Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Camila Xu and Version 1 by Natália Cruz-Martins.
Protein-tyrosine phosphatase 1B (PTP1B) negatively regulates insulin signaling pathways and plays an important role in type 2 diabetes mellitus (T2DM), as its overexpression may induce insulin resistance.
- protein-tyrosine phosphatase 1B
- type 2 diabetes mellitus
- insulin signaling pathways
1. Introduction
Diabetes mellitus (DM) is considered a major health problem worldwide [1]. Obesity and diabetes incidence still continue increasing due to globalization, mechanization, and changes in human lifestyle and daily routines [2]. According to the International Diabetes Federation (IDF), it was estimated that in 2017 there were 451 million (age 18–99 years) people with diabetes worldwide. These figures were expected to increase to 693 million by 2045 [3]. DM is a chronic metabolic disease that results from defects in insulin action, insulin secretion, or both, leading to persistent hyperglycemia [4].
Currently, type 2 diabetes mellitus (T2DM) represents a major threat to health [5]. Characterized by increased blood glucose levels, this is the underlying reason for several complications, including cardiovascular disorders, blindness, kidney failure, and peripheral nerve damages [6]. The development of T2DM and its complications are related, in most cases, to insulin resistance and postprandial hyperglycemic variations [7,8][7][8]. Thus, an effective drug for controlling insulin resistance may be beneficial in improving the quality life of T2DM patients. Several pharmacological strategies have been investigated on DM treatment, including insulin release stimulation, gluconeogenesis inhibition, glucose transport activity increase, and intestinal glucose absorption reduction [9]. Insulin supplements and other oral anti-diabetic drugs can be used alone or in combination to improve glycemic regulation [10]. However, some of the available anti-diabetic drugs have either the disadvantage of having low efficacy or serious side effects [11]. Thus, there is a continuous search for more effective and safer anti-hyperglycemic agents, especially from natural origins.
Insulin sensitizers, such as thiazolidinediones (TZDs or glitazones) have been used as effective drugs for T2DM treatment [12]. The identification of the enzyme responsible for the dephosphorylation of insulin receptors, called protein-tyrosine phosphatase 1B (PTP1B), showed that the inhibitors of such an enzyme could be employed as insulin sensitizer agents and, therefore, as promising anti-diabetic drugs [13]. This hypothesis was confirmed in mouse models, where it was found that PTP1B gene disruption can increase insulin sensitivity. Similar results were also obtained when PTP1B antisense nucleotides suppressed PTP1B gene expression [14].
Protein tyrosine phosphatases (PTPs) constitute a huge and structurally variable family of highly regulated enzymes. Most PTPs have been proposed to be targets for advanced drug discovery, and PTP1B is one of the well-established enzymes among PTPs [15,16,17][15][16][17]. It was the first isolated member of the PTP superfamily, and since then, growing evidence has linked it with insulin resistance, obesity, and T2DM. Numerous studies have shown that PTP1B can negatively regulate insulin and leptin signaling pathways. Indeed, PTP1B dephosphorylates both insulin receptor and its substrate IRS-1 in the insulin signaling pathway [18,19][18][19], whereas in the leptin pathway, PTP1B binds and dephosphorylates tyrosine kinase downstream of the Janus-Activated Kinase 2 (JAK2) leptin receptor [20]. In cell cultures, PTP1B overexpression causes a decrease in the insulin-stimulated phosphorylation of IR and IRS-1, while PTP1B raises insulin-initiated signaling level reduction [21,22][21][22]. The hypothesis that PTP1B expression can contribute to diabetes and obesity is supported by quantitative analysis of trait loci and mutations in the human PTP1B gene [23]. In in vivo studies, PTP1B knockout mice exhibited elevated resistance to high-fat diet-induced obesity and insulin sensitivity [24,25][24][25]. In addition, other studies on tissue-specific PTP1B knockout mice have shown that leptin action, adiposity, as well as body weight are controlled by neuronal PTP1B [26]. Generally, many studies suggest that PTP1B inhibitors constitute a highly promising approach for T2DM and obesity amelioration.
Aryl carboxylic acids, such as isoxazole [27], hydroxylpropionic [28], 2-oxalylamino benzoic (OBA) acids [29], and thiophene diacid [30], have been recognized as alternative phosphotyrosine (pTyr) surrogates to overcome the lack of cellular activity of highly charged phosphonates (Figure 1). Furthermore, it was reported that benzyl aryl α-ketoacid derivatives revealed significant PTP1B inhibitory effects in a non-competitive pattern, targeting conserved protein loop (WPD loop) open conformation [31]. It was also noticed that the existence of a benzyl group in these bioactive molecules may enhance PTP1B binding affinity, and being hydrophobic in nature, it also increases their cell membrane permeability. Recent studies also suggested that PTP1B may become an oncogene in breast cancer [16]. Accordingly, multiple studies have been conducted focusing on the development of new PTP1B inhibitors for the treatment of T2DM, obesity, and cancer, but to the authors’ knowledge there are no review articles published on this subject. In this sense, the present review aims to provide an overview of the role of PTP1B in T2DM insulin signaling and treatment, and to highlight the most recent findings on several compounds and extracts discovery from marine organisms and their relevance as upcoming PTP1B inhibitors.
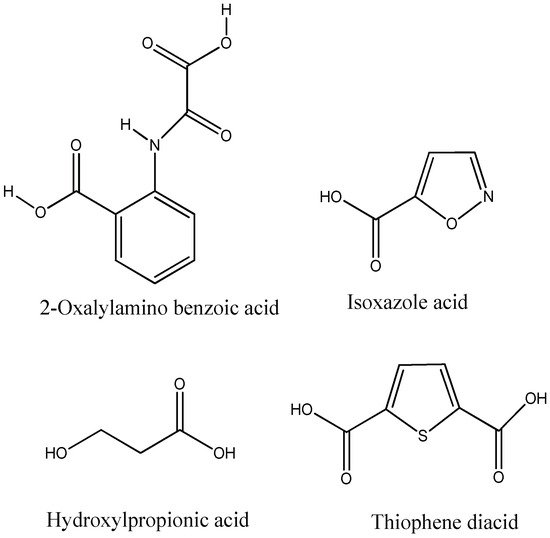
Figure 1.
Structures of phosphotyrosine (pTyr) surrogate acids.
2. Marine Sources as Upcoming Therapeutic Agents
The marine environment is considered a wide and relatively unexploited source of bioactive compounds with high biodiversity, including fatty acids (especially polyunsaturated fatty acids), proteins, polyphenols, sterols, sulfated polysaccharides, and pigments [32,33,34,35,36][32][33][34][35][36]. Indeed, marine algae has been increasingly exploited as renowned sources of metabolites with promising biological effects, including antioxidant, hypoglicemic, hypotensive, hypolipidemic, antibacterial, and antiviral activities [37,38][37][38]. Specifically, macroalgae are considered healthy foods as they are rich in minerals and dietary fibers. Traditionally, the Far East and Hawaiian Islands, Japan, Korea, and China consume marine algae as a common component of their diets. Macroalgae species can reach 9000 species and can be classified according to their pigment composition into three classes, i.e., Phaeophyta, Rhodophyta, and Chlorophyta (also known as brown, red, and green algae, respectively) [39].
Unique metabolites from diverse classes have been isolated from different marine plants, with in vivo remarkable pharmacological effects [40], such as anticancer, anti-hyperlipidemic, anti-diabetic, anti-hypertensive, antioxidant, anti-inflammatory, anticoagulant, anti-estrogenic, antibacterial, antifungal, antiviral, immunomodulatory, neuroprotective, and tissue healing properties [41]. More recently, as a result of the characterization of a large number of bioactive metabolites from marine macroalgae, there has been a growing interest in the search for potential applications of macroalgae and their metabolites as functional constituents for human and animal health benefits [42]. Functional constituents of macroalgae have been increasingly used as food supplements as well as for anti-diabetic purposes [40]. Hereby, the possible applications of marine macroalgae and/or macroalgae-derived bioactive metabolites for PTP1B inhibitory effects have been greatly expanded.
References
- Al-Lawati, J.A. Diabetes Mellitus: A Local and Global Public Health Emergency! Om. Med. J. 2017, 32, 177–179.
- Xiao, J.B.; Högger, P. Dietary polyphenols and type 2 diabetes: Current insights and future perspectives. Curr. Med. Chem. 2015, 22, 23–38.
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.D.; Ohlrogge, A.W.; Malanda, B. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281.
- American Diabetes Association. Classification and diagnosis of diabetes. Diabetes Care 2015, 40, S11–S24.
- Dubé, N.; Tremblay, M.L. Involvement of the small protein tyrosine phosphatases TC-PTP and PTP1B in signal transduction and diseases: From diabetes, obesity to cell cycle, and cancer. Biochim. Biophys. Acta 2005, 1754, 108–117.
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813–820.
- Lee, S.H.; Min, K.H.; Han, J.S.; Lee, D.H.; Park, D.B.; Jung, W.K.; Park, P.J.; Jeon, B.T.; Kim, S.K.; Jeon, Y.J. Effects of brown alga, Ecklonia cava on glucose and lipid metabolism in C57BL/KsJ- db/db mice, a model of type 2 diabetes mellitus. Food Chem. Toxicol. 2012, 50, 575–582.
- Pontiroli, A.E. Type 2 diabetes mellitus is becoming the most common type of diabetes in school children. Acta Diabetol. 2004, 41, 85–90.
- Thilagam, E.; Parimaladevi, B.; Kumarappan, C.; Mandal, S.C. α-Glucosidase and α-amylase inhibitory activity of Senna surattensis. J. Acupunct. Meridian Stud. 2013, 6, 24–30.
- Jung, M.; Park, M.; Lee, H.C.; Kang, Y.H.; Kang, E.S.; Kim, S.K. Antidiabetic agents from medicinal plants. Curr. Med. Chem. 2006, 13, 1203–1218.
- Ray, S.D. Side effects of drugs annual. In A Worldwide Yearly Survey of New Data in Adverse Drug Reactions; Elsevier: Waltham, MA, USA, 2017.
- Scheen, A.J.; Lefebvre, P.J. Troglitazone: Antihyperglycemic activity and potential role in the treatment of type 2 diabetes. Diabetes Care 1999, 22, 1568–1577.
- Ahmad, F.; Li, P.M.; Meyerovitch, J.; Goldstein, B.J. Osmotic loading of neutralizing antibodies demonstrates a role for protein-tyrosine phosphatase 1B in negative regulation of the insulin action pathway. J. Biol. Chem. 1995, 270, 20503–20508.
- Zinker, B.A.; Rondinone, C.M.; Trevillyan, J.M.; Gum, R.J.; Clampit, J.E.; Waring, J.F.; Xie, N.; Wilcox, D.; Jacobson, P.; Frost, L.; et al. PTP1B antisense oligonucleotide lowers PTP1B protein, normalizes blood glucose, and improves insulin sensitivity in diabetic mice. Proc. Nat. Acad. Sci. USA 2002, 99, 11357–11362.
- Johnson, T.O.; Ermolieff, J.; Jirousek, M.R. Protein tyrosine phosphatase 1B inhibitors for diabetes. Nat. Rev. Drug Discov. 2002, 1, 696–709.
- Zhang, S.; Zhang, Z.Y. PTP1B as a drug target: Recent developments in PTP1B inhibitor discovery. Drug Discov. Today 2007, 12, 373–381.
- Zhang, Z.Y.; Lee, S.Y. PTP1B inhibitors as potential therapeutics in the treatment of type 2 diabetes and obesity. Expert Opin. Investig. Drugs 2003, 12, 223–233.
- Bandyopadhyay, D.; Kusari, A.; Kenner, K.A.; Liu, F.; Chernoff, J.; Gustafson, T.A.; Kusari, J. Protein-tyrosine phosphatase 1B complexes with the insulin receptor in vivo and is tyrosine-phosphorylated in the presence of insulin. J. Biol. Chem. 1997, 272, 1639–1645.
- Goldstein, B.J.; Bittner-Kowalczyk, A.; White, M.F.; Harbeck, M. Tyrosine dephosphorylation and deactivation of insulin receptor substrate-1 by protein-tyrosine phosphatase 1B. Possible facilitation by the formation of a ternary complex with the Grb2 adaptor protein. J. Biol. Chem. 2000, 275, 4283–4289.
- Lund, I.K.; Hansen, J.A.; Andersen, H.S.; Moller, N.P.H.; Billestrup, N. Andersen, Mechanism of protein tyrosine phosphatase 1B-mediated inhibition of leptin signalling. J. Mol. Endocrinol. 2005, 34, 339–351.
- Byon, J.C.H.; Kusari, A.B.; Kusari, J. Protein-tyrosine phosphatase-1B acts as a negative regulator of insulin signal transduction. Mol. Cell. Biochem. 1998, 182, 101–108.
- Kenner, K.A.; Anyanwu, E.; Olefsky, J.M.; Kusari, J. Protein-tyrosine phosphatase 1B is a negative regulator of insulin- and insulin-like growth factor-I-stimulated signaling. J. Biol. Chem. 1996, 271, 19810–19816.
- Meshkani, R.; Taghikhani, M.; Al-Kateb, H.; Larijani, B.; Khatami, S.; Sidiropoulos, G.K.; Hegele, R.A.; Adeli, K. Polymorphisms within the protein tyrosine phosphatase 1B (PTPN1) gene promoter: Functional characterization and association with type 2 diabetes and related metabolic traits. Clin. Chem. 2007, 53, 1585–1592.
- Elchebly, M.; Payette, P.; Michaliszy, E.; Cromlish, W.; Collins, S.; Loy, A.L.; Normandin, D.; Cheng, A.; Himms-Hagen, J.; Chan, C.; et al. Increased insulin sensitivity and obesity resistance in mice lacking the protein tyrosine phosphatase-1B gene. Science 1999, 283, 1544–1548.
- Klaman, L.D.; Boss, O.; Peroni, O.D.; Kim, J.K.; Martino, J.L.; Zabolotny, J.M.; Moghal, N.; Lubkin, M.; Kim, Y.B.; Sharpe, A.H.; et al. Increased energy expenditure, decreased adiposity, and tissue-specific insulin sensitivity in proteintyrosine phosphatase 1B-deficient mice. Mol. Cell. Biol. 2000, 20, 5479–5489.
- Bence, K.K.; Delibegovic, M.; Xue, B.; Gorgun, C.Z.; Hotamisligil, G.S.; Neel, B.G.; Kahn, B.B. Neuronal PTP1B regulates body weight, adiposity and leptin action. Nat. Med. 2006, 12, 917–924.
- Zhao, H.; Liu, G.; Xin, Z.; Serby, M.D.; Pei, Z.; Szczepankiewicz, B.G.; Hajduk, P.J.; Abad-Zapatero, C.; Hutchins, C.W.; Lubben, T.H.; et al. Jirousek, Isoxazole carboxylic acids as protein tyrosine phosphatase 1B (PTP1B) inhibitors. Bioorgan. Med. Chem. Lett. 2004, 14, 5543–5546.
- Leung, K.W.; Posner, B.I.; Just, G. Periodinates: A new class of protein tyrosine phosphatase inhibitors. Bioorg. Med. Chem. Lett. 1999, 9, 353–356.
- Andersen, H.S.; Olsen, O.H.; Iversen, L.F.; Sorensen, A.L.P.; Mortensen, S.B.; Christensen, M.; Branner, S.; Hansen, T.K.; Lau, J.F.; Jeppesen, L.; et al. Discovery and SAR of a novel selective and orally bioavailable nonpeptide classical competitive inhibitor class of protein-tyrosine phosphatase 1B. J. Med. Chem. 2002, 45, 4443–4459.
- Wilson, D.P.; Wan, Z.K.; Xu, W.X.; Kirincich, S.J.; Follows, B.C.; Joseph-McCarthy, D.; Foreman, K.; Moretto, A.; Wu, J.; Zhu, M.; et al. Structure-based optimization of protein tyrosine phosphatase 1B inhibitors: From the active site to the second phosphotyrosine binding site. J. Med. Chem. 2007, 50, 4681–4698.
- Liu, S.; Zeng, L.F.; Wu, L.; Yu, X.; Xue, T.; Gunawan, A.M.; Long, Y.Q.; Zhang, Z.Y. Targeting inactive enzyme conformation: Aryl diketoacid derivatives as a new class of PTP1B inhibitors. J. Am. Chem. Soc. 2008, 130, 17075–17084.
- Lee, S.H.; Ko, S.C.; Kang, M.C.; Lee, D.H.; Jeon, Y.J. Octaphlorethol a, a marine algae product, exhibits antidiabetic effects in type 2 diabetic mice by activating amp-activated protein kinase and upregulating the expression of glucose transporter 4. Food Chem. Toxicol. 2016, 91, 58–64.
- Manikkam, V.; Vasiljevic, T.; Donkor, O.N.; Mathai, M.L. A review of potential marine-derived hypotensive and anti-obesity peptides. Crit. Rev. Food Sci. Nutr. 2016, 56, 92–112.
- Ruocco, N.; Costantini, S.; Guariniello, S.; Costantini, M. Polysaccharides from the marine environment with pharmacological, cosmeceutical and nutraceutical potential. Molecules 2016, 21, 551.
- Saleh, A.S.M.; Zhang, Q.; Shen, Q. Recent research in antihypertensive activity of food protein-derived hydrolyzates and peptides. Crit. Rev. Food Sci. Nutr. 2016, 56, 760–787.
- Suleria, H.Á.R.; Gobe, G.; Masci, P.; Osborne, S.A. Marine bioactive compounds and health promoting perspectives; innovation pathways for drug discovery. Trends Food Sci. Technol. 2016, 50, 44–55.
- Choochote, W.; Suklampoo, L.; Ochaikul, D. Evaluation of antioxidant capacities of green microalgae. J. Appl. Phycol. 2014, 26, 43–48.
- Zhao, C.; Wu, Y.J.; Yang, C.F.; Liu, B.; Huang, Y.F. Hypotensive, hypoglycemic and hypolipidemic effects of bioactive compounds from microalgae and marine microorganisms. Int. J. Food Sci. Technol. 2015, 50, 1705–1717.
- Khan, W.; Rayirath, U.P.; Subramanian, S.; Jithesh, M.N.; Rayorath, P.; Hodges, D.M.; Critchley, A.T.; Craigie, J.S.; Norrie, J.; Prithiviraj, B. Seaweed extracts as biostimulants of plant growth and development. J. Plant Growth Regul. 2009, 28, 386–399.
- Pangestuti, R.; Kim, S.K. Biological activities and health benefit effects of natural pigments derived from marine algae. J. Funct. Foods 2011, 3, 255–266.
- Mohamed, S.; Hashim, S.N.; Rahman, H.A. Seaweeds: A sustainable functional food for complementary and alternative therapy. Trends Food Sci. Technol. 2012, 23, 83–96.
- Gupta, S.; Abu-Ghannam, N. Bioactive potential and possible health effects of edible brown seaweeds. Trends Food Sci. Technol. 2011, 22, 315–326.
More
