Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Catherine Yang and Version 1 by Soo-young Na.
Capsule endoscopy (CE) has proven to be a valuable diagnostic modality for small bowel diseases over the past 20 years, particularly Crohn’s disease (CD), which can affect the entire gastrointestinal tract from the mouth to the anus. CE is not only used for the diagnosis of patients with suspected small bowel CD, but can also be used to assess disease activity, treat-to-target, and postoperative recurrence in patients with established small bowel CD. As CE can detect even mildly non-specific small bowel lesions, a high diagnostic yield is not necessarily indicative of high diagnostic accuracy.
- inflammatory bowel disease
- Crohn’s disease
- capsule endoscopy
1. Introduction
Capsule endoscopy (CE), which was first used to evaluate the small intestine in 10 healthy volunteers by Iddan et al. [1], has enabled direct visual observation of small bowel lesions and phenotypes that had been difficult to assess using conventional endoscopy [2]. As CE allows direct observation of the small intestine, it is able to visualize even mildly inflammatory mucosal lesions, such as erythema, erosion, and small ulcers, which are difficult to detect with radiological imaging modalities such as small bowel follow-through (SBFT), small bowel contrast ultrasound (SBCUS), CT enterography (CTE), and MR enterography (MRE) [3]. This advantage has aided in precision medicine-based diagnostic and therapeutic decision-making, especially in patients with suspected or established Crohn’s disease (CD) of the small intestine. The scope of use of CE has expanded over the past 20 years, allowing its application in patients with ulcerative colitis (UC), along with pan-enteric CD. This has been made possible with the subsequent development of colon capsule endoscopy (CCE), which enables visualization of both the small and large intestines [4].
The incidence of inflammatory bowel disease (IBD) is relatively high but rather stable in Western countries; however, the incidence and prevalence of IBD are rapidly increasing in Asia, Eastern Europe, and South America, and this shift in the epidemiology of IBD indicates that it has become a global disease, whose increasing incidence has been burdening health services in recent years [5,6,7,8][5][6][7][8]. Therefore, the use of CE for the diagnosis and management of IBD is becoming more frequent and its implementation is considered a priority in the field of IBD.
2. Crohn’s Disease
Patients with suspected CD (SCD) who cannot be diagnosed by radiological modalities can be diagnosed by small bowel CE (SBCE), since the latter can visualize even mildly superficial mucosal lesions that are rarely visible using radiological imaging techniques [9]. SBCE is also useful in assessing disease activity, treat-to-target, and postoperative recurrence in patients with established CD (ECD).
CD mainly involves terminal ileum, and the disease location is limited to the small bowel alone in about 30% of patients [10,11][10][11]. Ileocolonoscopy with biopsy is considered the first-line diagnostic modality in patients with suspected CD. However, lesions located proximal to the terminal ileum are difficult to diagnose by conventional ileocolonoscopy. Patients with small bowel CD (SBCD) have been diagnosed radiologically by SBFT or small bowel enteroclysis, which are modalities with reasonable diagnostic accuracy [12,13][12][13]. Recently, however, these modalities have been largely replaced by cross-sectional imaging modalities, which can better classify disease phenotype and behavior. Several meta-analyses have shown that CTE, MRE, and SBCUS have similar accuracy in the diagnosis of SBCD [14]. SBCE, a safe and painless endoscopic method for the evaluation of the small bowel, is useful diagnostic modality for SBCD. The results of a Spanish national survey showed that SBCE is widely used to manage IBD in patients with SCD (76.3%), to assess inflammatory activity (54.7%) and to evaluate the extent of disease (54.7%) [15]. In addition, CE has been shown to be useful in assessing mucosal CD activity in selected patients with colonic CD [16]. The typical CE findings in SBCD are shown in Figure 1A.
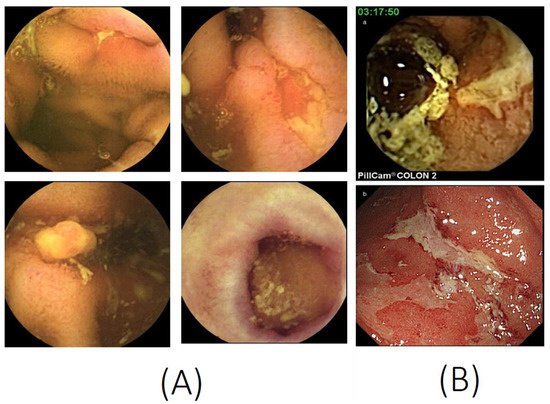
Figure 1. Capsule endoscopy images. (A) Small bowel capsule endoscopy features of Crohn’s disease, such as ulcers, longitudinal ulcers, inflammatory polyps, and scars. (B) A colon capsule endoscopy image (a) consistent with the conventional colonoscopy image (b) of ulcerative colitis (photocopies from Hosoe N, et al. J. Gastroenterol. Hepatol. 2013, 28, 1174–1179, with permission from John Wiley and Sons [17]).
2.1. Capsule Retention
Although CE is a simple and safe test, capsule retention (CR) in the gastrointestinal tract is a frequent complication. CE can be retained within the small bowel, usually in patients with stricturing disease. Passage of the CE through the gastrointestinal tract, including the pylorus and ileocecal valve, may be difficult in pediatric patients, but this method can be safely used in children over 9 years of age [18].
A meta-analysis that included 25 studies of 5876 patients with obscure gastrointestinal bleeding, nine studies of 968 patients with SCD, and 11 studies of 558 patients with ECD found that the pooled CR rates were 3.6% (95% confidence interval (CI), 1.7–8.6%) for SCD and 8.2% (95% CI, 6.0–11.0%) for ECD [19]. These CR rates decreased to 2.7% (95% CI, 1.1–6.4%) in subsequent CE after a patency capsule (PC) or CTE to exclude strictures, with the latter comparable to the CR rate for obscure gastrointestinal bleeding (2.1%; 95% CI, 1.5–2.8%) [19]. A more recent meta-analysis found that the CR rates were 2.35% (95% CI, 1.31–4.19%) in 1234 patients with SCD and 4.63% (95% CI, 3.42–6.25%) in 1720 patients with ECD [20]. Although these CR rates are significantly lower than those previously reported, this meta-analysis also found that the CR rate after PC in patients with ECD was 2.88% (95% CI, 1.74–4.74%) [20].
A meta-analysis that included five studies of 203 patients showed that PC had a sensitivity of 97% (95% CI, 93–99%), a specificity of 83% (95% CI, 65–95%), and an accuracy of 0.956 in diagnosing small bowel obstruction [21]. A recent meta-analysis found that the CR rates in patients with ECD were 2.32% (95% CI, 0.87–6.03%) after negative small bowel cross-sectional imaging and 2.88% (95% CI, 1.74–4.74%) after negative PC [20]. MRE showed a high sensitivity (>92%) and negative predictive value (NPV) (>96%) for PC retention in patients with ECD [22]. These results suggest that cross-sectional imaging of the small bowel should be performed prior to CE to determine the presence of strictures in patients with CD. However, a multicenter prospective study showed that CR rates are similar in low-risk (0.7%, 20/2942) and negative PC high-risk (0.7%, 1/151) patients, but are significantly higher in high-risk patients with negative cross-sectional imaging (CTE or MRE; 8.3%, 2/24; p = 0.049) [23]. Thus, even if cross-sectional imaging is normal, the PC procedure should be offered to patients at increased risk of CR.
As CR is usually asymptomatic, initial periodic monitoring is suggested. Even if CR has occurred in IBD patients, the European Society of Gastrointestinal Endoscopy guidelines recommend observation in patients with asymptomatic CR for the following reasons [24]: (1) 35–50% of CR patients spontaneously excreted the capsules after ≥15 days without further management, (2) a short course of medical therapy may allow capsule excretion, and (3) spontaneous excretion usually occurs 4–12 weeks after ingestion [25]. However, device-assisted enteroscopy or surgical removal should be considered if the capsule is not excreted after 3–6 months or if patients experience symptoms of acute obstruction [25].
2.2. Superior Diagnostic Yield to Other Imaging Modalities
CE provides high-resolution endoluminal images of the small bowel. As there is no standard modality that can be compared with CE to determine its diagnostic accuracy, the ‘diagnostic yield’ is determined in many studies of this modality.
CE showed a higher diagnostic yield than small bowel enteroclysis in the detection of small bowel lesions, in 27 patients with ECD (74.1% vs. 40.7%; p < 0.05) and in 20 patients with SCD (65% vs. 30%; p < 0.05) [26]. CE also showed a higher diagnostic yield than CT enterolysis in evaluating jejunal or ileal lesions in 41 patients with small bowel CD (61.0% vs. 29.3%; p < 0.004) but not in those with terminal/neonatal ileum [27]. CE was superior to MR enteroclysis in detecting inflammatory lesions in the proximal and middle parts of the small bowel (66.7% vs. 5.6%; p = 0.016) in 18 patients with SCD or ESD [28].
A meta-analysis that included only prospective studies found that the diagnostic yield of CE was significantly superior to that of SBFT, CTE, ileocolonoscopy, and push enteroscopy in the evaluation of SBCD [29]. The results of recent meta-analysis have revealed that the diagnostic yields of CE do not differ significantly from those of CTE and MRE in patients with both SCD and ECD [30]. The different results for CTE in these meta-analyses were due to differences in the inclusion criteria. Another recent meta-analysis found that the diagnostic yield of CE was similar to that of MRE in 10 studies involving 400 patients (odds ratio (OR), 1.17; 95% CI, 0.83–1.67) and SBCUS in five studies involving 142 patients (OR, 0.88; 95% CI, 0.51–1.53) with SCD and ECD [31]. In that analysis, however, CE was superior to MRE in seven studies involving 251 patients with proximal suspected small bowel disease (OR, 2.79; 95% CI, 1.2–6.48) [31]. Subsequent prospective studies comparing CE with MRE found that both modalities independently detected previously unrecognized proximal disease locations in 51% (29/56) and 26% (20/79) (p < 0.01), respectively, in patients with SBCD in clinical remission or with mild disease [32]. CE was also superior to MRE in detecting small bowel lesions (76.6% vs. 44.7%, p = 0.001) in SCD and ECD, with CE being superior to MRE in detecting lesions in the jejunum, ileum, and terminal ileum (all, p < 0.05) [9].
Although the diagnostic yield of CE was higher than that of other imaging diagnostic modalities, especially in patients with proximal small bowel disease, there are several barriers to the clinical use of CE. The higher diagnostic yield of CE does not directly indicate a higher diagnostic accuracy, as diagnostic yield can be affected by other factors, such as nonsteroidal anti-inflammatory drugs [33]. Minor mucosal injuries and erosions have been detected in up to 20% of healthy volunteers [33]. In addition, cost analyses suggest that the addition of CE as a third test after ileocolonoscopy and negative CTE or SBFT is not cost-effective [34].
2.3. Increasing Diagnostic Capability of Capsule Endoscopy
CE is being developed in several ways, including the development of CE instruments with higher frame rates and increased image resolution, which should increase the possibility of obtaining higher diagnostic yield and accuracy than in the past [35]. For example, the adaptive frame rate (AFR) technology with a movement sensor, which captures images depending on the speed of the capsule’s movement, of the PillCam SB3 (Medtronic, Ltd., Dublin, Ireland; 2–6 images/second) and PillCam Crohn’s capsule (PCC, Medtronic, Ltd.; 4–35 images/second) may increase the diagnostic yield of CD [36,37,38][36][37][38]. Non-white light imaging has been reported to improve the detection rate and visibility of small intestinal lesions by increasing the visualization of surface patterns and color differences in the presence of bile juice and blood [39]. For example, flexible spectral color enhancement (FICE, Fujifilm Corp., Tokyo, Japan) is a digital processing method of white light imaging that emphasizes specific ranges of wavelengths of light in the red, green, and blue spectrum [39]. The FICE wavelength settings were developed with the aims of reducing blue light interference (FICE1), accentuating blood (FICE 2), and strengthening the differences between bile and blood (FICE3) [39]. A contrast capsule (Olympus Corp., Tokyo, Japan), which increases brightness in the blue wavelength range by using a special instrument equipped with a light-emitting diode [39], enables easy detection of areas of bleeding by selecting green and blue data. However, clinical trials of FICE and the contrast capsule so far have yielded controversial results for the detection of gastrointestinal lesions [40,41][40][41]. Another optical-digital method similar to contrast capsule, termed narrow-band imaging (Olympus Corp.), which allows better visualization of mucosal surface patterns and superficial capillaries, is being applied to CE in device manufacturing research [42].
2.4. Clinical Suspicion of Crohn’s Disease with Negative Conventional Modalities
SBCE is a sensitive tool to detect mucosal abnormalities in the small bowel. Approximately 5–10% of patients have isolated small bowel disease that cannot be detected by conventional ileocolonoscopy [43]. The diagnostic yield of SBCE in patients with SCD has been reported to range from 40% to 70%. For example, SBCE diagnosed CD in 12 (71%) of 17 patients with symptoms such as abdominal pain, anemia, and diarrhea of unknown cause with normal appearance on conventional modalities [44]. SBCE in 20 SCD patients suspected of having small bowel lesions diagnosed CD in 13 patients (65%) [45], showing that CE is effective in diagnosing patients with SCD undetected by conventional diagnostic methods. SBCE detected lesions supporting the diagnosis of CD in 9 (43%) of 21 patients with clinical SCD [46]. This method also increased diagnostic yield by 24% in patients with perianal disease and negative conventional work up, including ileocolonoscopy [47]. These results showed that CE is a useful test for the diagnosis of CD in patients who have not been diagnosed by conventional modalities, such as gastroscopy, ileocolonoscopy, and SBFT.
The presence of biochemical markers in patients with SCD symptoms has been reported to increase the diagnostic yield of SBCE [48]. Although two retrospective studies showed the presence of small bowel inflammation in the majority of ECD patients in remission with biomarkers [49[49][50],50], a meta-analysis showed that the likelihood of a positive diagnosis is very low in SCD patients with fecal calprotectin (FC) < 50 μg/g [51]. A recent retrospective study revealed that FC was positively correlated with significant inflammatory activity (Lewis Score, LS ≥ 135; rank correlation = 0.56; p < 0.001) [52], further indicating that FC may be a useful marker to select patients with SCD for SBCE. Various FC cut-off values have been reported to be indicative of small bowel CD. For example, 33 (89.2%) of 37 patients with FC ≥ 100 µg/g were found to have an LS ≥ 135 [52]. A recent meta-analysis that included 14 studies suggested that FC ≥ 100 ug/g and LS ≥ 135 cut-off values had diagnostic odds ratios of 8.96 and 10.90, respectively [53].
As normal radiological imaging of the small bowel by SBFT, SBCUS, CTE, and MRE cannot entirely exclude small bowel involvement, SCD patients with normal radiological results but elevated FC and/or unexplained anemia should be considered for additional SBCE [9,13][9][13]. Careful monitoring without additional work up may be sufficient for asymptomatic CD patients with negative FC results [54]. Based on the above results, a diagnostic algorithm using CE in patients with SCD is illustrated in Figure 2.
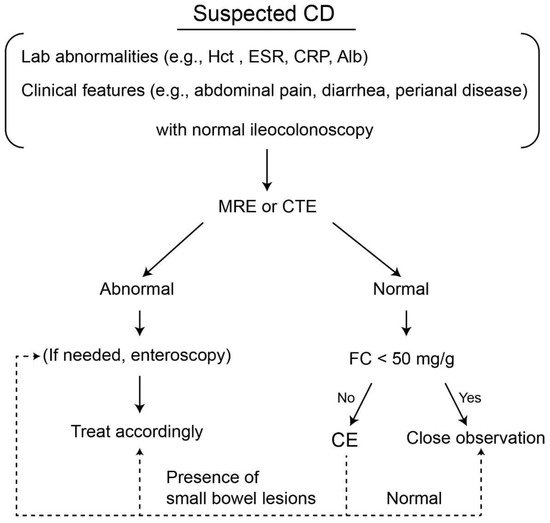
Figure 2. Suggested diagnostic algorithm for the use of small bowel capsule endoscopy in patients with suspected Crohn’s disease. CD, Crohn’s disease; Hct, hematocrit; CRP, C-reactive protein; Alb, albumin; MRE, MR enterography; CTE, CT enterography; FC, fecal calprotectin, CE, capsule endoscopy.
References
- Iddan, G.; Meron, G.; Glukhovsky, A.; Swain, P. Wireless capsule endoscopy. Nature 2000, 405, 417.
- Appleyard, M.; Glukhovsky, A.; Swain, P. Wireless-capsule diagnostic endoscopy for recurrent small-bowel bleeding. N. Engl. J. Med. 2001, 344, 232–233.
- Hong, S.M.; Jung, S.H.; Baek, D.H. Diagnostic Yields and Clinical Impacts of Capsule Endoscopy. Diagnostics 2021, 11, 1842.
- Eliakim, R.; Fireman, Z.; Gralnek, I.M.; Yassin, K.; Waterman, M.; Kopelman, Y.; Lachter, J.; Koslowsky, B.; Adler, S.N. Evaluation of the PillCam Colon capsule in the detection of colonic pathology: Results of the first multicenter, prospective, comparative study. Endoscopy 2006, 38, 963–970.
- Hong, S.W.; Ye, B.D. The first step to unveil the epidemiology of inflammatory bowel disease in Central Asia. Intest. Res. 2020, 18, 345–346.
- Yen, H.H.; Weng, M.T.; Tung, C.C.; Wang, Y.T.; Chang, Y.T.; Chang, C.H.; Shieh, M.J.; Wong, J.M.; Wei, S.C. Epidemiological trend in inflammatory bowel disease in Taiwan from 2001 to 2015: A nationwide populationbased study. Intest. Res. 2019, 17, 54–62.
- Sood, A.; Kaur, K.; Singh, A.; Midha, V.; Mahajan, R.; Bansal, N.; Mehta, V.; Singh, D. Trends of inflammatory bowel disease at a tertiary care center in northern India. Intest. Res. 2021, 19, 282–290.
- Sood, A.; Kaur, K.; Mahajan, R.; Midha, V.; Singh, A.; Sharma, S.; Puri, A.S.; Goswami, B.; Desai, D.; Pai, C.G.; et al. Colitis and Crohn’s Foundation (India): A first nationwide inflammatory bowel disease registry. Intest. Res. 2021, 19, 206–216.
- González-Suárez, B.; Rodriguez, S.; Ricart, E.; Ordás, I.; Rimola, J.; Díaz-González, Á.; Romero, C.; de Miguel, C.R.; Jáuregui, A.; Araujo, I.K.; et al. Comparison of Capsule Endoscopy and Magnetic Resonance Enterography for the Assessment of Small Bowel Lesions in Crohn’s Disease. Inflamm. Bowel Dis. 2018, 24, 775–780.
- Mekhjian, H.S.; Switz, D.M.; Melnyk, C.S.; Rankin, G.B.; Brooks, R.K. Clinical features and natural history of Crohn’s disease. Gastroenterology 1979, 77, 898–906.
- Steinhardt, H.J.; Loeschke, K.; Kasper, H.; Holtermüller, K.H.; Schäfer, H. European Cooperative Crohn’s Disease Study (ECCDS): Clinical features and natural history. Digestion 1985, 31, 97–108.
- Saibeni, S.; Rondonotti, E.; Iozzelli, A.; Spina, L.; Tontini, G.E.; Cavallaro, F.; Ciscato, C.; de Franchis, R.; Sardanelli, F.; Vecchi, M. Imaging of the small bowel in Crohn’s disease: A review of old and new techniques. World J. Gastroenterol. 2007, 13, 3279–3287.
- Chernish, S.M.; Maglinte, D.D.; O’Connor, K. Evaluation of the small intestine by enteroclysis for Crohn’s disease. Am. J. Gastroenterol. 1992, 87, 696–701.
- Lamb, C.A.; Kennedy, N.A.; Raine, T.; Hendy, P.A.; Smith, P.J.; Limdi, J.K.; Hayee, B.; Lomer, M.C.E.; Parkes, G.C.; Selinger, C.; et al. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut 2019, 68, s1–s106.
- González, A.E.; Castillejo, Ó.N.; Sainz, I.F.U.; López-García, A.; Pomares, Ó.M.; Abdo, Y.Z. Use of capsule endoscopy in inflammatory bowel disease in clinical practice in Spain. Results from a national survey. Gastroenterol. Hepatol. 2021, 44, 696–703.
- D’Haens, G.; Löwenberg, M.; Samaan, M.A.; Franchimont, D.; Ponsioen, C.; van den Brink, G.R.; Fockens, P.; Bossuyt, P.; Amininejad, L.; Rajamannar, G.; et al. Safety and Feasibility of Using the Second-Generation Pillcam Colon Capsule to Assess Active Colonic Crohn’s Disease. Clin. Gastroenterol. Hepatol. 2015, 13, 1480–1486.e1483.
- Hosoe, N.; Matsuoka, K.; Naganuma, M.; Ida, Y.; Ishibashi, Y.; Kimura, K.; Yoneno, K.; Usui, S.; Kashiwagi, K.; Hisamatsu, T.; et al. Applicability of second-generation colon capsule endoscope to ulcerative colitis: A clinical feasibility study. J. Gastroenterol. Hepatol. 2013, 28, 1174–1179.
- Seidman, E.G.; Sant’Anna, A.M.; Dirks, M.H. Potential applications of wireless capsule endoscopy in the pediatric age group. Gastrointest. Endosc. Clin. N. Am. 2004, 14, 207–217.
- Rezapour, M.; Amadi, C.; Gerson, L.B. Retention associated with video capsule endoscopy: Systematic review and meta-analysis. Gastrointest. Endosc. 2017, 85, 1157–1168.e1152.
- Pasha, S.F.; Pennazio, M.; Rondonotti, E.; Wolf, D.; Buras, M.R.; Albert, J.G.; Cohen, S.A.; Cotter, J.; D’Haens, G.; Eliakim, R.; et al. Capsule Retention in Crohn’s Disease: A Meta-analysis. Inflamm. Bowel Dis. 2020, 26, 33–42.
- Zhang, W.; Han, Z.L.; Cheng, Y.; Xu, Y.Z.; Xiao, K.; Li, A.M.; Wang, Y.D.; Li, Y.; Liu, S.D. Value of the patency capsule in pre-evaluation for capsule endoscopy in cases of intestinal obstruction. J. Dig. Dis. 2014, 15, 345–351.
- Rozendorn, N.; Klang, E.; Lahat, A.; Yablecovitch, D.; Kopylov, U.; Eliakim, A.; Ben-Horin, S.; Amitai, M.M. Prediction of patency capsule retention in known Crohn’s disease patients by using magnetic resonance imaging. Gastrointest. Endosc. 2016, 83, 182–187.
- Rondonotti, E.; Soncini, M.; Girelli, C.M.; Russo, A.; de Franchis, R. Short article: Negative small-bowel cross-sectional imaging does not exclude capsule retention in high-risk patients. Eur. J. Gastroenterol. Hepatol. 2016, 28, 871–875.
- Rondonotti, E.; Spada, C.; Adler, S.; May, A.; Despott, E.J.; Koulaouzidis, A.; Panter, S.; Domagk, D.; Fernandez-Urien, I.; Rahmi, G.; et al. Small-bowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) Technical Review. Endoscopy 2018, 50, 423–446.
- Rondonotti, E. Capsule retention: Prevention, diagnosis and management. Ann. Transl. Med. 2017, 5, 198.
- Efthymiou, A.; Viazis, N.; Vlachogiannakos, J.; Georgiadis, D.; Kalogeropoulos, I.; Mantzaris, G.; Karamanolis, D.G. Wireless capsule endoscopy versus enteroclysis in the diagnosis of small-bowel Crohn’s disease. Eur. J. Gastroenterol. Hepatol. 2009, 21, 866–871.
- Voderholzer, W.A.; Beinhoelzl, J.; Rogalla, P.; Murrer, S.; Schachschal, G.; Lochs, H.; Ortner, M.A. Small bowel involvement in Crohn’s disease: A prospective comparison of wireless capsule endoscopy and computed tomography enteroclysis. Gut 2005, 54, 369–373.
- Gölder, S.K.; Schreyer, A.G.; Endlicher, E.; Feuerbach, S.; Schölmerich, J.; Kullmann, F.; Seitz, J.; Rogler, G.; Herfarth, H. Comparison of capsule endoscopy and magnetic resonance (MR) enteroclysis in suspected small bowel disease. Int. J. Colorectal. Dis. 2006, 21, 97–104.
- Dionisio, P.M.; Gurudu, S.R.; Leighton, J.A.; Leontiadis, G.I.; Fleischer, D.E.; Hara, A.K.; Heigh, R.I.; Shiff, A.D.; Sharma, V.K. Capsule endoscopy has a significantly higher diagnostic yield in patients with suspected and established small-bowel Crohn’s disease: A meta-analysis. Am. J. Gastroenterol. 2010, 105, 1240–1248.
- Choi, M.; Lim, S.; Choi, M.G.; Shim, K.N.; Lee, S.H. Effectiveness of Capsule Endoscopy Compared with Other Diagnostic Modalities in Patients with Small Bowel Crohn’s Disease: A Meta-Analysis. Gut Liver 2017, 11, 62–72.
- Kopylov, U.; Yung, D.E.; Engel, T.; Vijayan, S.; Har-Noy, O.; Katz, L.; Oliva, S.; Avni, T.; Battat, R.; Eliakim, R.; et al. Diagnostic yield of capsule endoscopy versus magnetic resonance enterography and small bowel contrast ultrasound in the evaluation of small bowel Crohn’s disease: Systematic review and meta-analysis. Dig. Liver Dis. 2017, 49, 854–863.
- Greener, T.; Klang, E.; Yablecovitch, D.; Lahat, A.; Neuman, S.; Levhar, N.; Avidan, B.; Yanai, H.; Dotan, I.; Chowers, Y.; et al. The Impact of Magnetic Resonance Enterography and Capsule Endoscopy on the Re-classification of Disease in Patients with Known Crohn’s Disease: A Prospective Israeli IBD Research Nucleus (IIRN) Study. J. Crohn’s Colitis 2016, 10, 525–531.
- Pennazio, M.; Spada, C.; Eliakim, R.; Keuchel, M.; May, A.; Mulder, C.J.; Rondonotti, E.; Adler, S.N.; Albert, J.; Baltes, P.; et al. Small-bowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy 2015, 47, 352–376.
- Levesque, B.G.; Cipriano, L.E.; Chang, S.L.; Lee, K.K.; Owens, D.K.; Garber, A.M. Cost effectiveness of alternative imaging strategies for the diagnosis of small-bowel Crohn’s disease. Clin. Gastroenterol. Hepatol. 2010, 8, 261–267.
- Jiang, B.; Qian, Y.Y.; Pan, J.; Jiang, X.; Wang, Y.C.; Zhu, J.H.; Zou, W.B.; Zhou, W.; Li, Z.S.; Liao, Z. Second-generation magnetically controlled capsule gastroscopy with improved image resolution and frame rate: A randomized controlled clinical trial (with video). Gastrointest. Endosc. 2020, 91, 1379–1387.
- Monteiro, S.; de Castro, F.D.; Carvalho, P.B.; Moreira, M.J.; Rosa, B.; Cotter, J. PillCam® SB3 capsule: Does the increased frame rate eliminate the risk of missing lesions? World J. Gastroenterol. 2016, 22, 3066–3068.
- Hosoe, N.; Hayashi, Y.; Ogata, H. Colon Capsule Endoscopy for Inflammatory Bowel Disease. Clin. Endosc. 2020, 53, 550–554.
- PillCam Software V9. Available online: https://www.medtronic.com/covidien/en-us/support/software/gastrointestinal-products/pillcam-software-v9.html (accessed on 22 October 2021).
- Ogata, N.; Ohtsuka, K.; Ogawa, M.; Maeda, Y.; Ishida, F.; Kudo, S.-E. Image-Enhanced Capsule Endoscopy Improves the Identification of Small Intestinal Lesions. Diagnostics 2021, 11, 2122.
- Yung, D.E.; Boal Carvalho, P.; Giannakou, A.; Kopylov, U.; Rosa, B.; Rondonotti, E.; Toth, E.; Plevris, J.N.; Koulaouzidis, A. Clinical validity of flexible spectral imaging color enhancement (FICE) in small-bowel capsule endoscopy: A systematic review and meta-analysis. Endoscopy 2017, 49, 258–269.
- Koulaouzidis, A.; Douglas, S.; Plevris, J.N. Blue mode does not offer any benefit over white light when calculating Lewis score in small-bowel capsule endoscopy. World J. Gastrointest. Endosc. 2012, 4, 33–37.
- Kim, S.H.; Chun, H.J. Capsule Endoscopy: Pitfalls and Approaches to Overcome. Diagnostics 2021, 11, 1765.
- Dulai, P.S.; Singh, S.; Vande Casteele, N.; Boland, B.S.; Rivera-Nieves, J.; Ernst, P.B.; Eckmann, L.; Barrett, K.E.; Chang, J.T.; Sandborn, W.J. Should We Divide Crohn’s Disease into Ileum-Dominant and Isolated Colonic Diseases? Clin. Gastroenterol. Hepatol. 2019, 17, 2634–2643.
- Fireman, Z.; Mahajna, E.; Broide, E.; Shapiro, M.; Fich, L.; Sternberg, A.; Kopelman, Y.; Scapa, E. Diagnosing small bowel Crohn’s disease with wireless capsule endoscopy. Gut 2003, 52, 390–392.
- Ge, Z.Z.; Hu, Y.B.; Xiao, S.D. Capsule endoscopy in diagnosis of small bowel Crohn’s disease. World J. Gastroenterol. 2004, 10, 1349–1352.
- Herrerías, J.M.; Caunedo, A.; Rodríguez-Téllez, M.; Pellicer, F.; Herrerías, J.M., Jr. Capsule endoscopy in patients with suspected Crohn’s disease and negative endoscopy. Endoscopy 2003, 35, 564–568.
- Adler, S.N.; Yoav, M.; Eitan, S.; Yehuda, C.; Eliakim, R. Does capsule endoscopy have an added value in patients with perianal disease and a negative work up for Crohn’s disease? World J. Gastrointest. Endosc. 2012, 4, 185–188.
- De Bona, M.; Bellumat, A.; Cian, E.; Valiante, F.; Moschini, A.; De Boni, M. Capsule endoscopy findings in patients with suspected Crohn’s disease and biochemical markers of inflammation. Dig. Liver Dis. 2006, 38, 331–335.
- Kopylov, U.; Yablecovitch, D.; Lahat, A.; Neuman, S.; Levhar, N.; Greener, T.; Klang, E.; Rozendorn, N.; Amitai, M.M.; Ben-Horin, S.; et al. Detection of Small Bowel Mucosal Healing and Deep Remission in Patients with Known Small Bowel Crohn’s Disease Using Biomarkers, Capsule Endoscopy, and Imaging. Am. J. Gastroenterol. 2015, 110, 1316–1323.
- Kopylov, U.; Nemeth, A.; Koulaouzidis, A.; Makins, R.; Wild, G.; Afif, W.; Bitton, A.; Johansson, G.W.; Bessissow, T.; Eliakim, R.; et al. Small bowel capsule endoscopy in the management of established Crohn’s disease: Clinical impact, safety, and correlation with inflammatory biomarkers. Inflamm. Bowel Dis. 2015, 21, 93–100.
- Kopylov, U.; Yung, D.E.; Engel, T.; Avni, T.; Battat, R.; Ben-Horin, S.; Plevris, J.N.; Eliakim, R.; Koulaouzidis, A. Fecal calprotectin for the prediction of small-bowel Crohn’s disease by capsule endoscopy: A systematic review and meta-analysis. Eur. J. Gastroenterol. Hepatol. 2016, 28, 1137–1144.
- Monteiro, S.; Barbosa, M.; Cúrdia Gonçalves, T.; Boal Carvalho, P.; Moreira, M.J.; Rosa, B.; Cotter, J. Fecal Calprotectin as a Selection Tool for Small Bowel Capsule Endoscopy in Suspected Crohn’s Disease. Inflamm. Bowel Dis. 2018, 24, 2033–2038.
- Jung, E.S.; Lee, S.P.; Kae, S.H.; Kim, J.H.; Kim, H.S.; Jang, H.J. Diagnostic Accuracy of Fecal Calprotectin for the Detection of Small Bowel Crohn’s Disease through Capsule Endoscopy: An Updated Meta-Analysis and Systematic Review. Gut Liver 2021, 15, 732–741.
- Bar-Gil Shitrit, A.; Koslowsky, B.; Livovsky, D.M.; Shitrit, D.; Paz, K.; Adar, T.; Adler, S.N.; Goldin, E. A prospective study of fecal calprotectin and lactoferrin as predictors of small bowel Crohn’s disease in patients undergoing capsule endoscopy. Scand. J. Gastroenterol. 2017, 52, 328–333.
More
