Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Rita Xu and Version 1 by Julita Tokarek.
The aim of this study was to investigate the impact of gut microbiota on the prevalence of obesity and associated morbidities, taking into consideration underlying molecular mechanisms. In addition to exploring the relationship between obesity and fecal microorganisms with their metabolites, the study also focused on the factors that would be able to stimulate growth and remodeling of microbiota.
- obesity
- microbiota
- dysbiosis
- morbidities
- gastrointestinal microbiome
1. Introduction
1.1. Obesity Epidemic—Statistics, General Background, Causes and Effects
Obesity is now recognized as a global epidemic affecting both developed and poor-resource countries [1], which lowers life expectancy [2] and has extensive consequences for countries’ health care systems [1]. Body mass index (BMI), which is weight in kilograms divided by height in meters squared, is used to identify obesity. For adults, a BMI of 25.0 to 29.9 kg/m2 is defined as overweight and a BMI of 30 kg/m2 or higher is defined as obese [3]. In the last three decades, the worldwide prevalence of obesity has increased 27.5% for adults and 47.1% for children [3]. By 2030, 81% of men and 74.9% of women in the USA are projected to be obese or overweight, whereas it is expected that by 2030, approximately 39% of children and 46% of adolescents will have an abnormally high BMI [4]. Childhood obesity (defined as body mass index-for-age (or BMI-for-age) percentile greater than 95 percent) is an important risk factor for adult obesity: a meta-analysis of available data showed that half of obese children were still obese as adults; additionally, risk is more than twofold if both parents are obese [5].
The etiology of obesity is multifactorial and includes genetic, hormonal, socioeconomic, environmental, and cultural influences [3]. Comorbidities and their treatment might be a factor in the prevalence and progression of obesity as well. Primary and secondary disease-related causes of obesity are presented in Table 1.
Table 1. Primary and secondary disease-related causes of obesity.
| Primary Causes of Obesity | Secondary Causes of Obesity | |
|---|---|---|
| Genetic causes | Neurologic | |
| Monogenic disorders | Brain injury, brain tumor | |
| Melanocortin-4 receptor mutation | Consequences of cranial irradiation | |
| Leptin and leptin receptor deficiency | Hypothalamic obesity | |
| Prohormone convertase deficiency | Endocrine | |
| BDNF and TrkB insufficiency | Hypothyroidism | a |
| SIM 1 insufficiency | Cushing syndrome | |
| Proopiomelanocortin deficiency | Growth hormone deficiency | |
| Syndromes | Pseudohypoparathyroidism | |
| Prader–Willi | Psychological | |
| Bardet–Biedl | Eating disorders, depression | b |
| Cohen | Drug-induced | |
| Alström | Tricyclic antidepressants, antipsychotics | |
| Beckwith–Wiedemann | Oral contraceptives | |
| Froehlich | Anticonvulsants | |
| Carpenter | Glucocorticoids | |
| Sulfonylureas | ||
| Glitazones | ||
| Beta-blockers |
a Controversial whether hypothyroidism causes obesity or exacerbates obesity. b Depression associated with overeating or binging [3].
The primary causes of obesity can be classified as being related to genetic disorders in the form of monogenic diseases and genetic syndromes [6]. Monogenic obesity is a rare and severe early-onset obesity inherited in a Mendelian pattern with abnormal feeding behavior and endocrine abnormalities. It is mainly caused by autosomal recessive mutations in genes of leptin, pro-opiomelanocortin (POMC), pro-hormone convertase 1, and melanocortin 4 receptor (MC4R), which play a key role in the hypothalamic control of food intake. The predominant features of the genetic syndromes associated with obesity presented in Table 1 are physical characteristics, including dysmorphic features, developmental delay, and mental retardation. Existing genetic defects are often chromosomal abnormalities that usually involve multiple genes. Prader–Willi syndrome is one of the most common syndromic forms of obesity in children [6].
Secondary causes of obesity include diseases involving systemic dysfunctions and disorders of regulatory mechanisms causing metabolic changes in the body, which promote the development of obesity secondary to the primary disease. Disturbances in homeostasis resulting from diseases lead to the body’s inability to maintain energy balance, dysregulation in hormone synthesis and secretion, abnormal energy expenditure, and altered consumption behavior [7].
The association between gestational weight gain and obesity has been investigated, and maternal gestational weight increase is an independent predictor of obesity in infancy [8]. There is a strong association of genetics with obesity, which means that the involvement of multiple genes and their complex interaction can result in the manifestation of the disease, which can be a monogenic (5% of the cases) or a polygenic obesity type [9]. Obesity treatment consists of bariatric surgery and medical treatment, of which undergoing a surgery provides a longer life expectancy [10].
1.2. Association of Obesity with Other Diseases
Obesity increases the risk of other associated diseases, as it has been recognized as a key factor inhibiting DNA damage repair mechanisms. Cellular response to DNA damage can result in irreversible cell-cycle arrest, activation of several proteins that can induce adipocyte differentiation and hypertrophy, disturbances in cell metabolism, impairment of glucose metabolism, and promotion of the development of systemic insulin resistance [11]. The most well-established weight-related comorbidities are insulin resistance, type 2 diabetes, and cardiovascular disease, the risks of which are proportional to BMI [12]. Moreover, the obesity is linked with various immediate and long-term adverse health outcomes such as sleep apnea, hypertension, heart disease, stroke, osteoarthritis, and certain types of cancer and leads to psychosocial problems such as stigmatization and poor self-esteem [13].
The adverse effects of obesity on cardiometabolic health are indisputable; abdominal obesity is especially significant in the pathogenesis of cardiovascular disease and leads to well-identified disturbances in adipocyte biology and adipose tissue inflammation with direct systemic metabolic consequences such as endothelial dysfunction and atherogenesis [14].
Furthermore, multiple studies reveal a strong association between COVID-19 and obesity. COVID-19 patients with obesity have an enhanced hospitalization rate, more severe progression, and worse clinical outcomes [15].
Comorbidities of obesity are presented in Table 2.
Table 2. Comorbidities of obesity.
| Comorbidities of Obesity [15] | |
|---|---|
| Medical | Psychological |
| Dyslipidemia [16] | Negative mood [17] |
| Hypertension [16] | Attention deficit hyperactivity disorder [18] |
| Type 2 diabetes mellitus [16] | Depression [17] |
| Steatohepatitis and/or nonalcoholic fatty liver disease [18] | Poor self-esteem [17] |
| Polycystic ovary syndrome [18] | Eating disorders [18] |
| Gastro-esophageal reflux disease [18] | Internet addiction [18] |
| Obstructive sleep apnea [16] | Conduct issues or disorders [18] |
| Weight-related joint disease [16] | Reduced quality of life [18] |
| Benign intracranial hypertension [18] | |
1.3. Unhealthy Diet and Lifestyle and Their Relationship to Obesity
It is commonly stated that urbanization is one of the most important drivers of the worldwide rise in BMI because diet and lifestyle in cities lead to adiposity; however, in high-income and industrialized countries, a persistently higher rural BMI was noted, especially for women [19]. Sedentary behavior and time spent in front of TV are both related to a higher odds of obesity [20]. Among university students, being male, the family home not being in the university city, having a mother of low socioeconomic status, and finally, not studying a health-related course are the factors associated with a lower quality diet [21]. A positive correlation was found between BMI and consumption of a Westernized and high in protein/fat diet [22].
Studies conducted on adults showed that experimental reduction in sleep duration downregulates the satiety hormone, leptin, and upregulates the appetite-stimulating hormone, ghrelin, and increases hunger and appetite [23].
1.4. Gut Microbiome and Obesity
The body’s microbiome, bacteria, viruses, archaea, and eukaryotic microbes residing in and on the body have the potential to impact our physiology in several ways, including contributing to metabolic function. Studies have demonstrated that the composition of gut microbiome can among many other functions increase dietary energy intake, and therefore, promote the obese phenotype [3].
Throughout the gastrointestinal tract, differences in the composition of the microbial population have been observed. Due to the high motility of the esophagus and the acidic unfavorable environment of the stomach, there are quantitatively the fewest microorganisms, and the predominant bacteria come from the oral cavity (e.g., Streptococci and Lactobacilli). In the intestinal microbiota, there are already many more bacteria, e.g., in the initial part of the jejunum, the most numerous is the genus Streptococcus, while the ileocecal region is inhabited by the subgroup Bacillus bacteria (phylum Firmicutes, mainly Streptococcaceae), bacteria of the phylum Actinobacteria (especially the subgroups Actinomycinaeae and Corynebacteriaceae), Bacteroidetes, and Lachnospiraceae. The largest number of bacteria and the greatest microbial diversity are found in the distal segment of the ileum and colon because there is a more favorable pH for bacterial colonization, and in addition, due to antiperistaltic contractions, the food content is retained longer in the intestinal lumen. There are mainly the Gram-positive bacteria Bacteroides and Clostridium, Lactobacillus, Enterococcus, and Enterobacteriaceae.
2. Function and Physiology of Adipose Tissue
Adipose tissue is a compound and highly active metabolic and endocrine organ that consists of adipocytes, connective tissue, nerve tissue, and immune cells. These components together form an integrated and multifunctional unit [25][24].
There are two main types of adipose tissue distinguished in human body—white adipose tissue (WAT) and brown adipose tissue (BAT) [26][25]. Their content in the organism depends on genetic, metabolic, and environmental factors, but WAT makes up the majority of adipose tissue in adults [27][26].
2.1. Morphology
The differences between adipocytes in WAT and BAT can be observed via light microscope.
White adipocytes are spherical cells with a diameter of 10–100 μm [27][26]. Their organelles along with the compressed nucleus are in the perimeter of the cell due to a unilocular lipid droplet that occupies a major part of the cytosol [28][27].
Brown adipocytes typically present an ellipsoid shape that ranges from 15 to 50 μm in diameter and contains multilocular lipid droplets [27][26]. The nucleus of these cells is in the central part of the cytoplasm. BAT is also characterized by the expression of the proton transporter UCP (uncoupling protein) [28][27].
2.2. Function and Physiology
The adipose tissue is characterized by high plasticity and the ability to change volume as well as composition based on the energetic status of the body. An increase in the weight of adipose tissue may occur in both hypertrophy and hyperplasia [29][28].
The most important function of WAT is storage of triglycerides during increased energy supply and exploitation of these reserves during higher energy expenditure, while BAT plays a vital role in the process of thermogenesis, especially in infants [29][28].
Despite the differences in functions, both WAT and BAT present endocrine activity [30][29]. The factors secreted by the adipose tissue include leptin, adiponectin, complement components, plasminogen activator inhibitor-1, proteins of the renin–angiotensin system, and resistin. Moreover, metabolism of sex steroid hormones and glucocorticoids also takes place in adipocytes [25][24].
2.3. Endocrine Activity
Leptin was initially described as an anti-obesity hormone; however, current studies have proved that its function concentrates on energy sufficiency rather than on excess [31][30]. Therefore, lower energy intake results in a decrease in leptin levels, which is associated with physiological responses to starvation, such as increased appetite and restricted energy expenditure. This function complies with the evolutionary mechanism that prevents energy sufficiency but is not needed in current highly developed societies [25][24]. Furthermore, many forms of obesity result in elevated circulating leptin. However, both endo- and exogenous high leptin levels do not act against developing obesity. The mechanism for the occurrence of leptin resistance is unknown, but it may be a result of defective leptin signaling or transport across the blood–brain barrier [32][31].
Both adiponectin and resistin participate in the mechanism of insulin resistance.
There is an inverse association between adiponectin levels and insulin resistance—adiponectin levels are low with insulin resistance, and treatment with adiponectin may improve metabolic parameters in patients with obesity or lipodystrophy. Consequently, the increase in adiponectin levels occurs when insulin sensitivity improves [33][32].
On the other hand, resistin belongs to molecules that impair insulin-stimulated glucose uptake, which may lead to insulin resistance [34][33]. However, many epidemiological studies did not provide a valid link between resistin expression in adipose tissue or circulating resistin levels and the occurrence of this condition [35][34].
3. Function of Gut Microbiota
Gut microbiota consists of about 80 trillion bacteria that create a complex system that plays a vital role in maintenance of homeostasis in human organism. This includes, e.g., regulation of the host’s metabolism, provision of proper function of the intestinal barrier, and immunomodulation [36][35]. More than 100 bacterial species could be found in the intestines, and there are about ten times more bacterial cells present in gut microbiota than human cells in the body [37][36]. Most bacteria that form the gut microbiota belong to four bacterial phylotypes: Bacteroidetes, Firmicutes, Proteobacteria, and Actinobacteria, mainly anaerobic species [38,39][37][38]. However, various kinds of virus, protozoa, archaea, and fungi could also be a part of intestinal microflora [36][35].
The development of gut microbiota begins immediately at birth—the fetus is exposed to a bacterial population for the first time during passage through the birth canal [40][39]. That is the reason why infants’ microbiota can consist of similar bacteria that form the vaginal microbiota of their mothers [41][40]. Moreover, there is a proven difference in the composition of gut microbiota among infants delivered through caesarean section and those delivered vaginally [42][41].
The composition of intestinal microbiota undergoes many changes during a lifetime; nevertheless, the microbiota of one-year-old children tends to stabilize and begins to resemble that of young adults [41][40]. Due to this fact, the initial colonization of the gastrointestinal tract might be an important factor determining the composition of the microbiota in adulthood [40][39]. Studies on mono- and dizygotic twins have even proved that the bacterial colonization during birth had more impact on their adult microbiota than genetic factors [43][42]. However, the development of gut microbiota is a complex process and depends on many different factors (e.g., diet, usage of probiotics or antibiotics) and further studies are necessary to fully establish the parental role in determining the composition of the adult microbiota [40][39].
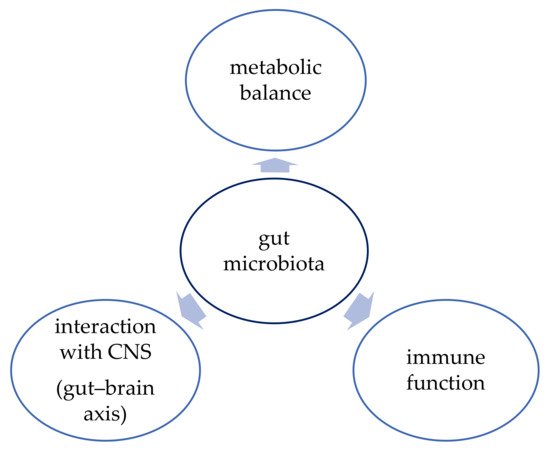
The function of human gut microbiota has been an object of interest of many studies conducted in the last few years. The function of the gut microbiota is shown in Figure 1. The microbiota proved to have a vital role in maintaining metabolic balance and proper function of the immune system, but there is also evidence that it can influence brain development and neurogenesis and that it interacts with the central nervous system (CNS) via the “gut–brain axis” [44][43]. Therefore, better understanding of this complex system might bring promising therapeutic options not only for obesity but also for depression, inflammatory bowel diseases (IBD), or cancer, for example [37][36].
3.1. Metabolic Balance
The bacteria present in the intestines create an extraordinary metabolic “organ” that can extract nutrients and energy from ingested food. For example, the gut microbiota enables the catabolism of dietary fiber that cannot be fully hydrolyzed by human enzymes during digestion. Short chain fatty acids (SCFAs) are the main product of this process, and they consist of fatty acids with fewer than six carbons [38][37]. Different studies proved that SCFAs could influence appetite regulation and the metabolism of lipids and glucose [45][44]. These molecules could also affect the integrity of the intestinal barrier by promoting the proper function of tight junctions (TJs) between epithelial cells and hence regulating the absorption of xenobiotics [38][37].
3.2. Immune Function
The gut microbiota contributes to the proper function of the immune system in two different ways [46][45]. First, it provides physical protection against enteropathogens by maintaining the structural integrity of the intestinal barrier [38][37]. Second, the intestinal microbiota plays an important role in immunomodulation. Symbiotic bacteria can influence the immune response of the host by improving the activity of macrophages and natural killer (NK) cells [46][45]. They also promote tolerogenic dendritic cells and regulate inflammatory-related pathways [38][37].
3.3. Gut–Brain Axis
The mechanism in which the bidirectional communication between the gut microbiota and the brain occurs has not been fully explained yet. However, the studies have shown there are many ways that allow this pathway to function, including neuroanatomical pathway and the endocrine, immune, and metabolic system [36][35]. This network allows the brain to affect gut movement and modify the sensory and secretion function of the intestines [36][35]. On the other hand, microorganisms present in the gut microbiota can produce neurotransmitters (e.g., dopamine) [44][43] and promote the release of gut hormones from gut enteroendocrine cells [47][46]. These transmitters provide the link to affect brain function [47][46].
References
- Kwaifa, I.K.; Bahari, H.; Yong, Y.K.; Noor, S.M. Endothelial Dysfunction in Obesity-Induced Inflammation: Molecular Mechanisms and Clinical Implications. Biomolecules 2020, 10, 291.
- Binkley, J.K.; Eales, J.; Jekanowski, M. The relation between dietary change and rising US obesity. Int. J. Obes. 2000, 24, 1032–1039.
- Apovian, C.M. Obesity: Definition, comorbidities, causes, and burden. Am. J. Manag. Care 2016, 22, S176–S185.
- Wang, Y.; Beydoun, M.A.; Min, J.; Xue, H.; Kaminsky, L.A.; Cheskin, L.J. Has the prevalence of overweight, obesity and central obesity levelled off in the United States? Trends, patterns, disparities, and future projections for the obesity epidemic. Int. J. Epidemiol. 2020, 49, 810–823.
- Maffeis, C.; Tatò, L. Long-term effects of childhood obesity on morbidity and mortality. Horm. Res. Paediatr. 2001, 55 (Suppl. S1), 42–45.
- Loos, R.J.F.; Yeo, G.S.H. The genetics of obesity: From discovery to biology. Nat. Rev. Genet. 2021, 1–14.
- Gadde, K.M.; Martin, C.K.; Berthoud, H.R.; Heymsfield, S.B. Obesity: Pathophysiology and Management. J. Am. Coll. Cardiol. 2018, 71, 69–84.
- Menghetti, E.; Strisciuglio, P.; Spagnolo, A.; Carletti, M.; Paciotti, G.; Muzzi, G.; Beltemacchi, M.; Concolino, D.; Strambi, M.; Rosano, A. Hypertension and obesity in Italian school children: The role of diet, lifestyle and family history. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 602–607.
- Sheikh, A.B.; Nasrullah, A.; Haq, S.; Akhtar, A.; Ghazanfar, H.; Nasir, A.; Afzal, R.M.; Bukhari, M.M.; Chaudhary, A.Y.; Naqvi, S.W. The Interplay of Genetics and Environmental Factors in the Development of Obesity. Cureus 2017, 9, e1435.
- Pontiroli, A.E.; Zakaria, A.S.; Fanchini, M.; Osio, C.; Tagliabue, E.; Micheletto, G.; Saibene, A.; Folli, F. A 23-year study of mortality and development of co-morbidities in patients with obesity undergoing bariatric surgery (laparoscopic gastric banding) in comparison with medical treatment of obesity. Cardiovasc. Diabetol. 2018, 17, 161.
- Lodarczyk, M.W.; Nowicka, G. Obesity, DNA damage, and development of obesity-related diseases. Int. J. Mol. Sci. 2019, 20, 1146.
- Pozza, C.; Isidori, A.M. What’s Behind the Obesity Epidemic. In Imaging in Bariatric Surgery; Laghi, A., Rengo, M., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 1–8.
- An, R. Diet quality and physical activity in relation to childhood obesity. Int. J. Adolesc. Med. Health 2017, 29.
- Antonopoulos, A.S.; Tousoulis, D. The molecular mechanisms of obesity paradox. Cardiovasc. Res. 2017, 113, 1074–1086.
- Ritter, A.; Kreis, N.-N.; Louwen, F.; Yuan, J. Obesity and COVID-19: Molecular Mechanisms Linking Both Pandemics. Int. J. Mol. Sci. 2020, 21, 5793.
- Pi-Sunyer, F.X. Comorbidities of overweight and obesity: Current evidence and research issues. Med. Sci. Sports Exerc. 1999, 31 (Suppl. S11), S602–S608.
- Fabricatore, A.N.; Wadden, T.A. Psychological aspects of obesity. Clin. Dermatol. 2004, 22, 332–337.
- Reinehr, T. Long-term effects of adolescent obesity: Time to act. Nat. Rev. Endocrinol. 2018, 14, 183–188.
- NCD Risk Factor Collaboration. Rising rural body-mass index is the main driver of the global obesity epidemic in adults. Nature 2019, 569, 260–264.
- Katzmarzyk, P.T.; Chaput, J.-P.; Fogelholm, M.; Hu, G.; Maher, C.; Maia, J.; Olds, T.; Sarmiento, O.L.; Standage, M.; Tremblay, M.S.; et al. International Study of Childhood Obesity, Lifestyle and the Environment (ISCOLE): Contributions to Understanding the Global Obesity Epidemic. Nutrients 2019, 11, 848.
- Martinez-Lacoba, R.; Pardo-Garcia, I.; Amo-Saus, E.; Escribano-Sotos, F. Socioeconomic, demographic and lifestyle-related factors associated with unhealthy diet: A cross-sectional study of university students. BMC Public Health 2018, 18, 1241.
- Gutiérrez-Pliego, L.E.; de Camarillo-Romero, E.; Montenegro-Morales, L.P.; de Garduño-García, J. Dietary patterns associated with body mass index (BMI) and lifestyle in Mexican adolescents. BMC Public Health 2016, 16, 850.
- Felső, R.; Lohner, S.; Hollódy, K.; Erhardt, É.; Molnár, D. Relationship between sleep duration and childhood obesity: Systematic review including the potential underlying mechanisms. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 751–761.
- Kershaw, E.E.; Flier, J.S. Adipose Tissue as an Endocrine Organ. J. Clin. Endocrinol. Metab. 2004, 89, 2548–2556.
- Acín-Perez, R.; Petcherski, A.; Veliova, M.; Benador, I.Y.; Assali, E.A.; Colleluori, G.; Cinti, S.; Brownstein, A.J.; Baghdasarian, S.; Livhits, M.J.; et al. Recruitment and remodeling of peridroplet mitochondria in human adipose tissue. Redox Biol. 2021, 46, 102087.
- Cinti, S. The adipose organ. Prostaglandins Leukot Essent Fat. Acids 2005, 73, 9–15.
- Sarjeant, K.; Stephens, J.M. Adipogenesis. Cold Spring Harb. Perspect. Biol. 2012, 4, a008417.
- Murawska-Ciałowicz, E. Adipose tissue-morphological and biochemical characteristic of different depots. Postepy. Hig. Med. Dosw. 2017, 71, 466–484.
- Cawthorn, W.P.; Scheller, E.L.; MacDougald, O.A. Adipose tissue stem cells meet preadipocyte commitment: Going back to the future. J. Lipid Res. 2012, 53, 227–246.
- Flier, J.S. Clinical review 94: What’s in a name? In search of leptin’s physiologic role. J. Clin. Endocrinol. Metab. 1998, 83, 1407–1413.
- Flier, J.S. Obesity wars: Molecular progress confronts an expanding epidemic. Cell 2004, 116, 337–350.
- Díez, J.J.; Iglesias, P. The role of the novel adipocyte-derived protein adiponectin in human disease: An update. Mini Rev. Med. Chem. 2010, 10, 856–869.
- Steppan, C.M.; Bailey, S.T.; Bhat, S.; Brown, E.J.; Banerjee, R.R.; Wright, C.M.; Patel, H.R.; Ahima, R.S.; Lazar, M.A. The hormone resistin links obesity to diabetes. Nature 2001, 409, 307–312.
- Banerjee, R.R.; Lazar, M.A. Resistin: Molecular history and prognosis. J. Mol. Med. 2003, 81, 218–226.
- Wang, H.-X.; Wang, Y.-P. Gut Microbiota-brain Axis. Chin. Med. J. 2016, 129, 2373–2380.
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 2012, 489, 220–230.
- Li, X.; Liu, L.; Cao, Z.; Li, W.; Li, H.; Lu, C.; Yang, X.; Liu, Y. Gut microbiota as an “invisible organ” that modulates the function of drugs. Biomed Pharm. 2020, 121, 109653.
- Grenham, S.; Clarke, G.; Cryan, J.F.; Dinan, T.G. Brain? Gut? Microbe Communication in Health and Disease. Front. Physio. 2011, 2, 94.
- Sekirov, I.; Russell, S.L.; Antunes, L.C.M.; Finlay, B.B. Gut Microbiota in Health and Disease. Physiol. Rev. 2010, 90, 859–904.
- Mändar, R.; Mikelsaar, M. Transmission of mother’s microflora to the newborn at birth. Biol. Neonatol. 1996, 69, 30–35.
- Huurre, A.; Kalliomäki, M.; Rautava, S.; Rinne, M.; Salminen, S.; Isolauri, E. Mode of delivery-effects on gut microbiota and humoral immunity. Neonatology 2008, 93, 236–240.
- Zoetendal, E.G.; Ak, A.D.L. The Host Genotype Affects the Bacterial Community in the Human Gastronintestinal Tract. Microb. Ecol. Health Dis. 2001, 13, 129–134.
- Strandwitz, P. Neurotransmitter modulation by the gut microbiota. Brain Res. 2018, 1693 Pt B, 128–133.
- Chambers, E.S.; Morrison, D.J.; Frost, G. Control of appetite and energy intake by SCFA: What are the potential underlying mechanisms? Proc. Nutr. Soc. 2015, 74, 328–336.
- Goldman, C.G.; Barrado, D.A.; Balcarce, N.; Rua, E.C.; Oshiro, M.; Calcagno, M.L.; Janjetic, M.; Fuda, J.; Weill, R.; Salgueiro, M.J.; et al. Effect of a probiotic food as an adjuvant to triple therapy for eradication of Helicobacter pylori infection in children. Nutrition 2006, 22, 984–988.
- Sun, L.-J.; Li, J.-N.; Nie, Y.-Z. Gut hormones in microbiota-gut-brain cross-talk. Chin. Med. J. 2020, 133, 826–833.
More