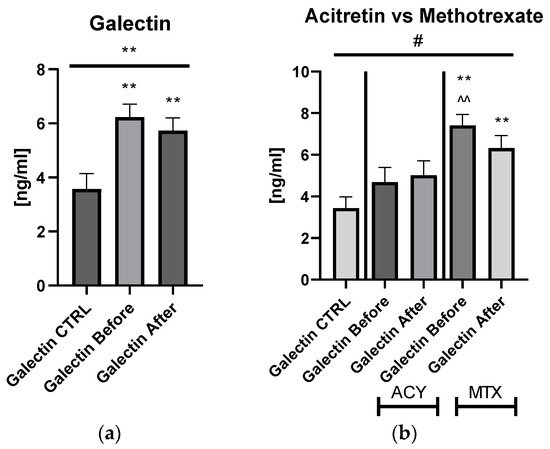
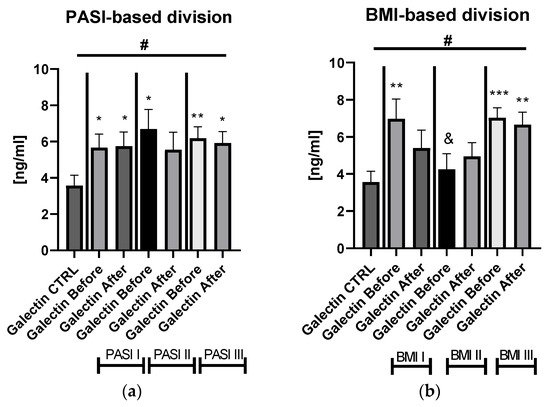
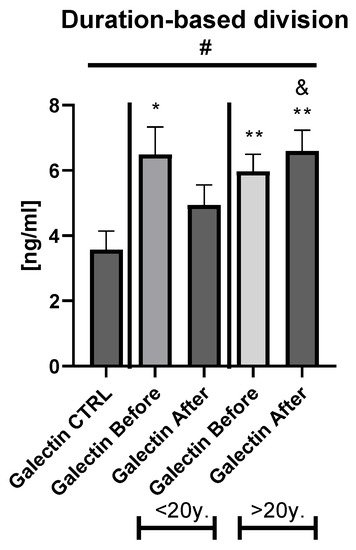
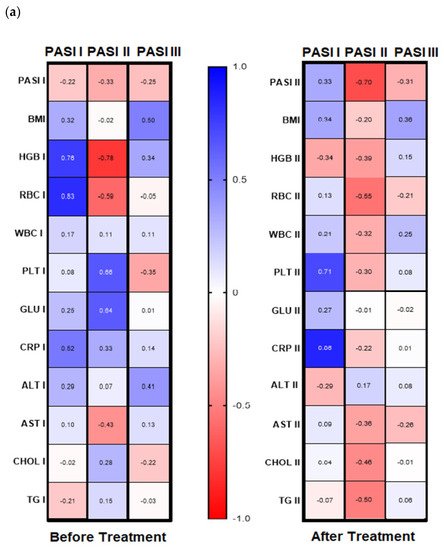
Galectin-3 (gal-3) regulates many different biological processes and diseases, which are common accompanying diseases of psoriasis. Psoriasis is one of the most common skin diseases. There is little information about potential diagnostic role of gal-3 in psoriasis. Serum gal-3 concentrations were measured before and after twelve weeks of antipsoriatic treatment in patients with psoriasis and compared to 11 persons without psoriasis (control group). Serum gal-3 level in patients with psoriasis was significantly higher compared to the control group. In obese patients and long-lasting psoriasis positive relations of gal-3 and index of psoriasis severity were noted. In psoriatics with low gal-3 levels, it was noted that the higher the gal-3, the higher the BMI and glucose level. In patients with long history of psoriasis it was observed that the higher gal-3, the lower the lipids levels. The Gal-3 level might be a factor affecting the course of psoriasis and useful in prediction of cardiometabolic comorbidities, especially in patients with a long history of the disease or obesity. Patients with low serum gal-3 and a short history of psoriasis may have greater risk of diabetes. In obese patients with long-lasting psoriasis, gal-3 may have a beneficial influence against abnormal lipid profiles or perhaps further cardiovascular disorder development.
1. Introduction
Psoriasis is a common chronic, autoimmune, and inflammatory disease affecting 2–4% of the global population and an incidence of 60.4 to 140 new cases per 100,000
[1,2][1][2]. In recent years the viewpoint on the pathogenesis of psoriasis has evolved significantly at the same time due to continuous enriched research in this field. Thus, currently psoriasis is definitely more than skin deep and closely related to numerous cardiometabolic disorders (CMDs), including obesity, diabetes mellitus (DM), dyslipidemia, and hypertension, which make up the metabolic syndrome (MS), as well as non-alcoholic fatty liver disease (NAFLD) and cardiovascular diseases (CVD)
[2,3,4][2][3][4]. It has been widely proven that the higher rate of comorbidities is related to psoriasis severity
[5,6][5][6]. Persons with psoriasis have shortened life expectancy of over 5 years mainly due to CVD and increased relative risk of mortality of 1.53 in comparison to 1.12 in the general population
[7]. Psoriatics with severe form present a higher rate of ischemic heart diseases but also have additional absolute risk of 6.2% of a major adverse cardiovascular event (MACE) within 10 years of psoriasis history compared to the healthy population
[2,8][2][8]. The multidirectional relationship of psoriasis with numerous comorbidities is translated by common genetic or immunological pathways, oxidative stress, as well as systemic metabolically driven inflammation which is important in psoriasis pathogenesis and consequently leads to the development of atherosclerosis, insulin resistance, and CMDs
[3,4,5][3][4][5]. For years, researchers have been constantly looking for novel modulators of metaflammation in psoriasis to reduce the risk of internal disorders or use as potential targets for therapeutic intervention. We also previously investigated various active proteins in serum of psoriatic patients and demonstrated them as novel indicators of inflammation or the metabolic complications development in psoriasis
[9,10][9][10].
According to recent accumulating evidence, galectins seem to be very promising molecular effectors mediating a wide range of biological processes in various tissues including cutaneous and involved in pathogenesis of various diseases
[11,12][11][12]. Galectins are a growing family of β-galactoside-binding lectins consisting of either one or two 130 amino-acid-long highly conserved carbohydrate recognition domains (CRD). At least 15 galectins have been described in mammalians so far and they have been divided into three groups based on their CRD number, structure, and properties
[11,13,14,15][11][13][14][15]. Galectins, by binding saccharides in glycoconjugates on the cell surface, exert multimodal biological roles, not yet fully known, in the regulation of cell–cell and cell–matrix interactions, immunity, expression of certain genes, regeneration, apoptosis, and cancer progression
[11,12,14,16,17][11][12][14][16][17]. It is noteworthy that the molecules can act opposite, depending on various conditions, serving as positive or negative modulators of the same processes
[12,13,14][12][13][14]. The plasticity of galectins and their wide distribution in tissues (not only in the cytoplasm and nuclei but also in the extracellular space) makes them extremely interesting for research but also clinical usability. Many galectins take part in cancers’ invasiveness, and have been related with higher rates of metastases and poorer survival outcome in different cancers
[11,18][11][18]. Galectin-7 has been linked not only with lung cancer, but also with being highly expressed in epithelial tissues, and related to the regulation of skin homeostasis or keratinocytes proliferation
[18,19][18][19]. Chen et al. demonstrated that galectin-7 mRNA was downregulated in human psoriatic lesions and a IL-23-induced psoriasis model that proves the role of the lectin in pathogenesis of psoriasis
[19].
Galectin-3 (gal-3) is the only representative of chimera-type galectin, of a 35-kDa weight, encoded by the LGALS3 gene located on chromosome 14
[13,14,20][13][14][20]. It has a wide tissue distribution with high expression in epithelial and myeloid cells, but also in these involved in immune response, such as neutrophils, eosinophils, monocytes, mast cells, Langerhans cells, dendritic cells, osteoclasts, chondrocytes or activated macrophages, and T- and B-cells
[13,15,16][13][15][16]. With reference to the skin, its expression is high in keratinocytes, hair follicles, and sebaceous and sweat glands, but also secreted by other resident skin cells such as melanocytes, fibroblasts, and monocytes
[15,17][15][17]. Depending on the localization of galectin-3—extra- or intracellular—it displays different functions
[13]. Gal-3, even most other galectins, is involved in cell adhesion to extracellular matrix, modulation of cell migration and adhesion, and inhibition of apoptosis. It is a significant regulator of signal transduction and immune surveillance, cell growth, and differentiation, including epidermal keratinocytes. Further, gal-3 promotes angiogenesis and induction of fibrosis
[16]. Similarly to other lectins, gal-3 is a pro-tumor factor enhancing oncogenesis and metastasis
[13]. Its higher expression was noted in various tumors such as of the thyroid gland, the central nervous system, the kidney, the liver, and lymphoma or melanoma
[13]. The multifunctional activity of gal-3, as well as in the skin, results in great interest over other galectins for its role in various clinical conditions and diseases, such as asthma, cancer, atherosclerosis, CVD, obesity, diabetes mellitus, kidney disorders, atopic dermatitis, and psoriasis, as well. Much research has demonstrated rapid expression of gal-3 in biological fluids from injured or inflammatory cells under diseased conditions indicating the lectin as a sensitive regulator of many critical and pathological processes and an active promotor of inflammation
[14,20,21][14][20][21]. Furthermore, gal-3 has been identified as a next-generation marker for detection of early stages of different diseases inter alia myocardial dysfunction and heart failure (HF) or cardiac degeneration in acute myocarditis
[14,22,23][14][22][23]. Numerous data proved that elevated serum galectin-3 is related with recurrent HF, poorer prognosis of coronary heart disease and increased risk of death
[13,20,23,24][13][20][23][24]. In 2014, the Food and Drug Administration (FDA) identified gal-3 as one of the validated cardiovascular biomarkers
[21]. Further, lectin is strongly linked with atherosclerosis which also is closely related to psoriasis
[20,21][20][21]. An increased gal-3 level was positively correlated with carotid intima media thickness which is again also greater in psoriasis especially with concomitant metabolic syndrome
[25,26][25][26]. Therefore, the gal-3 assessment might be extremely useful in diagnosis and prediction of CMDs in psoriasis. Data on this subject are scarce so far. Kotwica et al. suggested that enhanced profibrotic activity reflected by elevated serum gal-3 may be involved in subclinical myocardial impairment in psoriasis
[27]. Lacina et al. investigated the expression of several galectins in the psoriatic epithelium which did not express gal-3 contrary to normal epidermis
[28]. Interestingly, strong expression of galectin-3/galectin-3-reactive glycoligands in capillaries of psoriatic plaques pointed that altered galectin expression and binding patterns in psoriatic skin indicates its modifying impact on keratinocyte maturation in hyperactivated psoriatic epithelium and thus on the development of psoriatic skin lesions
[28].
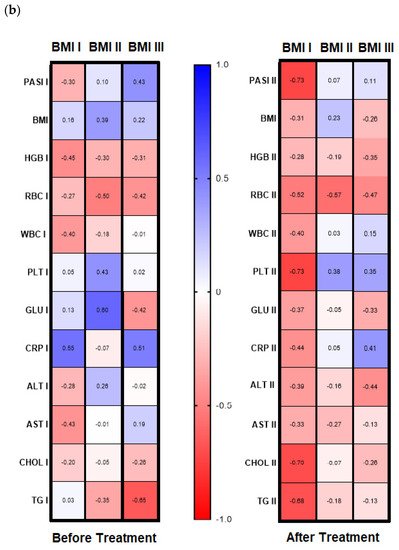
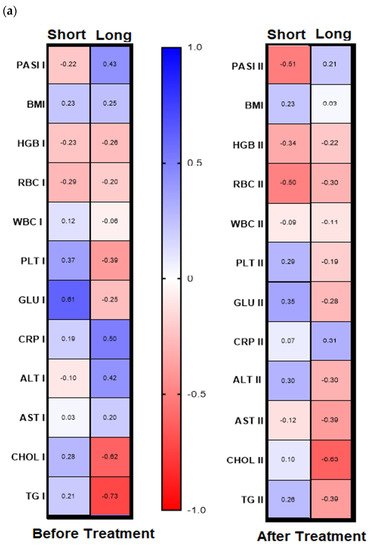
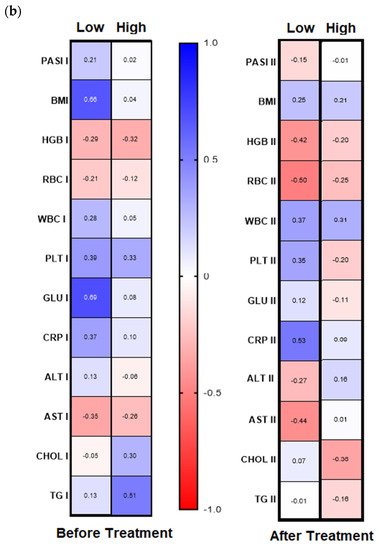
The wide range of multifunctionality of gal-3 inter alia being a biomarker of inflammation, atherosclerosis, fibrosis, oxidative stress, and many others, involved intensively in the pathogenesis of psoriasis, has prompted us to investigate the potential diagnostic and clinical usefulness of gal-3 in serum of psoriatics and its relationship with the disease severity, metabolic, or inflammatory indicators. We aimed to look for various interrelations between gal-3 levels and various parameters in terms of duration of psoriasis to clarify whether the lectin may be a factor stratifying the risk of developing complications. Furthermore, to the best of our knowledge, we are one of the first who evaluated the impact of classic systemic antipsoriatic treatment on gal-3 levels in order to assess its potency to assess the efficacy of certain drugs in psoriasis or perhaps contribute to develop newer therapeutic strategies.
2. Results
The study included 31 patients with a flare of plaque-type psoriasis, 12 women and 19 men, with the mean age of 51.4 ± 9.61, and 11 healthy volunteers of the mean age of 55.2 ± 15.6, matched for age, weight, and BMI. The mean value of BMI of the patients was 28.5 ± 6.53 kg/m
2. Severity of psoriatic skin lesions expressed by mean PASI score was 17.15 ± 7.92 before treatment and 4.31 ± 2.06 after therapy. Baseline characteristic of the control group and patients are summarized in
Table 1 and
Table 2. Additional, more accurate analysis of the psoriatic subjects subdivided with regard to the specific drug they were treated with, along with division according to BMI and PASI, has been placed in the
Supplementary Materials (could be found in https://www.
mdpi.com/2079-7737/11/1/88#supplementary).
Table 1. Baseline characteristics of patients and control group.
| Parameters |
Control Group (n = 11) |
Patients Group (n = 31) |
| Parameter |
Before Treatment |
After Treatment |
| Age [years] |
51.4 ± 9.61 |
55.2 ± 15.6 |
| PASI |
17.15 ± 7.92 |
| Height [cm] |
167 ± 9.3 |
172 ± 10.3 |
| 4.31 ± 2.06 *** |
Weight [kg] |
74.6 ± 21.3 |
85.2 ± 21.3 |
| BMI [kg/m2] |
25.6 ± 6.53 |
28.5 ± 6.53 |
Table 2. Basal characteristic of the patients’ group before and after total treatment.
| Hemoglobin [g/dL] |
13.55 ± 1.71 |
13.21 ± 1.48 |
| RBC [x103/mL] |
4.37 ± 0.57 |
4.29 ± 0.48 |
| WBC [x103/mL] |
7.64 ± 1.88 |
6.63 ± 1.61 * |
| PLT [x103/mL] |
255 ± 64.5 |
231.7 ± 59.2 |
| Glucose [mg/dL] |
85 (53–215) |
88.5 (55–140) |
| CRP [mg/L] |
5.13 (1–34.7) |
1.81 (0.5–15) ** |
| ALT [U/L] |
24.87 ± 9.05 |
21.03 ± 10.55 |
| AST [U/L] |
21.5 (14–86) |
19 (12–52) |
| Total Chol [mg/dL] |
171.6 ± 38.2 |
170.4 ± 35.6 |
| TG [mg/dL] |
137.1 ± 55.1 |
120.1 ± 46.62 |