Cashew Nut Shell Liquid (CNSL) is a promising non-edible renewable resource, directly extracted from the shell of the cashew nut. The interesting structure of CNSL and its components (cardanol, anacardic acid and cardol) lead to the synthesis of biobased surfactants.
1. Introduction
Numerous studies are bringing to light the possible toxicity of currently widely used products such as pesticides [1][3], and additives [2][3][4][4,5,6]. This has the effect of accelerating the search for alternatives that are as safe as possible for human beings and the planet on which humanwe live. It is in this momentum that sustainable and green chemistry is emerging and defined [5][6][7,8]. The aim and the definition of sustainability are to reconcile economic, environmental, and social dimensions with the aid of the 3Ps: People, Planet, and Profit, around the same objective, and to ensure its continuity over time [7][8][9,10].
In these times, when components and additives are deeply studied, scrutinized, and criticized, surfactants are crystallizing a certain focus for consumer interest. Present in the shampoos, detergents, household products, cosmetics, and also in the world of construction and materials, their omnipresence and their possible impact are worrying. Their proximity to houmanr body, theour food, and the water peoplewe consume make surfactants a potential source of pollution and danger for humans, but also for rivers, oceans, sea life, wildlife, and flora on which peoplewe depend, and for which peoplewe are responsible [9][10][11][11,12,13].
Many bioresources such as sugar
[12][13][14,15], vegetable oil
[14][15][16,17], or lignin
[16][18] have been already used to synthesize and produce efficient surfactants. Some of them such as Alkyl PolyGlycoside (APG), or glycerol esters are already commercially available. However, vegetable oil and sugar are feedstock and compete with animal and human alimentation. Moreover, the actual production of beetroot for sugar induces the use of neonicotinoid, poison for bees, and other important insect pollinizers
[17][18][19,20]. In addition, a so-called green or sustainable surfactant is, of course, a biobased molecule. However, the term “green” is not limited to the source of this raw material. It is good to remember that “bio-based” or “natural” does not equal “non-hazardous” or “non-toxic”. “Green” also relates to the manufacturing process, the low degree of dangers or toxicity of both reagents and final product, and the end of life of the product
[6][8].
CNSL (Cashew NutShell Liquid) is a versatile abundant bioresource already used to synthesize surfactants. Even now, CNSL is absent or barely mentioned in specialized reviews or chapters talking about synthetic biobased surfactant
[19][20][21][21,22,23]. A lot of work on this raw material has been undertaken in this field since the last, and only a brief, review focused on CNSL
[22][24]. Given the growing interest in this resource over the past decade, it seems relevant to summarize the work carried out.
2. Cashew NutShell Liquid
Cashew NutShell Liquid (CNSL) is an oil extracted from a by-product of cashew cultivation, currently considered as waste: its shell. Inedible, and not competing with food production, the tonnages of the nutshells are closely linked to that of the edible cashew kernels already produced. The annual world production of cashew nut shells is estimated at around 3M T/year and is located in geographical areas with a tropical climate such as South America (Brazil), Asia (India, Vietnam, etc.), and Africa (Côte d’Ivoire, Benin, Burkina Faso, Tanzania, etc.)
[23][24][25,26]. Various extraction methods make it possible to obtain CNSL (pyrolysis
[25][27], solvent extraction
[26][28], screw pressing
[27][29], and supercritical CO
2 extraction
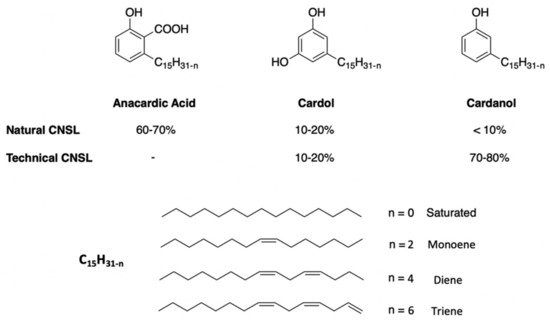
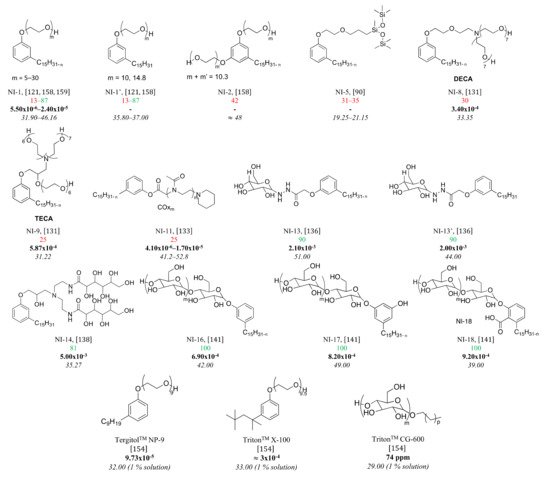
[28][29][30,31]) with mass yields oscillating between 15 and 30%. When talking about CNSL, two different types of oils must be distinguished by their composition: the CNSL says “natural” and the one called “technical” (
Figure 12).
Figure 2. Chemical structures and compositions of the natural and technical CNSL’s molecules.
Natural CNSL is obtained under gentle or low-temperature extraction conditions. The oil obtained is mainly composed of anacardic acid (60–70%), cardol (10–20%), and cardanol (<10%). Anacardic acid tends to decarboxylate into cardanol around 140 °C
[30][32]. This decarboxylation can take place during extraction under heating or pressing conditions. It yields a technical CNSL, mainly composed of cardanol (70–80%) and cardol (10–20%). In both CNSLs, there are also methyl cardol (<3%) and traces of urushiol. Some extraction processes also tend to promote the formation of residol, a polymeric residue from the polycondensation of CNSL phenolic compounds. It is possible to separate and purify the various compounds of CNSL by precipitation
[31][33], distillation
[32][34], liquid/liquid extraction
[31][33][33,35] or chromatographic column
[34][36].
The three main compounds of CNSL, anacardic acid, cardol, and cardanol can be each found in four forms: saturated, monoene, diene and triene. Unsaturated or mixed compounds are present in oil form, whereas hydrogenated compounds are in the solid form.
3. Synthesis of CNSL-Based Surfactants
3.1. Introduction
Surfactants are amphiphilic molecules widely used in industry. The global surfactant market is estimated at $41.3 billion in 2019
[35][72]. Their peculiar structure, composed of both lipophilic and hydrophilic parts in the form of nano-domains (micelles, etc.), to adsorb at interfaces where they reduce interfacial or surface tension. This property makes them very interesting for a wide range of applications such as household detergents, personal care, industrial and institutional cleaners, food and beverage, etc.,
[36][73].
3.2. Anionic Surfactants Based on CNSL
The CNSL-based anionic surfactants are divided into sulfonate and carboxylate compounds and are mainly based on cardanol with rare examples on anacardic acid derivatives.
3.2.1. Carboxylated CNSL-Based Surfactants
Two routes have been investigated to synthesize this class of surfactants, either starting from a natural phenol bearing a carboxylic acid (anacardic acid) or converting the natural phenol group into carboxylic acid by carboxymethylation.
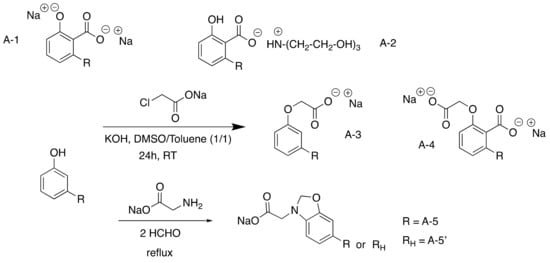
The simplest carboxylated surfactants were the anarcardate salts resulting from the deprotonation of the acid group by KOH (98% yield) (
Scheme 12). They were compared to 2-o-cardanyl acetate sodium salt, produced in a low yield (41%) by carboxymethylation using Williamson reaction between the phenol unit of cardanol and sodium chloroacetate
[37][38][74,75]. Compared to the first natural one, this second surfactant was 85% biobased and uses an irritant and environmentally toxic reactant. More recently benzoxazine surfactants were synthesized via a Mannich reaction using amino acids (glycine) and cardanol
[39][76].
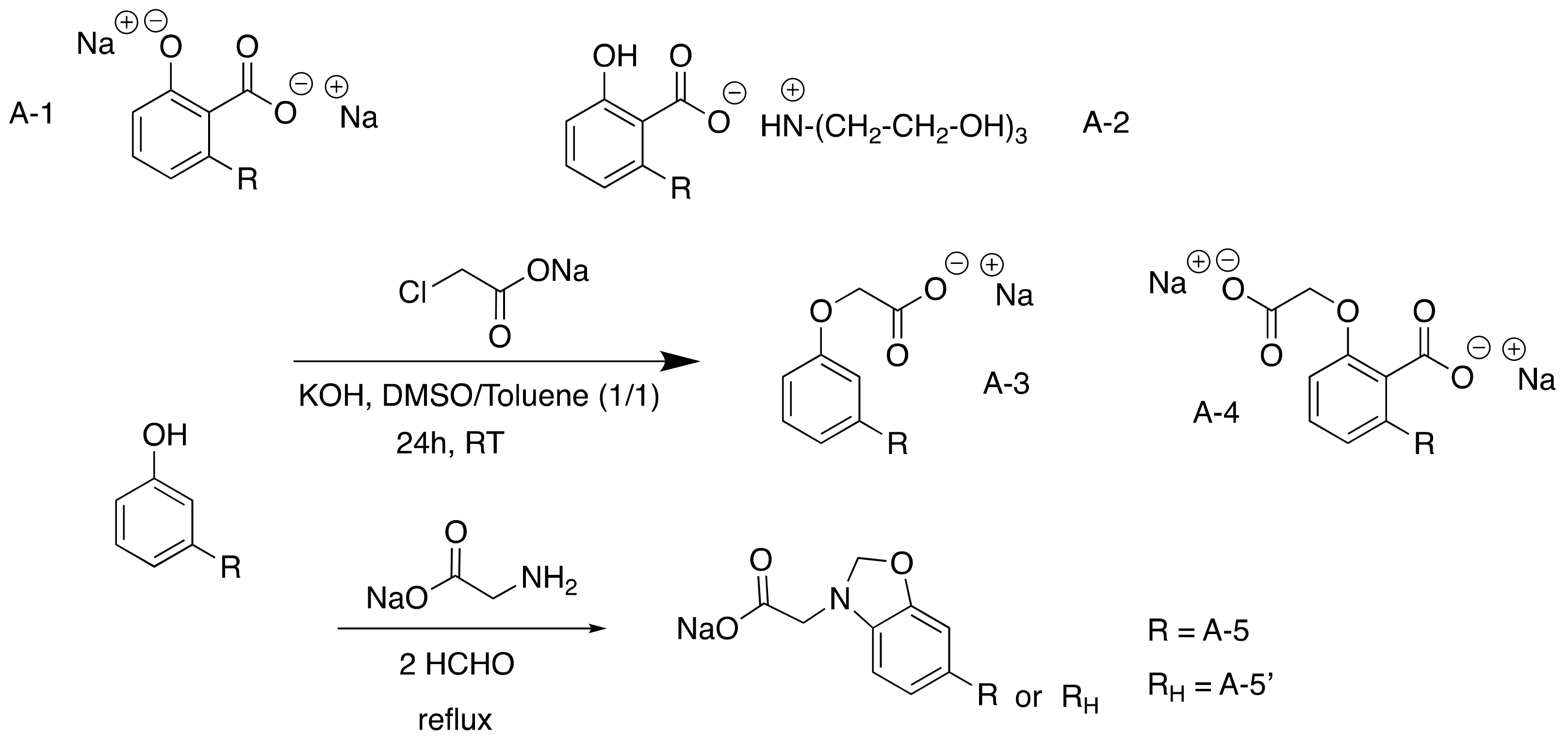
Scheme 2. Carboxylated CNSL-based surfactants.
3.2.2. Sulfonated CNSL-Based Surfactants
The most abundant anionic cardanol surfactants are the sulfonated ones with the polar head linked to the aromatic or at the end of an aliphatic chain (with a molecular or polymeric spacer between the cycle and the ionic head).
Aromatic Sulfonated Surfactants
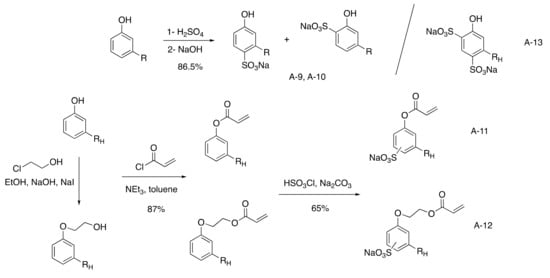
The most direct route to reach sulfonated cardanol surfactant was the sulfonation of the aromatic ring to afford a mixture of two isomers in ortho and para position in 86% yield with 80% biobased. (
Scheme 24)
[40][41][42][43][82,83,84,85]. It was compared to sodium n-alkylbenzenesulfonate as dodecylbenzene sulfonate (DDBS)
[44][86] and diversified into calcium
[45][87] and potassium
[46][88] salts. The phenol ring was also converted into an acrylate group to elaborate saturated cardanol surfmers using acrylate chloride (87% yield) followed by sulfonation using chlorosulfonic acid (65% yield)
[47][89]. An alternative was also described with the addition of a spacer between the phenyl ring and the ionic group using 2-chloroethanol, reducing the biobased content under 70% and using more toxic reactant for no real improvement.
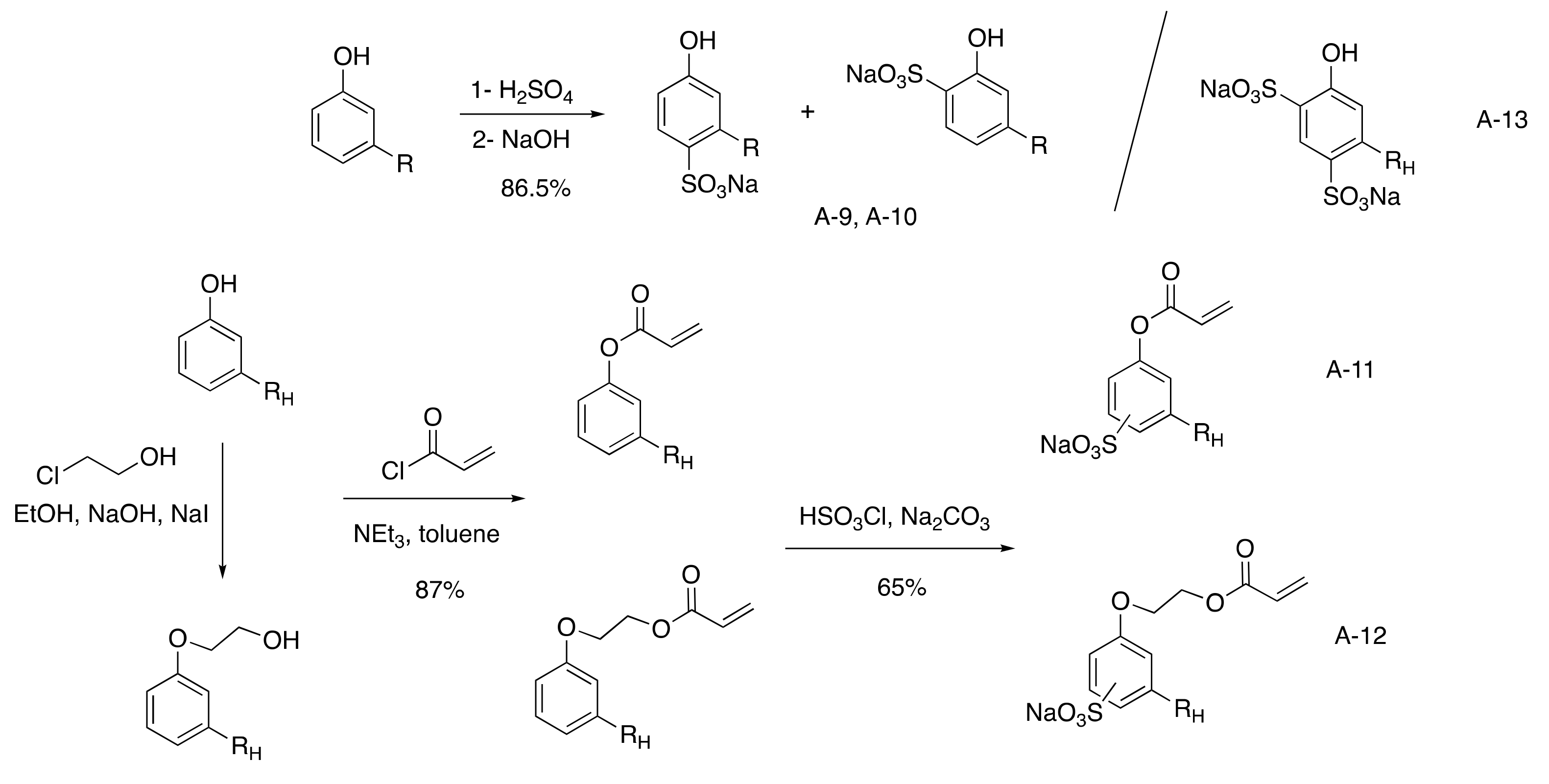
Scheme 4. Monosulfonated cardanol surfactants.
Gemini Aromatic Sulfonated Surfactants
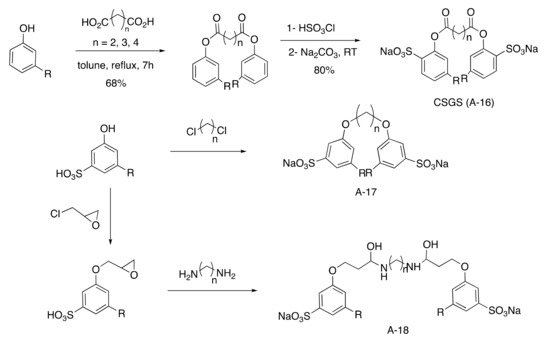
Ester-linked anionic Gemini surfactants were investigated using various lengths of diacid chain (from 4 to 6 carbons) by esterification of phenol followed by a regioselective sulfonation in the ortho position (
Scheme 36)
[41][83]. Otherwise, ether-linked anionic Gemini surfactants were studied after sulfonation of cardanol in presence of highly reactive oleum followed by etherification with various toxic dichloroalkanes
[48][93]. Another pathway was the reaction of epoxidized cardanol with various diamines producing a surfactant that was 50% biobased
[49][94].
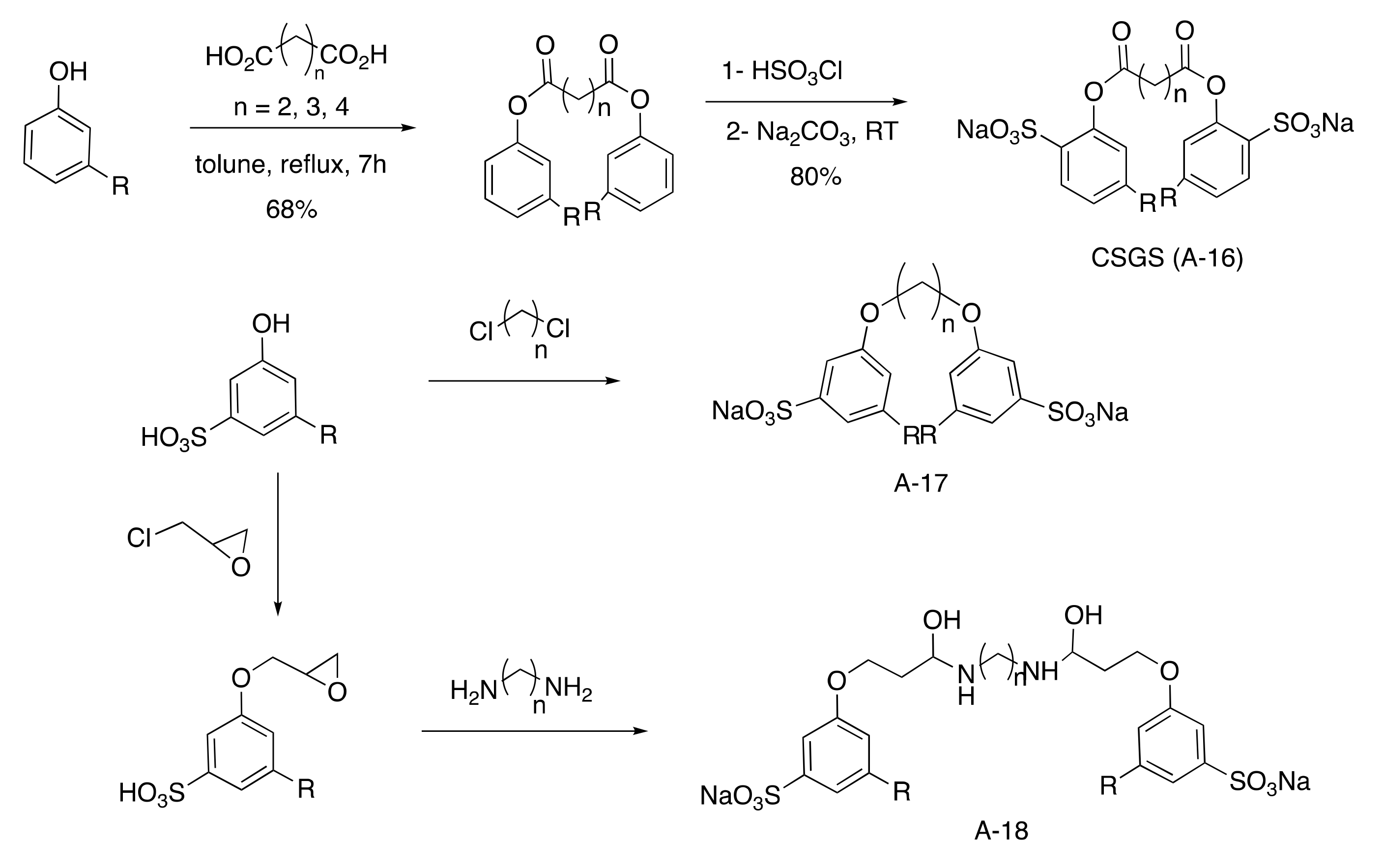
Scheme 6. Gemini aromatic sulfonated surfactants.
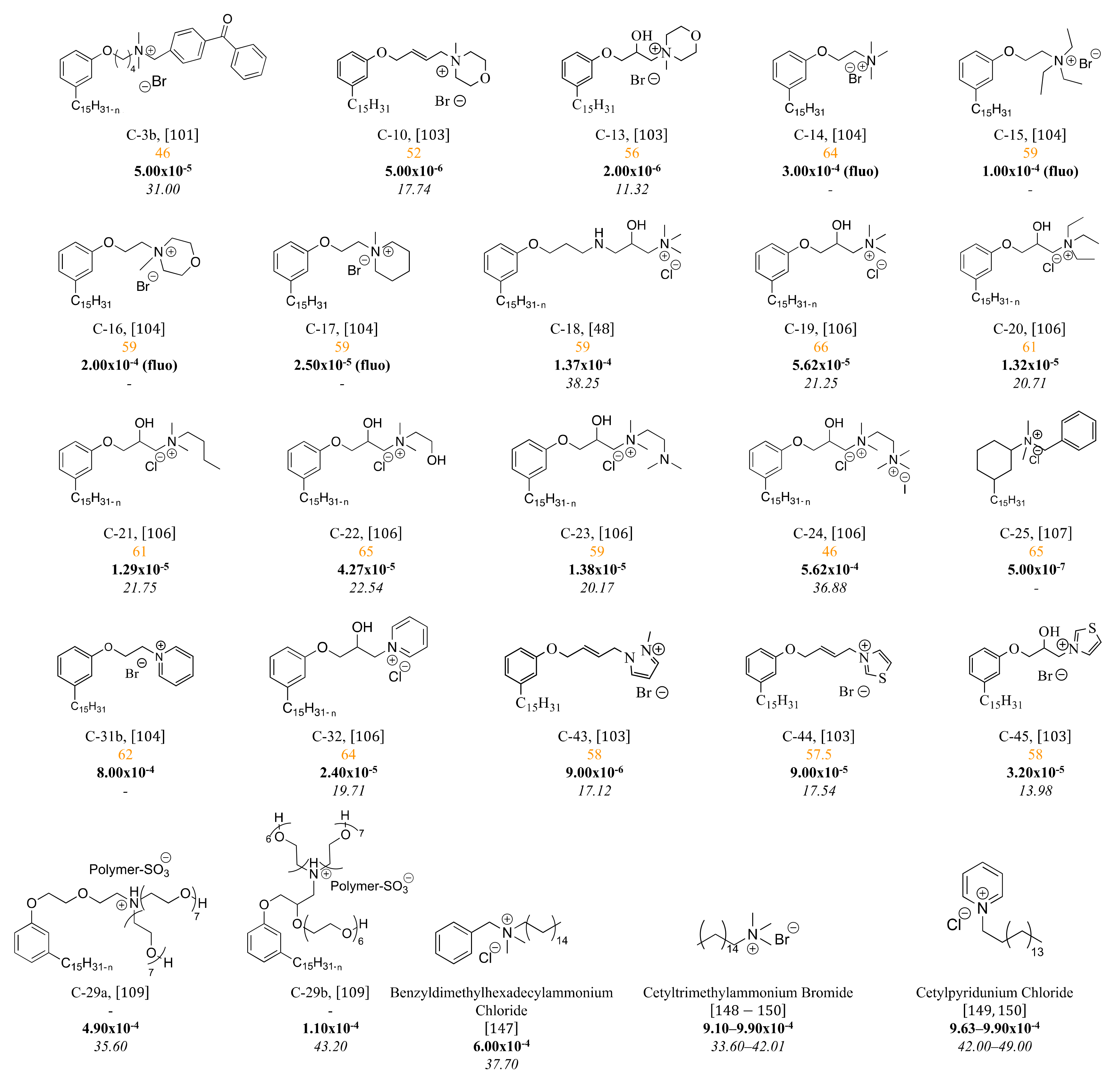
3.3. Cationic Surfactants Based on CNSL
3.3.1. Ammonium Derivatives of Cardanol
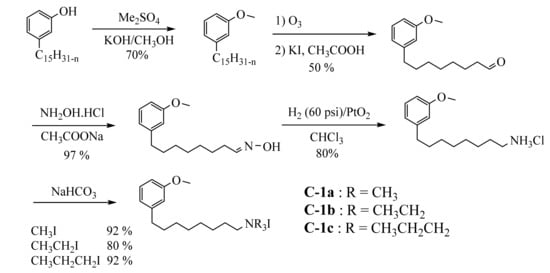
Cashew nutshell liquid (CNSL) has been used for the first time to prepare quaternary ammonium salts with the aim of obtaining water-soluble compounds with germicidal properties
[50][98].
Several decades later, quaternary ammonium salts used as a phase transfer agent were prepared in five steps starting from cardanol (
Scheme 49).
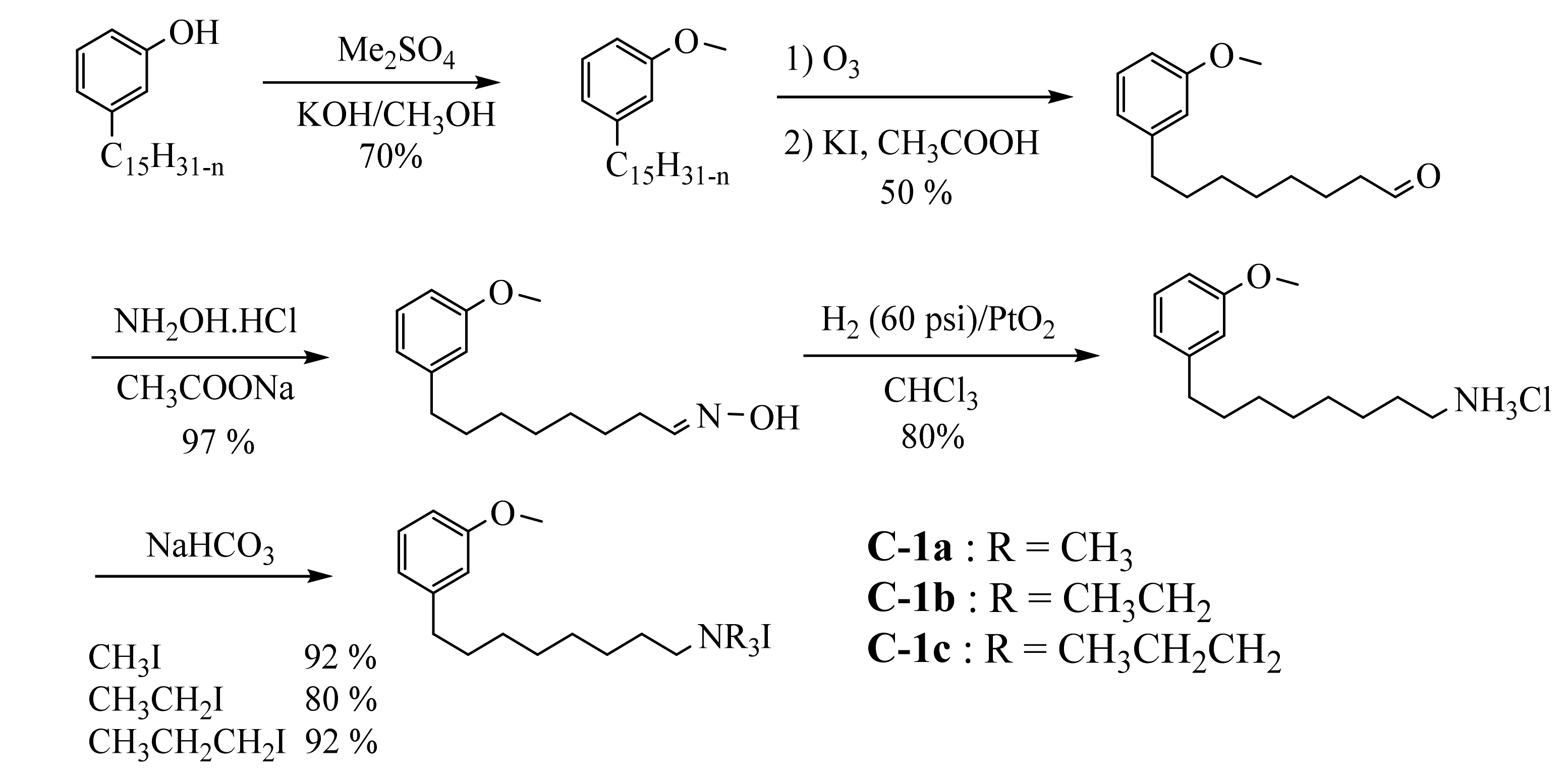
Scheme 9. Five steps synthesis of 3-methoxy-phenyl- N,N,N-trialkyl-1-N-octylammonium iodide surfactants from cardanol.
3.3.2. Macromolecular Ammonium CNSL-Based Surfactants
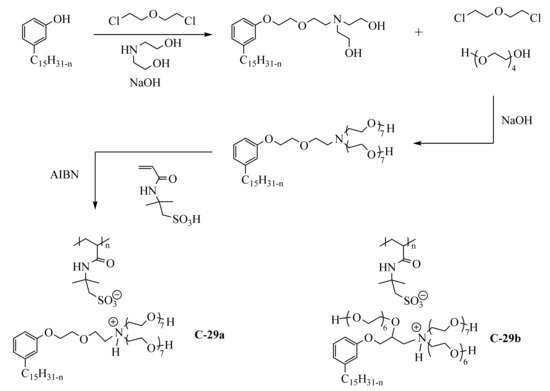
Amphiphilic poly(ionic liquid)s C-29a and C-29-b (
Scheme 519) were obtained from an acrylamide monomer bearing acid sulfonic group, the 2-acrylamido-2-methyl-1-propane-sulfonic acid
[51][109]. In the two first steps, the phenol group of cardanol was etherified with diethanolamine, ethanolamine, and tetraethylene glycol using a linking agent-based on bis(2-chloroethyl)ether. Then, the amine group was quaternized by acid-base reaction with the monomer and the radical polymerization was performed with 2,2′-azobis(2-methylpropionitrile) (AIBN) as initiator at 70 °C (C-29a). The surface activity of the polymer was studied with the aim to use it as a demulsifier for crude oil-water emulsions. This study reported the preparation of another amphiphilic poly(ionic liquid) bearing three hydrophilic chains (molecule C-29b not described
in th
ereis section).
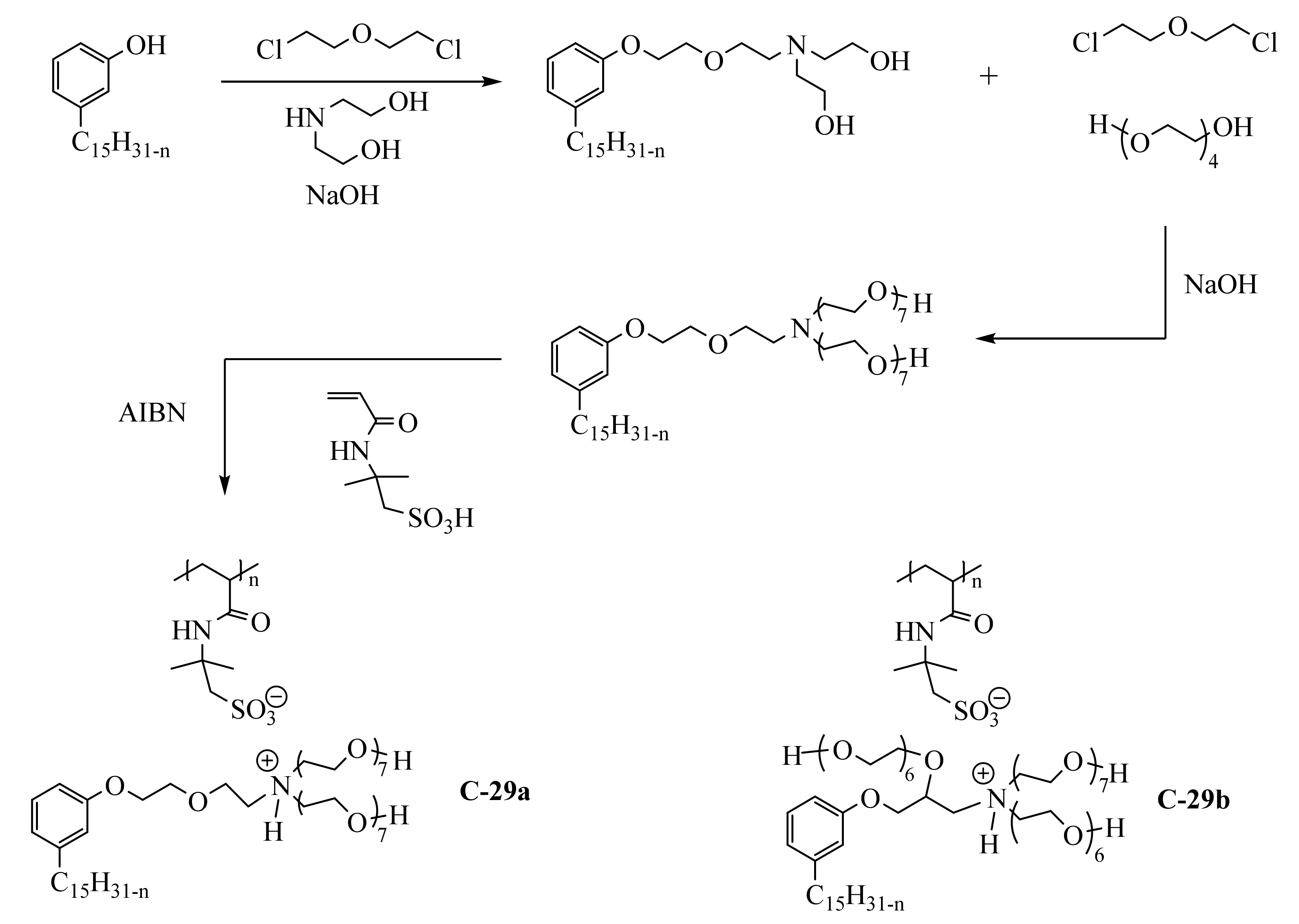
Scheme 19. Preparation of poly(ionic liquid)s based on cardanol and acrylamide monomer.
3.3.3. Pyridinium Derivatives of Cardanol
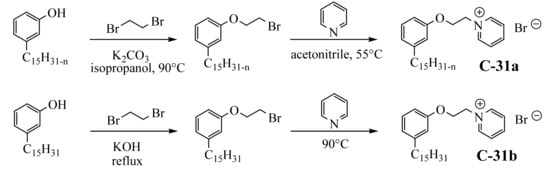
Two studies report a similar procedure for obtaining 62% biobased cationic surfactants by introducing the pyridinium group to cardanol in two steps
[52][53][102,104]. Firstly, cardanol was used for the nucleophilic substitution of an alkyl halide in the presence of a base (K
2CO
3 or KOH). Further, the pyridinium salts were obtained after reaction with pyridine, not the friendliest solvents (
Scheme 621).
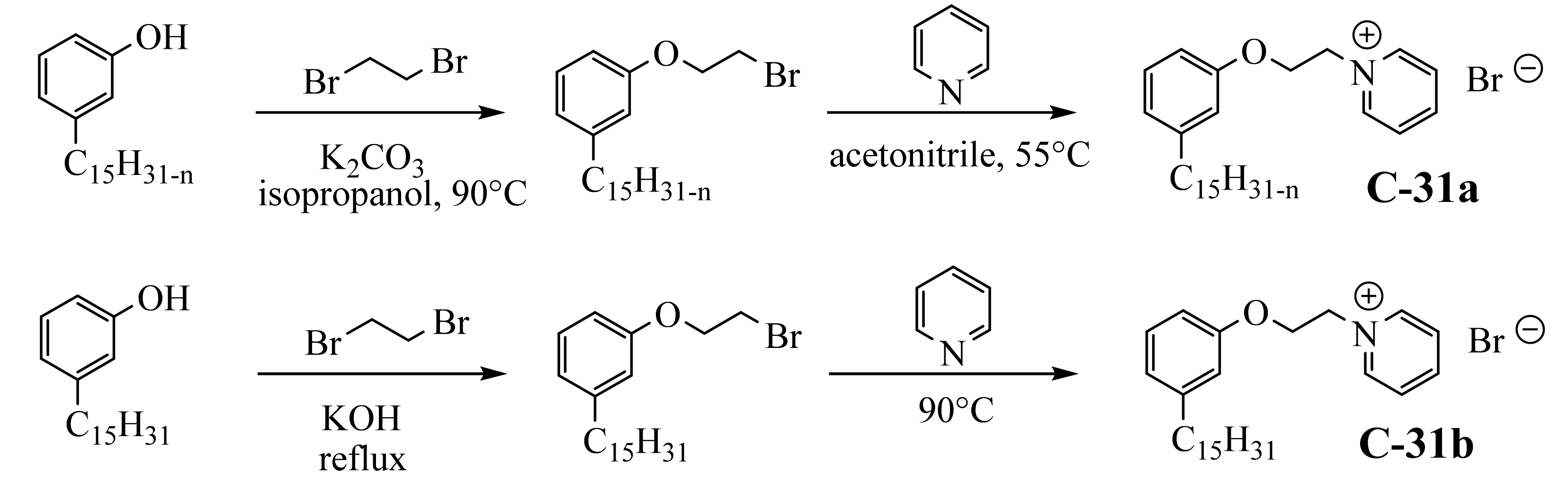
Scheme 21. Synthesis of pyridinium salt from cardanol and hydrogenated cardanol.
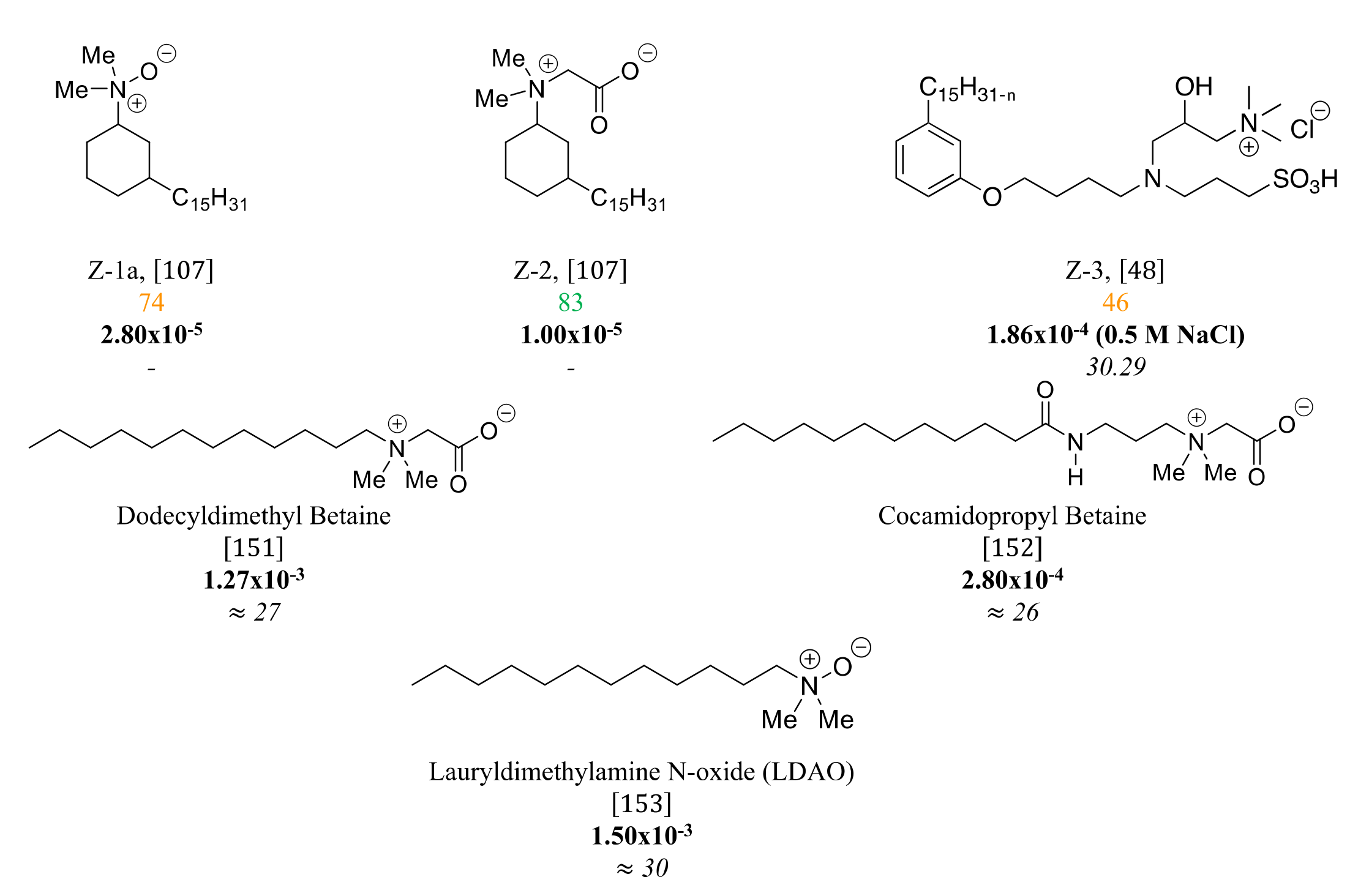
3.4. Zwitterionic Surfactants Based on CNSL
Zwitterionic surfactants are organic compounds exhibiting an apolar aliphatic chain and a polar head group with a positive and negative charge. This particularity provides them with very advantageous properties compared to conventional cationic or anionic surfactants. Usually, zwitterionic surfactants possess valuable antibacterial and fungicidal activity combined to good compatibility with a large range of surfactants. These salts are known for their robust structure, high tolerance to salinity and temperature. Moreover, zwitterionic surfactants have low toxicity which makes them almost non-irritating to the skin and eyes. Thus, these foaming agents can be used in mild shampoos and skin cleansers. Several other interesting and complementary properties should also be noted: these zwitterions are classified as antistatic and emulsifying agents with good dispersibility, wettability, and biocompatibility.
To date, very few examples have described zwitterionic surfactants derived from CNSL oil. All studies used cardanol as a raw material with a cationic part resulting from a quaternized amine or introduced by a ring-opening. Concerning the anionic part, a carboxylic or sulfonate anion was mainly used. To this end, phenol represents a very promising reactive function for developing zwitterionic surfactants by simple etherification.
3.5. Nonionic Surfactants Based on CNSL
Nonionic surfactants are molecules that do not undergo ionization when being dissolved in water. They exhibit high stability in solution, good solubility in both water and organic solvents, and excellent compatibility with other surfactants. This section describes the different syntheses of nonionic surfactants based on CNSL. All these surfactants have a hydrophilic part such as poly(ethylene oxide) or PEO, polyoxazoline, and/or sugar groups. According to the nature of the polar head, nonionic surfactants can be partially or totally biobased.
4. Structure-Property Relationship for CNSL-Based Surfactants
Indeed, CNSL surfactants are characterized by a large structural diversity, whether in terms of the nature of the polar head (anionic, cationic, zwitterionic, or nonionic) and/or the hydrophobic tail into the cardanol, cardol, or anacardic acid with the presence of alkenes on the C15 side chain combined with other hydrophilic/hydrophobic groups (alcohol, ether, amine, etc.). For example, several publications reported the preparation of Gemini (or dimeric) surfactants, which are composed of two polar heads and two hydrophobic tails, linked together by a spacer [54][55][141,142]. Gemini surfactants are known to be considerably more surface-active than conventional (single chain) surfactants.
Analyses of the structure-property relationship of CNSL surfactants were realized by comparing Critical Micelle Concentration (CMC), surface tension at CMC (γ
CMC), and sometimes Krafft temperature (T
K) or Cloud Point (T
C) of some CNSL-derivatives described in the synthesis section (cf.
Figure 210,
Figure 311,
Figure 412 and
Figure 513). Some trends related to the polar head (nature of the hydrophilic group, length) or the alkyl chain (nature of the phenolic compound, unsaturation) are considered here. However, one must bear in mind that experimental data about CNSL-based surfactants are often lacking. For example, very few studies deal with T
K or T
C of such molecules. As such, it can be difficult to conclude about the influence of a structural parameter on the surfactant properties.
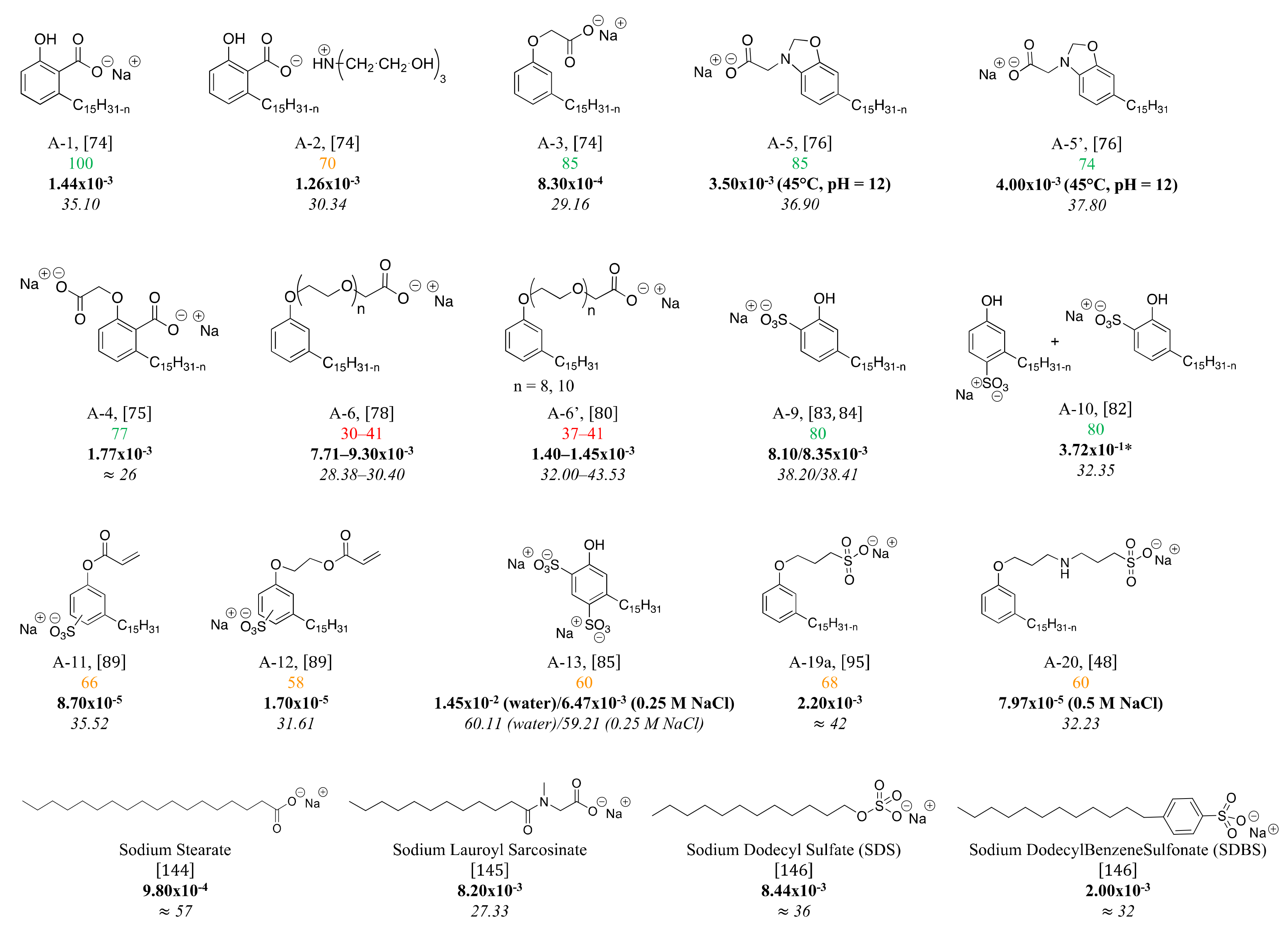
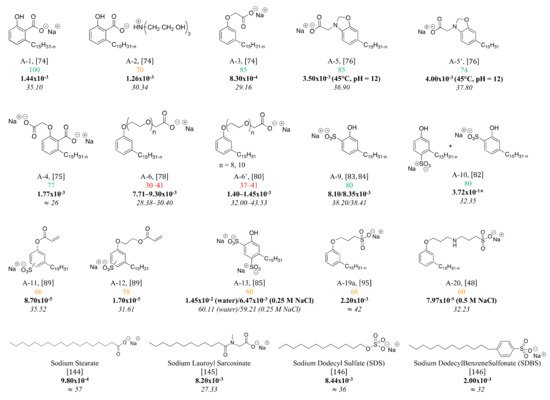
Figure 10. %
(green for % up to 75%, red for % under 50% and orange in between (50–75%), CMC (mol/L, bold) and surface tension (mN/m, italics) of anionic CNSL-based surfactants.
Figure 11. %
(orange in between (50–75%), CMC (mol/L, bold), and surface tension (mN/m, italics) of cationic CNSL-based surfactants.
Figure 12. %
(green for % up to 75%, and orange in between (50–75%), CMC (mol/L, bold) and surface tension (mN/m, italics) of zwitterionic CNSL-based surfactants.
Figure 513. %
bio (green for % up to 75%, red for % under 50%), CMC (mol/L, bold) and surface tension (mN/m, italics) of nonionic CNSL-based surfactants.
5. Conclusions
CNSL is a generous raw material with high potential. Generating many synthons in the field of polymers and additives; their application in the field of surfactants is no exception to the rule. From “A Brief Review of Cardanol-Based Surfactant” by Yang et al. in 2014
[22][24], the number of publications using one of the CNSL compounds for surfactant synthesis increased significantly. Numerous patents attest to the enthusiasm and interest of industries on this bio-based solution. It is also mainly patented from the Asian continent, which is very active in the production and valorization of these co-products of the cashew industry.
Considering a little more closely, the nature of the surfactants obtained or their synthesis, it can be noticed that, for the moment, it is far from an idyllic model. Even if the physicochemical properties (CMC, surface tension) of these surfactants compete with the commercial products currently on sale and in the process of substitution, many of these surfactants are biobased only from the hydrophobic part of their structure provided by the CNSL derivative. Indeed, except for some nonionic structures, where the polar head is a derivative of sugar, most come from compounds that are petrochemicals and/or use toxic/CMR reagents such as sultones or ethylene oxide.
CNSL is, therefore, a good track as synthons for the synthesis of effective surfactants. Much progress has been made over the last 10 years and efforts must be maintained to synthesize and produce non-hazardous surfactants that are the most biobased as possible. Synthesis routes still need to be optimized considering the fundamental principles of green chemistry. The future of this resource will be shaped by simple, inexpensive, and virtuous chemistry, involving the use of all CNSL compounds or even crude oil itself. CNSL provides the hydrophobic part of the surfactant, so it is necessary to imagine, as it is already the case for some nonionic surfactants, the use of bio-based polar heads, which also meet the criteria of non-toxicity.
ItWe is then able tocan then think of sugars, glycerol already used but also some amines, amino acids, acids, and diacids, etc.