Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Congling Shi and Version 3 by Peter Tang.
Aerogel is generated by the replacement of liquid inside a gel with gas by freeze-drying or supercritical drying technique. Three-dimensional graphene-based aerogels (3D GAs), combining the intrinsic properties of graphene and 3D porous structure, can be prepared via self-assembly method, template-guided method and sol-gel process. They have attracted increasing research interest in varied fields with potential applications in photoredox catalysis, biomedicine, energy storage, supercapacitor or other single aspect.
- three-dimensional
- graphene-based aerogels
- synthetic strategy
- application
1. Introduction
Aerogel, generated by the replacement of liquid inside a gel with gas by freeze-drying or supercritical drying technique, was first presented by S. Kistler in the 1930s [1]. As the lightest solid porous material in the world, it has attracted wide attention owing to its three-dimensional (3D) network structure, high specific surface area, extremely low-density, and thermal conductivity [2][3][4][2,3,4]. The precursor of aerogel can be selected among organic polymers [5], inorganic materials [6], and polymeric hybrid materials [7]. At the beginning, it underwent a long slow-development-stage because of the difficulty in synthesis and the lack of application. However, aerogels with richer types and application fields have been flourishing in the past decade. Such a porous material can be an enticing prospect for its application in the fields of aerospace, chemical engineering, construction, electrical equipment, water purification, and biomedicine [8][9][10][11][8,9,10,11]. Among them, graphene aerogels [12][13][12,13], carbon aerogels [14][15][14,15], and carbon nanotube (CNT) aerogels [16] are the most investigated topics, comprising more than 60% of the literature studies.
As the most important component of two-dimensional (2D) carbon-based material, graphene possesses superior thermal conductivity (~5000 W/m·K), high specific surface area (2630 m2/g), chemical stability, and high electron mobility, as well as excellent mechanical and optical properties, which has great application potential in many fields [17]. However, their direct application as excellent adsorbents, anode materials, and mechanical devices suffers from limitation, as a serious loss in surface area occurs due to the re-stacking problem resulting from π-π interaction between graphene layers and the Van der Waals force [18].
The past several years has witnessed explosive interest in constructing a series of composites based on the versatile platform of graphene. In order to overcome the problem and make full use of the characteristics of graphene sheets, the conversion of 2D sheet to 3D porous aerogel via self-assembling by various methods, including hydrothermal reduction, chemical reduction, crosslinking, and sol-gel processes, is an ideal choice [19]. Due to integration intrinsic properties, including high porosity, conductivity, and feasibility of manufacturing in an industrial scale of graphene and aerogel, 3D GAs have emerged as one of the most exciting and promising materials. The past almost two decades have witnessed a rapid development process in exploring the performance and application of GAs, as reflected in the increasing number of publications collected in the Web of Science database for the search criteria “graphene AND aerogel” (Figure 1).
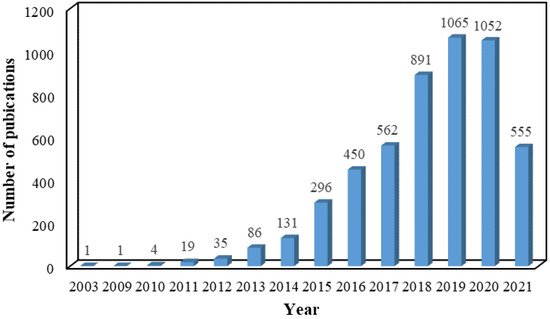
Figure 1.
Number of publications about GAs in the past almost two decades. Data are summarized on the Web of Science.
2. Preparation Strategies
With the further development of 3D GAs, expanded preparation approaches such as hydrothermal reduction [20][21], chemical reduction [21][22][22,23], electrochemical synthesis [23][24][24,25], self-assembly [25][26][26,27], emulsion technique [27][28], breath-figure method [28][29], chemical vapor deposition [29][30], and ink-printing technique [30][31] have been adopted to fabricate varied and unique microstructures. Among these, hydrothermal or chemical reduction is the most attractive one based on its low-cost and scalable production. Here, we have classified these methods into two major categories: template-free method and template-directed method.
The interfacial interaction and chemical composition of as-prepared functional GO and/or RGO-based aerogel can be characterized by various means. XPS analysis is performed to characterize both the chemical state and atomic ratio of every element in the GO and the graphene aerogels. For the C 1s XPS spectrum of GA, the peaks attributed to carbon atoms connecting with oxygenated groups, such as C–O and O–C=O, have disappeared as the oxygenated species are substantially removed on the reduction of GO to graphene [31][32]. It is also evidence for the deoxygenation process and successful preparation of GAs. The significant structural changes are also reflected in the Raman spectra. The G band, which resonates at a lower frequency and an increased D/G intensity ratio compared to that of GO, suggests a reduction of the exfoliated GO. The FT-IR spectra gives the information of various functional groups, proving the change from graphene oxide into a graphite-like structure due to the chemical reduction.
The graphene-based aerogels are mainly prepared from a GO precursor via the reduction process. The dispersion of graphene in substrate is always particularly important for improving the performance of aerogel. In order to avoid aggregation induced by strong a van der Waals force, many preparation methods involving covalent and/or non-covalent modification between GO and polymer matrix and appropriate ultrasonic dispersion are employed [32][33]. The dispersion of graphene can be reflected on the morphology of aerogel characterized by SEM, SEM-EDS and TEM, and the XRD pattern. Furthermore, it is feasible to study the dispersion state of graphene by characterizing the typical properties of aerogels based on the obvious effect of dispersion on performance.
2.1. Template-Free Approach
A template-free method is preferred in practical application due to the relatively simple synthesis procedures, low cost, and easy scaling up [33][34]. A series of template-free methods have been put forward for synthesizing 3D GA, in which the most typical and dominant is the self-assembly method. Taking advantage of inherent orderly stacking behavior via π-π interaction of graphene and GO nanosheets under appropriate conditions can produce various 3D structures [34][35][36][35,36,37] without the need and limitation of a template, making it novel and appealing. However, it is still important to develop suitable approaches to achieve the assembly and avoid the precipitation of graphene in a parallel arrangement while declining the repulsion forces of GO solution [37][38][39][38-40]. Currently, the widely used methods involve with chemical [40][41][42][43][44][45][46][23,41-46]/hydrothermal [47][48][49][50][51][47-51]/electrochemical [52][24] reduction-induced self-assembly and chemical/physical crosslinking-induced self-assembly [53][54][55][56][57][58][59][60][52-59].2.2. Template-Directed Approach
The template-directed strategy, including the emulsion technique, breath figure method, and ink-printing technique, is one of the mainstream approaches employed in the formation of 3D GA with ordered and hierarchical structures [61][60]. It uses a pre-existing guide (hard or soft template) to directly synthesize the target materials that are difficult to obtain by other tools. Meanwhile, it also inevitably limits the scalability of resulting aerogels owing to the hard accessibility of a well-organized and large-size template itself [62][46]. The general route for a templated-directed method mainly involves the following steps: (1) template preparation, (2) synthesis of target materials using the template, (3) template removal (if necessary). The obtained aerogels were widely used in lithium-ion batteries and proved to be excellent electrode materials with outstanding performance. The approach can be classified into hard template method [63][64][65][61-63] and soft template method [66][67][68][69][64-67].3. Application
Graphene aerogel, with characteristics of low density, high surface area and porosity, and good electrical and thermal conductivity, has attracted the attention of researchers and flourished in the recent decades [70][68]. They possesses potential application in diverse fields, such as sorption in environmental protection [25][71][26,69] electrode materials [72][73][70,71], electronic devices [74][72], flame-retardant and fire-warning materials [75][76][73,74], catalysis [77][75], energy storage [78][76], and microwave absorption [79][58].3.1. Aerogels for Absorption
Water contamination caused by harmful chemicals, particularly oils and soluble dyes and phosphate, has become an issue of serious global concern. Various technologies including chemical precipitation [80][81][77,78], biological treatment [82][79], membrane filtration [83][84][80,81], adsorption [85][82], and ion exchange [86][83] have been employed to remove organic contaminants from wastewater. Among them, the adsorption method has been recognized as the most promising candidate. Therefore, the preparation of novel absorbents with low density, water pickup and cost, high absorption capacity, and good recyclability is in urgent need.
Due to the super-hydrophobicity of GA, it is commonly considered as a competitive and efficient adsorbent for oil in water, with a higher adsorption capacity compared to other kinds of adsorbents [87][88][84,85].
