Lung cancer accounts for approximately 18.4% of the total cancer-related deaths, the highest of all cancer types. The prognosis of lung cancer is relatively unfavorable compared to that of other malignancies, and as a prognosis largely depends on the stage of onset, thus, the early diagnosis of lung cancer is very important. Pulmonary tuberculosis (TB) is a known risk factor for lung cancer.
- pulmonary tuberculosis
- lung cancer
- meta regression
- burden of tuberculosis
1. Introduction
Chronic inflammation resulting in pathological changes is a major risk factor in carcinogenesis. Inflammation is known to play a key role in carcinogenesis, such as infection with hepatitis B and C viruses in hepatocellular carcinoma, Helicobacter pylori in gastric cancer, and human papilloma virus in gynecological cancers [7][1]. Several meta-analyses have shown that previous inflammatory diseases in the lungs, such as pneumonia, chronic bronchitis, and pulmonary tuberculosis (TB), may increase the risk of lung cancer (relative risk ratio 1.36–1.90), independent of cigarette smoking [8,9][2][3]. According to forty-nine studies, pulmonary and extra-pulmonary TB infections increase the risk of 10 cancer types, including head and neck cancer, leukemia, lymphoma, gastrointestinal cancer, kidney cancer, bladder cancer, and lung cancer [10][4]. Thus, TB infection may influence the pathogenesis of lung cancer with or without cigarette smoking. To prevent the emergence of airborne transmittable TB and its progression to cancer, the control and prevention of TB is very important.
2. Pulmonary TB and Risk of Lung Cancer with All Eligible Studies


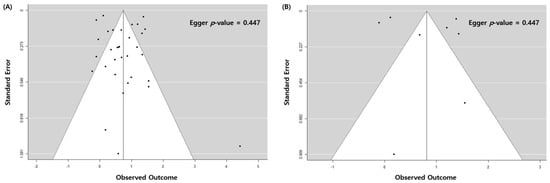
| Subgroup | No. of Cohorts * | OR (95% CI) | p | -Value | I2 | Value (%) | p | -Value | I2 | between Subgroups (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| I | 2 | Value (%) | I | 2 | between Subgroups (%) |
|||||
| All cohorts | 33 | 2.09 (1.62–2.69) | <0.001 | 95 |

| Study | Selection | Comparability | Outcome | Quality Score | |||||
|---|---|---|---|---|---|---|---|---|---|
| Adequacy of Case Definition | Degree of Representation of Cases | Selection of Controls | Definition of Controls | Comparability of Cases and Controls on the Basis of Design or Analysis | Confirmation of Exposure | Same Method of Confirmation for Cases and Controls | Non-Response Rate | ||
| Selection | Comparability | Outcome | Quality Score |
||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Study | Degree of Representation of the Exposed Cohort | Selection of the Non-Exposed Cohort | Confirmation of Exposure | Demonstration That the Current Outcome of Interest Is Absent at the Start of the Study | Comparability of Cohorts Based on Design or Analysis | Assessment of Outcome | Sufficiency of Follow-Up to Detect Outcomes | Adequacy of Follow-Up of Cohorts | |||||||||||||
| All studies | 8 | 2.26 (1.29–3.94) | 0.004 | 99 | |||||||||||||||||
| An et al. 2020 [20] | An et al. 2020 [5] | ||||||||||||||||||||
| Kim et al. 2020 [ | * | 19] | Kim et al. 2020 [31 | * | * | * | ] | ** | * | * | * | 9 | * | * | * | ** | * | * | 7 | ||
| TB burden of country | |||||||||||||||||||||
| Country of TB burden | |||||||||||||||||||||
| Yang et al. 2015 [22] | Yang et al. 2015 [6] | * | * | * | * | ** | * | * | |||||||||||||
| Oh et al. 2020 [21] | 8 | Oh et al. 2020 [32] | * | * | * | ** | * | * | * | 8 | Low | 18 | 1.77 (1.22–2.56) | Low | 0.003 | 97 | 7 | 2.04 (1.12–3.73) | 0.020 | ||
| Yang et al. 2015 [23 | 12 | ||||||||||||||||||||
| ] | Yang et al. 2015 [7] | 99 | 78 | ||||||||||||||||||
| * | * | * | * | * | * | * | * | 8 | |||||||||||||
| Simonsen et al. 2014 [24] | Simonsen et al. 2014 [33] | * | * | Medium | 6 | 2.48 (1.71–3.58) | <0.001 | 75 | |||||||||||||
| Medium | |||||||||||||||||||||
| 8 | |||||||||||||||||||||
| * | * | * | * | * | * | 8 | [8] | 1 | 4.18 (3.15–5.55) | <0.001 | - | ||||||||||
| Bae et al. 2013 [26] | Bae et al. 2013 [34] | * | * | * | ** | * | * | 7 | High | 9 | 2.57 (1.68–3.93) | <0.001 | 81 | ||||||||
| Region of country | |||||||||||||||||||||
| East Asia and Pacific | 19 | ||||||||||||||||||||
| * | * | * | * | * | High | 0 | - | - | - | ||||||||||||
| Shiels et al. 2011 [29] | Shiels et al. 2011 [35] | * | * | * | 2.49 (1.83–3.39) | <0.001 | |||||||||||||||
| 93 | 58 | ||||||||||||||||||||
| Europe and Central Asia | 7 | ||||||||||||||||||||
| Hosgood et al. 2013 [25] | Hosgood et al. 2013 | * | * | 7 | |||||||||||||||||
| Lo et al. 2013 [27] | Lo et al. 2013 [9] | * | * | * | * | ** | * | * | 8 | * | ** | * | * | * | 9 | Region of country | |||||
| Bodmer et al. 2012 [28] | Bodmer et al. 2012 [10 | ] | |||||||||||||||||||
| * | * | * | * | 1.60 (0.80–3.22) | 0.185 | 98 | |||||||||||||||
| ** | * | * | * | ||||||||||||||||||
| Yu et al. 2011 [30] | Yu et al. 2011 [36] | * | * | 9 | * | * | * | * | * | East Asia and Pacific | 4 | 2.79 (1.21–6.39) | 0.016 | 98 | 0 | ||||||
| 7 | Koshiol et al. 2010 [31] | Koshiol et al. 2010 [11] | * | * | * | * | ** | * | * | 8 | Europe and Central Asia | 4 | 1.79 (0.67–4.77) | 0.244 | 99 | ||||||
| Park et al. 2010 [32] | Park et al. 2010 [12] | North America | 7 | North America | 0 | - | - | - | |||||||||||||
Abbreviations: CI, confidence interval; No, Number; OR, odds ratio; TB, tuberculosis.
4. Stratified and Sensitivity Analysis
| * | ||||||||||||||||||||||||||||||
| * | ||||||||||||||||||||||||||||||
| * | ||||||||||||||||||||||||||||||
| * | ||||||||||||||||||||||||||||||
| ** | * | 7 | ||||||||||||||||||||||||||||
| 1.53 (1.11–2.12) | 0.010 | 0 | ||||||||||||||||||||||||||||
| Liang et al. 2009 [33] | Liang et al. 2009 [13] | * | * | * | * | ** | * | * | Economic status of country | |||||||||||||||||||||
| 8 | ||||||||||||||||||||||||||||||
| Wang et al. 2009 [34] | Wang et al. 2009 [14] | * | * | * | * | * | * | * | 7 | High-income | ||||||||||||||||||||
| Galeone et al. 2008 [35] | Galeone et al. 2008 [ | 24 | 15 | 1.91 (1.41–2.59) | ] | * | * | <0.001 | 96 | 20 | ||||||||||||||||||||
| * | * | ** | * | * | 8 | Upper-middle-income | 9 | 2.57 (1.68–3.93) | <0.001 | |||||||||||||||||||||
| Ramanakumar et al. 2006 [36] | Ramanakumar et al. 2006 [16] | 81 | ||||||||||||||||||||||||||||
| a | * | * | * | * | ** | * | * | 8 | Age | |||||||||||||||||||||
| Ramanakumar et al. 2006 [36] | Ramanakumar et al. 2006 [16] | b | * | * | * | * | ** | * | * | 8 | Adjusted | |||||||||||||||||||
| Zatloukal et al. 2003 [ | 29 | 37] | Zatloukal et al. 2003 | 2.00 (1.54–2.61) | [ | <0.001 | 95 | 14 | ||||||||||||||||||||||
| 17] | * | * | * | * | ** | * | * | 8 | Not adjusted | 4 | 3.84 (1.21–12.15) | 0.022 | 82 | |||||||||||||||||
| Chan-Yeung et al. 2003 [38] | Chan-Yeung et al. 2003 [18] | * | * | * | * | * | * | 7 | Sex | |||||||||||||||||||||
| * | ||||||||||||||||||||||||||||||
| Kreuzer et al. 2002 [39] | Kreuzer et al. 2002 [19] | * | * | * | * | * | * | * | Adjusted | 22 | 2.23 (1.60–3.11) | <0.001 | 96 | 0 | ||||||||||||||||
| 7 | ||||||||||||||||||||||||||||||
| Brenner et al. 2001 [40] | Brenner et al. 2001 [20] | * | * | * | * | ** | * | * | 8 | Not adjusted | 11 | 1.90 (1.47–2.46) | <0.001 | |||||||||||||||||
| Kreuzer et al. 2001 [41 | 61 | |||||||||||||||||||||||||||||
| ] | Kreuzer et al. 2001 [21] | Smoking | ||||||||||||||||||||||||||||
| Adjusted | 22 | 2.03 (1.51–2.73) | <0.001 | 90 | 0 | |||||||||||||||||||||||||
| Not adjusted | 11 | 2.19 (1.34–3.59) | 0.002 | 98 | ||||||||||||||||||||||||||
| Hypertension | ||||||||||||||||||||||||||||||
| Adjusted | 2 | 1.92 (0.66–5.57) | 0.230 | 99 | 0 | |||||||||||||||||||||||||
| Not adjusted | 31 | 2.10 (1.62–2.73) | <0.001 | 92 | ||||||||||||||||||||||||||
| Diabetes | ||||||||||||||||||||||||||||||
| Adjusted | 2 | 1.72 (0.48–6.20) | 0.404 | 99 | 0 | |||||||||||||||||||||||||
| Not adjusted | 31 | 2.13 (1.63–2.77) | <0.001 | 94 | ||||||||||||||||||||||||||
| Respiratory comorbidities | ||||||||||||||||||||||||||||||
| Adjusted | 8 | 1.32 (0.93–1.86) | 0.121 | 94 | 90 | |||||||||||||||||||||||||
| Not adjusted | 25 | 2.51 (2.04–3.08) | <0.001 | 78 | ||||||||||||||||||||||||||
| * | * | * | * | * | * | * | * | 8 | ||||||||||||||||||||||
| Zhou et al. 2000 [42] | Zhou et al. 2000 [22] | * | * | * | * | * | * | * | 7 | |||||||||||||||||||||
| Osann et al. 2000 [43] | Osann et al. 2000 [23] | * | * | * | * | ** | * | * | 8 | |||||||||||||||||||||
| Mayne et al. 1999 [44] | Mayne et al. 1999 [24] | * | * | * | * | * | * | * | 7 | |||||||||||||||||||||
| Ko et al. 1997 [45] | Ko et al. 1997 [25] | * | * | * | * | * | * | * | 7 | |||||||||||||||||||||
| Schwartz et al. 1996 [46] | Schwartz et al. 1996 [26] | * | * | * | * | ** | * | * | 8 | |||||||||||||||||||||
| Luo et al. 1996 [47] | Luo et al. 1996 [27] | * | * | * | * | * | * | * | 7 | |||||||||||||||||||||
| Wu et al. 1995 [48] | Wu et al. 1995 [28] | * | * | * | * | ** | * | * | 8 | |||||||||||||||||||||
| Alavanja et al. 1992 [49] | Alavanja et al. 1992 [29] | * | * | * | * | ** | * | * | Wu-Williams et al. 1990 [50] | Wu-Williams et al. 1990 [30] | * | * | * | * | ** | * | Cohort of the study | |||||||||||||
| * | 8 | Population-based | 23 | 1.95 (1.41–2.68) | <0.001 | 96 | 0 | |||||||||||||||||||||||
| Hospital-based | 10 | 2.36 (1.85–3.01) | <0.001 | 49 | ||||||||||||||||||||||||||
| Study design | ||||||||||||||||||||||||||||||
| Prospective cohort study | 4 | 1.96 (1.22–3.15) | 0.005 | 84 | 94 | |||||||||||||||||||||||||
| Retrospective cohort study | 2 | 3.95 (3.58–4.36) | <0.001 | 0 | ||||||||||||||||||||||||||
| Case-control study | 27 | 1.99 (1.56–2.53) | <0.001 | 89 | ||||||||||||||||||||||||||
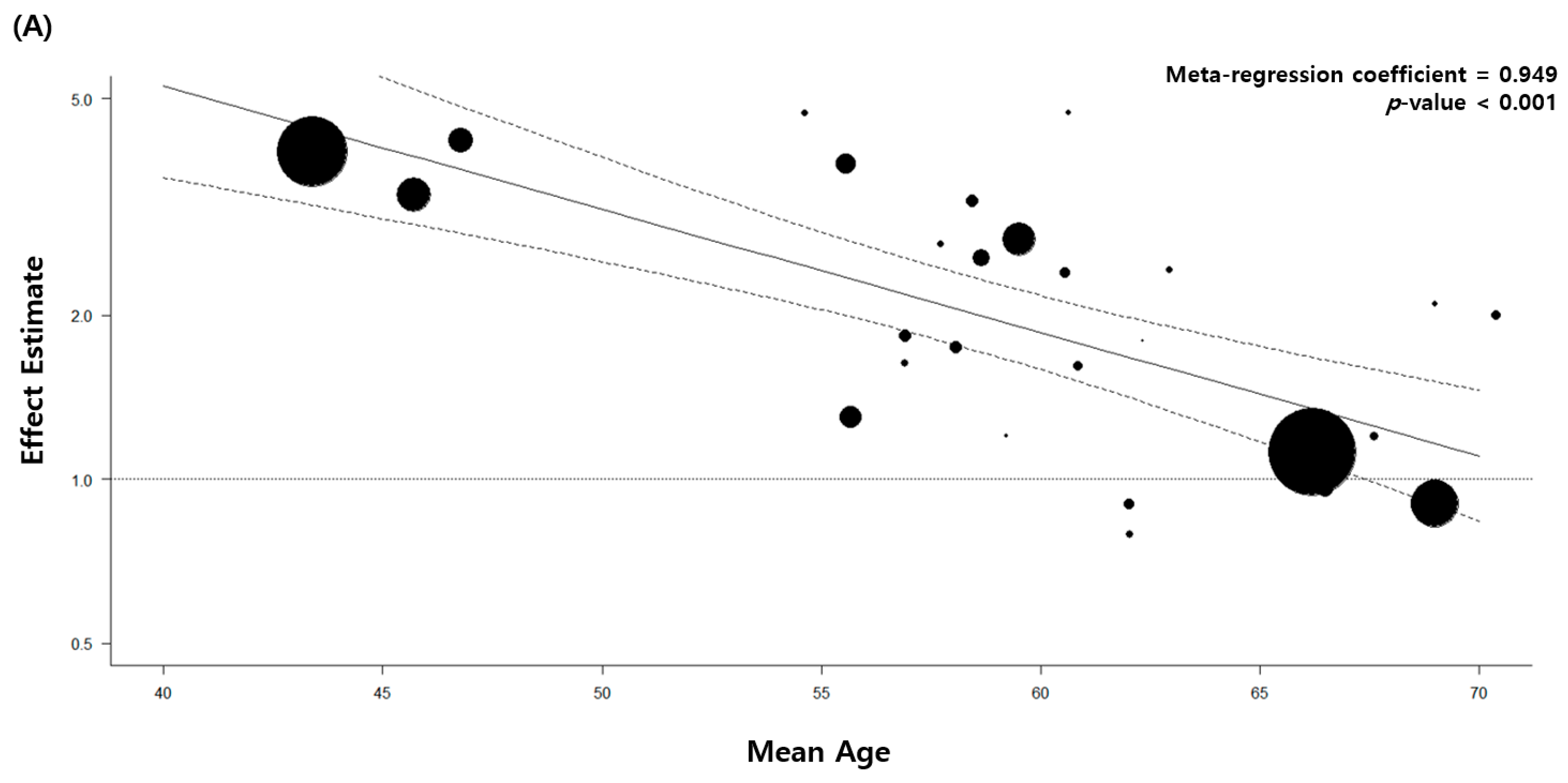
| Diagnostic method | of pulmonary TB | |||||||||||||||||||||||||||||
| Medical record | 8 | 2.26 (1.29–3.94) | 0.004 | 99 | 0 | |||||||||||||||||||||||||
| Imaging | 3 | 2.13 (1.16–3.92) | 0.015 | 80 | ||||||||||||||||||||||||||
| Self-report or physical examination |
22 | 1.96 (1.56–2.47) | <0.001 | 66 | ||||||||||||||||||||||||||
* Since two separate cohorts were reported in one article, a total of 33 eligible cohorts were extracted and analyzed from 32 enrolled studies. Abbreviations: CI, confidence interval; No, Number; OR, odds ratio; TB, tuberculosis.
3. Pulmonary TB and Risk of Lung Cancer with High-Quality Studies
| Subgroup | No. of Studies | OR (95% CI) |
|---|
A sItudy can be awarded a maximum of one star for each numbered item within the Selection and Outcome categories. A maximum of two stars can be given for Comparability.
References
- Coussens, L.M.; Werb, Z. Inflammation and Cancer. Nature 2002, 420, 860–867.
- Brenner, D.R.; Boffetta, P.; Duell, E.J.; Bickeböller, H.; Rosenberger, A.; McCormack, V.; Muscat, J.E.; Yang, P.; Wichmann, H.-E.; Brueske-Hohlfeld, I.; et al. Previous Lung Diseases and Lung Cancer Risk: A Pooled Analysis From the International Lung Cancer Consortium. Am. J. Epidemiol. 2012, 176, 573–585.
- Brenner, D.R.; McLaughlin, J.R.; Hung, R.J. Previous Lung Diseases and Lung Cancer Risk: A Systematic Review and Meta-Analysis. PLoS ONE 2011, 6, e17479.
- Leung, C.Y.; Huang, H.-L.; Rahman, M.; Nomura, S.; Abe, S.K.; Saito, E.; Shibuya, K. Cancer Incidence Attributable to Tuberculosis in 2015: Global, Regional, and National Estimates. BMC Cancer 2020, 20, 1–13.
- An, S.J.; Kim, Y.J.; Han, S.S.; Heo, J. Effects of Age on the Association between Pulmonary Tuberculosis and Lung Cancer in a South Korean Cohort. J. Thorac. Dis. 2020, 12, 375–382.
- Yang, L.; Lu, X.; Deng, J.; Zhou, Y.; Huang, N.; Qiu, F.; Yang, X.; Yang, R.; Fang, W.; Ran, P.; et al. Risk Factors Shared by COPD and Lung Cancer and Mediation Effect of COPD: Two Center Case–Control Studies. Cancer Causes Control 2014, 26, 11–24.
- Yang, T.-Y.; Lin, W.-M.; Lin, C.-L.; Sung, F.-C.; Kao, C.-H. Correlation between Use of Simvastatin and Lovastatin and Female Lung Cancer Risk: A Nationwide Case-Control Study. Int. J. Clin. Pract. 2014, 69, 571–576.
- HosgoodIII, H.D.; Chapman, R.S.; He, X.; Hu, W.; Tian, L.; Liu, L.Z.; Lai, H.; Chen, W.; Rothman, N.; Lan, Q. History of Lung Disease and Risk of Lung Cancer in a Population with High Household Fuel Combustion Exposures in Rural China. Lung Cancer 2013, 81, 343–346.
- Lo, Y.-L.; Hsiao, C.-F.; Chang, G.-C.; Tsai, Y.-H.; Huang, M.-S.; Su, W.-C.; Chen, Y.-M.; Hsin, C.-W.; Chang, C.-H.; Yang, P.-C.; et al. Risk Factors for Primary Lung Cancer among Never Smokers by Gender in a Matched Case–Control Study. Cancer Causes Control 2012, 24, 567–576.
- Bodmer, M.; Becker, C.; Jick, S.S.; Meier, C.R. Metformin Does Not Alter the Risk of Lung Cancer: A Case–Control Analysis. Lung Cancer 2012, 78, 133–137.
- Koshiol, J.; Rotunno, M.; Consonni, D.; Pesatori, A.C.; De Matteis, S.; Goldstein, A.M.; Chaturvedi, A.K.; Wacholder, S.; Landi, M.T.; Lubin, J.H.; et al. Lower Risk of Lung Cancer after Multiple Pneumonia Diagnoses. Cancer Epidemiol. Biomark. Prev. 2010, 19, 716–721.
- Park, S.K.; Cho, L.Y.; Yang, J.J.; Park, B.; Chang, S.H.; Lee, K.S.; Kim, H.; Yoo, K.Y.; Lee, C.T. Lung Cancer Risk and Cigarette Smoking, Lung Tuberculosis According to Histologic Type and Gender in a Population Based Case-Control Study. Lung Cancer 2010, 68, 20–26.
- Liang, H.; Guan, P.; Yin, Z.; Li, X.; He, Q.; Zhou, B. Risk of Lung Cancer following Nonmalignant Respiratory Conditions among Nonsmoking Women Living in Shenyang, Northeast China. J. Womens Health 2009, 18, 1989–1995.
- Wang, X.R.; Yu, I.T.; Chiu, Y.L.; Qiu, H.; Fu, Z.; Goggins, W.; Au, J.S.; Tse, L.A.; Wong, T.W. Previous Pulmonary Disease and Family Cancer History Increase the Risk of Lung Cancer among Hong Kong Women. Cancer Causes Control 2009, 20, 757–763.
- Galeone, C.; Pelucchi, C.; La Vecchia, C.; Negri, E.; Bosetti, C.; Hu, J. Indoor Air Pollution from Solid Fuel Use, Chronic Lung Diseases and Lung Cancer in Harbin, Northeast China. Eur. J. Cancer Prev. 2008, 17, 473–478.
- Ramanakumar, A.V.; Parent, M.E.; Menzies, D.; Siemiatycki, J. Risk of Lung Cancer Following Nonmalignant Respiratory Conditions: Evidence from Two Case-Control Studies in Montreal, Canada. Lung Cancer 2006, 53, 5–12.
- Zatloukal, P.; Kubík, A.; Pauk, N.; Tomásek, L.; Petruzelka, L. Adenocarcinoma of the Lung among Women: Risk Associated with Smoking, Prior Lung Disease, Diet and Menstrual and Pregnancy History. Lung Cancer 2003, 41, 283–293.
- Chan-Yeung, M.; Koo, L.C.; Ho, J.C.; Tsang, K.W.; Chau, W.S.; Chiu, S.W.; Ip, M.S.; Lam, W.K. Risk Factors Associated with Lung Cancer in Hong Kong. Lung Cancer 2003, 40, 131–140.
- Kreuzer, M.; Heinrich, J.; Kreienbrock, L.; Rosario, A.S.; Gerken, M.; Wichmann, H.E. Risk Factors for Lung Cancer among Nonsmoking Women. Int. J. Cancer 2002, 100, 706–713.
- Brenner, A.V.; Wang, Z.; Kleinerman, R.A.; Wang, L.; Zhang, S.; Metayer, C.; Chen, K.; Lei, S.; Cui, H.; Lubin, J.H. Previous Pulmonary Diseases and Risk of Lung Cancer in Gansu Province, China. Int. J. Epidemiol. 2001, 30, 118–124.
- Kreuzer, M.; Gerken, M.; Kreienbrock, L.; Wellmann, J.; Wichmann, H.E. Lung Cancer in Lifetime Nonsmoking Men-Results of a Case-Control Study in Germany. Br. J. Cancer 2001, 84, 134–140.
- Zhou, B.S.; Wang, T.J.; Guan, P.; Wu, J.M. Indoor Air Pollution and Pulmonary Adenocarcinoma among Females: A Case-Control Study in Shenyang, China. Oncol. Rep. 2000, 7, 1253–1259.
- Osann, K.E.; Lowery, J.T.; Schell, M.J. Small Cell Lung Cancer in Women: Risk Associated with Smoking, Prior Respiratory Disease, and Occupation. Lung Cancer 2000, 28, 1–10.
- Mayne, S.T.; Buenconsejo, J.; Janerich, D.T. Previous Lung Disease and Risk of Lung Cancer among Men and Women Nonsmokers. Am. J. Epidemiol. 1999, 149, 13–20.
- Ko, Y.C.; Lee, C.H.; Chen, M.J.; Huang, C.C.; Chang, W.Y.; Lin, H.J.; Wang, H.Z.; Chang, P.Y. Risk Factors for Primary Lung Cancer among Non-Smoking Women in Taiwan. Int. J. Epidemiol. 1997, 26, 24–31.
- Schwartz, A.G.; Yang, P.; Swanson, G.M. Familial Risk of Lung Cancer among Nonsmokers and their Relatives. Am. J. Epidemiol. 1996, 144, 554–562.
- Luo, R.X.; Wu, B.; Yi, Y.N.; Huang, Z.W.; Lin, R.T. Indoor Burning Coal Air Pollution and Lung Cancer—A Case-Control Study in Fuzhou, China. Lung Cancer 1996, 14 (Suppl. 1), S113–S119.
- Wu, A.H.; Fontham, E.T.; Reynolds, P.; Greenberg, R.S.; Buffler, P.; Liff, J.; Boyd, P.; Henderson, B.E.; Correa, P. Previous Lung Disease and Risk of Lung Cancer among Lifetime Nonsmoking Women in the United States. Am. J. Epidemiol. 1995, 141, 1023–1032.
- Alavanja, M.C.; Brownson, R.C.; Boice, J.D., Jr.; Hock, E. Preexisting Lung Disease and Lung Cancer among Nonsmoking Women. Am. J. Epidemiol. 1992, 136, 623–632.
- Wu-Williams, A.H.; Dai, X.D.; Blot, W.; Xu, Z.Y.; Sun, X.W.; Xiao, H.P.; Stone, B.J.; Yu, S.F.; Feng, Y.P.; Ershow, A.G.; et al. Lung Cancer among Women in North-East China. Br. J. Cancer 1990, 62, 982–987.
- Kim, H.; Kim, H.Y.; Goo, J.M.; Kim, Y. Lung Cancer CT Screening and Lung-Rads in a Tuberculosis-Endemic Country: The Korean Lung Cancer Screening Project (K-LUCAS). Radiology 2020, 296, 181–188.
- Oh, C.-M.; Roh, Y.-H.; Lim, D.; Kong, H.-J.; Cho, H.; Hwangbo, B.; Won, Y.-J.; Jung, K.-W.; Oh, K. Pulmonary Tuberculosis is Associated with Elevated Risk of Lung Cancer in Korea: The Nationwide Cohort Study. J. Cancer 2020, 11, 1899–1906.
- Simonsen, D.F.; Farkas, D.K.; Søgaard, M.; Horsburgh, C.R.; Sørensen, H.T.; Thomsen, R.W. Tuberculosis and Risk of Cancer: A Danish Nationwide Cohort Study. Int. J. Tuberc. Lung Dis. 2014, 18, 1211–1219.
- Bae, J.-M.; Li, Z.-M.; Shin, M.-H.; Kim, D.-H.; Lee, M.-S.; Ahn, Y.-O. Pulmonary Tuberculosis and Lung Cancer Risk in Current Smokers: The Seoul Male Cancer Cohort Study. J. Korean Med. Sci. 2013, 28, 896–900.
- Shiels, M.S.; Albanes, D.; Virtamo, J.; Engels, E.A. Increased Risk of Lung Cancer in Men with Tuberculosis in the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study. Cancer Epidemiol. Biomark. Prev. 2011, 20, 672–678.
- Yu, Y.H.; Liao, C.C.; Hsu, W.H.; Chen, H.J.; Liao, W.C.; Muo, C.H.; Sung, F.C.; Chen, C.Y. Increased Lung Cancer Risk among Patients with Pulmonary Tuberculosis: A Population Cohort study. J. Thorac. Oncol. 2011, 6, 32–37.
