Diabetic wWounds are severe injuries that are common in patients that suffer from diabetes. Most of the presently employed wound dressing scaffolds are inappropriate for treating dressing materials based on polymers have attracted much attention in the management of chronic wounds, especially diabetic woundinjuries. Improper treatment of diabetic wounds usually results in amputations. The shortcomings that are related to the currently used wound dressings include poor antimicrobial properties, inability to provide moisture, weak mechanical features, poor biodegradability, and biocompatibility, etc. To overcome the poor mechanical properties, polymer-based wound dressings have been designed from the combination of biopolymers (natural polymers) (e.g., chitosan, alginate, cellulose, chitin, gelatin, etc.) and synthetic polymers (e.g., poly (vinyl alcohol), poly (lactic-co-glycolic acid), polylactide, poly-glycolic acid, polyurethanes, etc.) to produce effective hybrid scaffolds for wound management.
1. Introduction
Diabetes is a chronic condition with a high incidence of mortality and numerous complications that include diabetic foot ulcers (DFU)
[1]. In 2013, it was reported that approximately 366 million individuals suffered from diabetes worldwide and in 2019, 1.5 million deaths were caused by diabetes
[2]. Diabetes is a medical condition due to the inability of the pancreas to produce sufficient insulin or the inability of the body to effectively use the insulin produced
[3]. Diabetic patients usually suffer from chronic injuries such as DFU and diabetic ulcers/leg ulcers. These wounds display features of a prolonged wound healing process and result in hospitalization and limb amputations
[4]. About 50–70% of limb amputations are caused by diabetic injuries, and it has been reported globally that one leg is amputated every 30 s because of diabetic wounds
[5,6][5][6]. Various factors result in delayed healing of diabetic wounds that are often taken into consideration by biomedical researchers, such as deformity, peripheral neuropathy, hanging on microcirculation function, macrovascular disease, peripheral arterial disease, cardiovascular events, kidney disease, and the disruption to growth factors (GFs) activity and expression
[7,8,9][7][8][9].
Wound dressing materials based on polymers have attracted much attention in the management of chronic wounds, especially diabetic injuries. These dressings demonstrate several interesting properties that can be beneficial for the management of chronic injuries. The properties of ideal polymeric dressings include high porosity and swelling ability, adequate water vapour transmission rate (WVTR), ability to offer moisture and warm environment to accelerate the wound healing process, gaseous permeation, excellent antimicrobial properties, excellent mechanical performance, and capability to deliver bioactive agents
[10,11,12][10][11][12]. Various polymers can be utilized for the formulation of ideal wound dressing materials. They are categorized as biopolymers and synthetic polymers. Examples of biopolymers (natural polymers) include alginate, dextran, hyaluronic acid (HA), chitosan, cellulose, gelatin, chitin, etc.
[13]. These polymers present excellent biomedical properties such as good biocompatibility, non-immunogenicity, non-toxicity, hemostatic effects, excellent biodegradability, antibacterial features, and wound healing properties
[14]. However, natural polymers also suffer from poor mechanical properties.
Natural-based polymeric wound dressing materials are usually cross-linked with synthetic polymers to enhance their mechanical performance
[14]. The synthetic polymers that can be cross-linked with natural polymers for wound dressing formulations include poly(vinyl alcohol) (PVA), poly(lactic-co-glycolic acid) (PLGA), polylactide (PLA), polyglycolic acid (PGA), polyurethanes (PUs), poly(ethylene oxide) (PEO)/poly(ethylene glycol) (PEG), poly(hydroxyethyl methacrylate) (PHEMA), and poly(vinyl pyrrolidone) (PVP)
[15]. The cross-linked dressings can also exhibit poor biological activities and weak wound healing properties, making them inappropriate for managing diabetic wounds. The encapsulation of bioactive agents in these polymer-based dressings has been reported to be a promising approach for wound care, precisely chronic wounds
[16]. The bioactive agents used in wound healing applications include antibiotics, GFs, stem cells, plant extracts, antioxidants, anti-inflammatory drugs (e.g., curcumin, etc.), and vitamins. Some polymeric wound dressings loaded with the above-mentioned bioactive agents are hydrogels, foams, membranes, films, nanofibers, transdermal patches, etc.
[17].
2. Polymer-Based Dressings Loaded with Bioactive Agents for Diabetic Wound Management
2.1. Nanofibers
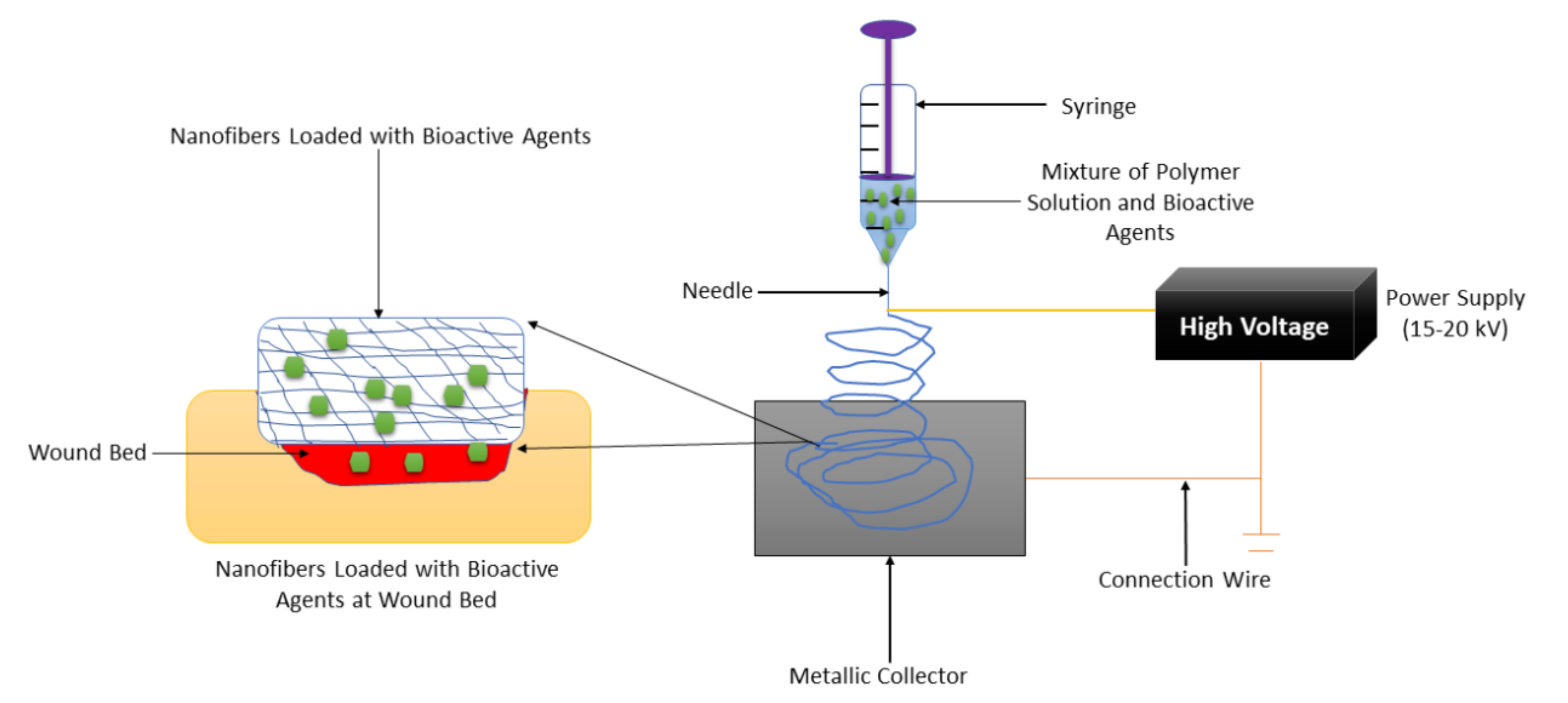
Nanofiber-based wound dressings have a mean diameter size of less than 1 micrometer
[50][18]. They are easily removed from the injury after their application. Among the preparation methods that are employed to fabricate nanofibers, electrospinning is the most employed one because of its several advantages such as adjustment of mechanical properties of nanofibers, control nanofiber porosity, cost-efficiency, simplicity, and versatility
[51,52,53][19][20][21]. The formulation of nanofibers using the electrospinning technique is shown in
Figure 31. Nanofibers present many advantages, including a small diameter, high porosity, narrow diameter distribution, gas permeation, and high-specific surface to area ratio
[54][22]. These wound dressings have been used for drug delivery, especially for the management of chronic injuries
[55][23]. They employ the accessories or excipients to transport the drugs to the wound with low toxicity and high efficiency. Nanofibrous wound dressings display related diameters with the extracellular matrix (ECM), making them suitable for improving wound healing and supporting cell proliferation and adhesion
[56][24]. Many research reports have discussed the therapeutic outcomes of nanofibers in diabetic wound management. Most of the nanofibers were formulated from poly (α-esters) (PLA, PGA, and PLGA), chitosan, gelatin, chitosan, HA, and alginate.
Figure 31.
Nanofiber formulation by the electrospinning technique.
Cam et al., fabricated bacterial cellulose-gelatin nanofibers co-loaded with glibenclamide and metformin for diabetic wound treatment
[57][25]. The scanning electron microscope (SEM) results of drug-encapsulated nanofibers exhibited bead-less and uniform structure (with a fiber diameter of 0.22 μm), mimicking that one of ECM. The cytotoxicity studies in vitro displayed high cell viability of mouse fibroblasts (L929) when they were incubated with plain nanofibers and glibenclamide/metformin-loaded nanofibers for 48 h, indicating non-toxicity and good biocompatibility of nanofibers
[57][25]. The encapsulation efficiencies of glibenclamide and metformin in hybrid nanofibers were ~78% and ~80%, respectively. The in vivo wound healing studies performed on drug-loaded fibers using type -1 diabetic Wistar rats displayed superior wound healing on full-thickness wounds than the pristine nanofibers, revealing that glibenclamide and metformin significantly accelerated diabetic wound healing
[57][25]. The co-loading of bioactive agents promoted an effective wound healing process. Choi et al., formulated PEG-PCL hybrid nanofibers encapsulated with human epidermal growth factor (EGF) using the electrospinning technique for the treatment of diabetic ulcers. The in vivo wound closure studies utilizing full-thickness injuries on streptozotocin (STZ)-induced diabetic mice demonstrated that the injury treated with EGF-encapsulated nanofibers was superior healed on day 7 than those dressed with plain nanofibers or EGF alone
[58][26]. The EGF-loaded significantly increased the rate of wound healing process resulting in complete wound closure in 7 days than the usual (14 or 15 days).
2.2. Films and Membranes
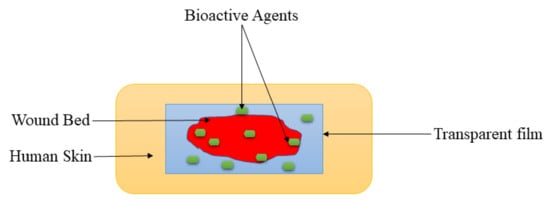
Films are wound dressing materials that are usually formulated adherent and transparent PU, which permits the permeation of gases such as oxygen, water vapor, and carbon dioxide between the injury and the surrounding
[73][27]. These dressing materials also useful for autolytic removal of dead tissues from the injury. The polymer-based films display excellent mechanical properties, including high elasticity and flexibility, leading to their ability to be altered to any shape of interest, and do not require extra tapping
[74][28]. The transparency of film dressings provides the inspection of the recovery process of the wound without removing the dressing (
Figure 42), making them appropriate for wound management of superficial wounds, epithelizing injury with low exudates, and shallow wounds
[75][29]. Tan et al., prepared sodium alginate-based hydrocolloid films incorporated with vicenin-2 for diabetic wound management
[76][30]. The in vivo wound closure studies employing diabetic STZ-induced SD rats showed that the lesions wrapped with vicenin-2-encapsulated films induced faster healing than those dressed with plain films. Also, the histological experiments indicated that pristine film-dressed diabetic wounds exhibited incomplete reepithelialization and poorly developed granulation tissue, while the vicenin-2 film-dressed diabetic rats showed moderate reepithelialization with well-developed granulation tissue after 2 weeks of treatment
[76][30]. The incorporation of vicenin into films significantly promoted the important processes (re-epithelization and granulation) of wound healing.
Figure 42.
Transparent film at the wound bed.
Colobatiu et al., reported chitosan-based films encapsulated with alcoholic extracts of various plants such as
Symphytum officinale, Plantago lanceolata, Tagetes patula, Arnica montana, Geum urbanum, and
Calendula officinalis for diabetic wound dressing application
[77][31]. These biopolymeric films displayed acceptable appearance, colour, structure, and flexibility as well as a good swelling ability, thus demonstrating a significant capability to prevent wound dehydration. The in vitro cytotoxicity experiments utilizing MTT assay displayed more than 80% cell viability of the Hs27 human fibroblast cells when incubated with bioactive extracts-loaded films, revealing good biocompatibility and non-toxicity. The in vivo experiments on diabetic STZ-induced Wister rats demonstrated that the injuries dressed with the bioactive-loaded films were observed to be almost fully closed (97.47%) on day 14, compared to the plain films that displayed only a 61.07% wound closure. Furthermore, histopathological analysis of chitosan-based films showed an important wound repairing ability, which could stimulate reepithelialization and hasten the wound healing mechanism in diabetic as well as normal wounds
[77][31]. The non-cytotoxic effects of films loaded with alcoholic extracts and other factors resulted in an improved diabetic wound healing process. The chitosan-based films were also formulated by Mizuno et al., that were loaded with fibroblast growth factors. The in vivo wound healing study showed high wound closure of full-thickness wounds on diabetic rats when treated with chitosan films when compared to control
[78][32]. The growth factors play a vital role in wound healing and accelerated the wound healing process.
2.3. Hydrogels
Hydrogels have attracted much attention in various biomedical applications in the past decades. They are 3-dimensional networks of cross-linked polymers (
Figure 53) which consist of more than 90% moisture content and are fabricated naturally, or through synthesis, via chemical or physical crosslinking methods
[96][33]. They have similarities with living tissues, adhesive nature, and they are malleable, and these characteristics make them considered as the best choice for wound dressing. Hydrogel dressings can accelerate the wound healing process since they can cool the wound through a gaseous exchange, reduce the pain by absorbing wound exudate, and preventing infections, and they can maintain a moist environment for cell migration. Furthermore, hydrogels can act as a delivery system that minimizes side effects and drug toxicity
[97,98,99][34][35][36]. There are several reports on the formulation of polymer-based bioactive hydrogels to improve the therapeutic effects of the currently used wound dressing materials to accelerate the wound healing process.
Figure 53.
Cross-linked hydrogel loaded with bioactive agents.
Wang et al., fabricated promising self-healing polypeptide-based hydrogel (denoted as FHE@exo hydrogel) with pH-responsive long-term exosomes release using Poly-ε-L-lysine (EPL), oxidative HA (OHA), and Pluronic (denoted as FHE hydrogel) by loading adipose mesenchymal stem cells (AMSCs)-derived exosomes through electrostatic interaction between EPL and exosomes
[100][37]. The in vivo studies of FHE@exo hydrogel, FHE hydrogel, and free exosomes were used with saline as a blank control demonstrated that all of them showed decreased diabetic wound size in all treated wounds within 14–21 days after surgery and FHE@exo hydrogel showed faster contraction rates with 88.67 ± 6.9% closure rate on day 14, compared to 36.3 ± 10.4% (saline), 64.3 ± 9.8% (FHE hydrogel) and 76.3 ± 3.2% (exosomes), respectively and at day 21 diabetic injuries treated with FHE@exo hydrogel were completely closed with remarkable hair growth
[100][37]. The loading of exosome into hydrogels significantly improve the wound healing process in vivo.
2.4. Foams and Wafers
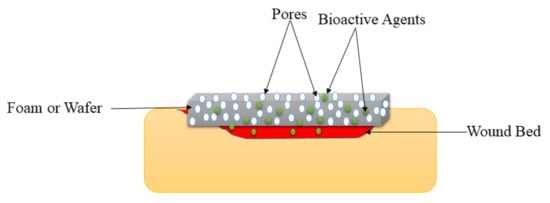
Foams are solid porous wound dressings (
Figure 64) that are made of hydrophobic and hydrophilic foam with bioadhesive boundaries
[42][38]. The external hydrophobic layer protects the injury from the liquid but allows gaseous exchange and water vapor permeation. These wound dressings can be sterilized and applied on injuries without resulting in pains to the patient if their parameters (such as mechanical properties, density, and thickness) are appropriately tailored. Foam wound dressings possess several advantages such as improved gaseous exchange, protect the wound from maceration, offer suitable moisture for the fast wound healing process, and absorb large amounts of exudate, making them appropriate for the management of burns, diabetic ulcers, traumatic wounds, etc.
[120][39]. The shortcoming of foam wound dressing materials is that they are inappropriate for dry wounds or injury with low exudates and dry scars
[121][40]. Pyun et al., formulated PU-based foams incorporated with recombinant human epidermal growth factor (rhEGF) for diabetic wound treatment. The FTIR spectrums confirmed the successful fabrication of the PU foam dressings. The water vapor transmission experiments of foams demonstrated a WVTR of about 3000 g/m
2/day, which is close to ideal wound dressings (2000–2500 g/m
2/day)
[122][41].
Figure 64.
Foam or wafer on the wound bed.
The cytotoxicity analysis in vitro exhibited very high cell proliferation and viability of CCD986-skin human fibroblast cell lines and HaCaT human keratinocyte when incubated with rhEGF-loaded foams, suggesting excellent biocompatibility of PU foams. The in vitro release profile displayed rapid release of rhEGF from the surface of foams in the first 24 h, followed by plateau release for 7 days. The in vivo studies using STZ induced diabetic SD rats showed that the full-thickness wounds were almost completely closed by more than 97% when treated with rhEGF-loaded foams. The histological analysis demonstrated that the diabetic wounds were completely resolved by regenerating the epithelial cell in the rats on day 21 after wounding
[122][41]. The moderate WVTR and release profile promoted enhanced healing of the diabetic wounds by inducing epithelial cell regeneration. Coutts et al., conducted clinical studies of PVA foam wound dressings co-loaded with gentian violet and methylene blue for bacterial-infected diabetic wounds
[123][42]. The outcomes of these studies presented enhancements in surface critical colonization and pain score at the end of the assessment period in some patients, especially in patients with DFUs. Furthermore, decreasing wound size was observed in 8 of the 14 patients at week 4
[123][42]. The other clinical studies reported by Moon et al., demonstrated that the wounds in diabetic patients dressed with Ag-incorporated PU foams were restored in 15.6 ± 3.8 days while those treated with plain foams healed in 14.4 ± 2.2 days, revealing that the presence of silver in the foams delayed the epithelialization of the diabetic injuries in patients. However, the difference was statistically significant in this study
[124][43].
Choi et al., fabricated PU foams loaded with Ag nanoparticles and rhEGF for bacteria-infected diabetic wound management
[125][44]. These foam wound dressings significantly demonstrated fluid retention, excellent absorbency, and fluid handling features. SEM micrographs exhibited that the PU foams demonstrated a relatively uniform pore size that ranges between 200–400 µm and it was not affected by the incorporation of bioactive agents, suggesting that these foams can provide high cell granulation rate and proliferation with an excellent gaseous exchange during wound healing. The in vitro cytotoxicity analysis utilizing MTT assay exhibited the high cell viability of L929 mouse fibroblasts when cultured with dual bioactive agent-loaded foams. The antimicrobial analysis using the inhibition zone method displayed that the PU foams loaded with Ag nanoparticles and rhEGF exhibited outstanding antibacterial efficacy (high inhibition zone) against
E. coli and
S. aureus, while unloaded foams did not display any inhibition effects. The in vivo experiments utilizing diabetic Balb/b mice demonstrated that injuries wrapped with the foams loaded with both Ag nanoparticles and rhEGF demonstrated excellent healing after 5 days of treatment than the gauze, suggesting a synergistic effect of incorporating bioactive agents together with growth factors
[125][44].
2.5. Sponges and Bandages
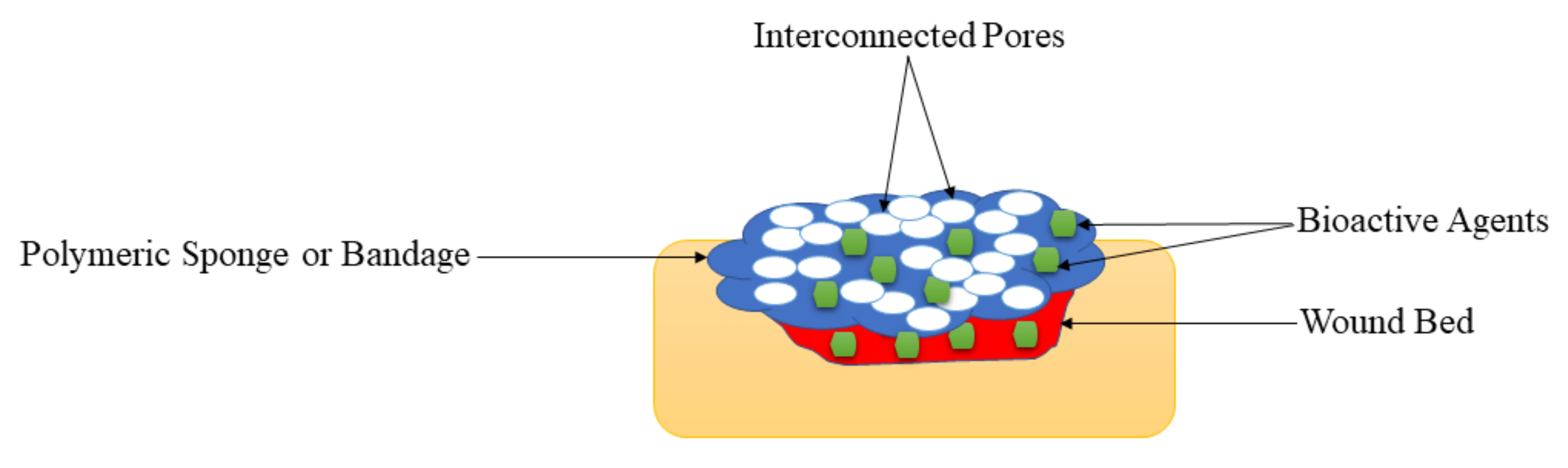
Sponges are wound dressings that possess interconnected porous structures (
Figure 75), soft and flexible
[135][45]. Their porous structure influences their high swelling capacity, making them appropriate for the management of exuding wounds. They also support cell migration and high water absorption capability appropriate for providing moisture to the wound bed while protecting the injury from bacterial infections
[136][46]. The sponge wound dressings formulated from PVA, alginate, chitosan, and graphene oxide demonstrated excellent antimicrobial efficacy
[137][47]. Several sponges have been prepared for the delivery of therapeutic agents for the treatment of diabetic wounds. Wang et al., formulated chitosan-cross-linked collagen sponges encapsulated with recombinant human acidic fibroblast growth factors to stimulate the diabetic wound healing process
[138][48]. These hybrid sponges exhibit several advantages required in an ideal wound dressing, such as uniform and porous ultrastructure, in vitro slow release of fibroblast GFs from the sponges, and high resistance to collagenase digestion
[138][48]. The remedial impact of the novel wound dressing comprising fibroblast growth factors on diabetic wound healing was studied in a type 1 diabetic rat model in which hyperglycemia was prompted by a single dosage of STZ and continued for a very long time. The diabetic wound healing was discovered to be significantly enhanced by chitosan-cross-linked collagen sponges loaded with fibroblast growth factor compared to the pristine sponges, revealing the capability of growth factor-loaded chitosan-cross linked collagen sponges wound dressings for diabetic wound healing
[138][48]. The presence of fibroblast growth factors and their sustained drug release mechanism from sponges significantly resulted in accelerated wound healing process.
Figure 75.
Sponges on the wound bed.
The polymer-based sponges and bandages loaded with therapeutic agents displayed high porosity that can promote high cell growth and attachment, which are suitable for the diabetic wound healing process. Also, the initial rapid drug release of bioactive agents from these scaffolds followed by sustained release is appropriate for improving the treatment of diabetic wounds (
Table 1 and
Table 2). Most of the reported sponges and bandages for the treatment of diabetic injuries were loaded with antibacterial agents (metallic nanoparticles and antibiotics) and they demonstrated excellent antibacterial activity against various antibiotic-resistant bacterial strains, suggesting that these are potential materials for the management of infected diabetic wounds. The encapsulation of bioactive agents into the sponges and bandages significantly accelerated the wound healing process of the diabetic injuries in vivo when compared with diabetic wounds dressed with plain scaffolds. Nevertheless, the very high porosity of polymeric sponges or bandages can result in high uptake of wound exudate and high WVTR that may cause dehydration of diabetic wounds. A dehydrated injury can lead to a delayed wound healing process.
Table 1.
Summary of polymer-based wound dressings loaded with bioactive agents for diabetic wounds.
| Types of Wound Dressing |
Used Polymers |
Loaded Bioactive Agents |
Results |
Ref |
Table 2.
Comparison of various types of wound dressings.
| Types of Wound Dressings |
Advantages |
Disadvantages |
Highlights |
| Nanofiber |
Gelatin and cellulose |
Glybenclamide and metformin |
Accelerated wound healing process and good biocompatibility |
[57] | [25] |
| Nanofiber |
PEG and PCL |
EGF |
Superior wound healing process |
The mechanical properties of films and membranes were like those of human skin, making them skin compatible and easily handled during diabetic wound management. | [58] | [26] |
| Nanofiber |
Polylactide |
Doxycycline |
Excellent mechanical performance, antibacterial effects, and excellent diabetic wound healing properties |
[59] | [49] |
| Hydrogels |
They are used as potential drug delivery systems in wound dressing applications and display other interesting properties such as high porosity, high swelling capacity, excellent biocompatibility, etc. |
The biopolymer hydrogel dressings demonstrate poor mechanical performance that makes them not compatible with the human skin. |
The drug release profiles were controlled release of bioactive agents from the polymeric hydrogels, resulting in an improved wound healing process. The high porosity of the hydrogels led to good swelling capability. |
Nanofiber |
PCL and gum tragacanth |
Curcumin |
Bead-free morphology and full wound closure on day 15. |
[60] | [50] |
| Foams and Wafers |
These wound dressings exhibit high porosity that could provide cell growth and adhesion to accelerate the wound healing process. |
They are not suitable for dry wounds. |
The WVTR experiments of foams and wafers loaded with drugs exhibited moderate WTVR that can provide appropriate moisture to accelerate the healing of diabetic wounds. |
Nanofiber |
PU and carboxymethylcellulose |
Malva sylvestris | plant extract |
Good diabetic wound healing rate |
[61] | [51] |
| Nanofiber |
Hydroxypropyl methylcellulose and PEO |
beta-glucan |
Non-toxic and accelerated wound closure. |
[62] | [52] |
| Nanofiber |
poly-N-acetyl glucosamine |
polydeoxyribonucleotide |
High rate of cell proliferation and angiogenesis. |
[63] | [53] |
| Nanofiber |
polyethersulfone |
henceforth CD34+ cells |
The fast diabetic wound healing process |
[64] | [54] |
| Nanofiber |
PCL |
Bixin |
Sustained drug release and accelerated wound healing. |
[65] | [55] |
| Sponges and Bandages |
These wound dressings are also displayed high porosity that could offer suitable gaseous permeation, superior cell proliferation, migration, and attachment for the accelerated wound healing process. |
The very high porosity of the polymeric sponges or bandages can result in high uptake of wound exudate and high WVTR that may cause wound dehydration. |
Nanofiber |
PLGA |
PDGF, vancomycin, and gentamicin |
Sustained drug release and accelerated wound healing. |
[66] | [56] |
| Nanofiber |
PCL |
Sodium percarbonate |
Superior vascularization. |
[67] | [57] |
| Nanofiber |
Cellulose acetate |
Ag nanoparticles |
High antibacterial efficacy and accelerated diabetic wound contraction. |
[68] | [58] |
| Nanofiber |
PCL |
Curcumin |
Excellent biocompatibility and increased rate of wound reduction |
[69] | [59] |
| Nanofiber |
PLGA |
Insulin |
Good mechanical performance and prolong drug release |
[70] | [60] |
| Nanofibers |
Chitosan and PVA |
ZnO |
Excellent antibacterial effects and accelerated diabetic wounds |
[71] | [61] |
| Nanofibers |
PVP and PCL |
Pioglitazone |
Non-toxicity and sustained drug release. |
[72] | [62] |
| Film |
Sodium alginate |
Vicenin-2 |
Faster diabetic wound recovery |
[76] | [30] |
| Film |
Chitosan |
Alcoholic extracts |
Excellent biocompatibility |
[77] | [31] |
| Film |
Chitosan |
Fibroblast growth factors |
High diabetic wound contraction rate |
[78] | [32] |
| Film |
Cellulose and PVA |
Propolis and vitamin C |
High swelling rate, controlled drug release, and accelerated diabetic wound healing |
[79] | [63] |
| Film |
Fibroin |
Aloe gel |
Excellent mechanical properties and fibroblast distribution and collagen fiber organization. |
[80] | [64] |
| Film |
Fibroin and chitosan |
ADSCs |
Good diabetic wound closure. |
[81] | [65] |
| Film |
PVA and cellulose |
Curcumin |
Good antibacterial effects and significantly diabetic wound closure. |
[82] | [66] |
| Polymeric sponges and bandages were mostly loaded with antibacterial agents for diabetic wound treatment, and they exhibited excellent antibacterial activity, demonstrating that these dressings are potential candidates for the management of infected diabetic wounds |
Film |
Chitosan |
Retinoic acid |
Increased wound reduction rate. |
[83] | [67] |
| Film |
Collagen |
Biotinylated GHK peptide |
Accelerated wound healing |
[84] | [68] |
| Film |
PVP and PVA |
Sodium fusidate |
Excellent mechanical performance |
[85] | [69] |
| Film |
Cellulose |
Selenium |
Fast diabetic wound healing rate |
[86] | [70] |
| Membrane |
PHBV |
Cerium Oxide nanoparticles |
Significant enhancement in cell infiltration and granulation tissue formation |
[88] | [71] |
| Membrane |
PVA and PLA |
GFs |
Excellent cell migration and proliferation |
[89] | [72] |
| Membrane |
HA |
Human keratinocytes |
The good clinical wound healing process |
[90] | [73] |
| Membrane |
PLGA and collagen |
Glucophage |
The faster wound healing process |
[91] | [74] |
| Membrane |
PLGA |
Metformin |
Enhanced the wound healing and re-epithelialization in diabetic rats |
[92] | [75] |
| Membrane |
PLLA |
Dimethyloxalylglycine |
Burst drug released followed by sustained drug release. |
[93] | [76] |
| Membrane |
Cellulose acetate |
Sesamol |
Improved diabetic wound healing |
[94] | [77] |
| Membrane |
PLGA and cellulose |
Neurotensin |
Sustained drug release and faster wound healing process |
[95] | [78] |
| Hydrogel |
Poly-ε-L-lysine, HA, and pluronic |
Adipose mesenchymal stem cells |
Increased diabetic wound rate |
[100] | [37] |
| Hydrogel |
HA and PEG |
Stem cell |
Good mechanical properties and faster diabetic wound healing. |
[101] | [79] |
| Hydrogel |
PEG and PVA |
Fibroblasts and insulin |
Accelerated wound repair |
[102] | [80] |
| Hydrogel |
HA |
Human adipose stem cells |
Improved wound closure rate |
[103] | [81] |
| Hydrogel |
Gelatin |
Chemotactic cytokines |
Accelerated wound healing |
[104] | [82] |
| Hydrogel |
Sodium carboxymethylcellulose |
B. orientale |
Fast wound recovery |
[105] | [83] |
| Hydrogel |
Pluronic F-127 |
ADSCs |
Accelerated wound healing |
[106] | [84] |
| Hydrogel |
PU |
AASCs |
Fast diabetic wound |
[107] | [85] |
| Hydrogel |
Chitosan |
Exosomes |
Accelerate angiogenesis and wound surface re-epithelialization |
[108] | [86] |
| Hydrogel |
PPCN |
SDF-1 |
Improved epithelial maturation and granulation tissue production |
[109] | [87] |
| Hydrogel |
Konjac glucomannan |
Avena sativa |
Support collagen expression, keratinocyte migration, fibroblast attachment, and proliferation |
[110] | [88] |
| Hydrogel |
Chitosan |
L-glutamic acid |
Promotes collagen deposition and accelerates vascularization |
[111] | [89] |
| Hydrogel |
Gelatin |
Curcumin |
Good cell migration |
[112] | [90] |
| Hydrogel |
Chitosan and PEG |
Ag nanoparticles |
Controlled drug release and diabetic wound stimulation. |
[113] | [91] |
| Hydrogel |
poly-(polyethyleneglycol citrate-co-N-isopropylacrylamide) |
Copper metal-organic framework |
Enhanced dermal cell migration and improved wound closure rates |
[114] | [92] |
| Hydrogel |
PVA |
Nitric Oxide |
Enhance diabetic wound healing |
[115] | [93] |
| Hydrogel |
HA |
DNA |
Enhanced development of granulation tissue |
[116] | [94] |
| Hydrogel |
poly (γ-glutamic acid) and chitosan |
Superoxide dismutase |
Good cytocompatibility and accelerated wound healing process |
[117] | [95] |
| Hydrogel |
Gelatin |
Cerium-containing bioactive glass nanoparticles |
Good antibacterial effects |
[118] | [96] |
| Hydrogel |
chitosan-dextran |
| Nanofibers |
They possess a structure that mimics ECM, making them suitable for skin wound healing and regeneration. They are frequently formulated using efficient and easily employed electrospinning techniques. |
| Ag nanoparticles |
| Broad-spectrum and long-lasting antibacterial activity |
| [ |
| 119 |
| ] |
| [ |
| 97 |
| ] |
| It is not easy to produce nanofibers less than 10 nm in diameter. |
| Foam |
| PU |
| RhEGF |
| Moderate WVTR and good biocompatibility |
| [ |
| 122 |
| ] |
| [ |
| 41 |
| ] |
| The SEM micrographs of nanofibers loaded with bioactive agents display bead-free morphology that mimics ECM, making these wound dressings appropriate for providing an environment for cell proliferation and adhesion to accelerate the diabetic wound healing process. |
| Foam |
| PVA |
| Gentian violet and methylene blue |
| High wound reduction rate |
| [ |
| 123 |
| ] |
| [ |
| 42 |
| ] |
| Foam |
| PU |
| Ag nanoparticle |
| Fast wound healing rate |
| [ |
| 124 | ] | [ | 43 | ] |
| Foam |
PU |
Ag nanoparticle |
Good antibacterial efficacy |
[125] | [44] |
| Foam |
PU |
Ag |
Good diabetic wound closure |
[126] | [98] |
| Foam |
Silk fibroin |
Gastrodia elata and tea tree oil |
High porosity and excellent biocompatibility |
[127] | [99] |
| Foam |
Chitosan |
Neurotensin |
High wound healing reduction |
[128] | [100] |
| Foam |
Silicone |
Silver |
Positive diabetic wound closure and reduction in size | [129] | [101] |
| Wafer |
Calcium alginate |
Ciprofloxacin |
High porosity and burst drug release followed the sustained release with good antibacterial efficacy |
[132] | [102] |
| Wafer |
Xanthan gum |
Silymarin |
Good cell migration |
[133] | [103] |
| Wafer |
Sodium alginate and gelatin |
Diosmin nanocrystals |
Sustained drug release and well-developed granulation tissue, well-organized dermal layers, complete re-epithelialization, and mature collagen bundles in diabetic wounds. |
[134] | [104] |
| Sponges |
Chitosan and collagen |
Recombinant human acidic fibroblast growth factors |
Enhanced diabetic wound healing |
[138] | [48] |
| Sponge |
HA and chitosan |
Ag nanoparticle |
Good antibacterial effects and good cytocompatibility |
[139] | [105] |
| Sponge |
Chitosan |
TMC nanoparticles |
Faster diabetic wound healing |
[140] | [106] |
| Sponge |
Chitosan and HA |
VEGFs |
Burst release of GFs followed by sustained release. |
[141] | [107] |
| Sponge |
HA and collagen |
EGF |
Promoted blood vascular formation and granulation tissue development. |
[142] | [108] |
| Sponge |
Chitosan and silk |
GMSC-derived exosomes |
Enhanced deposition, re-epithelialization, and remodeling of ECM |
[144] | [109] |
| Sponge |
Collagen |
Gementacin |
Good pathogen eradication in diabetic wound |
[143] | [110] |
| Sponge |
Chitosan and alginate |
Curcumin and honey |
Sustained drug release and faster wound healing |
[145] | [111] |
| Sponge |
Chitosan and collagen |
Thymosin beta 4 |
Enhanced diabetic cutaneous wound healing |
[146] | [112] |
| Bandages |
Chitosan |
ZnO nanoparticles |
Good cytocompatibility and antibacterial effects. |
[147] | [113] |
| Bandage |
Chitin |
ZnO nanoparticles | ] | [114] |
| Bandage |
| Films and Membranes |
These wound dressings are transparent, showing that the wound healing process can be observed without removing them. They also display good mechanical performance. |
excellent antibacterial activity and high cell adhesion and migration |
Sodium alginate |
EGF and curcumin |
Non-toxicity and good biocompatibility |
[148] | [115] |
| They are not suitable for exuding wounds due to their inability to absorb a large volume of biological fluids. |
[ | 149 |
Bandage |
Chitosan |
Fluconazole and ciprofloxacin |
High porosity and sustained drug release with good antimicrobial effects |
[150] | [116] |