Besides traditional risk factors, accumulated evidence suggested that a high inflammatory burden has emerged as a key characteristic modulating both the pathogenesis and progression of cardiovascular diseases, inclusive of atherosclerosis and myocardial infarction. To mechanistically elucidate the correlation, signalling pathways downstream to Toll-like receptors, nucleotide oligomerisation domain-like receptors, interleukins, tumour necrosis factor, and corresponding cytokines were raised as central mechanisms exerting the effect of inflammation. Other remarkable adjuvant factors include oxidative stress and secondary ferroptosis. These molecular discoveries have propelled pharmaceutical advancements. Statin was suggested to confer cardiovascular benefits not only by lowering cholesterol levels but also by attenuating inflammation. Colchicine was repurposed as an immunomodulator co-administered with coronary intervention. Novel interleukin-1β and −6 antagonists exhibited promising cardiac benefits in the recent trials as well. Moreover, manipulation of gut microbiota and associated metabolites was addressed to antagonise inflammation-related cardiovascular pathophysiology. The gut-cardio-renal axis was therein established to explain the mutual interrelationship.
- cardiovascular disease
- gut microbiota
- inflammation
1. Background
2. Inflammation and Cardiovascular Diseases
2.1. Atherosclerosis
2.2. Myocardial Infarction
3. Molecular Mechanism Underlying Inflammation
The molecular mechanism governing the role of inflammation in cardiovascular diseases has garnered considerable research attention. The inflammatory status can be activated by diverse aetiologies, such as myocardial ischaemia, viral myocarditis, hypertrophic or autoimmune cardiomyopathy, and genetic diseases. The injured myocardium triggers both innate and adaptive immunity [21][12]. In addition, cholesterol crystals, neutrophil extracellular traps, atheroprone flow, and hypoxia contribute to the activation of the inflammatory status. Binding of pathogen-associated molecular patterns or damage-associated molecular patterns to pattern recognition receptors, including TLRs and nucleotide oligomerisation domain (NOD)-like receptors, on local cardiomyocytes or residing immune cells, induces downstream signal transduction pathways. Eventually, the cascade leads to the upregulation of chemokines and inflammatory cytokines. The most commonly involved players are the IL family and TNF. Transcriptional profiling has also reported that the genetic landscape associated with inflammation is altered in patients with heart failure and myocardial ischaemia [22][13]. In addition, macrophages and neutrophils are recruited collectively to initiate adaptive immunity.4. Pharmaceutical Advancements
4.1. Colchicine
4.2. Interleukin-1 Antagonist
Meanwhile, other anti-inflammatory agents, apart from colchicine, were assessed for their cardiovascular effects. In the Canakinumab Anti-inflammatory Thrombosis Outcome Study (CANTOS) trail [47][17], 10,061 post-MI subjects with elevated CRP were enrolled. Targeting IL-1β with canakinumab markedly reduced the rate of recurrent cardiovascular events. Methotrexate, a widely used anti-inflammatory agent in rheumatological diseases, was evaluated but failed to improve the clinical efficacy of secondary prevention in patients with stable atherosclerosis. In the Cardiovascular Inflammation Reduction Trial (CIRT) [48][18], 4786 individuals with previous MI or multivessel coronary disease, concomitant diabetes mellitus (DM), or metabolic syndrome were enrolled. At the 2.3-year follow-up, methotrexate did not reduce the composite rate of cardiovascular mortality, nonfatal MI, or nonfatal stroke. Anakinra-mediated IL-1 blockade has been evaluated for its clinical efficacy as a potential therapeutic target. Abbate et al. conducted a pilot study with 30 patients sustaining STEMI [49][19]. The first dose of anakinra 100 mg was administered within 24 h of primary PCI and continued for 14 days. A significant decrease in serum CRP level and the rate of mortality or new-onset heart failure was documented, with no reduction in the level of CK-MB and MACE incidence.5. The Role of Gut Microbiota
A disorganised profile of the intestinal microorganism was proposed to be interrelated with inflammation under the context of cardiovascular diseases [60][20]. Amelioration of inflammatory status by modulating the gut microbiota composition is expected to serve as a new modality. Translocation of the microorganisms and shedding of the bacterial wall compound can contribute to the inflammatory status. Additionally, not only dysbiosis but also the altered metabolism gives rise to inflammation. First, the short-chain fatty acid generated by anaerobic fermentation of fibre is considered pivotal for initiating inflammatory signalling [61][21]. Second, butyrate is another end metabolite of fermentation that had been identified to govern regulatory T cells for orchestrating inflammation and recognised prerequisite to maintain the intestinal barrier [62][22]. A recent study with metagenome sequencing indicated the expression of the butyrate-encoding gene was negatively correlated with the level of inflammatory markers, which was proposed secondary to the altered abundance of Roseburia and Eubacterium in subjects with carotid atherosclerosis [63][23]. Third, lipopolysaccharide (LPS) shed from Gram-negative bacteria was also pointed out as the bridge of remodeled microorganisms to inflammation and eventually attributing to the pathogenesis of cardiovascular diseases. The enteric LPS invades systemic circulation and ignites inflammation after being recognised by TLRs. The elevated endotoxin level and pro-inflammatory status eventually compromise cardiac and vascular function [64][24]. Moreover, trimethylamine N-oxide (TMAO) was identified as another chief transmitter from dysbiosis to the inflammatory status impacting cardiovascular manifestations based on the metabolomic perspective. The quantity of TMAO was determined by age, body mass index, and especially the gut microbiome composition.6. Gut-Cardio-Renal Triplet
Based on the accumulating evidence of mutual interplay, the triplet of the gut-cardio-renal axis was conceptualised to modify traditional cardiorenal syndrome (Figure 21). The homeostasis of gut microbiota and its immunoregulatory effects are pivotal for maintaining the physiology of both the cardiac and renal systems. Regarding kidney physiology, impaired intestinal barrier function, microbial dysbiosis, compromised immunity, and toxin production were primary factors that correlated with altered gut microbiota and renal dysfunction [87][25]. Short-chain fatty acids have been identified as key players in immune regulation, signalling of G-protein-coupled receptors, and antagonists of histone deacetylases for epigenetic modulation [88][26]. Additionally, the pathogenesis of cardiorenal dysfunction implicates the effect of uraemic toxins, including indoxyl sulfate, p-cresyl sulfate, p-cresol, phenylacetic acid, indole-3-acetic acid, homocysteine, hippuric acid, and phenol [89][27]. Collectively, the search for an upstream therapeutic target for better-integrated care remains a priority. Linaclotide, a guanylate cyclase C agonist, was identified as a potential candidate. Low-dose linaclotide was suggested to prevent and manage cardiorenal syndrome by downregulating plasma TMAO, uraemic toxin, and colonic claudin-1 levels [90][28].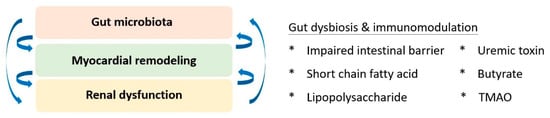
7. Conclusions
References
- Friuli, M.; Eramo, B.; Valenza, M.; Scuderi, C.; Provensi, G.; Romano, A. Targeting the Oxytocinergic System: A Possible Pharmacological Strategy for the Treatment of Inflammation Occurring in Different Chronic Diseases. Int. J. Mol. Sci. 2021, 22, 10250.
- Emerging Risk Factors Collaboration; Kaptoge, S.; Di Angelantonio, E.; Lowe, G.; Pepys, M.B.; Thompson, S.G.; Collins, R.; Danesh, J. C-reactive protein concentration and risk of coronary heart disease, stroke, and mortality: An individual participant meta-analysis. Lancet 2010, 375, 132–140.
- Stark, K.; Massberg, S. Interplay between inflammation and thrombosis in cardiovascular pathology. Nat. Rev. Cardiol. 2021, 18, 666–682.
- Frantz, S.; Falcao-Pires, I.; Balligand, J.-L.; Bauersachs, J.; Brutsaert, D.; Ciccarelli, M.; Dawson, D.; De Windt, L.J.; Giacca, M.; Hamdani, N.; et al. The innate immune system in chronic cardiomyopathy: A European Society of Cardiology (ESC) scientific statement from the Working Group on Myocardial Function of the ESC. Eur. J. Heart Fail. 2018, 20, 445–459.
- Zhu, Y.; Xian, X.; Wang, Z.; Bi, Y.; Chen, Q.; Han, X.; Tang, D.; Chen, R. Research Progress on the Relationship between Atherosclerosis and Inflammation. Biomolecules 2018, 8, 80.
- Chistiakov, D.A.; Melnichenko, A.A.; Grechko, A.V.; Myasoedova, V.A.; Orekhov, A.N. Potential of anti-inflammatory agents for treatment of atherosclerosis. Exp. Mol. Pathol. 2018, 104, 114–124.
- Zeng, X.; Guo, R.; Dong, M.; Zheng, J.; Lin, H.; Lu, H. Contribution of TLR4 signaling in intermittent hypoxia-mediated atherosclerosis progression. J. Transl. Med. 2018, 16, 106.
- Mouton, A.J.; DeLeon-Pennell, K.Y.; Rivera Gonzalez, O.J.; Flynn, E.R.; Freeman, T.C.; Saucerman, J.J.; Garrett, M.R.; Ma, Y.; Harmancey, R.; Lindsey, M.L. Mapping macrophage polarization over the myocardial infarction time continuum. Basic Res. Cardiol. 2018, 113, 26.
- Daseke, M.J., 2nd; Valerio, F.M.; Kalusche, W.J.; Ma, Y.; DeLeon-Pennell, K.Y.; Lindsey, M.L. Neutrophil proteome shifts over the myocardial infarction time continuum. Basic Res. Cardiol. 2019, 114, 37.
- Weinberg, S.E.; Sena, L.A.; Chandel, N.S. Mitochondria in the regulation of innate and adaptive immunity. Immunity 2015, 42, 406–417.
- Chang, M.Y.; Chan, C.K.; Braun, K.R.; Green, P.S.; O’Brien, K.D.; Chait, A.; Day, A.J.; Wight, T.N. Mono-cyte-to-macrophage differentiation: Synthesis and secretion of a complex extracellular matrix. J. Biol. Chem. 2012, 287, 14122–14135.
- Adamo, L.; Rocha-Resende, C.; Prabhu, S.D.; Mann, D.L. Reappraising the role of inflammation in heart failure. Nat. Rev. Cardiol. 2020, 17, 269–285.
- Mann, D.L.; Topkara, V.K.; Evans, S.; Barger, P.M. Innate Immunity in the Adult Mammalian Heart: For Whom the Cell Tolls. Trans. Am. Clin. Clim. Assoc. 2010, 121, 34–50.
- Tardif, J.-C.; Kouz, S.; Waters, D.D.; Bertrand, O.F.; Diaz, R.; Maggioni, A.P.; Pinto, F.J.; Ibrahim, R.; Gamra, H.; Kiwan, G.S.; et al. Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. N. Engl. J. Med. 2019, 381, 2497–2505.
- Nidorf, S.M.; Fiolet, A.T.L.; Mosterd, A.; Eikelboom, J.W.; Schut, A.; Opstal, T.S.J.; The, S.H.K.; Xu, X.F.; Ireland, M.A.; Lenderink, T.; et al. Colchicine in patients with chronic coronary disease. N. Engl. J. Med. 2020, 383, 1838–1847.
- Kalkman, D.N.; Aquino, M.; Claessen, B.E.; Baber, U.; Guedeney, P.; Sorrentino, S.; Vogel, B.; De Winter, R.J.; Sweeny, J.; Kovacic, J.C.; et al. Residual inflammatory risk and the impact on clinical outcomes in patients after percutaneous coronary interventions. Eur. Heart J. 2018, 39, 4101–4108.
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N. Engl. J. Med. 2017, 377, 1119–1131.
- Ridker, P.M.; Everett, B.M.; Pradhan, A.; MacFadyen, J.G.; Solomon, D.H.; Zaharris, E.; Mam, V.; Hasan, A.; Rosenberg, Y.; Iturriaga, E.; et al. Low-Dose Methotrexate for the Prevention of Atherosclerotic Events. N. Engl. J. Med. 2019, 380, 752–762.
- Abbate, A.; Van Tassell, B.W.; Biondi-Zoccai, G.; Kontos, M.C.; Grizzard, J.D.; Spillman, D.W.; Oddi, C.; Roberts, C.S.; Melchior, R.D.; Mueller, G.H.; et al. Effects of Interleukin-1 Blockade with Anakinra on Adverse Cardiac Remodeling and Heart Failure After Acute Myocardial Infarction . Am. J. Cardiol. 2013, 111, 1394–1400.
- Jie, Z.; Xia, H.; Zhong, S.-L.; Feng, Q.; Li, S.; Liang, S.; Zhong, H.; Liu, Z.; Gao, Y.; Zhao, H.; et al. The gut microbiome in atherosclerotic cardiovascular disease. Nat. Commun. 2017, 8, 845.
- Witkowski, M.; Weeks, T.L.; Hazen, S.L. Gut Microbiota and Cardiovascular Disease. Circ. Res. 2020, 127, 553–570.
- Furusawa, Y.; Obata, Y.; Fukuda, S.; Endo, T.A.; Nakato, G.; Takahashi, D.; Nakanishi, Y.; Uetake, C.; Kato, K.; Kato, T.; et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 2013, 504, 446–450.
- Karlsson, F.H.; Fåk, F.; Nookaew, I.; Tremaroli, V.; Fagerberg, B.; Petranovic, D.; Bäckhed, F.; Nielsen, J. Symptomatic atherosclerosis is associated with an altered gut metagenome. Nat. Commun. 2012, 3, 1245.
- Vatanen, T.; Kostic, A.D.; d’Hennezel, E.; Siljander, H.; Franzosa, E.A.; Yassour, M.; Kolde, R.; Vlamakis, H.; Arthur, T.D.; Hämäläinen, A.M.; et al. Variation in microbiome LPS immunogenicity contributes to autoimmunity in humans. Cell 2016, 165, 842–853.
- Lehto, M.; Groop, P.-H. The Gut-Kidney Axis: Putative Interconnections between Gastrointestinal and Renal Disorders. Front. Endocrinol. 2018, 9, 553.
- Huang, W.; Zhou, L.; Guo, H.; Xu, Y.; Xu, Y. The role of short-chain fatty acids in kidney injury induced by gut-derived inflammatory response. Metabolism 2017, 68, 20–30.
- Lekawanvijit, S. Role of gut-derived protein-bound uremic toxins in cardiorenal syndrome and potential treatment modalities. Circ. J. 2015, 79, 2088–2097.
- Nanto-Hara, F.; Kanemitsu, Y.; Fukuda, S.; Kikuchi, K.; Asaji, K.; Saigusa, D.; Iwasaki, T.; Ho, H.-J.; Mishima, E.; Suzuki, T.; et al. The guanylate cyclase C agonist linaclotide ameliorates the gut–cardio–renal axis in an adenine-induced mouse model of chronic kidney disease. Nephrol. Dial. Transpl. 2020, 35, 250–264.
