Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Yvaine Wei and Version 1 by Grazia Maugeri.
Pituitary Adenylate Cyclase-Activating Polypeptide (PACAP) is a neuropeptide with widespread distribution throughout the central and peripheral nervous system as well as in many other peripheral organs. It plays cytoprotective effects mediated mainly through the activation of specific receptors. PACAP is known to play pleiotropic effects on the eye, including the cornea, protecting it against different types of insult.
- PACAP
- eye
- cornea
- injury
- repair
1. Introduction
The cornea, the outermost part of the eye, is a transparent tissue with refractive and barrier functions [1]. Due to the direct connection of the cornea with the external environment, different types of insults, such as chemical, mechanical, and thermal damage, can cause its injury [2]. For this reason, corneal damage represents one of the major causes of blindness worldwide [3]. To date, corneal transplantation represents the most common and successful surgery by restoring good eyesight. However, the high cost, the high graft failure rate, the legal issues, and the lack of donors urge new options for treating, at least, some corneal lesions [4,5][4][5].
The neuropeptide pituitary adenylate cyclase-activating polypeptide (PACAP) was isolated for the first time in 1989 from sheep hypothalamic extracts [6]. It exists in two active forms, PACAP27 and PACAP38, including 27 and 38 amino acid residues, respectively, and with mostly comparable functions. PACAP belongs to the vasoactive intestinal polypeptide (VIP)/secretin/glucagon family peptides and shows a high degree of homology (~70%) to VIP. PACAP and VIP share three different receptors: the PAC1 receptor (PAC1R), which has a high affinity to PACAP as compared to VIP, and the VPAC1 and VPAC2 receptors, showing a comparable affinity for both peptides [7,8,9,10][7][8][9][10]. Alternative splicing occurring in the PAC1R gene generates different variants (Null, Hip, Hop1, Hop2, Hiphop1, Hiphop2, short and very short isoforms) that can activate the adenylate cyclase (AC) pathway forming cAMP as well as phospholipase C (PLC) pathway promoting the formation of protein kinase C (PKC) [11]. VPAC receptors are coupled to Gs proteins resulting in the activation of AC as well as other signaling cascades [12,13][12][13]. Some of the protective effects of PACAP are also mediated by the stimulation of an intracellular factor known as activity-dependent neuroprotective protein (ADNP) [14,15,16][14][15][16]. In accord, peptide activity scanning identified NAP (NAPVSIPQ), the smallest active element of ADNP, acts in synergy with PACAP by showing neuroprotective effect [17,18][17][18]. PACAP is widely distributed in the nervous system and is consequently implicated in different neurodegenerative diseases [19,20,21,22,23,24,25,26][19][20][21][22][23][24][25][26]. In addition, it plays a controversial function in various types of tumors by promoting or inhibiting its progression [27,28,29,30,31,32,33,34,35,36,37][27][28][29][30][31][32][33][34][35][36][37].
2. Overview on the Cornea Anatomy
The human cornea, together with the surrounding sclera, constitutes the protective outer barrier of the eye. In particular, it represents the outer covering of the anterior portion of the eyeball by exerting two essential functions: it protects from external physical trauma and provides about 70% refractive power of the eye. To perform these functions, the corneal tissue is both mechanically strong and transparent. The anterior surface of the cornea is convex and aspheric [41][38]. The cornea comprises five main layers: the epithelium, the Bowman’s membrane, the stroma, the Descemet’s membrane, and the endothelium [42,43][39][40] (Figure 1).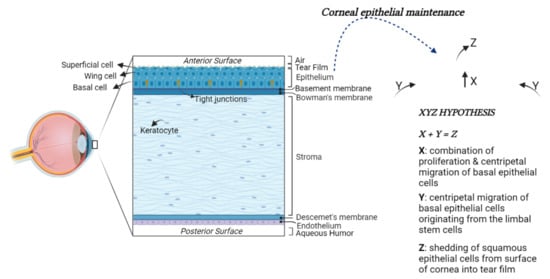
Figure 1. Structural anatomy of the human cornea. From left to right: (1) A diagram of human cornea structure; (2) The XYZ hypothesis. The asymmetric division of LESCs generates a stem-like daughter cell, remaining within the limbus, and a TAC, migrating in a centripetal direction (Y). TACs undergo multiple replications. In this process, they lose stemness, migrate anteriorly and differentiate to post-mitotic suprabasal wing cells (X), and progress in superficial squamous cells, which are lost during normal corneal surface exfoliation (Z) of the epithelial corneal maintenance.
The corneal epithelium is continuously subjected to a wide range of insults; therefore, its long-term maintenance is regulated by limbal epithelial stem cells (LESCs). The LESCs reside in an annular transition zone known as the limbus, laying at the junction area between the cornea and the sclera. They show typical characteristics of immature and undifferentiated cells [45,46][41][42]. In particular, they do not express the cytokeratin 3 and 12, commonly detected in mature, differentiated corneal epithelial cells, whereas they express cytokeratin 14 or TP63, which are stem markers of the immature or progenitor cells in various stratified epithelia. The LESCs give rise to transit-amplifying cells (TACs), which migrate and divide into basal corneal epithelial cells in normal homeostasis conditions or to replace those cells desquamated or lost by lesions (Figure 1) [47,48][43][44].
3. Role of PACAP and Its Receptors in the Eye
The presence of PACAP and its receptors has been largely shown in the eye [33,56][33][45]. PACAP positive expression was found in mammalian, teleost, turtle, and chicken retina [57][46]. In particular, in situ hybridization and immunohistochemical analysis have revealed the presence of PACAP in specific cell populations of retinal tissue samples. PACAP was positively expressed in the nerve fiber layer (NFL), the ganglion cell layer (GCL), the inner plexiform layer (IPL), and the pigment epithelium (PE). The immunoreactivity of PACAP appeared in the early phase of retinal development [58][47], as demonstrated by its presence in the chick inner nuclear layer (INL) from embryonic day 8 [59][48]. PACAP mRNA expression was detectable in the rat GCL at embryonic day 20 [60][49], whereas, in the zebrafish, PACAP immune-positive signal was found in the retina at 24 h post-fertilization [61][50]. PAC1R was strongly expressed in the GCL, in neuronal cell bodies of amacrine and horizontal cells localized in the INL and in the PE. On the contrary, PAC1R was weakly expressed in the IPL, outer plexiform layer (OPL), outer nuclear layer (ONL), and photoreceptor layer [62,63,64,65][51][52][53][54]. The expression profile of PAC1R splice variants (Null, Hip, Hop1, Hop2, Hiphop1, and Hiphop2) was described during retina development [11]. The expression of PAC1Rs at the subcellular level was identified at the plasma membrane, in the rough endoplasmic reticulum, in the cytoplasmic matrix of retinal ganglion cells (RGCs) and amacrine cells in the INL [66][55]. PAC1R immunoreactivity was also detected in retinal tissue and in rat primary cultures of Müller cells [67,68][56][57]. In the rat retina, the expression of VPAC1R and VPAC2R was demonstrated [69][58]. Moreover, Lakk et al. [11] showed the potential involvement of VPACRs at all stages of retinal development in the rat.
The protective effects of PACAP in the visual system have been widely studied in the neural and non-neuronal parts of the eye, including the cornea (Figure 2).
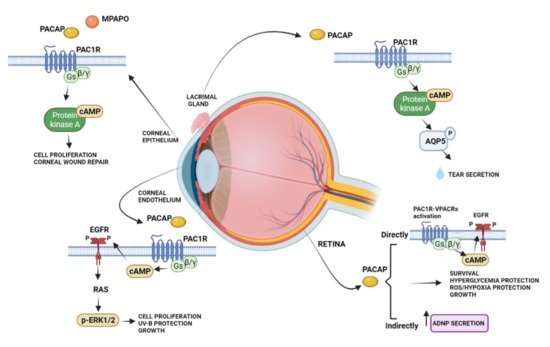 Figure 2. Schematic diagram showing the main pro-survival/antiapoptotic/protective intracellular pathways activated by PACAP in different ocular structures.
Several studies showed that PACAP has protective effects in the retina against toxic or ischemic insults, inflammation, hypoxia, oxygen-induced retinopathy, traumatic injuries, glaucoma, and diabetic retinopathy [70,71,72,73,74,75,76,77,78,79,80,81][59][60][61][62][63][64][65][66][67][68][69][70]. The protective role of PACAP was observed in different cell types, including bipolar neurons, amacrine, and pigment epithelial cells [82,83][71][72]. In particular, in the retinal pigment epithelial cells, PACAP counteracted oxidative stress and hypoxic insult by exerting protective and pro-survival effects [83,84,85,86][72][73][74][75].
Figure 2. Schematic diagram showing the main pro-survival/antiapoptotic/protective intracellular pathways activated by PACAP in different ocular structures.
Several studies showed that PACAP has protective effects in the retina against toxic or ischemic insults, inflammation, hypoxia, oxygen-induced retinopathy, traumatic injuries, glaucoma, and diabetic retinopathy [70,71,72,73,74,75,76,77,78,79,80,81][59][60][61][62][63][64][65][66][67][68][69][70]. The protective role of PACAP was observed in different cell types, including bipolar neurons, amacrine, and pigment epithelial cells [82,83][71][72]. In particular, in the retinal pigment epithelial cells, PACAP counteracted oxidative stress and hypoxic insult by exerting protective and pro-survival effects [83,84,85,86][72][73][74][75].

4. The Role of PACAP in the Cornea
The expression of PACAP and its receptors has been shown in the cornea. Here, PACAP positive cells were found in rabbit and human corneal epithelium, particularly in the basal cells. Moreover, the expression of PACAP was detected in the corneal endothelial layer and weakly in the stroma [98,99][76][77]. High expression levels of PAC1R were identified in the stromal and basal cells of the epithelium. Furthermore, the VPACRs were strongly expressed in all layers of the epithelium and in stromal cells of the rabbit cornea [99][77]. Previously, Wang [95][78] et al. detected PACAP immunoreactivity in nerve terminals running in the stroma and sending off some branches into the epithelium. Corneal injury is frequently associated with damage of the epithelium and its innervating fibers. In an in vivo experimental model of laser-assisted in situ keratomileuses (LASIK) surgery, PACAP showed to accelerate recovery of corneal sensitivity after the creation of a corneal flap. In more detail, the administration of 10 μM PACAP27 increased up to 75% the corneal sensitivity eight weeks after the operation [100][79]. In accord, it has been demonstrated that PACAP induced the growth of neuronal processes in cultured trigeminal ganglion cells. These neurons secrete various biologically active molecules enhancing the proliferation and differentiation of corneal epithelial cells as well as collagen VII production, important to maintaining and repairing the corneal epithelium [101][80]. The protective effect of PACAP was confirmed by Wu et al., 2015 [102][81], by showing that the peptide alone or in combination with the receptor protein of laminin, known as N-terminal agrin domain (NtA), significantly accelerated the process of repairing the mechanically injured corneal epithelial cells. It is well known that tear fluid contains different antibacterial proteins, growth factors, and secretory mucin important for corneal maintenance and its repairing [101,103][80][82]. For this reason, tear fluid reduction, occurring in dry eye syndrome, is an inducing factor in corneal keratinization. PACAP played an important role in protecting the corneal surface by stimulating tear secretion [104][83]. As described above, in the lacrimal gland, PACAP is an endogenous modulator of AQP5, involved in tear production [90][84]. In accord, PACAP null mice showed a reduction in the AQP5 expression, whereas the eye treatment with PACAP drops stimulated its transcription. Furthermore, PACAP null mice exhibited the dry eye syndrome phenotype with a corneal disorder associated with the reduction in tear volume [90][84].
5. Conclusions
The direct contact of the cornea with the external environment makes it frequently exposed to various types of injuries. The surgical replacement of lesioned cornea with healthy donor tissue is the frequently used therapeutic approach. To date, the actual challenge is linked to recruiting a sufficient number of donors, requiring alternatives to decrease this persistent demand. PACAP has shown important corneal protective and regenerative effects. Therefore, the development of innovative nanoformulation platforms for topical PACAP or PAC1R agonists delivery, as well as the synthesis of molecules able to increase PACAP endogenous expression, might represent a valid strategy for the treatment of some corneal diseases.References
- Avadhanam, V.S.; Liu, C.S. A brief review of Boston type-1 and osteo-odonto keratoprostheses. Br. J. Ophthalmol. 2015, 99, 878–887.
- Han, Y.; Li, X.; Zhang, Y.; Han, Y.; Chang, F.; Ding, J. Mesenchymal Stem Cells for Regenerative Medicine. Cells 2019, 8, 886.
- Flaxman, S.R.; Bourne, R.R.A.; Resnikoff, S.; Ackland, P.; Braithwaite, T.; Cicinelli, M.V.; Das, A.; Jonas, J.B.; Keeffe, J.; Kempen, J.H.; et al. Global causes of blindness and distance vision impairment 1990–2020: A systematic review and meta-analysis. Lancet Glob. Health 2017, 5, e1221–e1234.
- Gain, P.; Jullienne, R.; He, Z.; Aldossary, M.; Acquart, S.; Cognasse, F.; Thuret, G. Global Survey of Corneal Transplantation and Eye Banking. JAMA Ophthalmol. 2016, 134, 167–173.
- Golchet, G.; Carr, J.; Harris, M.G. Why don’t we have enough cornea donors? A literature review and survey. Optometry 2000, 71, 318–328.
- Miyata, A.; Arimura, A.; Dahl, R.R.; Minamino, N.; Uehara, A.; Jiang, L.; Culler, M.D.; Coy, D.H. Isolation of a novel 38 residue-hypothalamic polypeptide which stimulates adenylate cyclase in pituitary cells. Biochem. Biophys. Res. Commun. 1989, 164, 567–574.
- Arimura, A. Pituitary adenylate cyclase activating polypeptide (PACAP): Discovery and current status of research. Regul. Pept. 1992, 37, 287–303.
- Arimura, A.; Shioda, S. Pituitary adenylate cyclase activating polypeptide (PACAP) and its receptors: Neuroendocrine and endocrine interaction. Front. Neuroendocrinol. 1995, 16, 53–88.
- Sherwood, N.M.; Krueckl, S.L.; McRory, J.E. The origin and function of the pituitary adenylate cyclase-activating polypeptide (PACAP)/glucagon superfamily. Endocr. Rev. 2000, 21, 619–670.
- Harmar, A.J.; Fahrenkrug, J.; Gozes, I.; Laburthe, M.; May, V.; Pisegna, J.R.; Vaudry, D.; Vaudry, H.; Waschek, J.A.; Said, S.I. Pharmacology and functions of receptors for vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide: IUPHAR review 1. Br. J. Pharmacol. 2012, 166, 4–17.
- Dickson, L.; Finlayson, K. VPAC and PAC receptors: From ligands to function. Pharmacol. Ther. 2009, 121, 294–316.
- Somogyvári-Vigh, A.; Reglodi, D. Pituitary adenylate cyclase activating polypeptide: A potential neuroprotective peptide. Curr. Pharm. Des. 2004, 10, 2861–2889.
- Shioda, S.; Ohtaki, H.; Nakamachi, T.; Dohi, K.; Watanabe, J.; Nakajo, S.; Arata, S.; Kitamura, S.; Okuda, H.; Takenoya, F.; et al. Pleiotropic functions of PACAP in the CNS: Neuroprotection and neurodevelopment. Ann. N. Y. Acad. Sci. 2006, 1070, 550–560.
- Bassan, M.; Zamostiano, R.; Davidson, A.; Pinhasov, A.; Giladi, E.; Perl, O.; Bassan, H.; Blat, C.; Gibney, G.; Glazner, G.; et al. Complete sequence of a novel protein containing a femtomolar-activity-dependent neuroprotective peptide. J. Neurochem. 1999, 72, 1283–1293.
- Zamostiano, R.; Pinhasov, A.; Gelber, E.; Steingart, R.A.; Seroussi, E.; Giladi, E.; Bassan, M.; Wollman, Y.; Eyre, H.J.; Mulley, J.C.; et al. Cloning and characterization of the human activity-dependent neuroprotective protein. J. Biol. Chem. 2001, 276, 708–714.
- D’Amico, A.G.; Maugeri, G.; Musumeci, G.; Reglodi, D.; D’Agata, V. PACAP and NAP: Effect of Two Functionally Related Peptides in Diabetic Retinopathy. J. Mol. Neurosci. 2021, 71, 1525–1535.
- D’Amico, A.G.; Maugeri, G.; Rasà, D.; Federico, C.; Saccone, S.; Lazzara, F.; Fidilio, A.; Drago, F.; Bucolo, C.; D’Agata, V. NAP modulates hyperglycemic-inflammatory event of diabetic retina by counteracting outer blood retinal barrier damage. J. Cell. Physiol. 2019, 234, 5230–5240.
- D’Amico, A.G.; Scuderi, S.; Maugeri, G.; Cavallaro, S.; Drago, F.; D’Agata, V. NAP reduces murine microvascular endothelial cells proliferation induced by hyperglycemia. J. Mol. Neurosci. 2014, 54, 405–413.
- Solés-Tarrés, I.; Cabezas-Llobet, N.; Vaudry, D.; Xifró, X. Protective Effects of Pituitary Adenylate Cyclase-Activating Polypeptide and Vasoactive Intestinal Peptide against Cognitive Decline in Neurodegenerative Diseases. Front. Cell. Neurosci. 2020, 14, 221.
- Dejda, A.; Jolivel, V.; Bourgault, S.; Seaborn, T.; Fournier, A.; Vaudry, H.; Vaudry, D. Inhibitory effect of PACAP on caspase activity in neuronal apoptosis: A better understanding towards therapeutic applications in neurodegenerative diseases. J. Mol. Neurosci. 2008, 36, 26–37.
- Reglodi, D.; Kiss, P.; Lubics, A.; Tamas, A. Review on the protective effects of PACAP in models of neurodegenerative diseases in vitro and in vivo. Curr. Pharm. Des. 2011, 17, 962–972.
- Waschek, J.A. VIP and PACAP: Neuropeptide modulators of CNS inflammation, injury, and repair. Br. J. Pharmacol. 2013, 169, 512–523.
- D’Amico, A.G.; Maugeri, G.; Saccone, S.; Federico, C.; Cavallaro, S.; Reglodi, D.; D’Agata, V. PACAP Modulates the Autophagy Process in an In Vitro Model of Amyotrophic Lateral Sclerosis. Int. J. Mol. Sci. 2020, 21, 2943.
- Maugeri, G.; D’Amico, A.G.; Castrogiovanni, P.; Saccone, S.; Federico, C.; Reibaldi, M.; Russo, A.; Bonfiglio, V.; Avitabile, T.; Longo, A.; et al. PACAP through EGFR transactivation preserves human corneal endothelial integrity. J. Cell. Biochem. 2019, 120, 10097–10105.
- Maugeri, G.; D’Amico, A.G.; Morello, G.; Reglodi, D.; Cavallaro, S.; D’Agata, V. Differential Vulnerability of Oculomotor Versus Hypoglossal Nucleus During ALS: Involvement of PACAP. Front. Neurosci. 2020, 14, 805.
- Maugeri, G.; D’Amico, A.G.; Musumeci, G.; Reglodi, D.; D’Agata, V. Effects of Pacap on Schwann Cells: Focus on Nerve Injury. Int. J. Mol. Sci. 2020, 21, 8233.
- Moody, T.W.; Nuche-Berenguer, B.; Jensen, R.T. Vasoactive intestinal peptide/pituitary adenylate cyclase activating polypeptide, and their receptors and cancer. Curr. Opin. Endocrinol. Diabetes Obes. 2016, 23, 38–47.
- D’Amico, A.G.; Maugeri, G.; Bucolo, C.; Saccone, S.; Federico, C.; Cavallaro, S.; D’Agata, V. Nap Interferes with Hypoxia-Inducible Factors and VEGF Expression in Retina of Diabetic Rats. J. Mol. Neurosci. 2017, 61, 256–266.
- Maugeri, G.; D’Amico, A.G.; Rasà, D.M.; Saccone, S.; Federico, C.; Cavallaro, S.; D’Agata, V. PACAP and VIP regulate hypoxia-inducible factors in neuroblastoma cells exposed to hypoxia. Neuropeptides 2018, 69, 84–91.
- Maugeri, G.; D’Amico, A.G.; Reitano, R.; Magro, G.; Cavallaro, S.; Salomone, S.; D’Agata, V. PACAP and VIP Inhibit the Invasiveness of Glioblastoma Cells Exposed to Hypoxia through the Regulation of HIFs and EGFR Expression. Front. Pharmacol. 2016, 7, 139.
- Ferencz, S.; Reglodi, D.; Kaszas, B.; Bardosi, A.; Toth, D.; Vekony, Z.; Vicena, V.; Karadi, O.; Kelemen, D. PACAP and PAC1 receptor expression in pancreatic ductal carcinoma. Oncol. Lett. 2019, 18, 5725–5730.
- Bensalma, S.; Turpault, S.; Balandre, A.C.; De Boisvilliers, M.; Gaillard, A.; Chadéneau, C.; Muller, J.M. PKA at a Cross-Road of Signaling Pathways Involved in the Regulation of Glioblastoma Migration and Invasion by the Neuropeptides VIP and PACAP. Cancers 2019, 11, 123.
- Cavallaro, S.; D’Agata, V.; Guardabasso, V.; Travali, S.; Stivala, F.; Canonico, P.L. Differentiation induces pituitary adenylate cyclase-activating polypeptide receptor expression in PC-12 cells. Mol. Pharmacol. 1995, 48, 56–62.
- Castorina, A.; Giunta, S.; Scuderi, S.; D’Agata, V. Involvement of PACAP/ADNP signaling in the resistance to cell death in malignant peripheral nerve sheath tumor (MPNST) cells. J. Mol. Neurosci. 2012, 48, 674–683.
- Castorina, A.; Scuderi, S.; D’Amico, A.G.; Drago, F.; D’Agata, V. PACAP and VIP increase the expression of myelin-related proteins in rat schwannoma cells: Involvement of PAC1/VPAC2 receptor-mediated activation of PI3K/Akt signaling pathways. Exp. Cell Res. 2014, 322, 108–121.
- Castorina, A.; Tiralongo, A.; Giunta, S.; Carnazza, M.L.; Rasi, G.; D’Agata, V. PACAP and VIP prevent apoptosis in schwannoma cells. Brain Res. 2008, 1241, 29–35.
- D’Amico, A.G.; Scuderi, S.; Saccone, S.; Castorina, A.; Drago, F.; D’Agata, V. Antiproliferative effects of PACAP and VIP in serum-starved glioma cells. J. Mol. Neurosci. 2013, 51, 503–513.
- Feizi, S.; Jafarinasab, M.R.; Karimian, F.; Hasanpour, H.; Masudi, A. Central and peripheral corneal thickness measurement in normal and keratoconic eyes using three corneal pachymeters. J. Ophthalmic Vis. Res. 2014, 9, 296–304.
- Patel, S.; McLaren, J.; Hodge, D.; Bourne, W. Normal human keratocyte density and corneal thickness measurement by using confocal microscopy in vivo. Investig. Ophthalmol. Vis. Sci. 2001, 42, 333–339.
- Reinstein, D.Z.; Archer, T.J.; Gobbe, M.; Silverman, R.H.; Coleman, D.J. Stromal thickness in the normal cornea: Three-dimensional display with artemis very high-frequency digital ultrasound. J. Refract. Surg. 2009, 25, 776–786.
- Dziasko, M.A.; Daniels, J.T. Anatomical Features and Cell-Cell Interactions in the Human Limbal Epithelial Stem Cell Niche. Ocul. Surf. 2016, 14, 322–330.
- Notara, M.; Alatza, A.; Gilfillan, J.; Harris, A.R.; Levis, H.J.; Schrader, S.; Vernon, A.; Daniels, J.T. In sickness and in health: Corneal epithelial stem cell biology, pathology and therapy. Exp. Eye Res. 2010, 90, 188–195.
- Dorà, N.J.; Hill, R.E.; Collinson, J.M.; West, J.D. Lineage tracing in the adult mouse corneal epithelium supports the limbal epithelial stem cell hypothesis with intermittent periods of stem cell quiescence. Stem Cell Res. 2015, 15, 665–677.
- Thoft, R.A.; Friend, J. The X, Y, Z hypothesis of corneal epithelial maintenance. Investig. Ophthalmol. Vis. Sci. 1983, 24, 1442–1443.
- Atlasz, T.; Szabadfi, K.; Kiss, P.; Racz, B.; Gallyas, F.; Tamas, A.; Gaal, V.; Marton, Z.; Gabriel, R.; Reglodi, D. Pituitary adenylate cyclase activating polypeptide in the retina: Focus on the retinoprotective effects. Ann. N. Y. Acad. Sci. 2010, 1200, 128–139.
- Reglödi, D.; Somogyvári-Vigh, A.; Vígh, J.; Li, M.; Lengvári, I.; Arimura, A. Pituitary adenylate cyclase activating polypeptide is highly abundant in the nervous system of anoxia-tolerant turtle, Pseudemys scripta elegans. Peptides 2001, 22, 873–878.
- Fahrenkrug, J.; Nielsen, H.S.; Hannibal, J. Expression of melanopsin during development of the rat retina. Neuroreport 2004, 15, 781–784.
- Borba, J.C.; Henze, I.P.; Silveira, M.S.; Kubrusly, R.C.; Gardino, P.F.; de Mello, M.C.; Hokoç, J.N.; de Mello, F.G. Pituitary adenylate cyclase-activating polypeptide (PACAP) can act as determinant of the tyrosine hydroxylase phenotype of dopaminergic cells during retina development. Dev. Brain Res. 2005, 156, 193–201.
- Skoglösa, Y.; Takei, N.; Lindholm, D. Distribution of pituitary adenylate cyclase activating polypeptide mRNA in the developing rat brain. Brain Research. Mol. Brain Res. 1999, 65, 1–13.
- Mathieu, M.; Ciarlo, M.; Trucco, N.; Griffero, F.; Damonte, G.; Salis, A.; Vallarino, M. Pituitary adenylate cyclase-activating polypeptide in the brain, spinal cord and sensory organs of the zebrafish, Danio rerio, during development. Dev. Brain Res. 2004, 151, 169–185.
- Seki, T.; Shioda, S.; Ogino, D.; Nakai, Y.; Arimura, A.; Koide, R. Distribution and ultrastructural localization of a receptor for pituitary adenylate cyclase activating polypeptide and its mRNA in the rat retina. Neurosci. Lett. 1997, 238, 127–130.
- Seki, T.; Shioda, S.; Nakai, Y.; Arimura, A.; Koide, R. Distribution and ultrastructural localization of pituitary adenylate cyclase-activating polypeptide (PACAP) and its receptor in the rat retina. Ann. N. Y. Acad. Sci. 1998, 865, 408–411.
- Seki, T.; Izumi, S.; Shioda, S.; Zhou, C.J.; Arimura, A.; Koide, R. Gene expression for PACAP receptor mRNA in the rat retina by in situ hybridization and in situ RT-PCR. Ann. N. Y. Acad. Sci. 2000, 921, 366–369.
- Seki, T.; Shioda, S.; Izumi, S.; Arimura, A.; Koide, R. Electron microscopic observation of pituitary adenylate cyclase-activating polypeptide (PACAP)-containing neurons in the rat retina. Peptides 2000, 21, 109–113.
- Nakamachi, T.; Matkovits, A.; Seki, T.; Shioda, S. Distribution and protective function of pituitary adenylate cyclase-activating polypeptide in the retina. Front. Endocrinol. 2012, 3, 145.
- Dénes, V.; Czotter, N.; Lakk, M.; Berta, G.; Gábriel, R. PAC1-expressing structures of neural retina alter their PAC1 isoform splicing during postnatal development. Cell Tissue Res. 2014, 355, 279–288.
- Seki, T.; Hinohara, Y.; Taki, C.; Nakatani, M.; Ozawa, M.; Nishimura, S.; Takaki, A.; Itho, H.; Takenoya, F.; Shioda, S. PACAP stimulates the release of interleukin-6 in cultured rat Müller cells. Ann. N. Y. Acad. Sci. 2006, 1070, 535–539.
- D’Agata, V.; Cavallaro, S. Functional and molecular expression of PACAP/VIP receptors in the rat retina. Mol. Brain Res. 1998, 54, 161–164.
- Atlasz, T.; Werling, D.; Song, S.; Szabo, E.; Vaczy, A.; Kovari, P.; Tamas, A.; Reglodi, D.; Yu, R. Retinoprotective Effects of TAT-Bound Vasoactive Intestinal Peptide and Pituitary Adenylate Cyclase Activating Polypeptide. J. Mol. Neurosci. 2019, 68, 397–407.
- Ye, D.; Shi, Y.; Xu, Y.; Huang, J. PACAP Attenuates Optic Nerve Crush-Induced Retinal Ganglion Cell Apoptosis Via Activation of the CREB-Bcl-2 Pathway. J. Mol. Neurosci. 2019, 68, 475–484.
- Ye, D.; Yang, Y.; Lu, X.; Xu, Y.; Shi, Y.; Chen, H.; Huang, J. Spatiotemporal Expression Changes of PACAP and Its Receptors in Retinal Ganglion Cells After Optic Nerve Crush. J. Mol. Neurosci. 2019, 68, 465–474.
- Seki, T.; Itoh, H.; Nakamachi, T.; Shioda, S. Suppression of ganglion cell death by PACAP following optic nerve transection in the rat. J. Mol. Neurosci. 2008, 36, 57–60.
- Endo, K.; Nakamachi, T.; Seki, T.; Kagami, N.; Wada, Y.; Nakamura, K.; Kishimoto, K.; Hori, M.; Tsuchikawa, D.; Shinntani, N.; et al. Neuroprotective effect of PACAP against NMDA-induced retinal damage in the mouse. J. Mol. Neurosci. 2011, 43, 22–29.
- Szabadfi, K.; Reglodi, D.; Szabo, A.; Szalontai, B.; Valasek, A.; Setalo, G., Jr.; Kiss, P.; Tamas, A.; Wilhelm, M.; Gabriel, R. Pituitary Adenylate Cyclase Activating Polypeptide, A Potential Therapeutic Agent for Diabetic Retinopathy in Rats: Focus on the Vertical Information Processing Pathway. Neurotox. Res. 2016, 29, 432–446.
- Gábriel, R.; Pöstyéni, E.; Dénes, V. Neuroprotective Potential of Pituitary Adenylate Cyclase Activating Polypeptide in Retinal Degenerations of Metabolic Origin. Front. Neurosci. 2019, 13, 1031.
- D’Amico, A.G.; Maugeri, G.; Reitano, R.; Bucolo, C.; Saccone, S.; Drago, F.; D’Agata, V. PACAP Modulates Expression of Hypoxia-Inducible Factors in Streptozotocin-Induced Diabetic Rat Retina. J. Mol. Neurosci. 2015, 57, 501–509.
- D’Amico, A.G.; Maugeri, G.; Rasà, D.M.; Bucolo, C.; Saccone, S.; Federico, C.; Cavallaro, S.; D’Agata, V. Modulation of IL-1β and VEGF expression in rat diabetic retinopathy after PACAP administration. Peptides 2017, 97, 64–69.
- Giunta, S.; Castorina, A.; Bucolo, C.; Magro, G.; Drago, F.; D’Agata, V. Early changes in pituitary adenylate cyclase-activating peptide, vasoactive intestinal peptide and related receptors expression in retina of streptozotocin-induced diabetic rats. Peptides 2012, 37, 32–39.
- Kvarik, T.; Mammel, B.; Reglodi, D.; Kovacs, K.; Werling, D.; Bede, B.; Vaczy, A.; Fabian, E.; Toth, G.; Kiss, P.; et al. PACAP is protective in a rat model of retinopathy of prematurity. J. Mol. Neurosci. 2016, 60, 179–185.
- Szabo, E.; Patko, E.; Vaczy, A.; Molitor, D.; Csutak, A.; Toth, G.; Reglodi, D.; Atlasz, T. Retinoprotective effects of PACAP eye drops in microbead-induced glaucoma model in rats. Int. J. Mol. Sci. 2021, 22, 8825.
- Szabadfi, K.; Atlasz, T.; Kiss, P.; Reglodi, D.; Szabo, A.; Kovacs, K.; Szalontai, B.; Setalo, G., Jr.; Banki, E.; Csanaky, K.; et al. Protective effects of the neuropeptide PACAP in diabetic retinopathy. Cell Tissue Res. 2012, 348, 37–46.
- Fabian, E.; Reglodi, D.; Horvath, G.; Opper, B.; Toth, G.; Fazakas, C.; Vegh, A.G.; Wilhelm, I.; Krizbai, I.A. Pituitary adenylate cyclase activating polypeptide acts against neovascularization in retinal pigment epithelial cells. Ann. N. Y. Acad. Sci. 2019, 1455, 160–172.
- Maugeri, G.; D’Amico, A.G.; Saccone, S.; Federico, C.; Cavallaro, S.; D’Agata, V. PACAP and VIP Inhibit HIF-1α-Mediated VEGF Expression in a Model of Diabetic Macular Edema. J. Cell. Physiol. 2017, 232, 1209–1215.
- Maugeri, G.; D’Amico, A.G.; Gagliano, C.; Saccone, S.; Federico, C.; Cavallaro, S.; D’Agata, V. VIP Family Members Prevent Outer Blood Retinal Barrier Damage in a Model of Diabetic Macular Edema. J. Cell. Physiol. 2017, 232, 1079–1085.
- Mester, L.; Kovacs, K.; Racz, B.; Solti, I.; Atlasz, T.; Szabadfi, K.; Tamas, A.; Reglodi, D. Pituitary adenylate cyclase-activating polypeptide is protective against oxidative stress in human retinal pigment epithelial cells. J. Mol. Neurosci. 2011, 43, 35–43.
- Shioda, S.; Takenoya, F.; Wada, N.; Hirabayashi, T.; Seki, T.; Nakamachi, T. Pleiotropic and retinoprotective functions of PACAP. Anat. Sci. Int. 2016, 91, 313–324.
- Maugeri, G.; Longo, A.; D’Amico, A.G.; Rasà, D.M.; Reibaldi, M.; Russo, A.; Bonfiglio, V.; Avitabile, T.; D’Agata, V. Trophic effect of PACAP on human corneal endothelium. Peptides 2018, 99, 20–26.
- Wang, Z.Y.; Alm, P.; Håkanson, R. Distribution and effects of pituitary adenylate cyclase-activating peptide in the rabbit eye. Neuroscience 1995, 69, 297–308.
- Fukiage, C.; Nakajima, T.; Takayama, Y.; Minagawa, Y.; Shearer, T.R.; Azuma, M. PACAP induces neurite outgrowth in cultured trigeminal ganglion cells and recovery of corneal sensitivity after flap surgery in rabbits. Am. J. Ophthalmol. 2007, 143, 255–262.
- Dartt, D.A. Signal transduction and control of lacrimal gland protein secretion: A review. Curr. Eye Res. 1989, 8, 619–636.
- Wu, L.; Wang, J.; Chen, X.; Hong, A. Expression, identification and biological effects of the novel recombination protein, PACAP38-NtA, with high bioactivity. Int. J. Mol. Med. 2015, 35, 376–382.
- Il’inskiĭ, O.B.; Spevak, S.E.; Kochetkov, N.V.; Solov’eva, A.I.; Krasnikova, T.L. Participation of the lacrimal glands in wound healing processes. Bull. Exp. Biol. Med. 1985, 100, 91–93.
- Shioda, S.; Takenoya, F.; Hirabayashi, T.; Wada, N.; Seki, T.; Nonaka, N.; Nakamachi, T. Effects of PACAP on Dry Eye Symptoms, and Possible Use for Therapeutic Application. J. Mol. Neurosci. 2019, 68, 420–426.
- Nakamachi, T.; Ohtaki, H.; Seki, T.; Yofu, S.; Kagami, N.; Hashimoto, H.; Shintani, N.; Baba, A.; Mark, L.; Lanekoff, I.; et al. PACAP suppresses dry eye signs by stimulating tear secretion. Nat. Commun. 2016, 7, 12034.
More
