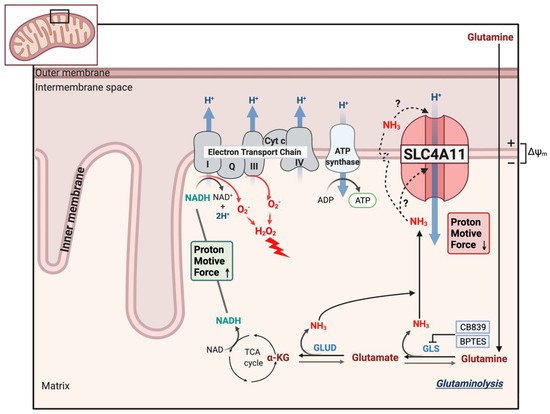
Solute-linked cotransporter, SLC4A11, a member of the bicarbonate transporter family, is an electrogenic H+ transporter activated by NH3 and alkaline pH. Although SLC4A11 does not transport bicarbonate, it shares many properties with other members of the SLC4 family. SLC4A11 mutations can lead to corneal endothelial dystrophy and hearing deficits that are recapitulated in SLC4A11 knock-out mice. SLC4A11, at the inner mitochondrial membrane, facilitates glutamine catabolism and suppresses the production of mitochondrial superoxide by providing ammonia-sensitive H+ uncoupling that reduces glutamine-driven mitochondrial membrane potential hyperpolarization. Mitochondrial oxidative stress in SLC4A11 KO also triggers dysfunctional autophagy and lysosomes, as well as ER stress. SLC4A11 expression is induced by oxidative stress through the transcription factor NRF2, the master regulator of antioxidant genes.
- glutamine
- ammonia
- corneal endothelial dystrophy
- lactate
- MCT4
1. Introduction
2. SLC4A11 Gene and Protein Characteristics
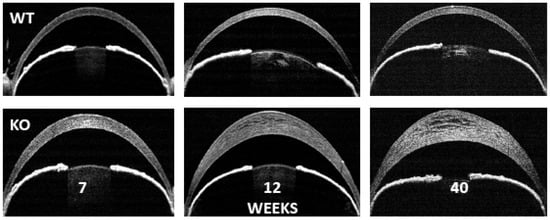
3. SLC4A11 Knock-Out Models
4. Gene Expression Changes in SLC4A11 Knock-Out
5. Ion Transport Function of SLC4A11
6. SLC4A11 Plasma Membrane Function
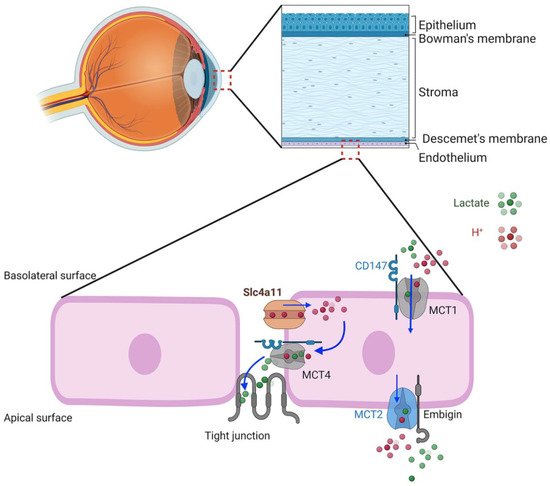
7. Facilitation of Glutamine Catabolism
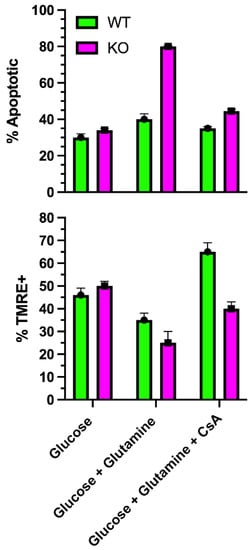
8. SLC4A11 Is a Mitochondrial Uncoupler
References
- Parker, M.D.; Ourmozdi, E.P.; Tanner, M.J. Human BTR1, a new bicarbonate transporter superfamily member and human AE4 from kidney. Biochem. Biophys. Res. Commun. 2001, 282, 1103–1109.
- Vilas, G.L.; Morgan, P.E.; Loganathan, S.K.; Quon, A.; Casey, J.R. A biochemical framework for SLC4A11, the plasma membrane protein defective in corneal dystrophies. Biochemistry 2011, 50, 2157–2169.
- Kao, L.; Azimov, R.; Shao, X.M.; Frausto, R.F.; Abuladze, N.; Newman, D.; Aldave, A.J.; Kurtz, I. Multifunctional Ion Transport Properties of Human SLC4A11: Comparison of the SLC4A11-B and SLC4A11-C Variants. Am. J. Physiol. Cell Physiol. 2016, 311, C820–C830.
- Damkier, H.H.; Nielsen, S.; Praetorius, J. Molecular expression of SLC4-derived Na+-dependent anion transporters in selected human tissues. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 293, R2136–R2146.
- Romero, M.F.; Chen, A.P.; Parker, M.D.; Boron, W.F. The SLC4 family of bicarbonate (HCO3−) transporters. Mol. Asp. Med. 2013, 34, 159–182.
- Qin, L.; Li, T.; Liu, Y. High SLC4A11 expression is an independent predictor for poor overall survival in grade 3/4 serous ovarian cancer. PLoS ONE 2017, 12, e0187385.
- Zahra, A.; Dong, Q.; Hall, M.; Jeyaneethi, J.; Silva, E.; Karteris, E.; Sisu, C. Identification of Potential Bisphenol A (BPA) Exposure Biomarkers in Ovarian Cancer. J. Clin. Med. 2021, 10, 1979.
- Jiao, X.; Sultana, A.; Garg, P.; Ramamurthy, B.; Vemuganti, G.K.; Gangopadhyay, N.; Hejtmancik, J.F.; Kannabiran, C. Autosomal recessive corneal endothelial dystrophy (CHED2) is associated with mutations in SLC4A11. J. Med. Genet. 2007, 44, 64–68.
- Kao, L.; Azimov, R.; Shao, X.M.; Abuladze, N.; Newman, D.; Zhekova, H.; Noskov, S.; Pushkin, A.; Kurtz, I. SLC4A11 function: Evidence for H+(OH−) and NH3-H+ transport. Am. J. Physiol. Cell Physiol. 2020, 318, C392–C405.
- Jalimarada, S.S.; Ogando, D.G.; Vithana, E.N.; Bonanno, J.A. Ion transport function of SLC4A11 in corneal endothelium. Investig. Ophthalmol. Vis. Sci. 2013, 54, 4330–4340.
- Loganathan, S.K.; Schneider, H.P.; Morgan, P.E.; Deitmer, J.W.; Casey, J.R. Functional assessment of SLC4A11, an integral membrane protein mutated in corneal dystrophies. Am. J. Physiol. Cell Physiol. 2016, 311, C735–C748.
- Ogando, D.G.; Jalimarada, S.S.; Zhang, W.; Vithana, E.N.; Bonanno, J.A. SLC4A11 is an EIPA-sensitive Na+ permeable pHi regulator. Am. J. Physiol. Cell Physiol. 2013, 305, C716–C727.
- Vithana, E.N.; Morgan, P.E.; Ramprasad, V.; Tan, D.T.; Yong, V.H.; Venkataraman, D.; Venkatraman, A.; Yam, G.H.; Nagasamy, S.; Law, R.W.; et al. SLC4A11 mutations in Fuchs endothelial corneal dystrophy. Hum. Mol. Genet. 2008, 17, 656–666.
- Loganathan, S.K.; Lukowski, C.M.; Casey, J.R. The cytoplasmic domain is essential for transport function of the integral membrane transport protein SLC4A11. Am. J. Physiol. Cell Physiol. 2016, 310, C161–C174.
- Vilas, G.L.; Loganathan, S.K.; Liu, J.; Riau, A.K.; Young, J.D.; Mehta, J.S.; Vithana, E.N.; Casey, J.R. Transmembrane water-flux through SLC4A11: A route defective in genetic corneal diseases. Hum. Mol. Genet. 2013, 22, 4579–4590.
- Kao, L.; Azimov, R.; Abuladze, N.; Newman, D.; Kurtz, I. Human SLC4A11-C functions as a DIDS-stimulatable H+(OH−) permeation pathway: Partial correction of R109H mutant transport. Am. J. Physiol. Cell Physiol. 2015, 308, C176–C188.
- Vilas, G.L.; Loganathan, S.K.; Quon, A.; Sundaresan, P.; Vithana, E.N.; Casey, J. Oligomerization of SLC4A11 protein and the severity of FECD and CHED2 corneal dystrophies caused by SLC4A11 mutations. Hum. Mutat. 2012, 33, 419–428.
- Park, M.; Li, Q.; Shcheynikov, N.; Muallem, S.; Zeng, W. Borate transport and cell growth and proliferation. Not only in plants. Cell Cycle 2005, 4, 24–26.
- Park, M.; Li, Q.; Shcheynikov, N.; Zeng, W.; Muallem, S. NaBC1 is a ubiquitous electrogenic Na+ -coupled borate transporter essential for cellular boron homeostasis and cell growth and proliferation. Mol. Cell 2004, 16, 331–341.
- Malhotra, D.; Loganathan, S.K.; Chiu, A.M.; Lukowski, C.M.; Casey, J.R. Human Corneal Expression of SLC4A11, a Gene Mutated in Endothelial Corneal Dystrophies. Sci. Rep. 2019, 9, 9681.
- Lopez, I.A.; Rosenblatt, M.I.; Kim, C.; Galbraith, G.C.; Jones, S.M.; Kao, L.; Newman, D.; Liu, W.; Yeh, S.; Pushkin, A.; et al. SLC4A11 gene disruption in mice: Cellular targets of sensorineuronal abnormalities. J. Biol. Chem. 2009, 284, 26882–26896.
- Groger, N.; Frohlich, H.; Maier, H.; Olbrich, A.; Kostin, S.; Braun, T.; Boettger, T. SLC4A11 prevents osmotic imbalance leading to corneal endothelial dystrophy, deafness, and polyuria. J. Biol. Chem. 2010, 285, 14467–14474.
- Han, S.B.; Ang, H.P.; Poh, R.; Chaurasia, S.S.; Peh, G.; Liu, J.; Tan, D.T.; Vithana, E.N.; Mehta, J.S. Mice with a targeted disruption of SLC4A11 model the progressive corneal changes of congenital hereditary endothelial dystrophy. Investig. Ophthalmol. Vis. Sci. 2013, 54, 6179–6189.
- Zhang, W.; Li, H.; Ogando, D.G.; Li, S.; Feng, M.; Price, F.W., Jr.; Tennessen, J.M.; Bonanno, J.A. Glutaminolysis is Essential for Energy Production and Ion Transport in Human Corneal Endothelium. EBioMedicine 2017, 16, 292–301.
- Ogando, D.G.; Choi, M.; Shyam, R.; Li, S.; Bonanno, J.A. Ammonia sensitive SLC4A11 mitochondrial uncoupling reduces glutamine induced oxidative stress. Redox Biol. 2019, 26, 101260.
- Shyam, R.; Ogando, D.G.; Choi, M.; Liton, P.B.; Bonanno, J.A. Mitochondrial ROS Induced Lysosomal Dysfunction and Autophagy Impairment in an Animal Model of Congenital Hereditary Endothelial Dystrophy. Investig. Ophthalmol. Vis. Sci. 2021, 62, 15.
- Shyam, R.; Ogando, D.G.; Kim, E.T.; Murugan, S.; Choi, M.; Bonanno, J.A. Rescue of the CHED Mouse Model by AAV-mediated SLC4A11 Replacement. Opthalmology Sci. 2021, 2.
- Li, S.; Kim, E.; Bonanno, J.A. Fluid transport by the cornea endothelium is dependent on buffering lactic acid efflux. Am. J. Physiol. Cell Physiol. 2016, 311, C116–C126.
- Li, S.; Kim, E.; Ogando, D.G.; Bonanno, J.A. Corneal Endothelial Pump Coupling to Lactic Acid Efflux in the Rabbit and Mouse. Investig. Ophthalmol. Vis. Sci. 2020, 61, 7.
- Ogando, D.G.; Bonanno, J.A. RNA sequencing uncovers alterations in corneal endothelial metabolism, pump and barrier functions of SLC4A11 KO mice. Exp. Eye Res. 2021, 214, 108884.
- Zhang, W.; Frausto, R.; Chung, D.D.; Griffis, C.G.; Kao, L.; Chen, A.; Azimov, R.; Sampath, A.P.; Kurtz, I.; Aldave, A.J. Energy Shortage in Human and Mouse Models of SLC4A11-Associated Corneal Endothelial Dystrophies. Investig. Ophthalmol. Vis. Sci. 2020, 61, 39.
- Ogando, D.G.; Shyam, R.; Kim, E.T.; Wang, Y.C.; Liu, C.Y.; Bonanno, J.A. Inducible SLC4A11 Knockout Triggers Corneal Edema Through Perturbation of Corneal Endothelial Pump. Investig. Ophthalmol. Vis. Sci. 2021, 62, 28.
- Zhang, W.; Ogando, D.G.; Bonanno, J.A.; Obukhov, A.G. Human SLC4A11 Is a Novel NH3/H+ Co-transporter. J. Biol. Chem. 2015, 290, 16894–16905.
- Myers, E.J.; Marshall, A.; Jennings, M.L.; Parker, M.D. Mouse SLC4A11 expressed in Xenopus oocytes is an ideally selective H+/OH− conductance pathway that is stimulated by rises in intracellular and extracellular pH. Am. J. Physiol. Cell Physiol. 2016, 311, C945–C959.
- Quade, B.N.; Parker, M.D.; Hoepflinger, M.C.; Phipps, S.; Bisson, M.A.; Foissner, I.; Beilby, M.J. The molecular identity of the characean OH− transporter: A candidate related to the SLC4 family of animal pH regulators. Protoplasma 2021, 1–2.
- Zhang, W.; Ogando, D.G.; Kim, E.T.; Choi, M.J.; Li, H.; Tenessen, J.M.; Bonanno, J.A. Conditionally Immortal Slc4a11−/− Mouse Corneal Endothelial Cell Line Recapitulates Disrupted Glutaminolysis Seen in Slc4a11−/− Mouse Model. Investig. Ophthalmol. Vis. Sci. 2017, 58, 3723–3731.
- Loganathan, S.K.; Casey, J.R. Corneal dystrophy-causing SLC4A11 mutants: Suitability for folding-correction therapy. Hum. Mutat. 2014, 35, 1082–1091.
- Chiu, A.M.; Mandziuk, J.J.; Loganathan, S.K.; Alka, K.; Casey, J.R. High Throughput Assay Identifies Glafenine as a Corrector for the Folding Defect in Corneal Dystrophy-Causing Mutants of SLC4A11. Investig. Ophthalmol. Vis. Sci. 2015, 56, 7739–7753.
- Nehrke, K. H(OH), H(OH), H(OH): A holiday perspective. Focus on “Mouse SLC4A11 expressed in Xenopus oocytes is an ideally selective H+/OH− conductance pathway that is stimulated by rises in intracellular and extracellular Ph”. Am. J. Physiol. Cell Physiol. 2016, 311, C942–C944.
- Contreras-Baeza, Y.; Sandoval, P.Y.; Alarcon, R.; Galaz, A.; Cortes-Molina, F.; Alegria, K.; Baeza-Lehnert, F.; Arce-Molina, R.; Guequen, A.; Flores, C.A.; et al. Monocarboxylate transporter 4 (MCT4) is a high affinity transporter capable of exporting lactate in high-lactate microenvironments. J. Biol. Chem. 2019, 294, 20135–20147.
- Mullen, A.R.; Wheaton, W.W.; Jin, E.S.; Chen, P.H.; Sullivan, L.B.; Cheng, T.; Yang, Y.; Linehan, W.M.; Chandel, N.S.; DeBerardinis, R.J. Reductive carboxylation supports growth in tumour cells with defective mitochondria. Nature 2011, 481, 385–388.
- Montoliu, C.; Cauli, O.; Urios, A.; ElMlili, N.; Serra, M.A.; Giner-Duran, R.; Gonzalez-Lopez, O.; Del Olmo, J.A.; Wassel, A.; Rodrigo, J.M.; et al. 3-nitro-tyrosine as a peripheral biomarker of minimal hepatic encephalopathy in patients with liver cirrhosis. Am. J. Gastroenterol. 2011, 106, 1629–1637.
- Alka, K.; Casey, J.R. Ophthalmic Nonsteroidal Anti-Inflammatory Drugs as a Therapy for Corneal Dystrophies Caused by SLC4A11 Mutation. Investig. Ophthalmol. Vis. Sci. 2018, 59, 4258–4267.
- Berry, B.J.; Trewin, A.J.; Amitrano, A.M.; Kim, M.; Wojtovich, A.P. Use the Protonmotive Force: Mitochondrial Uncoupling and Reactive Oxygen Species. J. Mol. Biol. 2018, 430, 3873–3891.
- Mailloux, R.J.; Harper, M.E. Uncoupling proteins and the control of mitochondrial reactive oxygen species production. Free Radic Biol. Med. 2011, 51, 1106–1115.
- Davuluri, G.; Allawy, A.; Thapaliya, S.; Rennison, J.H.; Singh, D.; Kumar, A.; Sandlers, Y.; Van Wagoner, D.R.; Flask, C.A.; Hoppel, C.; et al. Hyperammonaemia-induced skeletal muscle mitochondrial dysfunction results in cataplerosis and oxidative stress. J. Physiol. 2016, 594, 7341–7360.