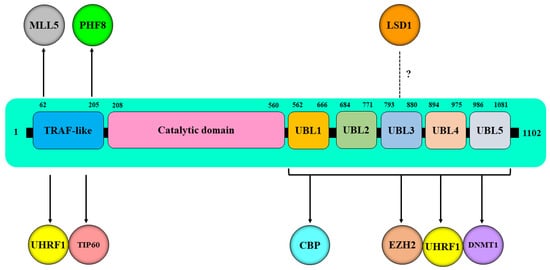
HAUSP (herpes virus-associated ubiquitin-specific protease), also known as Ubiquitin Specific Protease 7, plays critical roles in cellular processes, such as chromatin biology and epigenetics, through the regulation of different signaling pathways. HAUSP is a main partner of the “Epigenetic Code Replication Machinery,” ECREM, a large protein complex that includes several epigenetic players, such as the ubiquitin-like containing plant homeodomain (PHD) and an interesting new gene (RING), finger domains 1 (UHRF1), as well as DNA methyltransferase 1 (DNMT1), histone deacetylase 1 (HDAC1), histone methyltransferase G9a, and histone acetyltransferase TIP60. Due to its deubiquitinase activity and its ability to team up through direct interactions with several epigenetic regulators, mainly UHRF1, DNMT1, TIP60, the histone lysine methyltransferase EZH2, and the lysine-specific histone demethylase LSD1, HAUSP positions itself at the top of the regulatory hierarchies involved in epigenetic silencing of tumor suppressor genes in cancer.
- HAUSP
- UHRF1
- epigenetic
- cancer
1. Introduction
| Epi-Partner | Role of Epi-Partner | HAUSP Interaction Site | Epi-Partner Interaction Site | Related Epigenetic Events | Refs. | |||
|---|---|---|---|---|---|---|---|---|
| UHRF1 | Reader of both epigenetic marks (DNA methylation and histone code) | UBL1 domain (residues 560–664) |
A linker region encompassing amino acids 600–687 between the SRA and RING finger domains of UHRF1 | Promoting the stability of UHRF1 through HAUSP-dependent deubiquitination | [48] | |||
| TRAF-like domain | SRA domain | Promoting the stability of UHRF1 through HAUSP-dependent deubiquitination | [38] | |||||
| (UBL1-2) domains | A polybasic region (PBR) in the C terminus | Promoting the association of the TTD-PHD domains of UHRF1 with chromatin and, hence, efficient H3K9me3 binding | [57] | |||||
| DNMT1 | DNA methyltransferase 1 | C-terminal domain | Targeting sequence (TS) domain | Promoting the stability of DNMT1 through HAUSP-dependent deubiquitination | [38] | |||
| UBL domains (residues 560–1102) | KG linker (residues 1109–1119) | Promoting the stability of DNMT1 through acetylation of KG linker of DNMT1 | [49] | |||||
| TIP60 | Histone acetyltransferase | TRAF-like domain | Increased levels of H2AK 5 and H4K5 | [50] | ||||
| CBP | Histone acetyltransferase | Region encompassing amino acids 600–687 | CH3 domain | Increased levels of H3K56Ac | [51] | |||
| MLL5 | Histone lysine methyltransferase | TRAF domain | Multiple domains | Increased levels of H3K4m3 | [53] | |||
| EZH2 | Histone lysine N-methyltransferase | C-terminal region | 489 | PRKKKRK | 495 | region | Increased levels of H3K27m3 | [52] |
| LSD1 | Lysine specific demethylase 1 | [54] | ||||||
| A region encompassing amino acids 600–687 | Demethylation of H3K4me2 and H3K9me2 | [55] | ||||||
| PHF8 | Histone lysine demethylase | TRAF-like domain | The C-terminal region | Demethylation of H3K9me1,2, H3K27me2, and H4K20me1 | [56] |
2. Role of HAUSP in the Maintenance of DNA Methylation
2.1. HAUSP-Dependent Regulation of DNMT1 by Ubiquitination
The deubiquitinase activity of HAUSP regulates the stability and enzymatic activity of DNMT1 through several mechanisms. HAUSP can bind to DNMT1 and stabilize it through HAUSP-mediated deubiquitination [63][58]. High expression levels of DNMT1 protein have been found in human colon cancers, and this overexpression was correlated with HAUSP protein expression. Interestingly, the depletion of HAUSP in both human embryonic kidney cells and colorectal cancer cells resulted in an increase in DNMT1 ubiquitination and a reduction in DNMT1 protein expression. A similar increase in DNMT1 ubiquitination levels and a decrease in its protein expression were reported when HAUSP was knocked out in colorectal cancer cells [63][58]. By contrast, HAUSP overexpression in HAUSP knockout cells led to deubiquitination of DNMT1 and restoration of DNMT1 protein expression, suggesting that the deubiquitinase activity of HAUSP protects DNMT1 from proteasomal degradation, thereby promoting the stability and enzymatic activity of DNMT1 and the maintenance of DNA methylation [63][58].
One of the well-documented epigenetic regulators of the DNA methylation maintenance machinery is the epigenetic reader UHRF1, which has multiple functional domains [65,66,67][59][60][61]. Through its SRA domain, UHRF1 recognizes and binds hemi-methylated CpG islands, and via the same SRA domain, UHRF1 recruits DNMT1 to its correct position on chromatin to ensure faithful transmission of DNA methylation patterns during DNA replication [68,69,70][62][63][64].
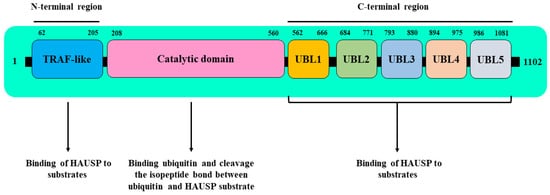
HAUSP binds to UHRF1 by its TRAF domain, but it also binds via its UBL domain (residues 560–664) to a linker region encompassing amino acids 600–687 between the SRA and RING finger domains of UHRF1, and this direct interaction is required for UHRF1 stability [48]. The downregulation of HAUSP decreased the expression levels of UHRF1 protein, whereas the overexpression of wild-type HAUSP, but not the catalytically inactive mutant HAUSP (C223S), significantly decreased the ubiquitination of UHRF1 and promoted its stability [48].
Obtaining a deep insight into how HAUSP deubiquitinase activity is involved in the stabilization of UHRF1 protein will help in understanding the regulatory role of HAUSP in the UHRF1-dependent maintenance of DNA methylation. In this regard, HAUSP was shown to regulate the stability of UHRF1 protein by targeting the ubiquitin ligase activity of the UHRF1 RING domain [38[38][59][65],65,81], which is used by UHRF1 to ubiquitinate itself (autoubiquitination) [65,66][59][60] or to ubiquitinate other substrates, mainly histone 3 [81,82,83][65][66][67]. Indeed, HAUSP, via its deubiquitinase activity, was shown to interfere with the E3 ubiquitin ligase activity of the RING domain of UHRF1, thereby eliminating the autoubiquitination of UHRF1 via the removal of ubiquitin adducts [38]. This led to the stabilization of UHRF1, indicating that HAUSP regulates the maintenance of DNA methylation through a direct association with UHRF1 [38].
2.2. HAUSP-Dependent Regulation of DNMT1 by Acetylation
3. Role of HAUSP in the Regulation of Histone Post-Translational Modifications
3.1. Role of HAUSP in the Regulation of Histone Monoubiquitination
3.2. Role of HAUSP in the Regulation of Histone Acetylation
The histone acetyltransferase TIP60 is a member of the ECREM complex that also includes HAUSP, UHRF1, and DNMT1, and each member has regulatory effects on the other partners (35, 49, 61, 70, 71, 73). TIP60, through its acetyltransferase, was shown to acetylate, destabilize, and trigger the ubiquitination of DNMT1 through the E3 ligase activity of UHRF1 [63][58]. TIP60 is also required for acetylation of histones [101,102,103][82][83][84] and of nonhistone proteins, such as the tumor suppressor p53 [104,105][85][86]. Thus, deciphering the mechanisms of TIP60 regulation is crucial for unraveling the complex role of this histone acetyltransferase in tumors. TIP60 is a target of the deubiquitinase activity of HAUSP [50], which interacts through its TRAF domain to deubiquitinate and stabilize TIP60, leading to the subsequent induction of p53-dependent apoptosis [50].3.3. Role of HAUSP in the Regulation of Histone Methylation
Histone lysine methyltransferases (KMTs), such as EZH2 [110][87] and MLL5, [53] and lysine specific histone demethylase (KDMs), including LSD1 [111,112][88][89] and PHF8 [113[90][91],114], are two families of enzymes involved in histone methylation and demethylation, respectively. Specifically, the trimethylation of lysine 27 of histone 3 (H3K27me3), mediated by EZH2, is considered one of histone changes that silence target genes, thereby promoting tumor growth and metastasis in many tumors [110,115,116,117][87][92][93][94]. EZH2 is a core subunit of the Polycomb Repressive Complex 2 (PRC2), which is associated with cancer [118,119][95][96].3.4. Role of HAUSP in the UHRF1-Mediated Readout of Histone H3 Lysine 9 Methylation
4. HAUSP Inhibitors as Promising Anticancer Agents
References
- Rawat, R.; Starczynowski, D.T.; Ntziachristos, P. Nuclear deubiquitination in the spotlight: The multifaceted nature of USP7 biology in disease. Curr. Opin. Cell Biol. 2019, 58, 85–94.
- Uckelmann, M.; Sixma, T.K. Histone ubiquitination in the DNA damage response. DNA Repair 2017, 56, 92–101.
- Liu, J.; Shaik, S.; Dai, X.; Wu, Q.; Zhou, X.; Wang, Z.; Wei, W. Targeting the ubiquitin pathway for cancer treatment. Biochim. Biophys. Acta 2015, 1855, 50–60.
- Yerlikaya, A.; Kanbur, E.; Stanley, B.A.; Tümer, E. The Ubiquitin-Proteasome Pathway and Epigenetic Modifications in Cancer. Anti-Cancer Agents Med. Chem. 2021, 21, 20–32.
- Komander, D.; Rape, M. The ubiquitin code. Annu. Rev. Biochem. 2012, 81, 203–229.
- Schulman, B.A.; Harper, J.W. Ubiquitin-like protein activation by E1 enzymes: The apex for downstream signalling pathways. Nat. Rev. Mol. Cell Biol. 2009, 10, 310–319.
- Ye, Y.; Rape, M. Building ubiquitin chains: E2 enzymes at work. Nat. Rev. Mol. Cell Biol. 2009, 10, 755–764.
- Mennerich, D.; Kubaichuk, K.; Kietzmann, T. DUBs, Hypoxia, and Cancer. Trends Cancer 2019, 5, 632–653.
- Poondla, N.; Chandrasekaran, A.P.; Kim, K.S.; Ramakrishna, S. Deubiquitinating enzymes as cancer biomarkers: New therapeutic opportunities? BMB Rep. 2019, 52, 181–189.
- Farshi, P.; Deshmukh, R.R.; Nwankwo, J.O.; Arkwright, R.T.; Cvek, B.; Liu, J.; Dou, Q.P. Deubiquitinases (DUBs) and DUB inhibitors: A patent review. Expert Opin. Ther. Pat. 2015, 25, 1191–1208.
- McClurg, U.L.; Robson, C.N. Deubiquitinating enzymes as oncotargets. Oncotarget 2015, 6, 9657–9668.
- Song, M.S.; Salmena, L.; Carracedo, A.; Egia, A.; Lo-Coco, F.; Teruya-Feldstein, J.; Pandolfi, P.P. The deubiquitinylation and localization of PTEN are regulated by a HAUSP-PML network. Nature 2008, 455, 813–817.
- Zhou, J.; Wang, J.; Chen, C.; Yuan, H.; Wen, X.; Sun, H. USP7, Target Validation and Drug Discovery for Cancer Therapy. Med. Chem. 2018, 14, 3–18.
- Bhattacharya, S.; Chakraborty, D.; Basu, M.; Ghosh, M.K. Emerging insights into HAUSP (USP7) in physiology, cancer and other diseases. Signal Transduct. Target. Ther. 2018, 3, 1–12.
- Cheng, C.D.; Dong, Y.F.; Niu, W.X.; Niu, C.S. HAUSP promoted the growth of glioma cells in vitro and in vivo via stabilizing NANOG. Pathol. Res. Pract. 2020, 216, 152883.
- Tavana, O.; Li, D.; Dai, C.; Lopez, G.; Banerjee, D.; Kon, N.; Chen, C.; Califano, A.; Yamashiro, D.J.; Sun, H.; et al. HAUSP deubiquitinates and stabilizes N-Myc in neuroblastoma. Nat. Med. 2016, 22, 1180–1186.
- Pozhidaeva, A.; Bezsonova, I. USP7, Structure, substrate specificity, and inhibition. DNA Repair 2019, 76, 30–39.
- Holowaty, M.N.; Sheng, Y.; Nguyen, T.; Arrowsmith, C.; Frappier, L. Protein interaction domains of the ubiquitin-specific protease, USP7/HAUSP. J. Biol. Chem. 2003, 278, 47753–47761.
- Jin, Q.; Martinez, C.A.; Arcipowski, K.M.; Zhu, Y.; Gutierrez-Diaz, B.T.; Wang, K.K.; Johnson, M.R.; Volk, A.G.; Wang, F.; Wu, J.; et al. USP7 Cooperates with NOTCH1 to Drive the Oncogenic Transcriptional Program in T-Cell Leukemia. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2019, 25, 222–239.
- Shan, H.; Li, X.; Xiao, X.; Dai, Y.; Huang, J.; Song, J.; Liu, M.; Yang, L.; Lei, H.; Tong, Y.; et al. USP7 deubiquitinates and stabilizes NOTCH1 in T-cell acute lymphoblastic leukemia. Signal Transduct. Target. Ther. 2018, 3, 29.
- Novellasdemunt, L.; Foglizzo, V.; Cuadrado, L.; Antas, P.; Kucharska, A.; Encheva, V.; Snijders, A.P.; Li, V.S.W. USP7 Is a Tumor-Specific WNT Activator for APC-Mutated Colorectal Cancer by Mediating β-Catenin Deubiquitination. Cell Rep. 2017, 21, 612–627.
- Franqui-Machin, R.; Hao, M.; Bai, H.; Gu, Z.; Zhan, X.; Habelhah, H.; Jethava, Y.; Qiu, L.; Frech, I.; Tricot, G.; et al. Destabilizing NEK2 overcomes resistance to proteasome inhibition in multiple myeloma. J. Clin. Investig. 2018, 128, 2877–2893.
- Tavana, O.; Sun, H.; Gu, W. Targeting HAUSP in both p53 wildtype and p53-mutant tumors. Cell Cycle 2018, 17, 823–828.
- Brooks, C.L.; Li, M.; Hu, M.; Shi, Y.; Gu, W. The p53–Mdm2–HAUSP complex is involved in p53 stabilization by HAUSP. Oncogene 2007, 26, 7262–7266.
- Bhattacharya, S.; Ghosh, M.K. HAUSP, a novel deubiquitinase for Rb-MDM2 the critical regulator. FEBS J. 2014, 281, 3061–3078.
- Meulmeester, E.; Pereg, Y.; Shiloh, Y.; Jochemsen, A.G. ATM-mediated phosphorylations inhibit Mdmx/Mdm2 stabilization by HAUSP in favor of p53 activation. Cell Cycle 2005, 4, 1166–1170.
- Harris, S.L.; Levine, A.J. The p53 pathway: Positive and negative feedback loops. Oncogene 2005, 24, 2899–2908.
- Karni-Schmidt, O.; Lokshin, M.; Prives, C. The Roles of MDM2 and MDMX in Cancer. Annu. Rev. Pathol. 2016, 11, 617–644.
- Marine, J.C.; Jochemsen, A.G. Mdmx as an essential regulator of p53 activity. Biochem. Biophys. Res. Commun. 2005, 331, 750–760.
- Melo, A.N.; Eischen, C.M. Protecting the genome from mdm2 and mdmx. Genes Cancer 2012, 3, 283–290.
- Li, Y.; Reverter, D. Molecular Mechanisms of DUBs Regulation in Signaling and Disease. Int. J. Mol. Sci. 2021, 22, 986.
- Alhosin, M.; Sharif, T.; Mousli, M.; Etienne-Selloum, N.; Fuhrmann, G.; Schini-Kerth, V.B.; Bronner, C. Down-regulation of UHRF1, associated with re-expression of tumor suppressor genes, is a common feature of natural compounds exhibiting anti-cancer properties. J. Exp. Clin. Cancer Res. 2011, CR 30, 41.
- Xue, B.; Zhao, J.; Feng, P.; Xing, J.; Wu, H.; Li, Y. Epigenetic mechanism and target therapy of UHRF1 protein complex in malignancies. OncoTargets Ther. 2019, 12, 549–559.
- Abdullah, O.; Omran, Z.; Hosawi, S.; Hamiche, A.; Bronner, C.; Alhosin, M. Thymoquinone Is a Multitarget Single Epidrug That Inhibits the UHRF1 Protein Complex. Genes 2021, 12, 622.
- Unoki, M. Current and potential anticancer drugs targeting members of the UHRF1 complex including epigenetic modifiers. Recent Pat. Anti-Cancer Drug Discov. 2011, 6, 116–130.
- Ahmad, T.; Ashraf, W.; Ibrahim, A.; Zaayter, L.; Muller, C.D.; Hamiche, A.; Mély, Y.; Bronner, C.; Mousli, M. TIP60 governs the auto-ubiquitination of UHRF1 through USP7 dissociation from the UHRF1/USP7 complex. Int. J. Oncol. 2021, 59, 89.
- Choudhry, H.; Zamzami, M.A.; Omran, Z.; Wu, W.; Mousli, M.; Bronner, C.; Alhosin, M. Targeting microRNA/UHRF1 pathways as a novel strategy for cancer therapy. Oncol. Lett. 2018, 15, 3–10.
- Felle, M.; Joppien, S.; Németh, A.; Diermeier, S.; Thalhammer, V.; Dobner, T.; Kremmer, E.; Kappler, R.; Längst, G. The USP7/Dnmt1 complex stimulates the DNA methylation activity of Dnmt1 and regulates the stability of UHRF1. Nucleic Acids Res. 2011, 39, 8355–8365.
- Bronner, C.; Alhosin, M.; Hamiche, A.; Mousli, M. Coordinated Dialogue between UHRF1 and DNMT1 to Ensure Faithful Inheritance of Methylated DNA Patterns. Genes 2019, 10, 65.
- Du, J.; Johnson, L.M.; Jacobsen, S.E.; Patel, D.J. DNA methylation pathways and their crosstalk with histone methylation. Nat. Rev. Mol. Cell Biol. 2015, 16, 519–532.
- Ren, W.; Fan, H.; Grimm, S.A.; Guo, Y.; Kim, J.J.; Yin, J.; Li, L.; Petell, C.J.; Tan, X.F.; Zhang, Z.M.; et al. Direct readout of heterochromatic H3K9me3 regulates DNMT1-mediated maintenance DNA methylation. Proc. Natl. Acad. Sci. USA 2020, 117, 18439–18447.
- Cedar, H.; Bergman, Y. Linking DNA methylation and histone modification: Patterns and paradigms. Nat. Rev. Genet. 2009, 10, 295–304.
- Bronner, C.; Krifa, M.; Mousli, M. Increasing role of UHRF1 in the reading and inheritance of the epigenetic code as well as in tumorogenesis. Biochem. Pharmacol. 2013, 86, 1643–1649.
- Kilin, V.; Gavvala, K.; Barthes, N.P.; Michel, B.Y.; Shin, D.; Boudier, C.; Mauffret, O.; Yashchuk, V.; Mousli, M.; Ruff, M.; et al. Dynamics of Methylated Cytosine Flipping by UHRF1. J. Am. Chem. Soc. 2017, 139, 2520–2528.
- Bronner, C.; Fuhrmann, G.; Chédin, F.L.; Macaluso, M.; Dhe-Paganon, S. UHRF1 Links the Histone code and DNA Methylation to ensure Faithful Epigenetic Memory Inheritance. Genet. Epigenet. 2010, 2009, 29–36.
- Li, T.; Wang, L.; Du, Y.; Xie, S.; Yang, X.; Lian, F.; Zhou, Z.; Qian, C. Structural and mechanistic insights into UHRF1-mediated DNMT1 activation in the maintenance DNA methylation. Nucleic Acids Res. 2018, 46, 3218–3231.
- Liu, X.; Gao, Q.; Li, P.; Zhao, Q.; Zhang, J.; Li, J.; Koseki, H.; Wong, J. UHRF1 targets DNMT1 for DNA methylation through cooperative binding of hemi-methylated DNA and methylated H3K9. Nat. Commun. 2013, 4, 1563.
- Ma, H.; Chen, H.; Guo, X.; Wang, Z.; Sowa, M.E.; Zheng, L.; Hu, S.; Zeng, P.; Guo, R.; Diao, J.; et al. M phase phosphorylation of the epigenetic regulator UHRF1 regulates its physical association with the deubiquitylase USP7 and stability. Proc. Natl. Acad. Sci. USA 2012, 109, 4828–4833.
- Cheng, J.; Yang, H.; Fang, J.; Ma, L.; Gong, R.; Wang, P.; Li, Z.; Xu, Y. Molecular mechanism for USP7-mediated DNMT1 stabilization by acetylation. Nat. Commun. 2015, 6, 7023.
- Dar, A.; Shibata, E.; Dutta, A. Deubiquitination of Tip60 by USP7 determines the activity of the p53-dependent apoptotic pathway. Mol. Cell. Biol. 2013, 33, 3309–3320.
- Wu, H.T.; Kuo, Y.C.; Hung, J.J.; Huang, C.H.; Chen, W.Y.; Chou, T.Y.; Chen, Y.; Chen, Y.J.; Chen, Y.J.; Cheng, W.C.; et al. K63-polyubiquitinated HAUSP deubiquitinates HIF-1α and dictates H3K56 acetylation promoting hypoxia-induced tumour progression. Nat. Commun. 2016, 7, 13644.
- Gagarina, V.; Bojagora, A.; Lacdao, I.K.; Luthra, N.; Pfoh, R.; Mohseni, S.; Chaharlangi, D.; Tan, N.; Saridakis, V. Structural Basis of the Interaction Between Ubiquitin Specific Protease 7 and Enhancer of Zeste Homolog 2. J. Mol. Biol. 2020, 432, 897–912.
- Ding, X.; Jiang, W.; Zhou, P.; Liu, L.; Wan, X.; Yuan, X.; Wang, X.; Chen, M.; Chen, J.; Yang, J.; et al. Mixed Lineage Leukemia 5 (MLL5) Protein Stability Is Cooperatively Regulated by O-GlcNac Transferase (OGT) and Ubiquitin Specific Protease 7 (USP7). PLoS ONE 2015, 10, e0145023.
- Yi, L.; Cui, Y.; Xu, Q.; Jiang, Y. Stabilization of LSD1 by deubiquitinating enzyme USP7 promotes glioblastoma cell tumorigenesis and metastasis through suppression of the p53 signaling pathway. Oncol. Rep. 2016, 36, 2935–2945.
- Liu, J.; Feng, J.; Li, L.; Lin, L.; Ji, J.; Lin, C.; Liu, L.; Zhang, N.; Duan, D.; Li, Z.; et al. Arginine methylation-dependent LSD1 stability promotes invasion and metastasis of breast cancer. EMBO Rep. 2020, 21, e48597.
- Wang, Q.; Ma, S.; Song, N.; Li, X.; Liu, L.; Yang, S.; Ding, X.; Shan, L.; Zhou, X.; Su, D.; et al. Stabilization of histone demethylase PHF8 by USP7 promotes breast carcinogenesis. J. Clin. Investig. 2016, 126, 2205–2220.
- Zhang, Z.M.; Rothbart, S.B.; Allison, D.F.; Cai, Q.; Harrison, J.S.; Li, L.; Wang, Y.; Strahl, B.D.; Wang, G.G.; Song, J. An Allosteric Interaction Links USP7 to Deubiquitination and Chromatin Targeting of UHRF1. Cell Rep. 2015, 12, 1400–1406.
- Du, Z.; Song, J.; Wang, Y.; Zhao, Y.; Guda, K.; Yang, S.; Kao, H.Y.; Xu, Y.; Willis, J.; Markowitz, S.D.; et al. DNMT1 stability is regulated by proteins coordinating deubiquitination and acetylation-driven ubiquitination. Sci. Signal. 2010, 3, ra80.
- Jenkins, Y.; Markovtsov, V.; Lang, W.; Sharma, P.; Pearsall, D.; Warner, J.; Franci, C.; Huang, B.; Huang, J.; Yam, G.C.; et al. Critical role of the ubiquitin ligase activity of UHRF1, a nuclear RING finger protein, in tumor cell growth. Mol. Biol. Cell 2005, 16, 5621–5629.
- Ibrahim, A.; Alhosin, M.; Papin, C.; Ouararhni, K.; Omran, Z.; Zamzami, M.A.; Al-Malki, A.L.; Choudhry, H.; Mély, Y.; Hamiche, A.; et al. Thymoquinone challenges UHRF1 to commit auto-ubiquitination: A key event for apoptosis induction in cancer cells. Oncotarget 2018, 9, 28599–28611.
- Hashimoto, H.; Horton, J.R.; Zhang, X.; Bostick, M.; Jacobsen, S.E.; Cheng, X. The SRA domain of UHRF1 flips 5-methylcytosine out of the DNA helix. Nature 2008, 455, 826–829.
- Avvakumov, G.V.; Walker, J.R.; Xue, S.; Li, Y.; Duan, S.; Bronner, C.; Arrowsmith, C.H.; Dhe-Paganon, S. Structural basis for recognition of hemi-methylated DNA by the SRA domain of human UHRF1. Nature 2008, 455, 822–825.
- Bostick, M.; Kim, J.K.; Estève, P.O.; Clark, A.; Pradhan, S.; Jacobsen, S.E. UHRF1 plays a role in maintaining DNA methylation in mammalian cells. Science 2007, 317, 1760–1764.
- Sharif, J.; Muto, M.; Takebayashi, S.; Suetake, I.; Iwamatsu, A.; Endo, T.A.; Shinga, J.; Mizutani-Koseki, Y.; Toyoda, T.; Okamura, K.; et al. The SRA protein Np95 mediates epigenetic inheritance by recruiting Dnmt1 to methylated DNA. Nature 2007, 450, 908–912.
- Nishiyama, A.; Yamaguchi, L.; Sharif, J.; Johmura, Y.; Kawamura, T.; Nakanishi, K.; Shimamura, S.; Arita, K.; Kodama, T.; Ishikawa, F.; et al. Uhrf1-dependent H3K23 ubiquitylation couples maintenance DNA methylation and replication. Nature 2013, 502, 249–253.
- Misaki, T.; Yamaguchi, L.; Sun, J.; Orii, M.; Nishiyama, A.; Nakanishi, M. The replication foci targeting sequence (RFTS) of DNMT1 functions as a potent histone H3 binding domain regulated by autoinhibition. Biochem. Biophys. Res. Commun. 2016, 470, 741–747.
- Qin, W.; Wolf, P.; Liu, N.; Link, S.; Smets, M.; La Mastra, F.; Forné, I.; Pichler, G.; Hörl, D.; Fellinger, K.; et al. DNA methylation requires a DNMT1 ubiquitin interacting motif (UIM) and histone ubiquitination. Cell Res. 2015, 25, 911–929.
- Qin, W.; Leonhardt, H.; Spada, F. Usp7 and Uhrf1 control ubiquitination and stability of the maintenance DNA methyltransferase Dnmt1. J. Cell. Biochem. 2011, 112, 439–444.
- Marsh, D.J.; Ma, Y.; Dickson, K.A. Histone Monoubiquitination in Chromatin Remodelling: Focus on the Histone H2B Interactome and Cancer. Cancers 2020, 12, 3462.
- Yan, Q.; Chen, B.J.; Hu, S.; Qi, S.L.; Li, L.Y.; Yang, J.F.; Zhou, H.; Yang, C.C.; Chen, L.J.; Du, J. Emerging role of RNF2 in cancer: From bench to bedside. J. Cell. Physiol. 2021, 236, 5453–5465.
- Marsh, D.J.; Dickson, K.A. Writing Histone Monoubiquitination in Human Malignancy—The Role of RING Finger E3 Ubiquitin Ligases. Genes 2019, 10, 67.
- Cohen, I.; Bar, C.; Ezhkova, E. Activity of PRC1 and Histone H2AK119 Monoubiquitination: Revising Popular Misconceptions. BioEssays News Rev. Mol. Cell. Dev. Biol. 2020, 42, e1900192.
- Benitz, S.; Regel, I.; Reinhard, T.; Popp, A.; Schäffer, I.; Raulefs, S.; Kong, B.; Esposito, I.; Michalski, C.W.; Kleeff, J. Polycomb repressor complex 1 promotes gene silencing through H2AK119 mono-ubiquitination in acinar-to-ductal metaplasia and pancreatic cancer cells. Oncotarget 2016, 7, 11424–11433.
- Sarkari, F.; Wheaton, K.; La Delfa, A.; Mohamed, M.; Shaikh, F.; Khatun, R.; Arrowsmith, C.H.; Frappier, L.; Saridakis, V.; Sheng, Y. Ubiquitin-specific protease 7 is a regulator of ubiquitin-conjugating enzyme UbE2E1. J. Biol. Chem. 2013, 288, 16975–16985.
- Zhang, Y.; Chan, H.L.; Garcia-Martinez, L.; Karl, D.L.; Weich, N.; Slingerland, J.M.; Verdun, R.E.; Morey, L. Estrogen induces dynamic ERα and RING1B recruitment to control gene and enhancer activities in luminal breast cancer. Sci. Adv. 2020, 6, eaaz7249.
- Chan, H.L.; Beckedorff, F.; Zhang, Y.; Garcia-Huidobro, J.; Jiang, H.; Colaprico, A.; Bilbao, D.; Figueroa, M.E.; LaCava, J.; Shiekhattar, R.; et al. Polycomb complexes associate with enhancers and promote oncogenic transcriptional programs in cancer through multiple mechanisms. Nat. Commun. 2018, 9, 3377.
- Benitz, S.; Straub, T.; Mahajan, U.M.; Mutter, J.; Czemmel, S.; Unruh, T.; Wingerath, B.; Deubler, S.; Fahr, L.; Cheng, T.; et al. Ring1b-dependent epigenetic remodelling is an essential prerequisite for pancreatic carcinogenesis. Gut 2019, 68, 2007–2018.
- Sánchez-Molina, S.; Figuerola-Bou, E.; Blanco, E.; Sánchez-Jiménez, M.; Táboas, P.; Gómez, S.; Ballaré, C.; García-Domínguez, D.J.; Prada, E.; Hontecillas-Prieto, L.; et al. RING1B recruits EWSR1-FLI1 and cooperates in the remodeling of chromatin necessary for Ewing sarcoma tumorigenesis. Sci. Adv. 2020, 6, eaba3058.
- de Bie, P.; Zaaroor-Regev, D.; Ciechanover, A. Regulation of the Polycomb protein RING1B ubiquitination by USP7. Biochem. Biophys. Res. Commun. 2010, 400, 389–395.
- Wang, H.; Wang, L.; Erdjument-Bromage, H.; Vidal, M.; Tempst, P.; Jones, R.S.; Zhang, Y. Role of histone H2A ubiquitination in Polycomb silencing. Nature 2004, 431, 873–878.
- Wheaton, K.; Sarkari, F.; Johns, B.S.; Davarinejad, H.; Egorova, O.; Kaustov, L.; Raught, B.; Saridakis, V.; Sheng, Y. UbE2E1/UBCH6 Is a Critical In Vivo E2 for the PRC1-catalyzed Ubiquitination of H2A at Lys-119. J. Biol. Chem. 2017, 292, 2893–2902.
- Squatrito, M.; Gorrini, C.; Amati, B. Tip60 in DNA damage response and growth control: Many tricks in one HAT. Trends Cell Biol. 2006, 16, 433–442.
- Judes, G.; Rifaï, K.; Ngollo, M.; Daures, M.; Bignon, Y.J.; Penault-Llorca, F.; Bernard-Gallon, D. A bivalent role of TIP60 histone acetyl transferase in human cancer. Epigenomics 2015, 7, 1351–1363.
- Judes, G.; Dubois, L.; Rifaï, K.; Idrissou, M.; Mishellany, F.; Pajon, A.; Besse, S.; Daures, M.; Degoul, F.; Bignon, Y.J.; et al. TIP60, an actor in acetylation of H3K4 and tumor development in breast cancer. Epigenomics 2018, 10, 1415–1430.
- Tang, Y.; Luo, J.; Zhang, W.; Gu, W. Tip60-dependent acetylation of p53 modulates the decision between cell-cycle arrest and apoptosis. Mol. Cell 2006, 24, 827–839.
- Sykes, S.M.; Mellert, H.S.; Holbert, M.A.; Li, K.; Marmorstein, R.; Lane, W.S.; McMahon, S.B. Acetylation of the p53 DNA-binding domain regulates apoptosis induction. Mol. Cell 2006, 24, 841–851.
- Yamagishi, M.; Uchimaru, K. Targeting EZH2 in cancer therapy. Curr. Opin. Oncol. 2017, 29, 375–381.
- Lim, S.; Janzer, A.; Becker, A.; Zimmer, A.; Schüle, R.; Buettner, R.; Kirfel, J. Lysine-specific demethylase 1 (LSD1) is highly expressed in ER-negative breast cancers and a biomarker predicting aggressive biology. Carcinogenesis 2010, 31, 512–520.
- Lan, F.; Nottke, A.C.; Shi, Y. Mechanisms involved in the regulation of histone lysine demethylases. Curr. Opin. Cell Biol. 2008, 20, 316–325.
- Tsukada, Y.; Ishitani, T.; Nakayama, K.I. KDM7 is a dual demethylase for histone H3 Lys 9 and Lys 27 and functions in brain development. Genes Dev. 2010, 24, 432–437.
- Feng, W.; Yonezawa, M.; Ye, J.; Jenuwein, T.; Grummt, I. PHF8 activates transcription of rRNA genes through H3K4me3 binding and H3K9me1/2 demethylation. Nat. Struct. Mol. Biol. 2010, 17, 445–450.
- Kim, K.H.; Roberts, C.W. Targeting EZH2 in cancer. Nat. Med. 2016, 22, 128–134.
- Duan, R.; Du, W.; Guo, W. EZH2, a novel target for cancer treatment. J. Hematol. Oncol. 2020, 13, 104.
- Varambally, S.; Dhanasekaran, S.M.; Zhou, M.; Barrette, T.R.; Kumar-Sinha, C.; Sanda, M.G.; Ghosh, D.; Pienta, K.J.; Sewalt, R.G.; Otte, A.P.; et al. The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature 2002, 419, 624–629.
- Kirmizis, A.; Bartley, S.M.; Kuzmichev, A.; Margueron, R.; Reinberg, D.; Green, R.; Farnham, P.J. Silencing of human polycomb target genes is associated with methylation of histone H3 Lys 27. Genes Dev. 2004, 18, 1592–1605.
- Lee, T.I.; Jenner, R.G.; Boyer, L.A.; Guenther, M.G.; Levine, S.S.; Kumar, R.M.; Chevalier, B.; Johnstone, S.E.; Cole, M.F.; Isono, K.; et al. Control of developmental regulators by Polycomb in human embryonic stem cells. Cell 2006, 125, 301–313.
- Houliston, R.S.; Lemak, A.; Iqbal, A.; Ivanochko, D.; Duan, S.; Kaustov, L.; Ong, M.S.; Fan, L.; Senisterra, G.; Brown, P.J.; et al. Conformational dynamics of the TTD-PHD histone reader module of the UHRF1 epigenetic regulator reveals multiple histone-binding states, allosteric regulation, and druggability. J. Biol. Chem. 2017, 292, 20947–20959.
- Cheng, J.; Yang, Y.; Fang, J.; Xiao, J.; Zhu, T.; Chen, F.; Wang, P.; Li, Z.; Yang, H.; Xu, Y. Structural insight into coordinated recognition of trimethylated histone H3 lysine 9 (H3K9me3) by the plant homeodomain (PHD) and tandem tudor domain (TTD) of UHRF1 (ubiquitin-like, containing PHD and RING finger domains, 1) protein. J. Biol. Chem. 2013, 288, 1329–1339.
- Gelato, K.A.; Tauber, M.; Ong, M.S.; Winter, S.; Hiragami-Hamada, K.; Sindlinger, J.; Lemak, A.; Bultsma, Y.; Houliston, S.; Schwarzer, D.; et al. Accessibility of different histone H3-binding domains of UHRF1 is allosterically regulated by phosphatidylinositol 5-phosphate. Mol. Cell 2014, 54, 905–919.
- Colland, F.; Formstecher, E.; Jacq, X.; Reverdy, C.; Planquette, C.; Conrath, S.; Trouplin, V.; Bianchi, J.; Aushev, V.N.; Camonis, J.; et al. Small-molecule inhibitor of USP7/HAUSP ubiquitin protease stabilizes and activates p53 in cells. Mol. Cancer Ther. 2009, 8, 2286–2295.
- Kategaya, L.; di Lello, P.; Rougé, L.; Pastor, R.; Clark, K.R.; Drummond, J.; Kleinheinz, T.; Lin, E.; Upton, J.P.; Prakash, S.; et al. USP7 small-molecule inhibitors interfere with ubiquitin binding. Nature 2009, 550, 534–538.
- Pozhidaeva, A.; Valles, G.; Wang, F.; Wu, J.; Sterner, D.E.; Nguyen, P.; Weinstock, J.; Kumar, K.G.S.; Kanyo, J.; Wright, D.; et al. USP7-Specific Inhibitors Target and Modify the Enzyme’s Active Site via Distinct Chemical Mechanisms. Cell Chem. Biol. 2017, 24, 1501–1512.
- Nininahazwe, L.; Liu, B.; He, C.; Zhang, H.; Chen, Z.S. The emerging nature of Ubiquitin-specific protease 2017, 7 (USP7): A new target in cancer therapy. Drug Discov. Today 2021, 26, 490–502.
- Altun, M.; Kramer, H.B.; Willems, L.I.; McDermott, J.L.; Leach, C.A.; Goldenberg, S.J.; Kumar, K.G.; Konietzny, R.; Fischer, R.; Kogan, E.; et al. Activity-based chemical proteomics accelerates inhibitor development for deubiquitylating enzymes. Chem. Biol. 2011, 18, 1401–1412.
