaltrials.gov. A Dose-Ranging Study to Evaluate the Safety and Efficacy of UNR844 in Subjects with Presbyopia. Available online: https://clinicaltrials.gov/ct2/show/NCT04806503?term=unr844&cond=Presbyopia&draw=2&rank=1 (accessed on 19 December 2021).
- Renna, A.; Alió, J.L.; Vejarano, L.F. Pharmacological treatments of presbyopia: A review of modern perspectives. Eye Vis. 2017, 4, 3.
- AbbVie VUITYTM (Pilocarpine HCI Ophthalmic Solution) 1.25%, the First and Only FDA-Approved Eye Drop to Treat Age-Related Blurry Near Vision (Presbyopia), Is Now, Available|AbbVie News Center. Available online: https://news.abbvie.com/news/press-releases/vuity-pilocarpine-hci-ophthalmic-solution-125-first-and-only-fda-approved-eye-drop-to-treat-age-related-blurry-near-vision-presbyopia-is-now-available.htm (accessed on 19 January 2022).
- Clinicaltrials.gov. Efficacy Study of Pilocarpine HCl Ophthalmic Solution (AGN-190584) in Participants with Presbyopia (GEMINI 1). Available online: https://clinicaltrials.gov/ct2/show/NCT03804268 (accessed on 19 January 2022).
- Clinicaltrials.gov. A Phase 3 Efficacy Study of Pilocarpine HCl Ophthalmic Solution (AGN-190584) in Participants with Presbyopia (GEMINI 2). Available online: https://clinicaltrials.gov/ct2/show/results/NCT03857542?term=agn-190584&cond=Presbyopia&draw=2&rank=5 (accessed on 14 January 2022).
- Waring IV, G.O.; McCabe, C.M.; Wirta, D.L.; Safyan, E.; Guo, Q.; Robinson, M.R. PA031—GEMINI 1 and 2 Pooled Phase 3 Safety and Efficacy: AGN-190584 Primary and Key Secondary Endpoints. Available online: https://registration.experientevent.com/showaao211/flow/Attendee#!/registrant//ShowItems/ (accessed on 15 January 2022).
- Pepose, J.S.; Pitlick, B.; Meyer, A.; Jaber, R.; Charizanis, K.; Slonim, C.; Lazar, E.; McDonald, M.; Sooch, M. Phentolamine mesylate ophthalmic solution provides long lasting pupil modulation and improves visual acuity. Investig. Ophthalmol. Vis. Sci. 2020, 61, 5100.
- Garner, W.H.; Garner, M.H. Protein Disulfide Levels and Lens Elasticity Modulation: Applications for Presbyopia. Investig. Opthalmol. Vis. Sci. 2016, 57, 2851.
- Cagini, C.; Leontiadis, A.; Ricci, M.A.; Bartolini, A.; Dragoni, A.; Pellegrino, R.M. Study of alpha-lipoic acid penetration in the human aqueous after topical administration. Clin. Experiment. Ophthalmol. 2010, 38, 572–576.
- Korenfeld, M.S.; Robertson, S.M.; Stein, J.M.; Evans, D.G.; Rauchman, S.H.; Sall, K.N.; Venkataraman, S.; Chen, B.-L.; Wuttke, M.; Burns, W. Topical lipoic acid choline ester eye drop for improvement of near visual acuity in subjects with presbyopia: A safety and preliminary efficacy trial. Eye 2021, 35, 3292–3301.
