White meat is the nutritional term for lighter-colored meat that contains less myoglobin than red meat, which contains a great deal. White meat includes poultry (e.g., chicken, duck, goose and turkey), fish, reptiles (e.g., land snail), amphibians (e.g., frog), crustaceans (e.g., shrimp and crab) and bivalves (e.g., oyster and clam), but it excludes all mammal flesh such as beef, pork, and lamb. White meat has high nutritional value and plays an important role in human diet. The production and sale of white meat need to meet specific quality and safety standards. Fluorescence spectroscopy, color imaging and multispectral imaging (MSI) have emerged as effective analytical methods for the non-destructive detection of quality attributes of various white meat products such as fish, shrimp, chicken, duck and goose.
- white meat
- multispectral imaging
- fluorescence spectroscopy
- convolutional neural network
- quality detection
1. Introduction
|
Technology |
Product |
Target Attributes |
Reference |
|---|---|---|---|
|
MSI |
Meat |
Adulteration |
Ropodi et al. [12] |
|
MSI, HSI |
Meat |
Defects |
Feng et al. [13] |
|
MSI |
Food |
Quality |
Su and Sun |
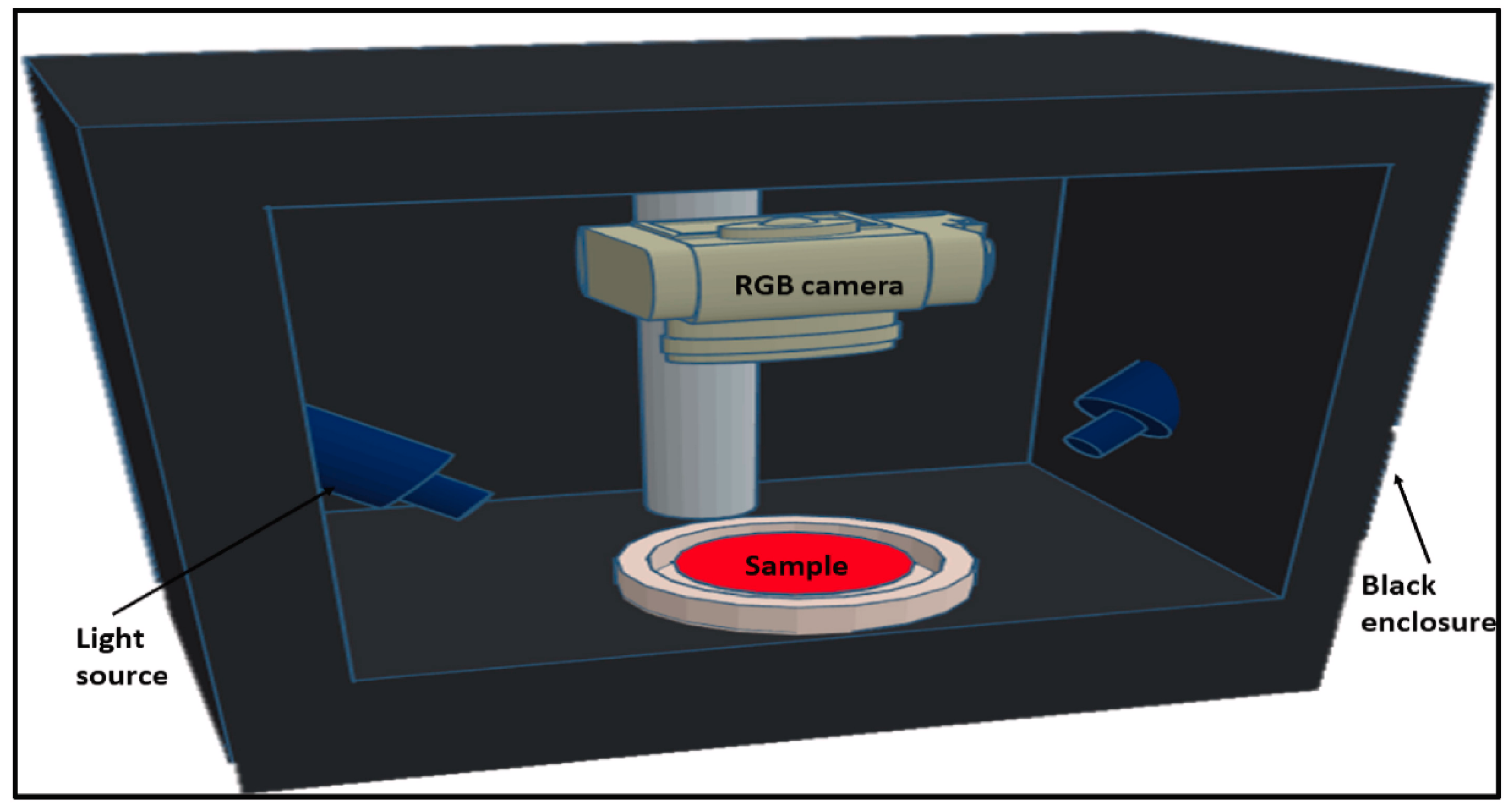
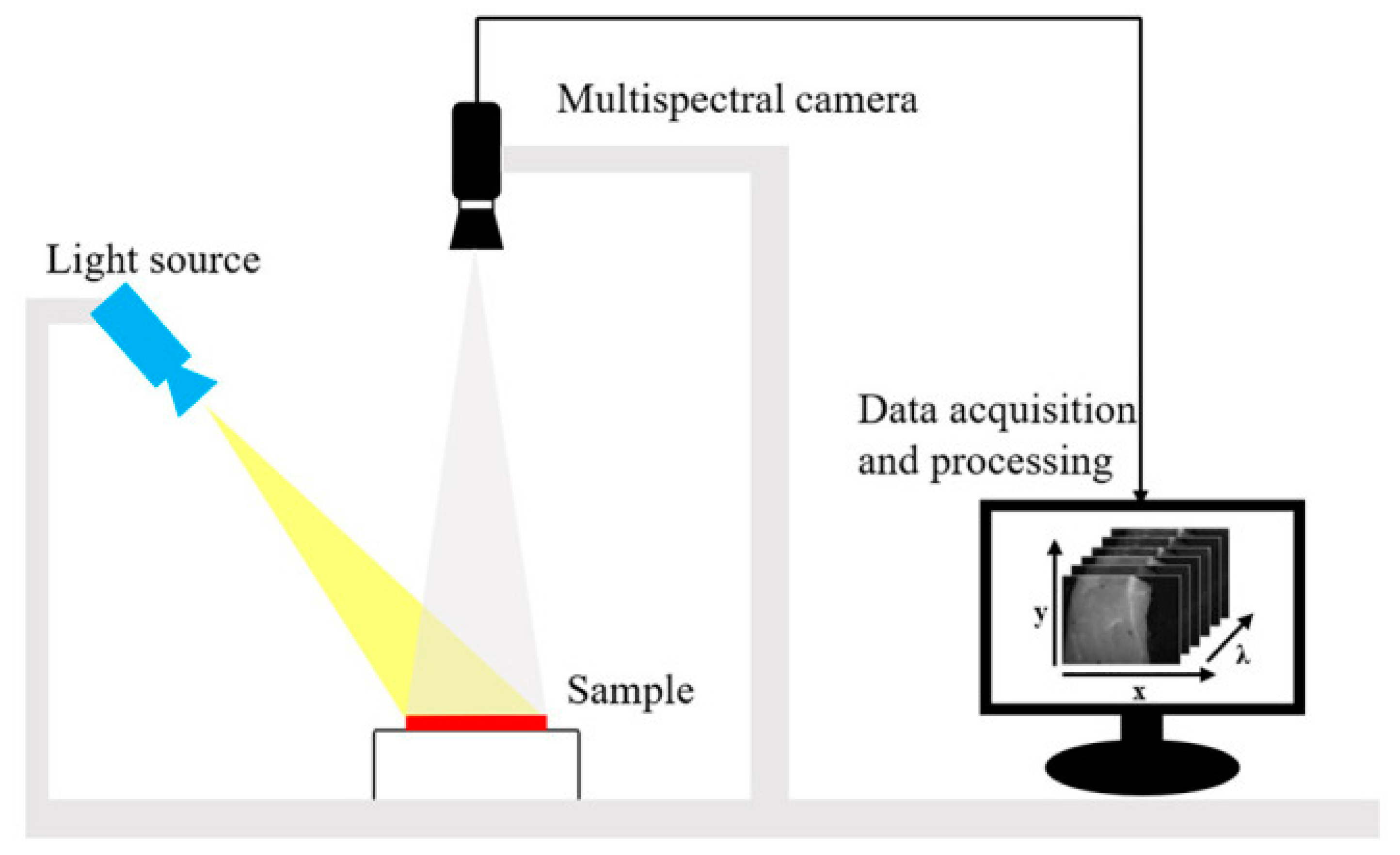
3. Quality Evaluation of White Meat
|
White Meat |
Module |
Quality Parameters |
Accuracy |
Reference |
||||
|---|---|---|---|---|---|---|---|---|
|
Fish |
MSI |
TVB-N, PPC |
R2p = 0.862 for TVB-N, R2p = 0.921 for PPC |
Khoshnoudi-Nia and Moosavi-Nasab [39], Khoshnoudi-Nia and Moosavi-Nasab [40] |
||||
|
Fish |
MSI |
TVC |
R2 = 0.62 |
Govari, et al. [41] |
||||
[ | ] | |||||||
|
Fish |
MSI |
TVC |
R2 = 0.683 |
Fengou, et al. [42] |
MSI, IRS, SERS, LIBS and HSI |
|||
|
Fish | Food |
MSI Quality |
Astaxanthin concentration Wang et al. [15] |
|||||
R | 2 | = 0.86 |
Dissing, et al. [43] |
MSI, HSI and VS |
Food |
|||
|
Fish |
MSI |
Authenticity, quality and safety |
TVB-N, TBARS, K Ropodi et al. [16] |
|||||
R | 2 | p = 0.922 for TVB-N, R2p = 0.867 for TBARS, R2p = 0.936 for K |
Cheng, et al. [44] |
Fluorescence spectroscopy |
||||
|
Fish |
Food |
Quality |
MSI Karoui and Blecker [ |
A ‘standard freshness index’ of K 17] |
||||
R | 2 | = 0.94, |
Omwange, et al. [45] |
Fluorescence spectroscopy |
Food |
|||
|
Fish |
Fluorescence spectroscopy |
Quality |
A ‘standard freshness index’ of K Strasburg and Ludescher [18] |
|||||
R | 2 | = 0.92 |
Omwange, et al. [46] |
Visible/Infrared, Raman and Fluorescence spectroscopy |
||||
|
Fish |
Raw and processed food |
Fluorescence spectroscopy Quality |
A ‘standard freshness index’ of K He and Sun [19] |
|||||
R | 2 | = 0.95 |
Liao, et al. [47] |
Fluorescence spectroscopy |
Food |
Quality |
||
|
Fish |
Fluorescence spectroscopy | Ahmad et al. [20] |
||||||
AEC; |
NADH |
R2 = 0.90 for AEC, R2 = 0.85 for NADH |
Rahman, et al. [ |
Fluorescence spectroscopy |
Dairy products |
Quality and safety |
Shaikh and O’Donnell [21] |
|
] | ||||||||
|
Fish |
Fluorescence spectroscopy |
NADH |
90.5% |
Hassoun and Karoui [49] |
Fluorescence spectroscopy |
Fresh and frozen-thawed muscle foods |
Muscle classification |
|
|
Fish |
RGB imaging |
Classification performance |
Hassoun [22] |
|||||
99.5% | Park, et al. [50] |
RGB-Imaging |
||||||
|
Fish |
Meat |
RGB imaging Quality and safety |
Astaxanthin concentration Taheri-Garavand et al. [23] |
|||||
R | 2 | = 0.66 |
Dissing et al. [43 |
RGB-Imaging |
Fish |
Quality |
Dowlati et al. [24] |
|
|
RGB-Imaging |
Food |
Quality |
Gomes and Leta [25] |
|||||
|
RGB-Imaging |
Food |
Quality |
Amani et al. [26] |
MSI––Multispectral imaging; HSI––Hyperspectral imaging; IRS––Infrared spectroscopy; SERS––Surface-Enhanced Raman Spectroscopy; LIBS––Laser induced breakdown spectroscopy; VS––Vibrational Spectroscopy.
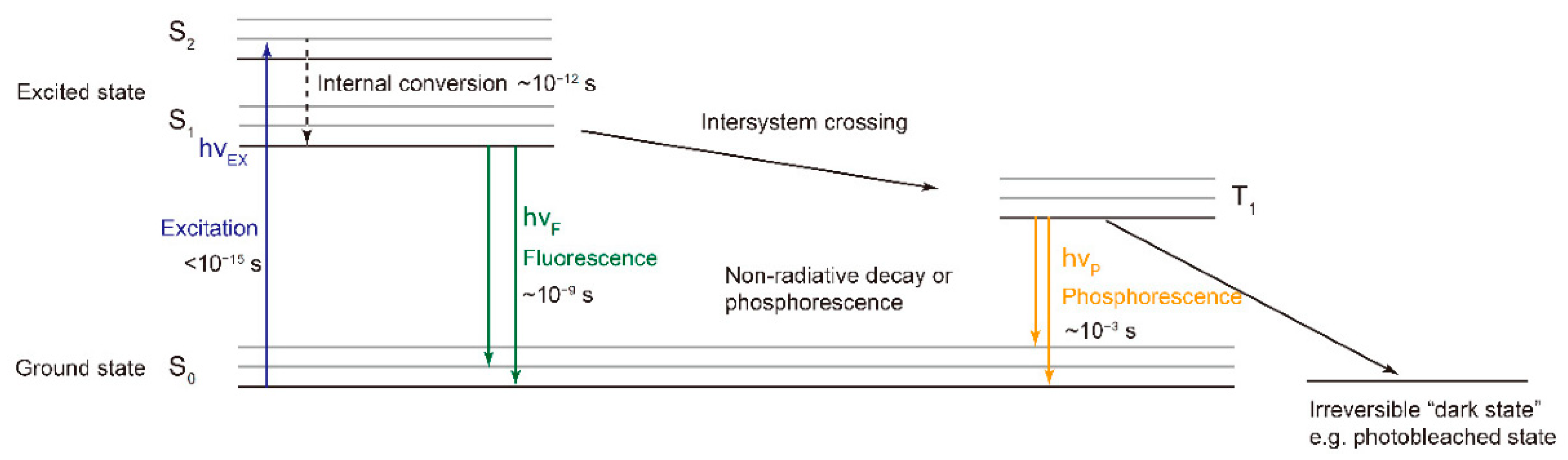
2. Fluorescence Spectroscopy, RGB- and Multispectral-Imaging
] | ||||
Fish | ||||
RGB imaging | ||||
Freshness of tuna meat cuts | 86.67% |
Lugatiman, et al. [51] |
||
|
Fish |
RGB imaging |
The main color of the sample |
75% |
Mateo, et al. [52] |
|
Fish |
RGB imaging |
Texture features |
86.3% |
Gu, et al. [53] |
|
Fish |
RGB imaging |
Color of Salmon Fillets |
R = 0.95 |
Quevedo, et al. [54] |
|
Fish |
RGB imaging |
Gill and eye color changes in the sparus aurata |
R2 = 0.994 |
Dowlati, et al. [55] |
|
Fish |
RGB imaging |
Body color of carp |
94.97% |
Taheri-Garavand, et al. [56] |
|
Fish |
RGB imaging |
Freshness |
98.2% |
Rocculi, et al. [57] |
|
Shrimp |
Fluorescence spectroscopy |
4-hexylresorcinol |
81.6% |
Jonker and Dekker [58] |
|
Shrimp |
Fluorescence spectroscopy |
K, pH |
R2 = 0.80 |
Rahman, et al. [59] |
|
Shrimp |
RGB imaging |
pH |
100% |
Witjaksono, et al. [60] |
|
Shrimp |
RGB imaging |
Identification accuracy of the proposed ShrimpNet for shrimp |
95.48% |
Hu, et al. [61] |
|
Shrimp |
RGB imaging |
Shrimp dehydration levels |
R = 0.86 |
Mohebbi, et al. [62] |
|
Shrimp |
RGB imaging |
Color changes in the head, legs and tail of pacific white shrimp (litopenaeus vannamei) |
90% |
Ghasemi-Varnamkhasti, et al. [63] |
|
Chicken |
Fluorescence spectroscopy |
Hydroxyproline concentration |
R2 = 0.82 |
Monago-Maraña, et al. [64] |
|
Chicken |
MSI |
Skin tumors |
86% |
Chao, et al. [65] |
|
Chicken |
MSI |
TVC |
90.4% |
Spyrelli, et al. [66] |
|
Chicken |
MSI |
pork-chicken adulteration |
90.00% for fresh samples, 86.67% for frozen-thawed samples |
Fengou, et al. [67] |
|
Chicken |
MSI |
Sepsis in chickens |
98.6% for septic chickens, 96.3% for healthy chickens |
Yang, et al. [68] |
|
Chicken |
MSI |
Contamination detection |
96% |
Park, et al. [69] |
|
Chicken |
MSI |
Chicken heart disease characterization |
100% |
Chao, et al. [70] |
|
Chicken |
MSI; Fluorescence spectroscopy |
Contamination detection |
92.5% |
Seo, et al. [71] |
|
Chicken |
Fluorescence spectroscopy |
Lipid oxidation |
R = 0.73 |
Gatellier, et al. [72] |
|
Chicken |
Fluorescence spectroscopy |
P. aeruginosa concentration |
96% |
Abdel-Salam, et al. [73] |
|
Chicken |
Fluorescence spectroscopy |
chicken meat tenderness |
R = 0.870 |
Yu, et al. [74] |
|
Chicken |
Fluorescence spectroscopy |
Contamination detection |
96.6% |
Cho, et al. [75] |
|
Chicken |
Fluorescence spectroscopy |
Measurement of lipid oxidation |
98% |
Wold and Kvaal [76] |
|
Chicken |
RGB imaging |
Avian flu infected chickens |
97.43% |
Cuan, et al. [77] |
|
Chicken |
RGB im-aging |
Color |
94% |
Yumono, et al. [78] |
|
Chicken |
RGB im-aging |
Freshness |
R = 0.987 |
Taheri-Garavand, et al. [79] |
|
Duck |
Fluorescence spectroscopy |
Gentamicin Residual in Duck Meat |
R = 0.996 |
Wang, et al. [80] |
|
Duck |
Fluorescence spectroscopy |
Doxycycline content in duck meat |
R = 0.998 |
Wang, et al. [81] |
|
Duck |
Fluorescence spectroscopy |
Carbaryl residue in duck meat |
R = 0.976 |
Xiao et al. [10] |
|
Duck |
Fluorescence spectroscopy |
Tetracycline content |
R = 0.952 |
Zhao, et al. [82] |
|
Duck |
Fluorescence spectroscopy |
Triazophos content |
R2p = 0.974, |
Zhao, et al. [83] |
|
Duck |
Fluorescence spectroscopy |
Neomycin residue |
R = 0.999 |
Jiang, et al. [84] |
|
Duck |
Fluorescence spectroscopy |
Carbofuran residue |
R2p = 0.999 |
XIAO, et al. [85] |
References
- Xiong, Z.; Xie, A.; Sun, D.-W.; Zeng, X.-A.; Liu, D. Applications of Hyperspectral Imaging in Chicken Meat Safety and Quality Detection and Evaluation: A Review. Crit. Rev. Food Sci. Nutr. 2014, 55, 1287–1301.
- Aykan, N.F. Red meat subtypes and colorectal cancer risk. Int. J. Cancer 2015, 137, 1788.
- Tsagkatakis, G.; Nikolidakis, S.; Petra, E.; Kapantagakis, A.; Grigorakis, K.; Katselis, G.; Vlahos, N.; Tsakalides, P.J.E.I. Fish Freshness Estimation though analysis of Multispectral Images with Convolutional Neural Networks. IST Int. Symp. Electron. Imaging 2020, 2020, 171.
- Schneider, M.J.; Vazquez-Moreno, L.; Bermudez-Almada, M.D.C.; Guardado, R.B.; Ortega-Nieblas, M.J.J.O.A.I. Multiresidue Determination of Fluoroquinolones in Shrimp by Liquid Chromatography-Fluorescence-Mass Spectrometryn. J. AOAC Int. 2005, 88, 1160–1166.
- Xiong, Z.; Sun, D.-W.; Pu, H.; Gao, W.; Dai, Q. Applications of emerging imaging techniques for meat quality and safety detection and evaluation: A review. Crit. Rev. Food Sci. Nutr. 2017, 57, 755–768.
- Kamruzzaman, M.; Makino, Y.; Oshita, S. Non-invasive analytical technology for the detection of contamination, adulteration, and authenticity of meat, poultry, and fish: A review. Anal. Chim. Acta 2015, 853, 19–29.
- Peng, Y.; Dhakal, S. Optical Methods and Techniques for Meat Quality Inspection. Trans. ASABE 2015, 58, 1371–1386.
- Qin, J.; Kim, M.S.; Chao, K.; Dhakal, S.; Lee, H.; Cho, B.-K.; Mo, C. Detection and quantification of adulterants in milk powder using a high-throughput Raman chemical imaging technique. Food Addit. Contam. Part A 2017, 34, 152–161.
- Yang, C.-C.; Chao, K.; Chen, Y.-R. Development of multispectral image processing algorithms for identification of wholesome, septicemic, and inflammatory process chickens. J. Food Eng. 2005, 69, 225–234.
- Xiao, H.-B.; Liu, M.-H.; Yuan, H.-C.; Xu, J.; Zhao, J.-H. Study on determination of carbaryl content in duck meat based on synchronous fluorescence spectroscopy. Spectrosc. Spectr. Anal. 2012, 32, 3058–3062.
- Wang, W.; Peng, Y.; Sun, H.; Zheng, X.; Wei, W. Spectral Detection Techniques for Non-Destructively Monitoring the Quality, Safety, and Classification of Fresh Red Meat. Food Anal. Methods 2018, 11, 2707–2730.
- Ropodi, A.I.; Panagou, E.Z.; Nychas, G.-J.E. Multispectral imaging (MSI): A promising method for the detection of minced beef adulteration with horsemeat. Food Control 2017, 73, 57–63.
- Feng, C.-H.; Makino, Y.; Oshita, S.; Martín, J.F.G. Hyperspectral imaging and multispectral imaging as the novel techniques for detecting defects in raw and processed meat products: Current state-of-the-art research advances. Food Control 2018, 84, 165–176.
- Su, W.-H.; Sun, D.-W. Multispectral Imaging for Plant Food Quality Analysis and Visualization. Compr. Rev. Food Sci. Food Saf. 2018, 17, 220–239.
- Wang, K.; Pu, H.; Sun, D.-W. Emerging Spectroscopic and Spectral Imaging Techniques for the Rapid Detection of Microorganisms: An Overview. Compr. Rev. Food Sci. Food Saf. 2018, 17, 256–273.
- Ropodi, A.; Panagou, E.; Nychas, G.-J. Data mining derived from food analyses using non-invasive/non-destructive analytical techniques; determination of food authenticity, quality & safety in tandem with computer science disciplines. Trends Food Sci. Technol. 2016, 50, 11–25.
- Karoui, R.; Blecker, C. Fluorescence Spectroscopy Measurement for Quality Assessment of Food Systems—A Review. Food Bioprocess Technol. 2011, 4, 364–386.
- Strasburg, G.M.; Ludescher, R. Theory and applications of fluorescence spectroscopy in food research. Trends Food Sci. Technol. 1995, 6, 69–75.
- He, H.-J.; Sun, D.-W. Microbial evaluation of raw and processed food products by Visible/Infrared, Raman and Fluorescence spectroscopy. Trends Food Sci. Technol. 2015, 46, 199–210.
- Ahmad, M.H.; Sahar, A.; Hitzmann, B. Fluorescence Spectroscopy for the Monitoring of Food Processes. Meas. Modeling Autom. Adv. Food Processing 2017, 161, 121–151.
- Shaikh, S.; O’Donnell, C. Applications of fluorescence spectroscopy in dairy processing: A review. Curr. Opin. Food Sci. 2017, 17, 16–24.
- Hassoun, A. Exploring the Potential of Fluorescence Spectroscopy for the Discrimination between Fresh and Frozen-Thawed Muscle Foods. Photochem 2021, 1, 247–263.
- Taheri-Garavand, A.; Fatahi, S.; Omid, M.; Makino, Y. Meat quality evaluation based on computer vision technique: A review. Meat Sci. 2019, 156, 183–195.
- Dowlati, M.; de la Guardia, M.; Mohtasebi, S.S. Application of machine-vision techniques to fish-quality assessment. TrAC Trends Anal. Chem. 2012, 40, 168–179.
- Gomes, J.F.S.; Leta, F.R. Applications of computer vision techniques in the agriculture and food industry: A review. Eur. Food Res. Technol. 2012, 235, 989–1000.
- Amani, H.; Badak-Kerti, K.; Khaneghah, A.M. Current progress in the utilization of smartphone-based imaging for quality assessment of food products: A review. Crit. Rev. Food Sci. Nutr. 2020, 1–13.
- Dufour, E.; Frencia, J.P.; Kane, E. Development of a rapid method based on front-face fluorescence spectroscopy for the monitoring of fish freshness. Food Res. Int. 2003, 36, 415–423.
- Hassoun, A.; Sahar, A.; Lakhal, L.; Aït-Kaddour, A. Fluorescence spectroscopy as a rapid and non-destructive method for monitoring quality and authenticity of fish and meat products: Impact of different preservation conditions. LWT 2019, 103, 279–292.
- Yokota, H.; Fukasawa, A.; Hirano, M.; Ide, T. Low-Light Photodetectors for Fluorescence Microscopy. Appl. Sci. 2021, 11, 2773.
- Karbiwnyk, C.M.; Carr, L.E.; Turnipseed, S.B.; Andersen, W.C.; Miller, K.E. Determination of quinolone residues in shrimp using liquid chromatography with fluorescence detection and residue confirmation by mass spectrometry. Anal. Chim. Acta 2007, 596, 257–263.
- Liu, Z.; Zhong, Y.; Hu, Y.; Yuan, L.; Luo, R.; Chen, D.; Wu, M.; Huang, H.; Li, Y. Fluorescence strategy for sensitive detection of adenosine triphosphate in terms of evaluating meat freshness. Food Chem. 2019, 270, 573–578.
- Mohd Ali, M.; Hashim, N.; Khairunniza-Bejo, S.; Shamsudin, R.; Wan Sembak, W. RGB imaging system for monitoring quality changes of seedless watermelon during storage. In Proceedings of the III International Conference on Agricultural and Food Engineering 1152, Kuala Lumpur, Malaysia, 13 May 2016; pp. 361–366.
- Rady, A.M.; Adedeji, A.; Watson, N.J.J.J.O.A.; Research, F. Feasibility of utilizing color imaging and machine learning for adulteration detection in minced meat. J. Agric. Food Res. 2021, 6, 100251.
- Barbin, D.F.; Mastelini, S.M.; Barbon, S.; Campos, G.F.C.; Barbon, A.P.A.C.; Shimokomaki, M. Digital image analyses as an alternative tool for chicken quality assessment. Biosyst. Eng. 2016, 144, 85–93.
- Taghizadeh, M.; Gowen, A.A.; O’Donnell, C.P. Comparison of hyperspectral imaging with conventional RGB imaging for quality evaluation of Agaricus bisporus mushrooms. Biosyst. Eng. 2011, 108, 191–194.
- Bandara, W.; Prabhath, G.; Dissanayake, D.; Herath, H.; Godaliyadda, G.; Ekanayake, M.; Vithana, S.; Demini, S.; Madhujith, T. A multispectral imaging system to assess meat quality. In Proceedings of the 2018 IEEE Region 10 Humanitarian Technology Conference (R10-HTC), Malambe, Sri Lanka, 6–8 December 2018; pp. 1–6.
- Jayasundara, D.; Ramanayake, L.; Senarath, N.; Herath, S.; Godaliyadda, R.; Ekanayake, P.; Herath, V.; Ariyawansha, S. Multispectral Imaging for Automated Fish Quality Grading. In Proceedings of the 2020 IEEE 15th International Conference on Industrial and Information Systems (ICIIS), Rupnagar, India, 26–28 November 2020; pp. 321–326.
- Li, A.; Li, C.; Gao, M.; Yang, S.; Liu, R.; Chen, W.; Xu, K. Beef Cut Classification Using Multispectral Imaging and Machine Learning Method. Front. Nutr. 2021, 8.
- Khoshnoudi-Nia, S.; Moosavi-Nasab, M. Prediction of various freshness indicators in fish fillets by one multispectral imaging system. Sci. Rep. 2019, 9, 14704.
- Khoshnoudi-Nia, S.; Moosavi-Nasab, M. Nondestructive Determination of Microbial, Biochemical, and Chemical Changes in Rainbow Trout (Oncorhynchus mykiss) During Refrigerated Storage Using Hyperspectral Imaging Technique. Food Anal. Methods 2019, 12, 1635–1647.
- Govari, M.; Tryfinopoulou, P.; Parlapani, F.; Boziaris, I.S.; Panagou, E.Z.; Nychas, G.-J. Quest of Intelligent Research Tools for Rapid Evaluation of Fish Quality: FTIR Spectroscopy and Multispectral Imaging Versus Microbiological Analysis. Foods 2021, 10, 264.
- Fengou, L.-C.; Lianou, A.; Tsakanikas, P.; Gkana, E.N.; Panagou, E.Z.; Nychas, G.-J.E. Evaluation of Fourier transform infrared spectroscopy and multispectral imaging as means of estimating the microbiological spoilage of farmed sea bream. Food Microbiol. 2019, 79, 27–34.
- Dissing, B.S.; Nielsen, M.C.E.; Ersbøll, B.K.; Frosch, S. Multispectral Imaging for Determination of Astaxanthin Concentration in Salmonids. PLoS ONE 2011, 6, e19032.
- Cheng, J.-H.; Sun, D.-W.; Qu, J.-H.; Pu, H.-B.; Zhang, X.-C.; Song, Z.; Chen, X.; Zhang, H. Developing a multispectral imaging for simultaneous prediction of freshness indicators during chemical spoilage of grass carp fish fillet. J. Food Eng. 2016, 182, 9–17.
- Omwange, K.A.; Saito, Y.; Zichen, H.; Khaliduzzaman, A.; Kuramoto, M.; Ogawa, Y.; Kondo, N.; Suzuki, T. Evaluating Japanese dace (Tribolodon hakonensis) fish freshness during storage using multispectral images from visible and UV excited fluorescence. LWT 2021, 151, 112207.
- Omwange, K.A.; Al Riza, D.F.; Sen, N.; Shiigi, T.; Kuramoto, M.; Ogawa, Y.; Kondo, N.; Suzuki, T. Fish freshness monitoring using UV-fluorescence imaging on Japanese dace (Tribolodon hakonensis) fisheye. J. Food Eng. 2020, 287, 110111.
- Liao, Q.H.; Suzuki, T.; Yasushi, K.; Al Riza, D.F.; Kuramoto, M.; Kondo, N. Monitoring Red Sea Bream Scale Fluorescence as a Freshness Indicator. Fishes 2017, 2, 10.
- Rahman, M.M.; Shibata, M.; ElMasry, G.; Nakazawa, N.; Nakauchi, S.; Hagiwara, T.; Osako, K.; Okazaki, E. Expeditious prediction of post-mortem changes in frozen fish meat using three-dimensional fluorescence fingerprints. Biosci. Biotechnol. Biochem. 2019, 83, 901–913.
- Hassoun, A.; Karoui, R. Front-face fluorescence spectroscopy coupled with chemometric tools for monitoring fish freshness stored under different refrigerated conditions. Food Control 2015, 54, 240–249.
- Park, J.-H.; Hwang, K.-B.; Park, H.-M.; Choi, Y.-K.J.J.O.T.K.I.O.I.; Engineering, C. Application of CNN for fish species classification. J. Korea Inst. Inf. Commun. Eng. 2019, 23, 39–46.
- Lugatiman, K.; Fabiana, C.; Echavia, J.; Adtoon, J.J. Tuna Meat Freshness Classification through Computer Vision. In Proceedings of the 2019 IEEE 11th International Conference on Humanoid, Nanotechnology, Information Technology, Communication and Control, Environment, and Management (HNICEM), Laoag, Philippines, 29 November–1 December 2019; pp. 1–6.
- Mateo-Aroca, A.; Soto, F.; Villarejo, J.A.; Roca-Dorda, J.; De La Gándara, F.; García, A. Quality analysis of tuna meat using an automated color inspection system. Aquac. Eng. 2006, 35, 1–13.
- Gu, J.; He, N.; Wu, X. A new detection method for fish freshness. In Proceedings of the 2014 Seventh International Symposium on Computational Intelligence and Design, Hangzhou, China, 13–14 December 2014; pp. 555–558.
- Quevedo, R.A.; Aguilera, J.M.; Pedreschi, F. Color of Salmon Fillets By Computer Vision and Sensory Panel. Food Bioprocess Technol. 2008, 3, 637–643.
- Dowlati, M.; Mohtasebi, S.S.; Omid, M.; Razavi, S.H.; Jamzad, M.; de la Guardia, M. Freshness assessment of gilthead sea bream (Sparus aurata) by machine vision based on gill and eye color changes. J. Food Eng. 2013, 119, 277–287.
- Taheri-Garavand, A.; Fatahi, S.; Banan, A.; Makino, Y. Real-time nondestructive monitoring of Common Carp Fish freshness using robust vision-based intelligent modeling approaches. Comput. Electron. Agric. 2019, 159, 16–27.
- Rocculi, P.; Cevoli, C.; Tappi, S.; Genovese, J.; Urbinati, E.; Picone, G.; Fabbri, A.; Capozzi, F.; Dalla Rosa, M. Freshness assessment of European hake (Merluccius merluccius) through the evaluation of eye chromatic and morphological characteristics. Food Res. Int. 2019, 115, 234–240.
- Jonker, K.M.; Dekker, C.P. Determination of 4-Hexylresorcinol in Shrimp by Liquid Chromatography with Fluorescence Detection. J. AOAC Int. 2000, 83, 241–244.
- Rahman, M.; Bui, M.V.; Shibata, M.; Nakazawa, N.; Rithu, M.N.A.; Yamashita, H.; Sadayasu, K.; Tsuchiyama, K.; Nakauchi, S.; Hagiwara, T.; et al. Rapid noninvasive monitoring of freshness variation in frozen shrimp using multidimensional fluorescence imaging coupled with chemometrics. Talanta 2021, 224, 121871.
- Witjaksono, G.; Hussin, N.H.F.B.M.; Rabih, A.A.S.; Alfa, S. Real time chromametry measurement for food quality detection using mobile device. In Proceedings of the IOP Conference Series: Materials Science and Engineering, Busan, Korea, 25–27 August 2017; p. 12024.
- Hu, W.-C.; Wu, H.-T.; Zhang, Y.-F.; Zhang, S.-H.; Lo, C.-H. Shrimp recognition using ShrimpNet based on convolutional neural network. J. Ambient. Intell. Humaniz. Comput. 2020, 1–8.
- Mohebbi, M.; Akbarzadeh-T, M.-R.; Shahidi, F.; Moussavi, M.; Ghoddusi, H.-B. Computer vision systems (CVS) for moisture content estimation in dehydrated shrimp. Comput. Electron. Agric. 2009, 69, 128–134.
- Ghasemi-Varnamkhasti, M.; Goli, R.; Forina, M.; Mohtasebi, S.S.; Shafiee, S.; Naderi-Boldaji, M. Application of Image Analysis Combined with Computational Expert Approaches for Shrimp Freshness Evaluation. Int. J. Food Prop. 2016, 19, 2202–2222.
- Monago-Maraña, O.; Wold, J.P.; Rødbotten, R.; Dankel, K.R.; Afseth, N.K. Raman, near-infrared and fluorescence spectroscopy for determination of collagen content in ground meat and poultry by-products. LWT 2021, 140, 110592.
- Chao, K.; Mehl, P.M.; Kim, M.S.; Chen, Y.-R. Detection of chicken skin tumors by mutlispectral imaging. In Proceedings of the Photonic Detection and Intervention Technologies for Safe Food, Bellingham, WA, USA, 5–6 November 2000; pp. 214–223.
- Spyrelli, E.D.; Ozcan, O.; Mohareb, F.; Panagou, E.Z.; Nychas, G.J.E. Spoilage assessment of chicken breast fillets by means of fourier transform infrared spectroscopy and multispectral image analysis. Curr. Res. Food Sci. 2021, 4, 121–131.
- Fengou, L.-C.; Lianou, A.; Tsakanikas, P.; Mohareb, F.; Nychas, G.-J.E. Detection of Meat Adulteration Using Spectroscopy-Based Sensors. Foods 2021, 10, 861.
- Yang, C.-C.; Chao, K.; Chen, Y.-R.; Kim, M.S. Application of Multispectral Imaging for Identification of Systemically Diseased Chicken. In Proceedings of the 2004 ASAE Annual Meeting, Ottawa, Canada, 1–4 August 2004; p. 1.
- Park, B.; Kise, M.; Lawrence, K.C.; Windham, W.R.; Smith, D.P.; Thai, C.N. Real-time multispectral imaging system for online poultry fecal inspection using unified modeling language. Sens. Instrum. Food Qual. Saf. 2007, 1, 45–54.
- Chao, K.; Chen, Y.R.; Hruschka, W.R.; Park, B. Chicken Heart Disease Characterization by Multi-spectral Imaging. Appl. Eng. Agric. 2001, 17, 99–106.
- Seo, Y.; Lee, H.; Mo, C.; Kim, M.S.; Baek, I.; Lee, J.; Cho, B.-K. Multispectral Fluorescence Imaging Technique for On-Line Inspection of Fecal Residues on Poultry Carcasses. Sensors 2019, 19, 3483.
- Gatellier, P.; Gomez, S.; Gigaud, V.; Berri, C.; Le Bihan-Duval, E.; Santé-Lhoutellier, V. Use of a fluorescence front face technique for measurement of lipid oxidation during refrigerated storage of chicken meat. Meat Sci. 2007, 76, 543–547.
- Abdel-Salam, Z.; Abdel-Salam, S.A.M.; Harith, M.A. Application of Laser Spectrochemical Analytical Techniques to Follow Up Spoilage of White Meat in Chicken. Food Anal. Methods 2017, 10, 2365–2372.
- Yu, F.; Xue, L.; Liu, M.-h.; Li, J. Preliminary study of laser-induced fluorescence spectroscopy detect chicken meat tenderness. In Proceedings of the 2nd International Conference on Information Science and Engineering, Hangzhou, China, 4–6 December 2010; pp. 6771–6774.
- Cho, B.-K.; Kim, M.S.; Chao, K.; Lefcourt, A.M.; Lawrence, K.; Park, B. Detection of Fecal Residue on Poultry Carcasses by Laser Induced Fluorescence Imaging. J. Food Sci. 2009, 74, E154–E159.
- Wold, J.P.; Kvaal, K. Mapping Lipid Oxidation in Chicken Meat by Multispectral Imaging of Autofluorescence. Appl. Spectrosc. 2000, 54, 900–909.
- Cuan, K.X.; Zhang, T.M.; Huang, J.D.; Fang, C.; Guan, Y. Detection of avian influenza-infected chickens based on a chicken sound convolutional neural network. Comput. Electron. Agric. 2020, 178, 105688.
- Yumono, F.; Subroto, I.M.I.; Prasetyowati, S.A.D. Artificial Neural Network for Healthy Chicken Meat Identification. IAES Int. J. Artif. Intell. (IJ-AI) 2018, 7, 63–70.
- Taheri-Garavand, A.; Fatahi, S.; Shahbazi, F.; De La Guardia, M. A nondestructive intelligent approach to real-time evaluation of chicken meat freshness based on computer vision technique. J. Food Process. Eng. 2019, 42, e13039.
- Wang, X.; Xu, J.; Liu, M.H.; Zhao, J.H.; Hong, Q. Determination of Gentamicin Residual in Duck Meat Using Fluorescence Analysis Method. Adv. Mater. Res. 2014, 1033–1034, 638–642.
- Wang, P.; Hong, Q.; Liu, M.; Yuan, H.; Peng, Y.; Zhao, J.; Pengwei, W.; Qian, H.; Muhua, L.; Haichao, Y.; et al. Rapid detection of doxycycline content in duck meat by using silver nanoparticles and alkylphenols polyoxyethylene enhanced fluorescence of europium complex. Spectrosc. Lett. 2016, 49, 563–567.
- Zhao, J.-H.; Yuan, H.-C.; Liu, M.-H.; Xiao, H.-B.; Hong, Q.; Xu, J. Rapid determination of tetracycline content in duck meat using particle swarm optimization algorithm and synchronous fluorescence spectrum. Spectrosc. Spect. Anal. 2013, 33, 3050–3054.
- Zhao, J.H.; Bin Xiao, H.; Yuan, H.C.; Hong, Q.; Liu, M.H. Application of Three-Dimensional Fluorescence Spectroscopy Coupled with ATLD in Rapid Determination of Triazophos Content in Duck Meat. Appl. Mech. Mater. 2014, 651–653, 362–366.
- Jiang, X.; Muhua, L.; Haichao, Y. A study on determination of neomycin residue in duck by fluorescence method. Acta Agric. Univ. Jiangxiensis 2013, 35, 635–640.
- Xiao, H.-B.; Zhao, J.-H.; Yuan, H.-C.; Xu, J.; Li, Q.; Liu, M.-H. Prediction of Carbofuran Residue in Duck Meat by Synchronous Fluorescence Spectroscopy Based on Support Vector Regression (SVR). J. Instrum. Anal. 2013, 3, 357–361.