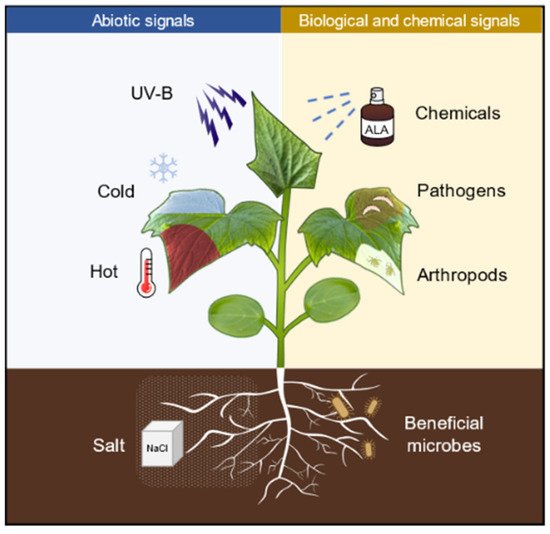
Priming is an adaptive strategy that improves plant defenses against biotic and abiotic stresses. Stimuli from chemicals, abiotic cues, and pathogens can trigger the establishment of priming state. Priming with 5-aminolevulinic acid (ALA), a potential plant growth regulator, can enhance plant tolerance to the subsequent abiotic stresses, including salinity, drought, heat, cold, and UV-B. However, the molecular mechanisms underlying the remarkable effects of ALA priming on plant physiology remain to be elucidated.
- 5-ALA
- defense priming
- abiotic stress
- plant hormone
1. Introduction
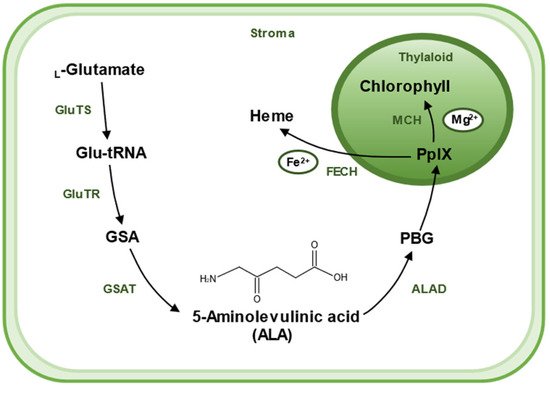
2. Biosynthesis of 5-Aminolevulinic Acid
There are two pathways for the biosynthesis of ALA, the C4 pathway (or Shemin pathway) and the C5 pathway (or Beale pathway) [17][15]. The C4 pathway is found in animals, fungi, and some algae and bacteria. In this pathway, ALA is produced by direct condensation of succinyl-CoA and Gly catalyzed by ALA synthase. The C5 pathway is mainly found in plants and archaea, which consists of a three-step enzymatic reaction [18][16]. Firstly, L-glutamate is ligated to tRNAGlu, which is catalyzed by glutamyl–tRNA synthetase (GluTS) to form L-glutamy–tRNA. Secondly, the carboxyl group of Glu-tRNA is reduced to a formyl group and L-Glu-tRNA is converted to L-glutamic acid 1-semialdehyde (GSA). GluTR plays a key role during the synthesis pathway of ALA. Lastly, GSA undergoes an isomerization reaction catalyzed by glutamate-1-semialdehyde aminotransferase (GSAT) to form ALA. These reactions are located in the chloroplast stroma [19][17] (Figure 2).
3. ALA Priming Enhances Plant Resistance to Abiotic Stresses
3.1. ALA Priming Alleviates Salt Stress
Priming with ALA increases the transcripts and protein accumulations of SOS1 (Na+/H+ antiporter) and HA3 (proton pump) on the plasma membrane (PM) as well as NHX1 (Na+/H+ antiporter) and VHA-A (proton pump) on the vesicle membrane compared with the unprimed cucumber (Cucumis sativus) in response to salt stress. The ion transporter proteins SOS1 and NHX1, with the energy provided by proton pump HA3 and VAH-A, help cucumber excrete Na+ from the cytoplasm or transfer it to the vesicles, resulting in a high-low-high osmotic potential in the vesicle-protoplast-exosome, and thus alleviating ion toxicity induced by salt stress. Pretreatment with ALA upregulates the expression of high-affinity K+ transporter protein 1 (HKT1) that regulates Na+/K+ homeostasis in cucumber cells and maintains normal metabolic activities in cells under salt stress conditions [27,28][22][23]. Proline accumulates in response to salinity and is a common compatible osmolyte in higher plants. Exogenous application of ALA upregulates delta-1-pyrroline-5-carboxylate synthase (P5CS) that controls the rate-limiting step of glutamate-derived proline biosynthesis in Oilseed rape (Brassica napus) and enhances tolerance to salt stress [26,29][24][25]. In addition, priming with ALA relieves cell oxidation stress caused by salt stress by improving the activity of antioxidant enzymes, including superoxide dismutase (SOD), catalase (CAT), and peroxidase (POD), and promoting the activity of enzymes involved in the ascorbate-glutathione cycle (AsA-GSH), including ascorbic acid oxidase (AAO), ascorbate peroxidase (APX), glutathione reductase (GR), dehydroascorbic acid reductase (DHAR), and monodehydroascorbic acid reductase (MDHAR) [30,31,32,33][26][27][28][29]. In addition to coping with osmotic stress and oxidative stress caused by salt stress, priming with ALA improves plant salt tolerance by increasing photosynthetic assimilation and promoting nitrogen metabolism. Cassia seed (Cassia obtusifolia), peach (Prunnus persica), and oilseed rape treated with ALA showed an increase in the net photosynthetic rate (Pn) and transpiration rate (Tr), as well as the photochemical efficiency of photosystem II (Fv/Fm) and the non-photochemical quenching (NPQ) during salt stress [29,31,34][25][27][30]. The induction of salt tolerance in plants by ALA may be achieved through nitric oxide (NO). ALA treatment increased NO and NOS activity in leaves, suggesting that ALA triggers NO synthesis by activating NOS, and thus improves salt tolerance in maize (Zea mays) [38][31].3.2. ALA Priming Increases Plant Tolerance to Extreme Temperature
ALA-pretreated cucumber leaves had higher antioxidant enzyme activity, higher levels of proline and soluble sugar content, and weaker growth inhibition under high-temperature stress conditions [43][32]. Priming with ALA increases germination and seedling emergence in red pepper (Capsicum annuum) and reduces tissue electrolyte leakage in rice (Oryza sativa) under cold stress [44,45][33][34]. Pretreatment with ALA also increases chlorophyll content and photosynthetic capacity of cucumber and enhances ribulose-1,5-bisphosphate (RuBP) carboxylase activity in maize under cold stress conditions [46,47,48][35][36][37]. Furthermore, ALA treatment also improved the antioxidant capacity of plants in response to cold stress by increasing the activities of SOD, APX, GR, CAT, and heme oxygenase-1 (HO-1) in red pepper, drooping wild ryegrass (Elymus nutans), and soybean plants (Glycine max) [49,50,51][38][39][40]. Interestingly, ALA priming upregulates the expression levels of respiratory burst oxidase homologue1 (RBOH1) in tomato (Solanum lycopersicum) and leads to the production of H2O2, which serves as a signaling molecule to activate defense against cold stress [52][41].3.3. ALA Priming Mitigates Drought-Induced Damage
3.4. ALA Priming Attenuates UV-B-Induced Damage
Ultraviolet-B (UV-B) radiation is a component of sunlight that induces several plant photomorphogenic responses, including hypocotyl growth inhibition and cotyledon curling [68][52]. High-intensity UV-B injures plants by damaging DNA, impaired photosynthesis, and cell death, and triggering the accumulation of ROS [69][53]. Priming with ALA was reported to significantly reduce plant damage from UV-B radiation by promoting photosynthesis, enhancing antioxidant capacity, and improving nitrogen metabolism. As a key precursor of chlorophyll biosynthesis, ALA alleviated the deficiency of chlorophyll biosynthesis during UV-B stress; ALA pretreatment upregulates the expression of genes involved in chlorophyll biosynthesis such as glutamyl-tRNA reductase (HEMA1), Mg-chelatase (CHLH), and protochlorophyllide oxidoreductase (POR) in pigeon pea (Cajanus cajan), thus promoting plant photosynthesis during UV-B stress [8,70][8][54]. In addition, ALA priming-increased activities of antioxidant enzymes are essential for lettuce (Lactuca sativa) resistance to UV-B stress [71][55]. In addition to enzymatic antioxidants, ALA also increases the content of non-enzymatic antioxidants such as flavonoids and phenolics [8]. Under UV-B stress conditions, ALA priming significantly improves the activities of nitrate reductase (NR), nitrite reductase (NiR), glutamine synthetase (GS), and glutamate synthase (GOGAT), and then increases the levels of NO3− and NO2− in the seedlings of pigeon pea [70][54]. Collectively, ALA priming contributes to UV-B tolerance by regulating photosynthesis, antioxidant, and nitrogen metabolism in plants.
References
- Martinez-Medina, A.; Flors, V.; Heil, M.; Mauch-Mani, B.; Pieterse, C.M.J.; Pozo, M.J.; Ton, J.; van Dam, N.M.; Conrath, U. Recognizing Plant Defense Priming. Trends Plant Sci. 2016, 21, 818–822.
- Mauch-Mani, B.; Baccelli, I.; Luna, E.; Flors, V. Defense Priming: An Adaptive Part of Induced Resistance. Annu. Rev. Plant Biol. 2017, 68, 485–512.
- Díaz-Valle, A.; López-Calleja, A.C.; Alvarez-Venegas, R. Enhancement of Pathogen Resistance in Common Bean Plants by Inoculation With Rhizobium etli. Front. Plant Sci. 2019, 10, 1317.
- Kim, J.I.; Baek, D.; Park, H.C.; Chun, H.J.; Oh, D.H.; Lee, M.K.; Cha, J.Y.; Kim, W.Y.; Kim, M.C.; Chung, W.S.; et al. Overexpression of Arabidopsis YUCCA6 in potato results in high-auxin developmental phenotypes and enhanced resistance to water deficit. Mol. Plant 2013, 6, 337–349.
- Singh, P.; Yekondi, S.; Chen, P.-W.; Tsai, C.-H.; Yu, C.-W.; Wu, K.; Zimmerli, L. Environmental History Modulates Arabidopsis Pattern-Triggered Immunity in a HISTONE ACETYLTRANSFERASE1–Dependent Manner. Plant Cell 2014, 26, 2676–2688.
- Savvides, A.; Ali, S.; Tester, M.; Fotopoulos, V. Chemical Priming of Plants Against Multiple Abiotic Stresses: Mission Possible? Trends Plant Sci. 2016, 21, 329–340.
- Mohamed, S.; Hangqi, S.; Jungui, X.; Yajing, G.; Wenjian, S.; Jin, H. Seed polyamines metabolism induced by seed priming with spermidine and 5-aminolevulinic acid for chilling tolerance improvement in rice (Oryza sativa L.) seedlings. Environ. Exp. Bot. 2017, 137, 58–72.
- Gupta, D.; Prasad, S.M. Priming with 5-aminolevulinic acid (ALA) attenuates UV-B induced damaging effects in two varieties of Cajanus cajan L. seedlings by regulating photosynthetic and antioxidant systems. S. Afr. J. Bot. 2021, 138, 129–140.
- Conrath, U.; Beckers, G.J.M.; Langenbach, C.J.G.; Jaskiewicz, M.R. Priming for Enhanced Defense. Annu. Rev. Phytopathol. 2015, 53, 97–119.
- Tanou, G.; Filippou, P.; Belghazi, M.; Job, D.; Diamantidis, G.; Fotopoulos, V.; Molassiotis, A. Oxidative and nitrosative-based signaling and associated post-translational modifications orchestrate the acclimation of citrus plants to salinity stress. Plant J. 2012, 72, 585–599.
- Li, M.; Yu, G.; Ma, J.; Liu, P. Interactions of importers in long-distance transmission of wound-induced jasmonate. Plant Signal. Behav. 2021, 16, 1886490.
- Lee, H.-R.; Lee, S.; Park, S.; van Kleeff, P.J.M.; Schuurink, R.C.; Ryu, C.-M. Transient Expression of Whitefly Effectors in Nicotiana benthamiana Leaves Activates Systemic Immunity Against the Leaf Pathogen Pseudomonas syringae and Soil-Borne Pathogen Ralstonia solanacearum. Front. Ecol. Evol. 2018, 6, 90.
- Ramírez-Carrasco, G.; Martínez-Aguilar, K.; Alvarez-Venegas, R. Transgenerational Defense Priming for Crop Protection against Plant Pathogens: A Hypothesis. Front. Plant Sci. 2017, 8, 696.
- Antoniou, C.; Savvides, A.; Christou, A.; Fotopoulos, V. Unravelling chemical priming machinery in plants: The role of reactive oxygen–nitrogen–sulfur species in abiotic stress tolerance enhancement. Curr. Opin. Plant Biol. 2016, 33, 101–107.
- Iida, K.; Kajiwara, M. Carbon Source Dependence of the Ratio of δ-Aminolevulinic Acid Biosynthesis via the C5 and Shemin Pathways in Euglena Gracilis (Euglenophyceae)1. J. Phycol. 2008, 44, 292–298.
- Wu, Y.; Liao, W.; Dawuda, M.M.; Hu, L.; Yu, J. 5-Aminolevulinic acid (ALA) biosynthetic and metabolic pathways and its role in higher plants: A review. Plant Growth Regul. 2019, 87, 357–374.
- Senge, M.O.; Ryan, A.A.; Letchford, K.A.; MacGowan, S.A.; Mielke, T. Chlorophylls, Symmetry, Chirality, and Photosynthesis. Symmetry 2014, 6, 781–843.
- Kang, Z.; Zhang, J.; Zhou, J.; Qi, Q.; Du, G.; Chen, J. Recent advances in microbial production of δ-aminolevulinic acid and vitamin B12. Biotechnol. Adv. 2012, 30, 1533–1542.
- Yang, P.; Liu, W.; Cheng, X.; Wang, J.; Wang, Q.; Qi, Q.; Liu, S.-J. A New Strategy for Production of 5-Aminolevulinic Acid in Recombinant Corynebacterium glutamicum with High Yield. Appl. Environ. Microbiol. 2016, 82, 2709–2717.
- Noh, M.H.; Lim, H.G.; Park, S.; Seo, S.W.; Jung, G.Y. Precise flux redistribution to glyoxylate cycle for 5-aminolevulinic acid production in Escherichia coli. Metab. Eng. 2017, 43, 1–8.
- Chen, B.; Li, J.; Feng, Y.; Le, K.; Zai, Y.; Tang, X.; Sun, Y.; Zeng, X.; Lin, L. Green and mild production of 5-aminolevulinic acid from algal biomass. Korean J. Chem. Eng. 2021, 38, 899–905.
- Anirban, B. Strawberries under salt stress: ALA and ROS to the rescue. Physiol. Plant. 2019, 167, 2–4.
- Yue, W.; Na, L.; Linli, H.; Weibiao, L.; Zhongqi, T.; Xuemei, X.; Jian, L.; Jianming, X.; Alejandro, C.-U.; Jihua, Y. 5-Aminolevulinic Acid Improves Morphogenesis and Na+ Subcellular Distribution in the Apical Cells of Cucumis sativus L. under Salinity Stress. Front. Plant Sci. 2021, 12, 404.
- Liyun, L.; Nguyen Tran, N.; Akihiro, U.; Hirofumi, S. Effects of 5-aminolevulinic acid on Swiss chard (Beta vulgaris L. subsp. cicla) seedling growth under saline conditions. Plant Growth Regul. 2014, 74, 219–228.
- Jun-Lan, X.; Hang-Chao, W.; Xiao-Yu, T.; Chun-Lei, Z.; Muhammad Shahbaz, N. 5-aminolevulinic acid improves salt tolerance mediated by regulation of tetrapyrrole and proline metabolism in Brassica napus L. seedlings under NaCl stress. Plant Physiol. Biochem. 2018, 124, 88–99.
- Nudrat Aisha, A.; Muhammad, A.; Al-Qurainy, F. Aminolevulinic acid-induced changes in some key physiological attributes and activities of antioxidant enzymes in sunflower (Helianthus annuus L.) plants under saline regimes. Sci. Hortic-Amst. 2012, 142, 143–148.
- Jia, Y.; Qiang, C.; Ting, T.; Gui, W.; Feng, X. Promotive effects of 5-Aminolevulinic acid on growth, photosynthetic gas exchange, chlorophyll, and antioxidative enzymes under salinity stress in Prunnus persica (L.) Batseh Seedling. Emir. J. Food Agric. 2016, 786–795.
- Mohamed, E.-E.; Ibrahim, A.; Abdulaziz, A.; Hayssam, A.; Aisha, A.; Jacques, W.; Margaret, A. Genetic Variation and Alleviation of Salinity Stress in Barley (Hordeum vulgare L.). Molecules 2018, 23, 2488.
- Yue, W.; Linli, H.; Weibiao, L.; Mohammed Mujitaba, D.; Jian, L.; Jianming, X.; Zhi, F.; Alejandro, C.-U.; Jihua, Y. Foliar application of 5-aminolevulinic acid (ALA) alleviates NaCl stress in cucumber (Cucumis sativus L.) seedlings through the enhancement of ascorbate-glutathione cycle. Sci. Hortic-Amst. 2019, 257, 108761.
- Chun-Ping, Z.; Yi-Cun, L.; Feng-Gang, Y.; Shi-Jun, H.; Hai-Ying, L.; Ping, H. Role of 5-aminolevulinic acid in the salinity stress response of the seeds and seedlings of the medicinal plant Cassia obtusifolia L. Bot. Stud. 2013, 54, 1–13.
- Cengiz, K.; Muhammad, A. Nitric Oxide is Required for Aminolevulinic Acid-Induced Salt Tolerance by Lowering Oxidative Stress in Maize (Zea mays). J. Plant Growth Regul. 2020, 40, 617–627.
- Zhang, J.; Li, D.M.; Gao, Y.; Yu, B.; Xia, C.X.; Bai, J.G. Pretreatment with 5-aminolevulinic acid mitigates heat stress of cucumber leaves. Biol. Plant. 2012, 56, 780–784.
- Ahmet, K.; Yakup, K. Promotion by 5-aminolevulenic acid of pepper seed germination and seedling emergence under low-temperature stress. Sci. Hortic-Amst. 2009, 119, 98–102.
- Hotta, Y.; Tanaka, T.; Luo, B.; Takeuchi, Y.; Konnai, M. Improvement of Cold Resistance in Rice Seedlings by 5-Aminolevulinic Acid. J. Pestic. Sci. 1998, 23, 29–33.
- Wang, Y.; Li, J.; Gu, W.; Zhang, Q.; Tian, L.; Guo, S.; Wei, S. Exogenous application of 5-aminolevulinic acid improves low-temperature stress tolerance of maize seedlings. Crop Pasture Sci. 2018, 69, 587–593.
- Ali, A.; Yan, Y.; Yumei, L.; Yansu, L.; Xianchang, Y. 5-Aminolevulinic Acid Improves Nutrient Uptake and Endogenous Hormone Accumulation, Enhancing Low-Temperature Stress Tolerance in Cucumbers. Int. J. Mol. Sci. 2018, 19, 3379.
- Ali, A.; Jun, W.; Xianchang, Y.; Chaoxing, H.; Yansu, L. Substrate Application of 5-Aminolevulinic Acid Enhanced Low-temperature and Weak-light Stress Tolerance in Cucumber (Cucumis sativus L.). Agronomy 2020, 10, 472.
- Karina, B.B.; María, L.T.; Alcira, B.; Guillermo, O.N. The role of 5-aminolevulinic acid in the response to cold stress in soybean plants. Phytochemistry 2010, 71, 2038–2045.
- Ahmet, K.; Yakup, K.; Ali Rıza, D. Enhancing chilling stress tolerance of pepper seedlings by exogenous application of 5-aminolevulinic acid. Environ. Exp. Bot. 2010, 67, 495–501.
- Juanjuan, F.; Yongfang, S.; Xitong, C.; Yuefei, X.; Tianming, H. Exogenous 5-aminolevulenic acid promotes seed germination in Elymus nutans against oxidative damage induced by cold stress. PLoS ONE 2014, 9, e107152.
- Liu, T.; Xu, J.; Zhang, J.; Li, J.; Hu, X. Exogenous 5-aminolevulinic acid pretreatment ameliorates oxidative stress triggered by low-temperature stress of Solanum lycopersicum. Acta Physiol Plant 2018, 40, 210.
- Kuiju, N.; Xiang, M.; Guoling, L.; Huiling, M.; Zhifeng, J.; Wenhui, L.; Qianqian, Y. 5-Aminolevulinic acid modulates antioxidant defense systems and mitigates drought-induced damage in Kentucky bluegrass seedlings. Protoplasma 2017, 254, 2083–2094.
- Dan, L.; Lingtong, W.; Muhammad Shahbaz, N.; Hongbo, L.; Xiangqin, D.; Ling, X.; Fan, Z.; Weijun, Z. 5-Aminolevulinic acid enhances photosynthetic gas exchange, chlorophyll fluorescence and antioxidant system in oilseed rape under drought stress. Acta Physiol. Plant 2013, 35, 2747–2759.
- Al-Thabet, S.S. Promotive Effect of 5-amino Levulinic Acid on Growth and Yield of Wheat Grown under Dry Conditions. J. Agron. 2006, 5, 45–49.
- Nudrat Aisha, A.; Shamim, K.; Naila, F.; Muhammad, A.; Fahad, A.-Q. 5−Aminolevulinic Acid Induces Regulation in Growth, Yield and Physio-Biochemical Characteristics of Wheat under Water Stress. Sains Malays. 2018, 47, 661–670.
- Kosar, F.; Akram, N.A.; Ashraf, M. Exogenously-applied 5-aminolevulinic acid modulates some key physiological characteristics and antioxidative defense system in spring wheat (Triticum aestivum L.) seedlings under water stress. S. Afr. J. Bot. 2015, 96, 71–77.
- Ahmad, S.; Anum Samreen, T.; Abdul, S.; Ahmad, N.; Abdul, Q.; Shabir, H.; Abdul, M. Foliage application of 5-aminolevulinic acid alleviates drought stress in sunflower (Helianthus annuus L.) through improving stay green and antioxidant enzymes activities. Acta Physiol. Plant 2021, 43, 1–7.
- Ostrowska, A.; Biesaga-Kościelniak, J.; Grzesiak, M.T.; Hura, T. Physiological responses of spring wheat to 5-aminolevulinic acid under water stress applied at seedling stage. Cereal Res. Commun. 2019, 47, 32–41.
- Rizwan, R.; Humaira, Y.; Iqbal, H.; Muhammad, I.; Muhammad Arslan, A.; Abida, P. Exogenously applied 5-aminolevulinic acid modulates growth, secondary metabolism and oxidative defense in sunflower under water deficit stress. Physiol. Mol. Biol. Plants 2020, 26, 489–499.
- Liu, D.; Hu, L.Y.; Ali, B.; Yang, A.G.; Wan, G.L.; Xu, L.; Zhou, W.J. Influence of 5-aminolevulinic acid on photosynthetically related parameters and gene expression in Brassica napus L. under drought stress. Soil Sci. Plant Nutr. 2016, 62, 254–262.
- Yuyan, A.; Lin, Q.; Liangju, W. ALA Pretreatment Improves Waterlogging Tolerance of Fig Plants. PLoS ONE 2016, 11, e0147202.
- Dotto, M.; Casati, P. Developmental reprogramming by UV-B radiation in plants. Plant Sci. 2017, 264, 96–101.
- Shi, C.; Liu, H. How plants protect themselves from ultraviolet-B radiation stress. Plant Physiol. 2021, 187, 1096–1103.
- Divya, G.; Sheo Mohan, P. 5-aminolevulinic acid (ALA) regulates photosynthetic performance and nitrogen metabolism status in UV-B challenged Cajanus cajan L. seedlings. J. Plant Biochem. Biot. 2021.
- Ozkan, A.; Omer Faruk, A.; Feyza Icoglu, A.; Ferhunde, A. Exogenous 5-aminolevulinic acid alleviates the detrimental effects of UV-B stress on lettuce (Lactuca sativa L) seedlings. Acta Physiol Plant 2017, 39, 55.
