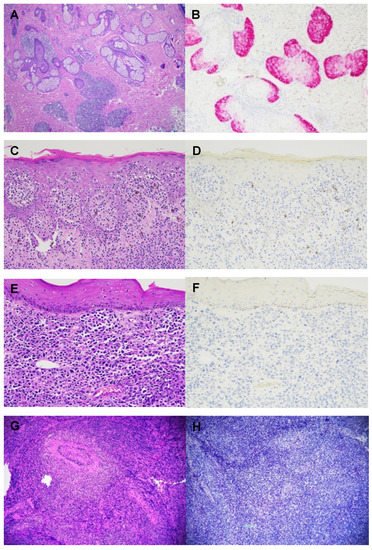
Cutaneous T-cell lymphomas (CTCLs) are rare tumors with no established markers that can reliably distinguish between benign and malignant lesions. Preferentially Expressed Antigen in Melanoma (PRAME) is a cancer/testis antigen that is found in many solid and hematologic malignancies. PRAME overexpression typically portends a poor prognosis and lower chemotherapeutic response. To date, no studies have established a role for PRAME in CTCL.
An analysis was performed on 47 cases definitively diagnosed as CTCL: 25 mycosis fungoides, 2 Sezary syndrome, 5 CD30+ lymphoproliferative disorder, 7 primary cutaneous anaplastic large T-cell lymphoma, 3 primary cutaneous CD4+ small/medium T-cell lymphoproliferative disorder, 1 subcutaneous panniculitis-like T-cell lymphoma, and 4 angiocentric T-cell lymphoma. PRAME immunohistochemistry was completely negative in all cases. PRAME expression was not found in any CTCL subtypes, suggesting that the pathogenesis of CTCL is not mediated by PRAME. Further study is required to identify biomarkers that might aid in the diagnosis and prognostication of CTCLs.
- cutaneous T-cell lymphoma
- PRAME
1. Background
2. Current Insight on Cutaneous T-Cell Lymphomas
| CTCLs | Number of Cases (%) | PRAME Immunohistochemistry |
|---|---|---|
| MF | 25 (53.2) | Negative |
| MF with CD8 immunophenotype | 2 (8.0) | Negative |
| MF with large cell transformation | 3 (12.0) | Negative |
| MF with blast cell transformation | 1 (4.0) | Negative |
| Sezary Syndrome | 2 (4.3) | Negative |
| CD30+ lymphoproliferative disorder | 5 (10.6) | Negative |
| Primary cutaneous anaplastic large T-cell lymphoma | 7 (14.9) | Negative |
| Primary cutaneous CD4+ small/medium T-cell Lymphoproliferative disorder | 3 (6.4) | Negative |
| Angiocentric T-cell lymphoma | 4 (8.5) | Negative |
| Subcutaneous panniculitis-like T-cell lymphoma | 1 (2.1) | Negative |
References
- Rodd, A.L.; Ververis, K.; Karagiannis, T.C. Current and Emerging Therapeutics for Cutaneous T-Cell Lymphoma: Histone Deacetylase Inhibitors. Lymphoma 2012, 2012, 290685.
- Eklund, Y.; Aronsson, A.; Schmidtchen, A.; Relander, T. Mycosis Fungoides: A Retrospective Study of 44 Swedish Cases. Acta Derm. Venereol. 2016, 96, 669–673.
- Bagherani, N.; Smoller, B.R. An overview of cutaneous T cell lymphomas. F1000Res 2016, 5, 1882.
- Wilcox, R.A. Cutaneous T-cell lymphoma: 2017 update on diagnosis, risk-stratification, and management. Am. J. Hematol. 2017, 92, 1085–1102.
- Willemze, R.; Cerroni, L.; Kempf, W.; Berti, E.; Facchetti, F.; Swerdlow, S.H.; Jaffe, E.S. The 2018 update of the WHO-EORTC classification for primary cutaneous lymphomas. Blood 2019, 133, 1703–1714, Correction in Blood 2019, 134, 1112.
- Olsen, E.; Vonderheid, E.; Pimpinelli, N.; Willemze, R.; Kim, Y.; Knobler, R.; Zackheim, H.; Duvic, M.; Estrach, T.; Lamberg, S.; et al. Revisions to the staging and classification of mycosis fungoides and Sezary syndrome: A proposal of the International Society for Cutaneous Lymphomas (ISCL) and the cutaneous lymphoma task force of the European Organization of Research and Treatment of Cancer (EORTC). Blood 2007, 110, 1713–1722, Erratum in Blood 2008, 111, 4830.
- Vandergriff, T.; Nezafati, K.A.; Susa, J.; Karai, L.; Sanguinetti, A.; Hynan, L.S.; Ambruzs, J.M.; Oliver, D.H.; Pandya, A.G. Defining early mycosis fungoides: Validation of a diagnostic algorithm proposed by the International Society for Cutaneous Lymphomas. J. Cutan. Pathol. 2015, 42, 318–328.
- Peterson, E.; Weed, J.; Lo Sicco, K.; Latkowski, J.A. Cutaneous T Cell Lymphoma: A Difficult Diagnosis Demystified. Dermatol. Clin. 2019, 37, 455–469.
- Foss, F.M.; Girardi, M. Mycosis Fungoides and Sezary Syndrome. Hematol. Oncol. Clin. N. Am. 2017, 31, 297–315.
- Hodak, E.; Amitay-Laish, I. Mycosis fungoides: A great imitator. Clin. Dermatol. 2019, 37, 255–267.
- Pulitzer, M. Cutaneous T-cell Lymphoma. Clin. Lab. Med. 2017, 37, 527–546.
- Geller, S.; Myskowski, P.L.; Pulitzer, M.; Horwitz, S.M.; Moskowitz, A.J. Cutaneous T-cell lymphoma (CTCL), rare subtypes: Five case presentations and review of the literature. Chin. Clin. Oncol. 2019, 8, 5.
- Cocks, M.; Porcu, P.; Wick, M.R.; Gru, A.A. Recent Advances in Cutaneous T-cell Lymphoma: Diagnostic and Prognostic Considerations. Surg. Pathol. Clin. 2019, 12, 783–803.
- Jawed, S.I.; Myskowski, P.L.; Horwitz, S.; Moskowitz, A.; Querfeld, C. Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome): Part I. Diagnosis: Clinical and histopathologic features and new molecular and biologic markers. J. Am. Acad. Dermatol. 2014, 70, 205.e1–205.e16.
- Kirsch, I.R.; Watanabe, R.; O’Malley, J.T.; Williamson, D.W.; Scott, L.-L.; Elco, C.P.; Teague, J.E.; Gehad, A.; Lowry, E.L.; LeBoeuf, N.R.; et al. TCR sequencing facilitates diagnosis and identifies mature T cells as the cell of origin in CTCL. Sci. Transl. Med. 2015, 7, 308ra158.
- Xu, Y.; Zou, R.; Wang, J.; Wang, Z.; Zhu, X. The Role of the Cancer Testis Antigen PRAME in Tumorigenesis and Immunotherapy in Human Cancer. Cell Prolif. 2020, 53, e12770.
- Ikeda, H.; Lethé, B.; Lehmann, F.; Van Baren, N.; Baurain, J.-F.; De Smet, C.; Chambost, H.; Vitale, M.; Moretta, A.; Boon, T.; et al. Characterization of an antigen that is recognized on a melanoma showing partial HLA loss by CTL expressing an NK inhibitory receptor. Immunity 1997, 6, 199–208.
- Quintarelli, C.; Dotti, G.; Hasan, S.T.; De Angelis, B.; Hoyos, V.; Errichiello, S.; Mims, M.; Luciano, L.; Shafer, J.; Leen, A.M.; et al. High-avidity cytotoxic T lymphocytes specific for a new PRAME-derived peptide can target leukemic and leukemic-precursor cells. Blood 2011, 117, 3353–3362.
- Salmaninejad, A.; Zamani, M.R.; Pourvahedi, M.; Golchehre, Z.; Hosseini Bereshneh, A.; Rezaei, N. Cancer/Testis Antigens: Expression, Regulation, Tumor Invasion, and Use in Immunotherapy of Cancers. Immunol. Investig. 2016, 45, 619–640.
- Lezcano, C.; Jungbluth, A.A.; Busam, K.J. PRAME Immunohistochemistry as an Ancillary Test for the Assessment of Melanocytic Lesions. Surg. Pathol. Clin. 2021, 14, 165–175.
- Wei, R.; Dean, D.C.; Thanindratarn, P.; Hornicek, F.J.; Guo, W.; Duan, Z. Cancer testis antigens in sarcoma: Expression, function and immunotherapeutic application. Cancer Lett. 2020, 479, 54–60.
- Zhu, H.; Wang, J.; Yin, J.; Lu, B.; Yang, Q.; Wan, Y.; Jia, C. Downregulation of PRAME Suppresses Proliferation and Promotes Apoptosis in Hepatocellular Carcinoma Through the Activation of P53 Mediated Pathway. Cell Physiol. Biochem. 2018, 45, 1121–1135, Epub 7 February 2018.
- Dyrskjøt, L.; Zieger, K.; Kissow Lildal, T.; Reinert, T.; Gruselle, O.; Coche, T.; Borre, M.; Ørntoft, T.F. Expression of MAGE-A3, NY-ESO-1, LAGE-1 and PRAME in urothelial carcinoma. Br. J. Cancer 2012, 107, 116–122.
- Zhang, W.; Barger, C.J.; Eng, K.H.; Klinkebiel, D.; Link, P.A.; Omilian, A.; Bshara, W.; Odunsi, K.; Karpf, A.R. PRAME expression and promoter hypomethylation in epithelial ovarian cancer. Oncotarget 2016, 7, 45352–45369.
- Mitsuhashi, K.; Masuda, A.; Wang, Y.H.; Shiseki, M.; Motoji, T. Prognostic significance of PRAME expression based on immunohistochemistry for diffuse large B-cell lymphoma patients treated with R-CHOP therapy. Int. J. Hematol. 2014, 100, 88–95.
- Ercolak, V.; Paydas, S.; Bagir, E.; Ergin, M.; Seydaoglu, G.; Celik, H.; Yavu, B.; Tanriverdi, K.; Gunaldi, M.; Afsar, C.U.; et al. PRAME Expression and Its Clinical Relevance in Hodgkin’s Lymphoma. Acta Haematol. 2015, 134, 199–207.
- Proto-Siqueira, R.; Figueiredo-Pontes, L.L.; Panepucci, R.A.; Garcia, A.B.; Rizzatti, E.G.; Nascimento, F.M.; Ishikawa, H.C.; Larson, R.E.; Falcão, R.P.; Simpson, A.J.; et al. PRAME is a membrane and cytoplasmic protein aberrantly expressed in chronic lymphocytic leukemia and mantle cell lymphoma. Leuk. Res. 2006, 30, 1333–1339.
- Shiseki, M.; Ishii, M.; Ohwashi, M.; Wang, Y.H.; Tanaka, N.; Osanai, S.; Yoshinaga, K.; Mori, N.; Tanaka, J. High PRAME expression is associated with poor survival and early disease progression in myelodysplastic syndromes with a low bone marrow blast percentage. Leuk. Lymphoma 2021, 62, 2448–2456.
- Arons, E.; Suntum, T.; Margulies, I.; Yuan, C.; Stetler-Stevenson, M.; Kreitman, R.J. PRAME expression in hairy cell leukemia. Leuk. Res. 2008, 32, 1400–1406.
- Beà, S.; Salaverria, I.; Armengol, L.; Pinyol, M.; Fernández, V.; Hartmann, E.M.; Jares, P.; Amador, V.; Hernández, L.; Navarro, A.; et al. Uniparental disomies, homozygous deletions, amplifications, and target genes in mantle cell lymphoma revealed by integrative high-resolution whole-genome profiling. Blood 2009, 113, 3059–3069.
- Epping, M.T.; Hart, A.A.; Glas, A.M.; Krijgsman, O.; Bernards, R. PRAME expression and clinical outcome of breast cancer. Br. J. Cancer 2008, 99, 398–403.
- Albertsmeier, M.; Altendorf-Hofmann, A.; Lindner, L.H.; Issels, R.D.; Kampmann, E.; Dürr, H.R.; Schubert-Fritschle, G.; Angele, M.K.; Kirchner, T.; Jungbluth, A.A.; et al. Cancer Testis Antigens and Immunotherapy: Expression of PRAME Is Associated with Prognosis in Soft Tissue Sarcoma. Cancers 2020, 12, 3612.
- Tanaka, N.; Wang, Y.H.; Shiseki, M.; Takanashi, M.; Motoji, T. Inhibition of PRAME expression causes cell cycle arrest and apoptosis in leukemic cells. Leuk. Res. 2011, 35, 1219–1225.
- Radich, J.P.; Dai, H.; Mao, M.; Oehler, V.; Schelter, J.; Druker, B.; Sawyers, C.; Shah, N.; Stock, W.; Willman, C.L.; et al. Gene expression changes associated with progression and response in chronic myeloid leukemia. Proc. Natl. Acad. Sci. USA 2006, 103, 2794–2799.
- Al-Khadairi, G.; Decock, J. Cancer Testis Antigens and Immunotherapy: Where Do We Stand in the Targeting of PRAME? Cancers 2019, 11, 984.
- Gezgin, G.; Luk, S.J.; Cao, J.; Dogrusöz, M.; van der Steen, D.M.; Hagedoorn, R.S.; Krijgsman, D.; van der Velden, P.A.; Field, M.G.; Luyten, G.P.M.; et al. PRAME as a Potential Target for Immunotherapy in Metastatic Uveal Melanoma. JAMA Ophthalmol. 2017, 135, 541–549.
- Matsushita, M.; Yamazaki, R.; Ikeda, H.; Kawakami, Y. Preferentially expressed antigen of melanoma (PRAME) in the development of diagnostic and therapeutic methods for hematological malignancies. Leuk. Lymphoma 2003, 44, 439–444.
- Roszik, J.; Wang, W.L.; Livingston, J.A.; Roland, C.L.; Ravi, V.; Yee, C.; Hwu, P.; Futreal, A.; Lazar, A.J.; Patel, S.R.; et al. Overexpressed PRAME is a potential immunotherapy target in sarcoma subtypes. Clin. Sarcoma Res. 2017, 7, 11.
- Tajeddine, N.; Louis, M.; Vermylen, C.; Gala, J.L.; Tombal, B.; Gailly, P. Tumor associated antigen PRAME is a marker of favorable prognosis in childhood acute myeloid leukemia patients and modifies the expression of S100A4, Hsp 27, p21, IL-8 and IGFBP-2 in vitro and in vivo. Leuk. Lymphoma 2008, 49, 1123–1131.
- Steinbach, D.; Hermann, J.; Viehmann, S.; Zintl, F.; Gruhn, B. Clinical implications of PRAME gene expression in childhood acute myeloid leukemia. Cancer Genet. Cytogenet. 2002, 133, 118–123.
- Steinbach, D.; Viehmann, S.; Zintl, F.; Gruhn, B. PRAME gene expression in childhood acute lymphoblastic leukemia. Cancer Genet. Cytogenet. 2002, 138, 89–91.
- Greiner, J.; Schmitt, M.; Li, L.; Giannopoulos, K.; Bosch, K.; Schmitt, A.; Dohner, K.; Schlenk, R.F.; Pollack, J.R.; Dohner, H.; et al. Expression of tumor-associated antigens in acute myeloid leukemia: Implications for specific immunotherapeutic approaches. Blood 2006, 108, 4109–4117.
- de Masson, A.; O’Malley, J.T.; Elco, C.P.; Garcia, S.S.; Divito, S.J.; Lowry, E.L.; Tawa, M.; Fisher, D.C.; Devlin, P.M.; Teague, J.E.; et al. High-throughput sequencing of the T cell receptor β gene identifies aggressive early-stage mycosis fungoides. Sci. Transl. Med. 2018, 10, eaar5894.
