It is generally believed that more than 700 Conus species have evolved during the last 50 million years. With the highest species abundance occurring in southeast Asia, most Conus can be found in the shallow waters of tropical and subtropical oceans. Conus is widely distributed in rocky shores, sandy beaches, coral reefs and intertidal waters, with depths reaching up to over 600 m. Nowadays, Conus species are generally overexploited, and some species are now endangered. Exploring these waters of potentially high species diversity could enrich our understanding of their population’s genetic structure and provide the missing pieces for clarifying Conus evolution. As the conotoxin compounds vary greatly throughout the growth stages and across geolocations within the same species, further investigation of these species-specific regional distribution differences may provide crucial insights for artificial breeding and harvesting specific bioactive compounds in the future.
1. Introduction
It is generally believed that more than 700
Conus species have evolved during the last 50 million years
[1,2][1][2]. With the highest species abundance occurring in southeast Asia, most
Conus can be found in the shallow waters of tropical and subtropical oceans
[3].
Conus is widely distributed in rocky shores, sandy beaches, coral reefs and intertidal waters, with depths reaching up to over 600 m
[4]. However, geophysical environment is not the only factor that influences the
Conus species distribution. For example, despite similar oceanic and climate conditions, more than 77
Conus species have been discovered in Indian coastal waters, especially in the Tamil Nadu Coast and the Andaman Islands
[4[4][5][6],
5,6], with fewer than 20 species reported on the eastern side of the Andaman Sea
[7]. Nowadays,
Conus species are generally overexploited, and some species are now endangered.
Exploring these waters of potentially high species diversity could enrich our understanding of their population’s genetic structure and provide the missing pieces for clarifying Conus evolution. As the conotoxin compounds vary greatly throughout the growth stages and across geolocations within the same species, further investigation of these species-specific regional distribution differences may provide crucial insights for artificial breeding and harvesting specific bioactive compounds in the future.
With intraspecies variation of the venom cocktails, a huge array of remarkably diverse conopeptides, ranging from 50,000 to 140,000 venom peptides, are estimated to be produced in the
Conus genus
[8]. With a unique repertoire of 100–200 venom peptides for each species,
Conus could presumably specifically target a wide range of vertebrate and invertebrate physiological receptors, assisting their prey hunting or defense from potential predators and competitors
[9]. Different prey targets come along with different venom recipes. Yet, despite the species diversity of
Conus, their feeding habits can be categorized into three main types, often with associated differences in the radula morphology
[6,10][6][10]. The radula of the vermivorous (V)
Conus usually have two barbs or the second barb replaced by a blade; spur and serration are present, with or without waist. Spur is absent in the molluscivous (M) radula; serrations are longer and terminate at a prominent cusp. The radula of the piscivorous (P)
Conus has three barbs, with a recurved tip on the third barb with no serration, waist, cusp or spur
[6]. Exceptionally, in some vermivore species, the radula was found to be similar to the molluscivous radula, indicating a more complex relationship between the radula morphology and feeding habit
[11].
Alongside radula changes,
Conus also developed complex venom compositions and envenom system. The venom gland of the sea snails can be divided into three sections: bulb, duct and radular sac
[12]. To hunt and defend themselves, each
Conus species can produce a cocktail of conotoxins (reaching hundreds of small polypeptides with very complex post-transcriptional modifications, alongside some paralytic small molecules) in their venom glands and inject the precise amount of venom cocktail through the duct anchored on its prey by the harpoon
[13,14,15][13][14][15]. The prey will be instantly paralyzed through rapid stabbing with such an efficient venom cocktail
[16]. Previous studies also proposed that modern
Conus species can produce venom peptides with different functions at different parts of the venom gland, even for different parts of the venom duct. For example, the distinct defense cabal was identified from bulb-proximal and predation-evoked cabal from the bulb-distal end of the specialized venom ducts
[17,18][17][18]. The diverse conotoxins allow
Conus to precisely adjust their cocktail recipes for each attack (hunting, defending or threatening competitors)
[19] or become specialized in a local prey according to geographic variation
[20]. Recent studies further illustrated that the defensive compositions of venom cocktails of the worm-hunting species may be the driving factor for the emergences of fish- and mollusc-hunting species
[21,22][21][22]. Consequently, conotoxin compositions are also expected to vary from lab culture to natural conditions.
In general, the small polypeptides (mostly 10 to 40 amino acid residues) are rich in disulphide bonds and act on the nervous system of the target organism, interfering with signal transmission in cells and neurons
[23]. Specific types of cysteine residues and disulphide bond arrangements have been found in different gene superfamilies, with each venom gene superfamily usually having unique corresponding binding sites from different pharmacological target families (
Table 1)
[3,24][3][24]. The targets of conotoxins include: (i) presynaptic membrane calcium channel or G protein receptor, (ii) voltage-gated potassium channel, (iii) adrenal hormone receptor, (iv) serotonin receptor, (v) somatostatin receptor, (vi) norepinephrine receptor, (vii) voltage-gated calcium ion channels and (viii) other targets
[25,26][25][26]. These receptors are ubiquitous when it comes to drug research. Conopeptides, with their abundant diversity, novel chemical structure, biological sensitivity and target selectiveness, are of great potential as drug precursors, as well as an important neuroscience tool
[27].
Table 1. Conopeptides gene superfamilies, cysteine framework and their pharmacological families [3,28,29]. Conopeptides gene superfamilies, cysteine framework and their pharmacological families [3][28][29].
| Conotoxin Family |
Definition |
Gene Superfamily |
Cysteine Framework |
| α (ALPHA) |
Nicotinic acetylcholine receptor (nAChR) |
A, B3, D, J, L, M, S |
I, II, III, IV, VIII, XIV, XX, XXV |
| γ (GAMMA) |
Neuronal pacemaker cation currents (inward cation current) |
O1, O2 |
VI/VII |
| δ (DELTA) |
Voltage-gated Na Channel (agonist, delay inactivation) |
O1 |
VI/VII |
| ε (EPISILON) |
Presynaptic calcium channels or G protein-coupled presynaptic receptor |
T |
V |
| I (IOTA) |
Voltage-gated Na Channel (agonist, no delayed inactivation) |
I1, M |
III, XI |
| κ (KAPPA) |
Voltage-gated K Channel (blocker) |
A, I2, J, M, O1 |
III, IV, VI/VII, XI, XIV |
| μ (MU) |
Voltage-gated Na Channel (antagonist, blocker) |
M, O1,T |
III, IV, V, VI, VII |
| ρ (RHO) |
Alpha 1 adrenoreceptor (GPCR) |
A |
I |
| σ (SIGMA) |
Serotonin-gated ion channels (GPCR) |
S |
VIII |
| τ (TAU) |
Somatostatin receptor |
T |
V |
| χ (CHI) |
Neuronal noradrenaline transporter |
T |
X |
| ω (OMEGA) |
Voltage-gated calcium channel |
O1, O2 |
VI/VII, XVI, XXVI |
Additionally, nerve system agents and anticancer drugs are research hotspots, and conotoxins are drawing more attention after the entry of Ziconotide (Prialt
®) on the market
[30]. Because the physiological system of fish is more similar to other vertebrates, such as human
[31], in comparison to benthic organisms, such as worms and mollusks, many studies focus exclusively on the piscivorous species and their venom production. To better categorize the complex conopeptides, the primary structure of 222 conotoxins has been identified and classified into several gene superfamilies, according to precursor signal peptides and amino acid sequences
[25,32][25][32]. Apart from their genetic diversity, the precursor RNA splicing process contributes greatly to the conotoxin diversity. With just over 30 gene superfamilies, more than 7000 conopeptides have been discovered
[33,34,35][33][34][35]. Not all of these peptides, however, are of equal importance. Compounds from A, M, O1 and T gene superfamilies account for the bulk of venom cocktails. Moreover, the expression of some conopeptides is so low that their presence cannot even be detected in traditional proteomic experiments
[18,36][18][36]. Perhaps it is the seemingly redundant venom compounds that are providing abundant raw materials for natural selection and contributing to the rapid speciation and quick adaptation of the
Conus. Therefore, to understand the rich proteomic and toxicological resources in
Conus, genomic and transcriptomic approaches are required to assist conotoxin identification and classification
[37], as they are more sensitive and accurate for capturing the trace amount of conopeptides. Hence, the biggest obstacles now hindering the use of multi-omic approaches in
Conus studies are data analysis and the scarcity of well-annotated genomes.
2. Feeding Habits and Evolution Path
Apart from the 190 species from three other genera (1 from
Californiconus, 159 from
Conasprella and 30 from
Profundiconus) in the Conidae family, there are currently 728 species recognized in the
Conus genus, making it one of the most diversified species in the Conoidea superfamily (
Supplementary Table S1, could be found in https://www.mdpi.com/1660-3397/20/2/105#supplementary). In this study, the feeding habits of known species are categorized based on radula morphology, field reports and dissection of digestive chambers. Only 506 species in the Conidae family were classified into the staple feeding habits. For
Conus, it is obvious that worm eating is the dominant feeding behavior, accounting for more than 72% of total species diversity, including most basal species, and thus likely to have first emerged in their evolutionary history. This trend was followed by the most recent fish-eating group (20%) and molluscivore group (12%). Interestingly, several polyphagous species have also been found scattered among the three monophagous groups, indicating a more complex hunting behavior of the
Conus species in natural environments.
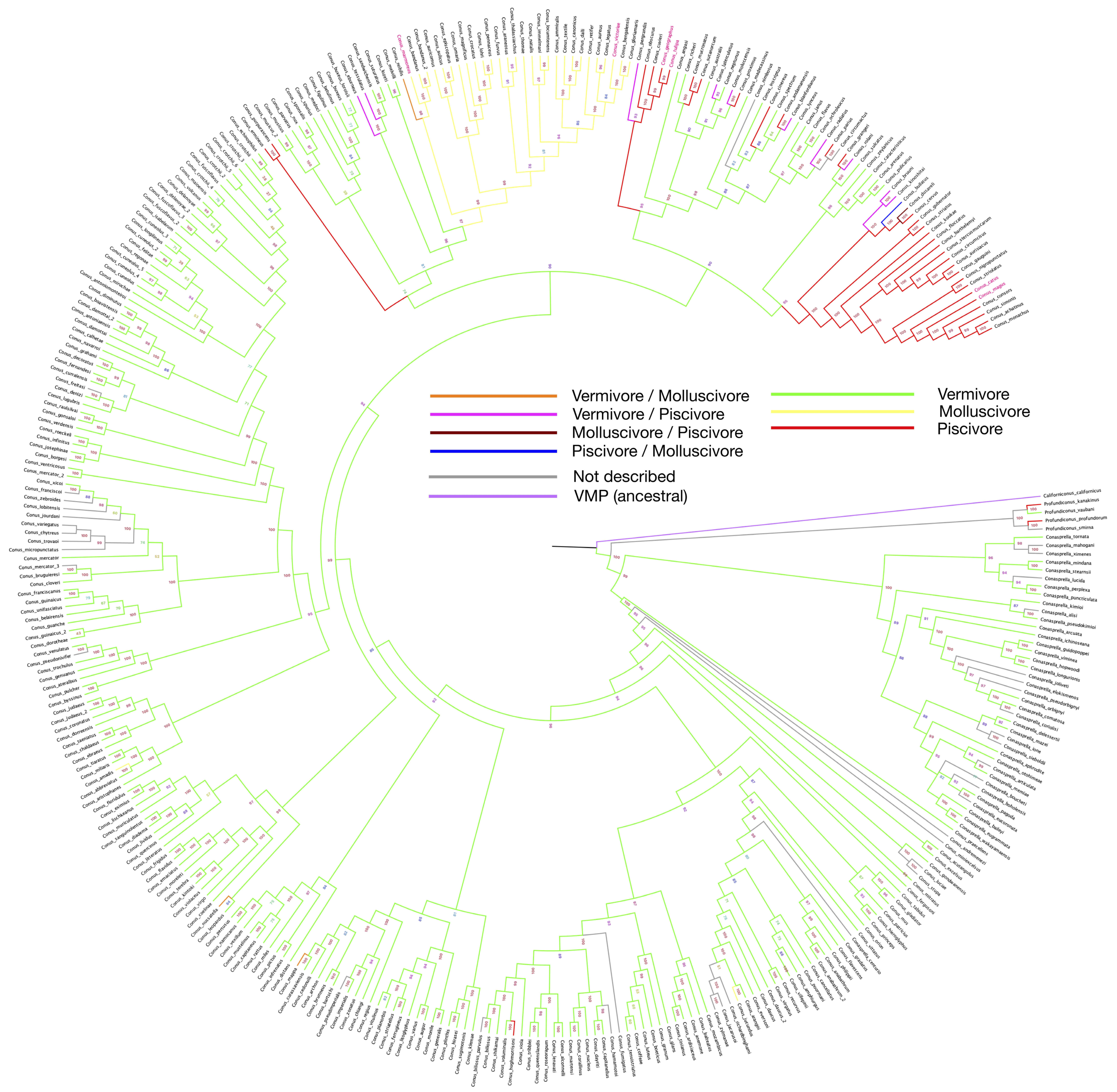
The phylogeny tree constructed with barcode sequences (12S, 16S and COI) is consistent with previous studies, and
wresearche
rs further highlighted the feeding habits using a color-coded pattern across the various species studied here with the same feeding habits (
Figure 1). As shown in the phylogenetic tree, following the vermivorous group, the first batch of fish-hunting species appeared in the Neogene period (around 23 million years ago)
[48][38]. Unlike the cluster of molluscivorous
Conus species that share one collective root on the phylogenetic tree, the emergence of the piscivores seems to be independent and recurrent along with speciation.
Figure 1. Phylogeny tree of 335
Conus species, constructed by using barcode sequences (12S, 16S and COI). Branch colors are set according to the feeding habit from
Supplementary Table S1. Bootstrap values are presented by the numbers next to each branch. Species with feeding habits found have been assigned with branch colors accordingly. Names of species that have been involved in drug development are labeled in red.
According to the latest taxonomy update from the WoRMS website, several species have been categorized into the same species, as indicated by trailers in the graph (
Figure 1). As shown in the tree, polyphagy is most likely to occur on restricted branches, where fish-hunting species have secondarily recurrently emerged from the worm-eating species. As a true omnivore,
Californiconus californicus not only feeds on marine worms, fish and mollusks; it is also a scavenger. Positioned on the proximal branch of the phylogenetic root, the mentioned
Californiconus californicus is considered as an ancestral state species to others in the family. Therefore, it is interesting to see that the majority of
Conus species become specialized in only one particular prey category, which makes the few polyphagous species outliers, or more interestingly, transitive species.
Many studies have suggested that the venom cocktail composition for each preference is highly distinguishable. The venoms of fish-hunting species are well selected to target vertebrate nervous system, in which receptors are very different from invertebrates. If we look at
Conus alone, vermivores are obviously plesiomorphic, and molluscivores and piscivores originate from them. Furthermore, the distinguishable evolutionary patterns between the fish- and mollusc-hunting species indicate that the determining factors for different feeding habits may be separately located in the genomes, where gene silencing or gene lost mechanism could play an important role. Otherwise, it would be difficult to explain the seemingly random occurrences of the fish-hunting species.
However, there are also cases of one
Conus species hunting various preys, or even some individual showing omnivorous behavior. This suggests that
Conus feeding habit is not monophagous and far more complex than simple hunting/defense venom recipes. Indeed, other factors, such as environmental pressure and prey abundance, could also play important roles in venom secretion.
Conus evolution is subject to both convergence and divergence. Different groups in
Conus could share similar evolutionary patterns due to comparable selection pressure in a confined geographical region. Previous molecular phylogenetic analysis has confirmed that
Conus species constitute a largely heterogeneous group, despite their overall morphological homogeneity
[49][39]. This may explain that, although the selective pressure on the venom is acting in different directions, vermivorous species, which are close to the fish-hunting clade, show great potential in accepting both worm and fish as prey.
On the phylogeny tree, some vermivorous sister species exhibit a fish-eating behavior or can also feed on other molluscs, suggesting a loose hunting strategy. Given that the roots of most divergent species are very recent, it indicates an ongoing adaptive process of some
Conus clusters
[50][40]. This phenomenon could be caused by both genetic plasticity and environmental pressure. It is also intriguing that fish-hunting and mollusc-hunting strategies were both developed at the same period of geological time, and the two clusters are genetically close to each other.
The origin of the Conidae family dates back to around 59 (73–55) million years ago, and the radiation of some analyzed species within the
Conus genus was estimated to have occurred between 24–15 (30–12) million years ago
[51][41]. The explosive fish speciation movement
[52][42] in this period (Neogene) became the ultimate driving force and selection pressure for some clades of
Conus. The emerge of massive Pleistomollusca species
[53][43] occupied ecological niches and stiffened the competition, potentially leading to the clade of molluscivores.
However, questions remain on the
Conus feeding habits. Normally, most specimens were dissected right after being collected for further multi-omics studies, and stomach contents and radula shapes were kept for feeding habit analysis. Only a small number of individual species have been kept for feeding behavior studies. The phylogeny and feeding habit results described in this work suggest a more complicated relationship between feeding habits and morphology.
Replicates are rare for most transcriptome datasets online, and gene expression can be influenced by environmental conditions, individual sample difference and other batch effects, making it challenging to compare datasets from various projects. Therefore, designed experiments should be carried out to retrieve precise transcriptome data for different tissues, and most importantly, the venom glands. Further studies could also explore venom cocktail composition difference induced by different pressures, such as constant defensive pressure, and how
Conus would cope with the pressure when food options are limited, especially for the polyphagous clades labeled on the phylogenetic tree (
Figure 1).
3. Distribution
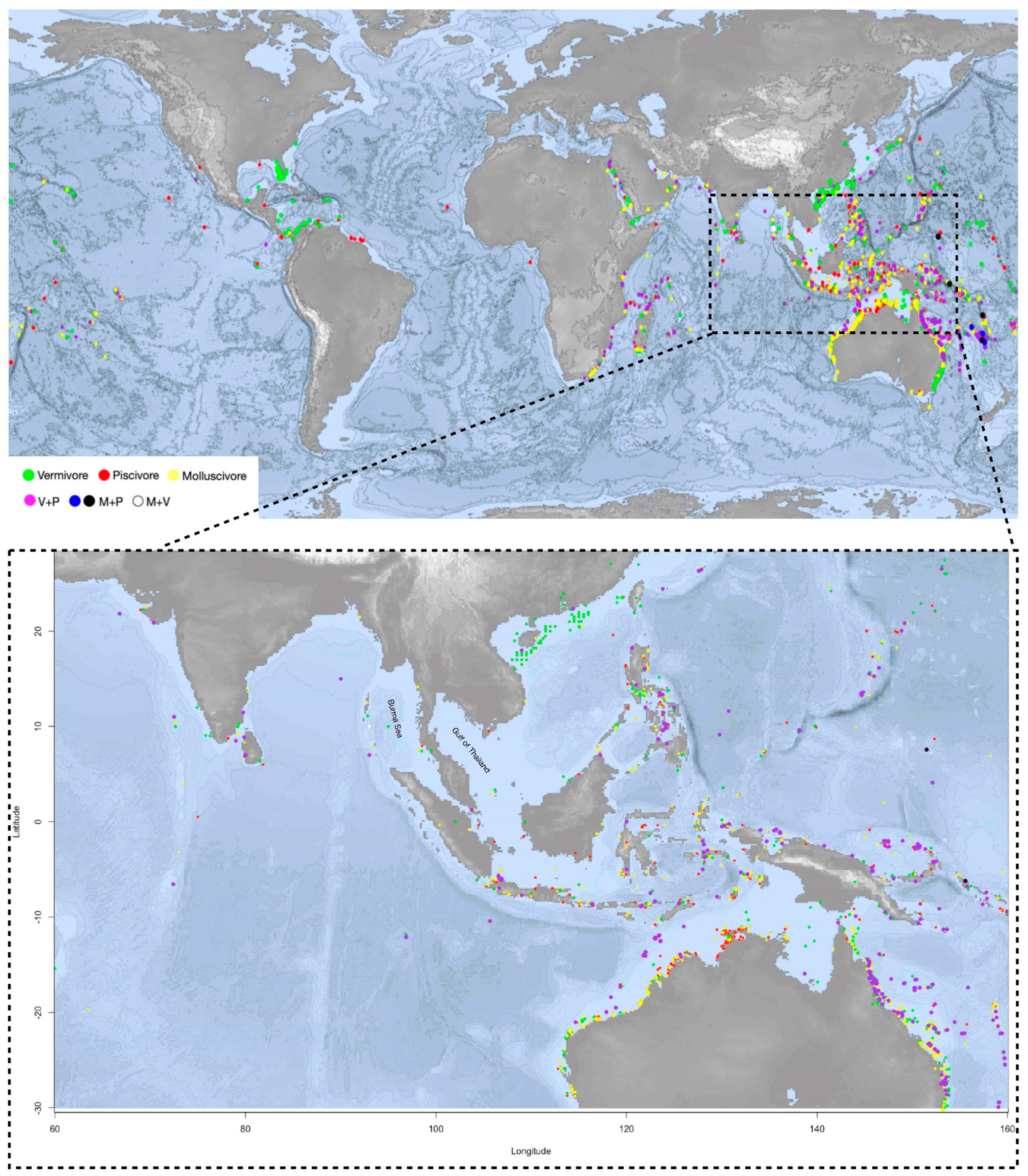
Geographical coordinates of the
Conus species from the last clade on the phylogeny tree are pinpointed on the map. On the map,
Conus species that prey on both worm and fish (V+P) are spread largely in the shallow waters of southeast Asia, Australia and African east coast, corresponding to the distribution areas of the most diversified
Conus species
[3]. Apart from ocean currents, which may prevent the species from dispersing globally, the distribution pattern of V+P species could result from a high competitive pressure and diversity of potential food sources in these areas. The geographical landform of islands has also accelerated evolutionary diversification
[50][40].
Interestingly, despite geological and hydrologic conditions being very similar to the surrounding waters, in south and southeast Asia, comprising the most species-rich area, few
Conus species have been reported on the east coast of the Burma (or Andaman) Sea and the Gulf of Thailand (
Figure 2). Little research and few scientific reports have been conducted in these areas, which could flag a potentially species-rich region for further studies.
Figure 2. The upper map shows the distribution of the monophagous and polyphagous
Conus species clades from the phylogenetic tree (species listed in the
Supplementary Table S2). Orange for molluscivous (M), red for piscivorous (P), green for vermivorous (V). Purple-color dots indicate the feeding habit of V+P (vermivorous and piscivorous); blue and black dots are for M+P (molluscivous and piscivorous); and the white dot is V+M (vermivorous and molluscivous).
Conus species V+P are spread largely in the shallow waters of southeast Asia and Australia and African east coast, corresponding to the distribution areas of the most diversified
Conus species. The distribution pattern of V+P species could result from a high competitive pressure and diversity of potential food sources in these areas. The lower map highlights the region where
Conus species are most abundant and diversified, including the north coast of Australia and south and southeast Asia.
4. Conservation Status
Despite
Conus resources being relatively abundant, it is of great ecological and biological necessity to protect them. Shell hunting of the
Conus has already driven some of the rare species to the edge of extinction. According to the International Union for the Conservation of Nature (IUCN) Red List, 26
Conus species are Near Threatened, 27 species are Vulnerable, and 14 more species have already been Endangered or Critically Endangered, accounting for 9.2% of the total
Conus species globally. The number seems to be relatively small, but some regions are suffering huge depletion. For instance, all three of the Critically Endangered species, along with another four (out of 11) Endangered species, are located in the Cape Verde waters; 45.3% of the 53 species are assessed as Threatened or Near Threatened with extinction. It is shocking to find that 61% of the 41
Conus species are threatened with extinction in the west Africa area
[54,55][44][45]. Shell hunting and habitat degradation are the main threatening factors for the
Conus. Human activities, such as tourism and industrial and residential effluent pollution, have severely affected local
Conus communities. In west Africa, there has been an observable reduction in species abundance and body size.
5. Drug Discovery
Venomous animals are widely distributed in the biosphere, and over time, a positive selection between venom and its target has been driven by the predator and prey arms race. Venoms are usually composed of novel chemical structures with various targets. Therefore, they encompass a pool of broad molecular structures highly relevant for novel drug discovery.
Marine toxins are one of the most rapidly growing research areas in bioactive substances. As unique chemical structures are more often found in venoms, the structure–function relationship of marine toxins is a valuable clue for discovering new targets and pathways. The most common marine toxins can be found in pufferfish and crabs; some toxic shellfish can also produce paralytic neurotoxins. Polyether toxins can be easily transmitted to humans through the food chain and cause food poisoning, such as palytoxin, dinophysistoxin and ciguatoxin. Other marine toxins include alkaloids, for example, saxitoxin. Among all the marine toxins, polypeptide toxins are so far the most effective and deadly (tetrodotoxin and conotoxin). They are usually highly selective with the nervous system, digestive system, cardiovascular system and cell membrane, with especially strong cardiotoxicity and cytotoxicity. This makes venom peptides very prominent for drug discovery. As a widely studied marine invertebrate, its diversified conopeptides make
Conus promising animals for the discovery of antimicrobial peptides, analgesics and anti-cancer drugs.
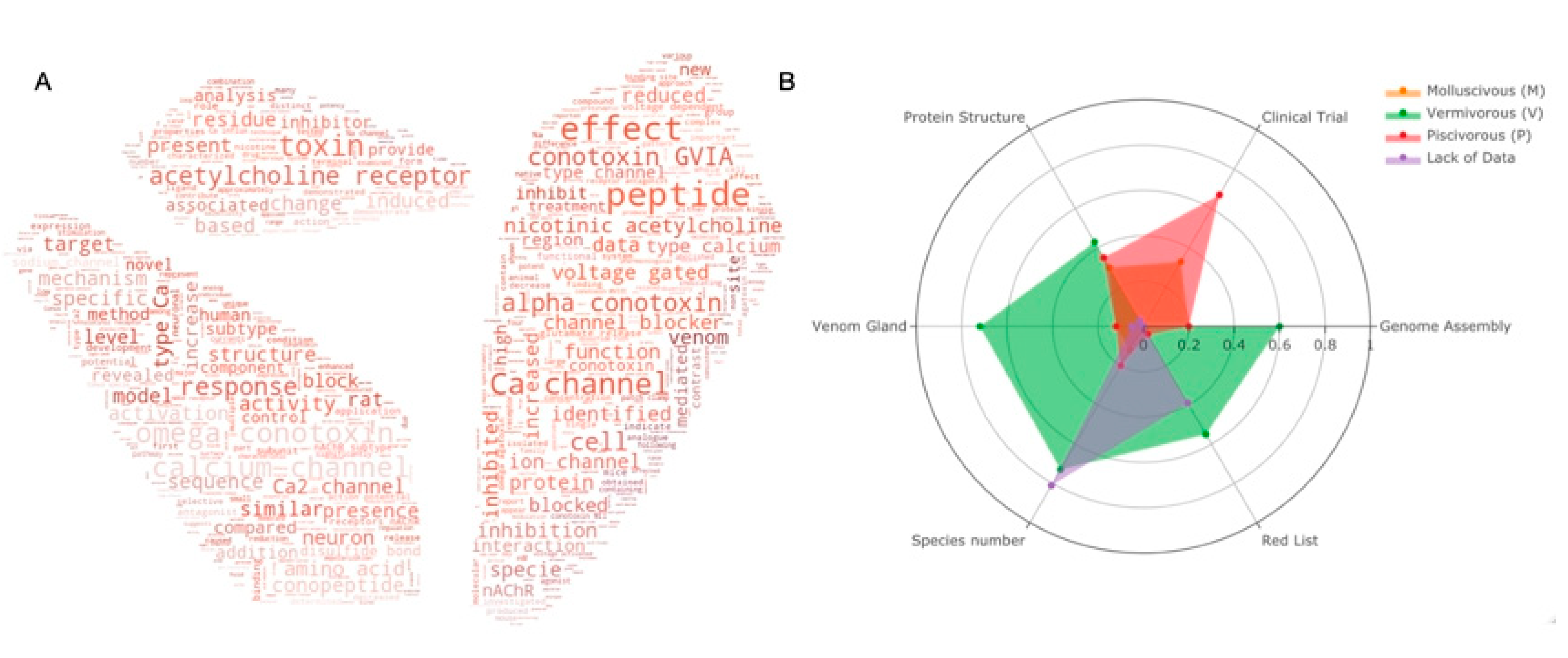
Many works have committed to decipher the secrets of conopeptides.
Figure 3A shows the top 100 keywords in the field for the past two decades. It is obvious to see that most publications are related to the ion channels (Ca
2+, sodium, voltage gated) and peptides–receptor interactions (acetylcholine receptor, inhibition, block). The most studied conotoxins are from alpha and omega pharmaceutical families. Protein structures and animal-trials-related topics are less reported.
Figure 3. (
A) Keyword cloud from the abstract of
Conus studies over the past 20 years; (
B) Statistics of current
Conus studies. Ratio of six categories (genome assemblies, species under clinical trials, characterized protein structures, venom glands, species number and red list species) for
Conus species of each feeding habit are presented in the radar plot. Orange for molluscivous (M), red for piscivorous (P), green for vermivorous (V) and species with insufficient data are presented in purple. Species with more than one feeding habit are counted separately in each group. Raw statistics can be seen in
Supplementary Tables S3 and S4.
As mentioned, worm-eating
Conus species (V) is the largest group of the three genera. It also has the highest number of venom glands (31 out of 43 transcriptomes,
Supplementary Table S4) and genome assemblies (2 out of 3), and it is the most endangered species. In contrast, there is no genome assembly so far in the mollusc-hunting group. Even though most of the data are for the worm-hunting species, mollusc- and fish-hunting species are more favored in clinical research. It is notable that a number of species are in danger with little data reported.
In the past 20 years, with Ziconotide being approved by the FDA, a number of conotoxins have been selected for clinical research (
Table 2). This is justified, since the prey for piscivorous
Conus species is vertebrate, whose physiology system is more similar to the human system. Therefore, most drug-related research has focused on this group. Interestingly, mollusc-hunting
Conus sourced molecules are also seen on the list, even if their prey is invertebrate. An important question to investigate now is whether the worm-hunting species, believed to be less promising for drug discovery, underestimated.
Table 2. Drug development with conotoxins.
| |
Conopeptide |
Commercial Name |
Comment |
Target |
Stage |
Company |
Conus | Species (**) |
Reference |
| 1 |
α-Vc1.1 |
ACV1 |
Neuropathic pain |
nAChR (α9α10) |
Phase II * |
Metabolic Pharmaceuticals, Melbourne, Australia |
Conus victoriae | (m) |
[3] |
| 2 |
ω-CVID |
AM336 |
Neuropathic pain |
Ca | 2+ | channel (CaV2.2) N-type calcium channels/blocker |
Phase IIa * |
Relevare Pharmaceuticals LTD., Australia |
Conus catus | (p) |
[3] |
| 3 |
μO-MrVIB |
CGX-1002 |
Neuropathic pain |
Sodium channels/subtype selective blocker |
Preclinical * |
Cognetix Inc, Salt Lake City, USA |
Conus tulipa | (p) |
[3] |
| 4 |
Conantokin-G |
CGX-1007 |
Intractable epilepsy / pain |
NMDA receptor (NR2B) |
Preclinical * |
Cognetix Inc, Salt Lake City, USA |
Conus geographus | (p) |
[3] |
| 5 |
Contulakin-G |
CGX-1160 |
Neuropathic pain |
Neurotensin receptor |
Phase Ib * |
Cognetix Inc, Salt Lake City, USA |
Conus geographus | (p) |
[3] |
| 6 |
ω-MVIIA |
SNX-III, C1002, Ziconotide, Prialt |
Intractable pain |
Ca | 2+ | channel (CaV2.2) N-type calcium channels/blocker |
FDA-approved |
Elan Corporation (Elan Pharmaceuticals), CA, USA |
Conus magus | (p) |
[3] |
| 7 |
χ-MrIA |
Xen2174 |
Neuropathic pain |
Norepinephrine transporter/inhibitor |
Phase IIa * |
Xenome, Ltd., Brisbane, Qld., Australia |
n.a. |
[3] |
| 8 |
κ-PVIIA |
CGX-1051 |
Acute Myocardial Infarct, Cardioprotection |
K | + | channel (KV1)/blocker |
Preclinical |
n.a. |
n.a. |
[64] | [46] |
| 9 |
n.a. |
CGX-1204 |
Muscle relaxer / pain |
Nicotinic acetylcholine receptors/antagonist |
Preclinical |
n.a. |
n.a. |
[64] | [46] |
| 10 |
μ-SIIIA |
PEG-SIIIA |
Inflammatory pain |
Sodium channels/blocker |
Preclinical |
n.a. |
n.a. |
[64] | [46] |
| 11 |
ρ-Conotoxin TIA |
n.a. |
n.a. |
α-1 adrenergic receptors |
Preclinical |
Xenome, Ltd., Brisbane, Qld., Australia |
Conus tulipa | (p) |
[65] | [47] |
| 12 |
χ-conopeptides (χ-CTX MrIA/B) |
n.a. |
Neuropathic pain |
Neurotransmitter transporters |
Preclinical |
Xenome, Ltd., Brisbane, Qld., Australia |
Conus marmoreus | (m) |
[65] | [47] |