“Post-acute sequelae of SARS-CoV-2 infection” (PASC), otherwise referred to as “long COVID” or “long-haul COVID”, refers to persistent and prolonged effects after acute COVID-19 and describes the persistence of symptoms or development of sequelae beyond 3 weeks from the onset of acute symptoms of SARS-CoV-2 infection. Symptoms commonly experienced by PASC patients include fatigue, palpitations, chest pain, dyspnea, reduced exercise tolerance, and “brain fog”. Additionally, symptoms of orthostatic intolerance and syncope suggest the involvement of the autonomic nervous system.
- COVID-19
- cardiovascular autonomic dysfunction
- PASC
- postural orthostatic tachycardia
- post-acute sequelae
- autoimmunity
1. Post-Acute Sequelae of COVID-19
COVID-19 survivors have been shown to experience a constellation of symptoms such as fatigue, dyspnea or breathlessness [8[1][2],15], palpitations [16,17][3][4], brain fog, lack of concentration [18[5][6],19], sleep disturbances (i.e., insomnia) [20][7], headache [17[4][8][9][10][11][12],21,22,23,24,25], orthostatic intolerance [25][12], anxiety and post-traumatic stress disorder [20[7][10][13][14][15],23,26,27,28], chest pain, joint pain [8][1], sore throat [17][4], and hair loss [15][2] persisting >4 weeks after recovery. These clinical sequelae share similarities with post-acute symptoms reported after severe acute respiratory syndrome (SARS) and Middle East Respiratory Syndrome (MERS) epidemics, caused by previous coronaviruses [14][16]. Such heterogeneous postmorbid clinical presentation, with a plethora of specific signs and symptoms often being interpreted as non-physiologic or related to mental-health-related conditions, lends itself to the potential marginalization of patients [28][15].
Fatigue is by far the most commonly reported symptom among subjects with previous COVID-19 infection. Severe fatigue is more frequently reported by female and younger patients [32][17], and was associated with a high prevalence of anxiety [33][18] and post-traumatic stress disorder. Notably, fatigue described by PASC patients shows some distinct features from chronic fatigue syndrome (CFS). Studies on CFS patients showed impaired cerebral blood flow [34][19] and reduced heart rate variability [35[20][21],36], although these findings could not be confirmed in PASC patients [33][18]. Furthermore, less than 50% of patients with PASC-related fatigue met the diagnostic criteria for CFS [37][22]. This suggests that PASC is not just a COVID-19-related manifestation of CFS but a specific pathophysiological entity with a specific CVAD phenotype.
2. COVID-19 Infection and Dysautonomia
3. Postural Orthostatic Tachycardia Syndrome after COVID-19
Beyond appropriate sinus tachycardia, which comprises both physiological and pathologic causes of increased heart rate, a possible explanation for palpitations and fatigue in COVID-19 survivors is POTS, which is a dysautonomia characterized by an excess in heart rate increase on standing and orthostatic intolerance [49][31]; it is more frequent in females (80% of cases), and the young (especially 15–45 years old). POTS is misdiagnosed in up to 75% of patients and viral infection is recognized as a trigger in up to 41% of cases [63][32]. In March 2021, the American Autonomic Society (AAS) issued a statement concerning the clinical overlap of PASC with POTS [4][33]. The AAS stated that most PASC symptoms can be suggestive of POTS when the patient also suffers from excessive orthostatic tachycardia, and that further, in-depth studies are needed to elucidate the duration of post-COVID CVAD, its underlying pathophysiology, and optimum treatment. There is increasing evidence of incident cases of postural tachycardia following COVID-19. In an international online survey on 802 COVID-19 survivors who received a post-acute diagnosis, 19% reported receiving a POTS diagnosis [54][34]. In this cohort, POTS was the second most common reported diagnosis in the aftermath of COVID-19, following migraine (27%).4. How Does SARS-CoV-2 Infection Cause Long-Term Dysautonomia?
Infections are known triggers of dysautonomia, particularly in POTS. Among various mechanisms by which SARS-CoV-2 may induce dysautonomia, three possible conditions have been proposed and gained support from preliminary evidence: hypovolemia, brainstem involvement, and autoimmunity [64][35]. Hypovolemia has been widely recognized in PASC patients with dysautonomic features [25,52][12][36]. Hypovolemia may trigger hyperadrenergic POTS and lead to cerebral hypoperfusion and the impairment of central autonomic networks [56][37]. Preliminary evidence on post-COVID POTS supports the correction of hypovolemia by the liberal intake of water and salt [16][3] and other nonpharmacological interventions aimed at intravascular volume expansion and increasing venous return [65][38] (Figure 1).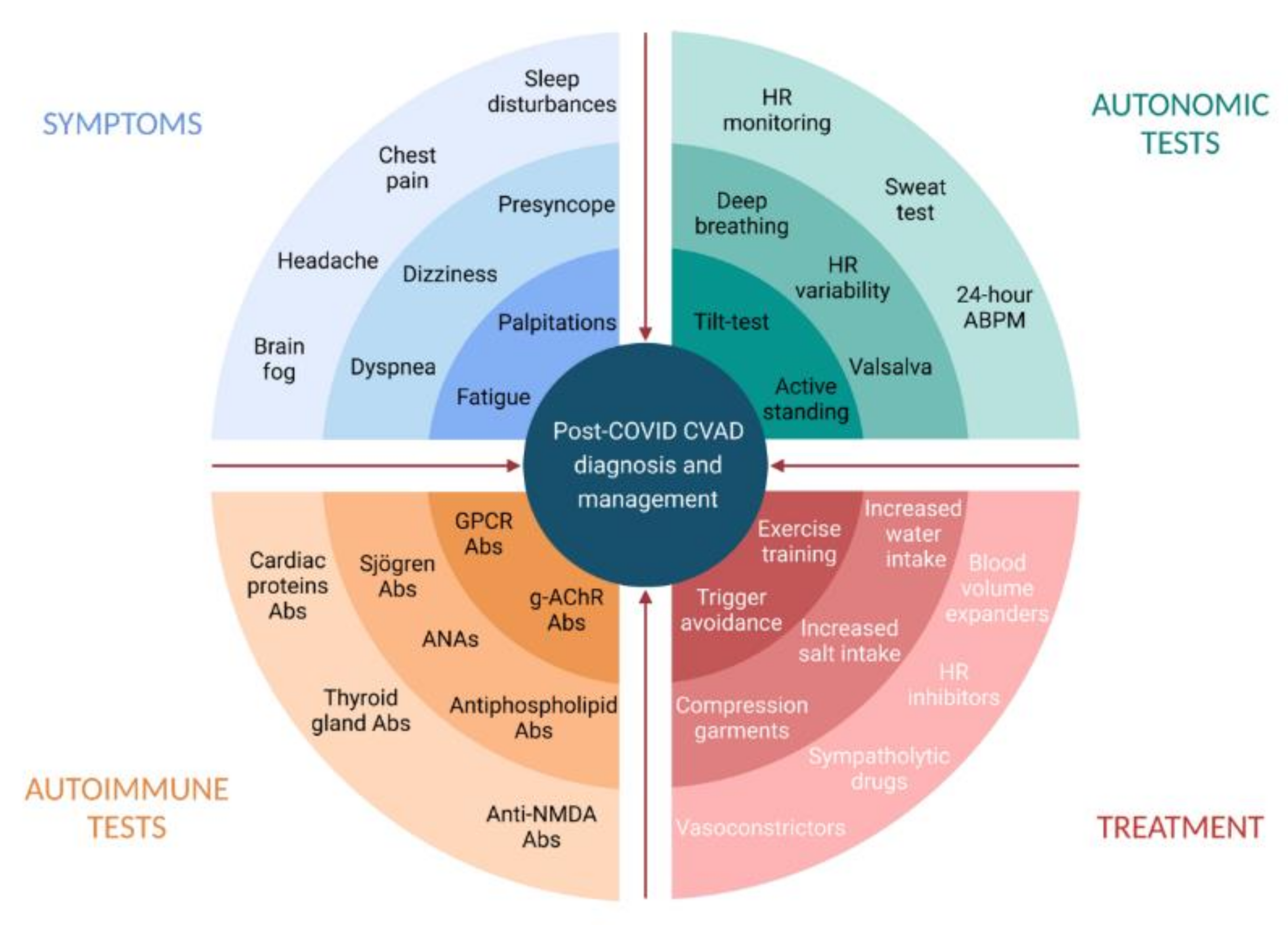
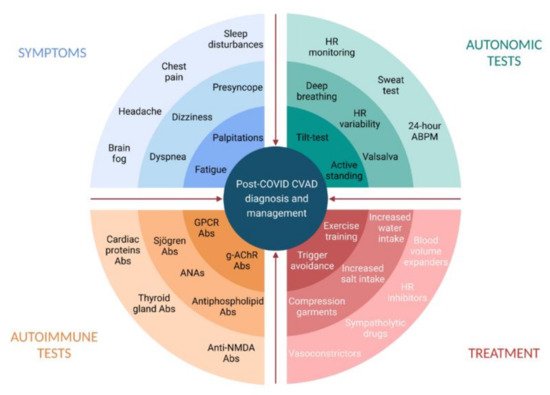 Figure 1. Post-COVID cardiovascular autonomic dysfunction: diagnosis and management. Abs, antibodies; ANAs, antinuclear antibodies; CVAD, cardiovascular autonomic dysfunction; g-AChR, ganglionic neuronal nicotinic acetylcholine receptor; GPCR, G-Protein coupled receptor; HR, heart rate. Created with BioRender.com.
Figure 1. Post-COVID cardiovascular autonomic dysfunction: diagnosis and management. Abs, antibodies; ANAs, antinuclear antibodies; CVAD, cardiovascular autonomic dysfunction; g-AChR, ganglionic neuronal nicotinic acetylcholine receptor; GPCR, G-Protein coupled receptor; HR, heart rate. Created with BioRender.com.5. Diagnosis of Post-COVID Dysautonomia
The diagnosis of post-COVID dysautonomia requires the comprehensive assessment of previous medical history, the careful evaluation and characterization of symptoms, and expert interpretation of neurological and cardiovascular autonomic investigations [4,16,83][33][3][53]. Patients’ medical histories and their recollection of symptoms are essential in detecting the presence of dysautonomia [84][54]. The histories of SARS-CoV-2 survivors with persistent autonomic dysfunction may reveal frequent fainting episodes, dizziness, lightheadedness, and/or palpitations [63][32] prior to SARS-CoV-2 infection, revealing underlying hypotensive susceptibility or previous orthostatic intolerance syndromes. Frequent comorbidities include migraine, irritable bowel syndrome, chronic fatigue syndrome, and autoimmune diseases [73,77][46][51]. Lightheadedness, tachycardia, dyspnea, headache, and poor concentration are the most frequent symptoms in PASC patients. Fatigue is the single most frequent symptom, although it is not specific for dysautonomia [33][18]. Severe symptoms may last for weeks or even months after infection and negatively affect overall quality of life [23][10].References
- Carfi, A.; Bernabei, R.; Landi, F. Persistent Symptoms in Patients After Acute COVID-19. JAMA 2020, 324, 603–605.
- Cheng, D.; Calderwood, C.; Skyllberg, E.; Ainley, A. Clinical characteristics and outcomes of adult patients admitted with COVID-19 in East London: A retrospective cohort analysis. BMJ Open Respir. Res. 2021, 8, e000813.
- Dani, M.; Dirksen, A.; Taraborrelli, P.; Torocastro, M.; Panagopoulos, D.; Sutton, R.; Lim, P.B. Autonomic dysfunction in ‘long COVID’: Rationale, physiology and management strategies. Clin. Med. 2021, 21, e63–e67.
- Ludvigsson, J.F. Case report and systematic review suggest that children may experience similar long-term effects to adults after clinical COVID-19. Acta Paediatr. 2021, 110, 914–921.
- Ostergaard, L. SARS CoV-2 related microvascular damage and symptoms during and after COVID-19: Consequences of capillary transit-time changes, tissue hypoxia and inflammation. Physiol. Rep. 2021, 9, e14726.
- Theoharides, T.C.; Cholevas, C.; Polyzoidis, K.; Politis, A. Long-COVID syndrome-associated brain fog and chemofog: Luteolin to the rescue. Biofactors 2021, 47, 232–241.
- Mazza, M.G.; De Lorenzo, R.; Conte, C.; Poletti, S.; Vai, B.; Bollettini, I.; Melloni, E.M.T.; Furlan, R.; Ciceri, F.; Rovere-Querini, P.; et al. Anxiety and depression in COVID-19 survivors: Role of inflammatory and clinical predictors. Brain Behav. Immun. 2020, 89, 594–600.
- Dennis, A.; Wamil, M.; Alberts, J.; Oben, J.; Cuthbertson, D.J.; Wootton, D.; Crooks, M.; Gabbay, M.; Brady, M.; Hishmeh, L.; et al. Multiorgan impairment in low-risk individuals with post-COVID-19 syndrome: A prospective, community-based study. BMJ Open 2021, 11, e048391.
- Osikomaiya, B.; Erinoso, O.; Wright, K.O.; Odusola, A.O.; Thomas, B.; Adeyemi, O.; Bowale, A.; Adejumo, O.; Falana, A.; Abdus-Salam, I.; et al. ‘Long COVID’: Persistent COVID-19 symptoms in survivors managed in Lagos State, Nigeria. BMC Infect. Dis. 2021, 21, 304.
- Peluso, M.J.; Kelly, J.D.; Lu, S.; Goldberg, S.A.; Davidson, M.C.; Mathur, S.; Durstenfeld, M.S.; Spinelli, M.A.; Hoh, R.; Tai, V.; et al. Rapid implementation of a cohort for the study of post-acute sequelae of SARS-CoV-2 infection/COVID-19. medRxiv 2021.
- Perlis, R.H.; Green, J.; Santillana, M.; Lazer, D.; Ognyanova, K.; Simonson, M.; Baum, M.A.; Quintana, A.; Chwe, H.; Druckman, J.; et al. Persistence of symptoms up to 10 months following acute COVID-19 illness. medRxiv 2021.
- Shouman, K.; Vanichkachorn, G.; Cheshire, W.P.; Suarez, M.D.; Shelly, S.; Lamotte, G.J.; Sandroni, P.; Benarroch, E.E.; Berini, S.E.; Cutsforth-Gregory, J.K.; et al. Autonomic dysfunction following COVID-19 infection: An early experience. Clin. Auton. Res. 2021.
- Gaber, T.A.K.; Ashish, A.; Unsworth, A. Persistent post-covid symptoms in healthcare workers. Occup. Med. 2021.
- Townsend, L.; Dyer, A.H.; Jones, K.; Dunne, J.; Mooney, A.; Gaffney, F.; O’Connor, L.; Leavy, D.; O’Brien, K.; Dowds, J.; et al. Persistent fatigue following SARS-CoV-2 infection is common and independent of severity of initial infection. PLoS ONE 2020, 15, e0240784.
- Auwaerter, P.G. The Race to Understand Post–COVID-19 Conditions. Ann. Intern. Med. 2021.
- O’Sullivan, O. Long-term sequelae following previous coronavirus epidemics. Clin. Med. 2021, 21, e68–e70.
- Halpin, S.J.; McIvor, C.; Whyatt, G.; Adams, A.; Harvey, O.; McLean, L.; Walshaw, C.; Kemp, S.; Corrado, J.; Singh, R.; et al. Postdischarge symptoms and rehabilitation needs in survivors of COVID-19 infection: A cross-sectional evaluation. J. Med. Virol. 2021, 93, 1013–1022.
- Townsend, L.; Moloney, D.; Finucane, C.; McCarthy, K.; Bergin, C.; Bannan, C.; Kenny, R.-A. Fatigue following COVID-19 infection is not associated with autonomic dysfunction. PLoS ONE 2021, 16, e0247280.
- van Campen, C.; Verheugt, F.W.A.; Rowe, P.C.; Visser, F.C. Cerebral blood flow is reduced in ME/CFS during head-up tilt testing even in the absence of hypotension or tachycardia: A quantitative, controlled study using Doppler echography. Clin. Neurophysiol. Pract. 2020, 5, 50–58.
- Escorihuela, R.M.; Capdevila, L.; Castro, J.R.; Zaragozà, M.C.; Maurel, S.; Alegre, J.; Castro-Marrero, J. Reduced heart rate variability predicts fatigue severity in individuals with chronic fatigue syndrome/myalgic encephalomyelitis. J. Transl. Med. 2020, 18, 4.
- Nelson, M.J.; Bahl, J.S.; Buckley, J.D.; Thomson, R.L.; Davison, K. Evidence of altered cardiac autonomic regulation in myalgic encephalomyelitis/chronic fatigue syndrome: A systematic review and meta-analysis. Medicine 2019, 98, e17600.
- Kedor, C.; Freitag, H.; Meyer-Arndt, L.; Wittke, K.; Zoller, T.; Steinbeis, F.; Haffke, M.; Rudolf, G.; Heidecker, B.; Volk, H.; et al. Chronic COVID-19 Syndrome and Chronic Fatigue Syndrome (ME/CFS) following the first pandemic wave in Germany—a first analysis of a prospective observational study. medRxiv 2021.
- Johansson, M.; Stahlberg, M.; Runold, M.; Nygren-Bonnier, M.; Nilsson, J.; Olshansky, B.; Bruchfeld, J.; Fedorowski, A. Long-Haul Post-COVID-19 Symptoms Presenting as a Variant of Postural Orthostatic Tachycardia Syndrome: The Swedish Experience. JACC Case Rep. 2021.
- Lo, Y.L. COVID-19, fatigue, and dysautonomia. J. Med. Virol. 2021, 93, 1213.
- Ghosh, R.; Roy, D.; Sengupta, S.; Benito-Leon, J. Autonomic dysfunction heralding acute motor axonal neuropathy in COVID-19. J. Neurovirol. 2020, 26, 964–966.
- Kanjwal, K.; Jamal, S.; Kichloo, A.; Grubb, B.P. New-onset Postural Orthostatic Tachycardia Syndrome Following Coronavirus Disease 2019 Infection. J. Innov. Card Rhythm. Manag. 2020, 11, 4302–4304.
- Moreno-Escobar, M.C.; Kataria, S.; Khan, E.; Subedi, R.; Tandon, M.; Peshwe, K.; Kramer, J.; Niaze, F.; Sriwastava, S. Acute transverse myelitis with Dysautonomia following SARS-CoV-2 infection: A case report and review of literature. J. Neuroimmunol. 2021, 353, 577523.
- Umapathi, T.; Poh, M.Q.W.; Fan, B.E.; Li, K.F.C.; George, J.; Tan, J.Y.L. Acute hyperhidrosis and postural tachycardia in a COVID-19 patient. Clin. Auton. Res. 2020, 30, 571–573.
- Loa, Y.L.; Leonga, H.N.; Hsua, L.Y.; Tana, T.T.; Kurupa, A.; Fook-Chonga, S.; Tana, B.H. Autonomic Dysfunction in Recovered Severe Acute Respiratory Syndrome Patients. Can. J. Neurol. Sci. 2005, 32, 264.
- Ståhlberg, M.; Reistam, U.; Fedorowski, A.; Villacorta, H.; Horiuchi, Y.; Bax, J.; Pitt, B.; Matskeplishvili, S.; Lüscher, T.F.; Weichert, I.; et al. Post-COVID-19 Tachycardia Syndrome: A distinct phenotype of Post-acute COVID-19 Syndrome. Am. J. Med. 2021.
- Fedorowski, A. Postural orthostatic tachycardia syndrome: Clinical presentation, aetiology and management. J. Intern. Med. 2019, 285, 352–366.
- Shaw, B.H.; Stiles, L.E.; Bourne, K.; Green, E.A.; Shibao, C.A.; Okamoto, L.E.; Garland, E.M.; Gamboa, A.; Diedrich, A.; Raj, V.; et al. The face of postural tachycardia syndrome—Insights from a large cross-sectional online community-based survey. J. Intern. Med. 2019, 286, 438–448.
- Raj, S.R.; Arnold, A.C.; Barboi, A.; Claydon, V.E.; Limberg, J.K.; Lucci, V.M.; Numan, M.; Peltier, A.; Snapper, H.; Vernino, S.; et al. Long-COVID postural tachycardia syndrome: An American Autonomic Society statement. Clin. Auton. Res. 2021.
- Davis, H.E.; Assaf, G.S.; McCorkell, L.; Wei, H.; Low, R.J.; Re’em, Y.; Redfield, S.; Austin, J.P.; Akrami, A. Characterizing Long COVID in an International Cohort: 7 Months of Symptoms and Their Impact. medRxiv 2021.
- Goldstein, D.S. The possible association between COVID-19 and postural tachycardia syndrome. Heart Rhythm 2021, 18, 508–509.
- Schofield, J.R. Persistent Antiphospholipid Antibodies, Mast Cell Activation Syndrome, Postural Orthostatic Tachycardia Syndrome and Post-COVID Syndrome: 1 Year On. Eur. J. Case Rep. Intern. Med. 2021, 8, 002378.
- Blitshteyn, S. Is postural orthostatic tachycardia syndrome (POTS) a central nervous system disorder? J. Neurol. 2021, 1–8.
- Bryarly, M.; Phillips, L.T.; Fu, Q.; Vernino, S.; Levine, B.D. Postural Orthostatic Tachycardia Syndrome: JACC Focus Seminar. J. Am. Coll. Cardiol. 2019, 73, 1207–1228.
- Yong, S.J. Persistent Brainstem Dysfunction in Long-COVID: A Hypothesis. ACS Chem. Neurosci. 2021, 12, 573–580.
- Lionetti, V.; Bollini, S.; Coppini, R.; Gerbino, A.; Ghigo, A.; Iaccarino, G.; Madonna, R.; Mangiacapra, F.; Miragoli, M.; Moccia, F.; et al. Understanding the heart-brain axis response in COVID-19 patients: A suggestive perspective for therapeutic development. Pharmacol. Res. 2021, 168, 105581.
- Barnden, L.R.; Crouch, B.; Kwiatek, R.; Burnet, R.; Mernone, A.; Chryssidis, S.; Scroop, G.; Del Fante, P. A brain MRI study of chronic fatigue syndrome: Evidence of brainstem dysfunction and altered homeostasis. NMR Biomed. 2011, 24, 1302–1312.
- Saeed, S.; Tadic, M.; Larsen, T.H.; Grassi, G.; Mancia, G. Coronavirus disease 2019 and cardiovascular complications: Focused clinical review. J. Hypertens. 2021, 39, 1282–1292.
- Barbieri, L.; Galli, F.; Conconi, B.; Gregorini, T.; Lucreziotti, S.; Mafrici, A.; Pravettoni, G.; Sommaruga, M.; Carugo, S. Takotsubo syndrome in COVID-19 era: Is psychological distress the key? J. Psychosom. Res. 2021, 140, 110297.
- Kir, D.; Beer, N.; De Marchena, E.J. Takotsubo cardiomyopathy caused by emotional stressors in the coronavirus disease 2019 (COVID-19) pandemic era. J. Card. Surg. 2021, 36, 764–769.
- Suzuki, H.; Yasuda, S.; Shimokawa, H. Brain-heart connection in Takotsubo syndrome before onset. Eur. Heart J. 2021, 42, 1909–1911.
- Vernino, S.; Stiles, L.E. Autoimmunity in postural orthostatic tachycardia syndrome: Current understanding. Auton. Neurosci. 2018, 215, 78–82.
- Kharraziha, I.; Axelsson, J.; Ricci, F.; Martino, G.D.; Persson, M.; Sutton, R.; Fedorowski, A.; Hamrefors, V. Serum Activity Against G Protein-Coupled Receptors and Severity of Orthostatic Symptoms in Postural Orthostatic Tachycardia Syndrome. J. Am. Heart Assoc. 2020, 9, e015989.
- Li, H.; Zhang, G.; Forsythe, E.; Okamoto, L.E.; Yu, X. Implications of Antimuscarinic Autoantibodies in Postural Tachycardia Syndrome. J. Cardiovasc. Transl. Res. 2021.
- Wallukat, G.; Hohberger, B.; Wenzel, K.; Fürst, J.; Schulze-Rothe, S.; Wallukat, A.; Hönicke, A.S.; Müller, J. Functional autoantibodies against G-protein coupled receptors in patients with persistent post-COVID-19 symptoms. J. Transl. Autoimmun. 2021, 4, 100100.
- Molina, V.; Shoenfeld, Y. Infection, vaccines and other environmental triggers of autoimmunity. Autoimmunity 2005, 38, 235–245.
- Dotan, A.; Muller, S.; Kanduc, D.; David, P.; Halpert, G.; Shoenfeld, Y. The SARS-CoV-2 as an instrumental trigger of autoimmunity. Autoimmun. Rev. 2021, 20, 102792.
- Kohno, R.; Cannom, D.S.; Olshansky, B.; Xi, S.C.; Krishnappa, D.; Adkisson, W.O.; Norby, F.L.; Fedorowski, A.; Benditt, D.G. Mast Cell Activation Disorder and Postural Orthostatic Tachycardia Syndrome: A Clinical Association. J. Am. Heart Assoc. 2021, e021002.
- Figueroa, J.J.; Cheshire, W.P.; Claydon, V.E.; Norcliffe-Kaufmann, L.; Peltier, A.; Singer, W.; Snapper, H.; Vernino, S.; Raj, S.R. Autonomic function testing in the COVID-19 pandemic: An American Autonomic Society position statement. Clin. Auton. Res. 2020, 30, 295–297.
- Brignole, M.; Moya, A.; de Lange, F.J.; Deharo, J.-C.; Elliott, P.M.; Fanciulli, A.; Fedorowski, A.; Furlan, R.; Kenny, R.A.; Martín, A.; et al. Practical Instructions for the 2018 ESC Guidelines for the diagnosis and management of syncope. Eur. Heart J. 2018, 39, e43–e80.
