Produced water (PW) is the most significant waste stream generated in the oil and gas industries. The generated PW has the potential to be a useful water source rather than waste. While a variety of technologies can be used for the treatment of PW for reuse, biological-based technologies are an effective and sustainable remediation method. Specifically, microalgae, which are a cost-effective and sustainable process that use nutrients to eliminate organic pollutants from PW during the bioremediation process. In these treatment processes, microalgae grow in PW free of charge, eliminate pollutants, and generate clean water that can be recycled and reused. This helps to reduce CO2 levels in the atmosphere while simultaneously producing biofuels, other useful chemicals, and added-value products.
- produced water
- oil and gas production
- BTEX removal
- biological treatment
- microalgae
1. Introduction
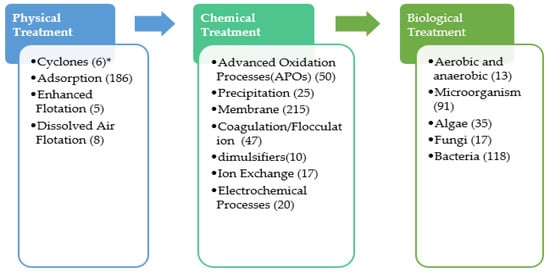
23. Produced Water
2.1. Produced Water Generation
3.1. Produced Water Generation
| Sr |
| + |
| Composition | Concentration Range (mg/L) | References |
| Chemical oxygen demand (COD) | 1220–2600 | [4][6][8][26][27] |
| Sodium ions (Na+) | 0–150,000 | |
| Total suspended solids (TSS) | 1.2–1000 | [4][6][8][19][26][27] |
| Calcium ion (Ca2+) | 0–74,000 | |
| Total polar compounds | 9.7–600 | [6][8][28] |
| Boron (B) | 5–95 | |
| Total dissolved solids (TDS) | 100–400,000 | [8][28] |
| Chlorine (Cl−) | 0–270,000 | |
| BTEX; benzene (B), toluene (T), ethylbenzene (E), and xylenes (X) | 0.73–24.1 | [4][26] |
| Magnesium (Mg2+) | 8–6000 | |
| Total organic compound (TOC) | 0–1500 | [4][6][19][26][27] |
| Iron(II) (Fe2+) | 0.1–1100 | |
| Total oil and grease | 2–565 | [28] |
| Barium ion (Ba2+) | 0–850 | |
| Phenol | 0.009–23 | [6][26][28] |
| Potassium ion (K+) | 24–4300 | |
| pH | 4.3–10 | [4] |
| Strontium ion (Sr+) | 0–6250 | |
| Total organic acids | 0.001–10,000 | [4] |
| Aluminium (Al3+) | 310–340 | |
| Lithium (Li+) | 3–40 | [4][26] |
| Lead (Pb) | 0.008–0.08 | |
| Bicarbonate (HCO−3) | 0–15,000 | [8] |
| Arsenic (As) | 0.002–11 | |
| Sulfate (SO2−4) | 0–15,000 | [4][6][8] |
| Manganese (Mn) | 0.004–175 | |
| Titanium (Ti) | 0.01–0.7 | [4][6] |
| Parameter | Raw Produced Water | Filtered Water |
|---|---|---|
| Total organic carbon (mg/L) | 389.1 | 317 |
| Total nitrogen (mg/L) | 35.77 | 27.6 |
| Total phosphorus (μg/L) | 277.78 | 180 |
| Benzene (mg/L) | 21 | 16.1 |
| Toluene (mg/L) | 3.8 | 3.21 |
| Ethylbenzene (mg/L) | 1.22 | 1.05 |
| Xylene (mg/L) | 3.43 | 3.11 |
2.2. Characteristics of Produced Water
3.2. Characteristics of Produced Water
Produced water characteristics vary between regions and a specific study for each area should be conducted to investigate the effects of PW discharge on the environment [8][17][8,18]. Further, PW contains a complex composition of physical and chemical properties, dependent on the geological formation, geographic field [5], extraction method, and the type of extracted hydrocarbon [6]. Rahman et al. [1] detail a list of PW parameters and their typical range. It was observed that the toxicity of the PW generated from gas wells is 10 times greater than the toxicity produced from oil wells [5]. Given that, special treatment should be taken for PW from oil wells. The composition of PW from oil fields is summarized in Table 23. The primary constitutes found in PW are total dissolved solids (TDS), salts, benzene (B), toluene (T), ethylbenzene (E), and xylenes (X) (denoted as BTEX), polyaromatic hydrocarbons (PAHs), oil and grease (O&G). The BTEX are volatile organic compounds that naturally occur in oil and gas wells, including gasoline and natural gas. The BTEX compounds also freely escape into the atmosphere during PW treatment [25][31]. Additionally, traces of natural organic and inorganic compounds, phenol, organic acids, and chemical additives added during the drilling process can be found in PW and contribute to its total toxicity [5].| Zinc (Zn) |
| 0.01–35 |
| Composition | Concentration Range (mg/L) | References |
|---|
| Microalgae Species | Type of Nutrients | Removal Efficiency% | References | ||
|---|---|---|---|---|---|
| Chemical oxygen demand (COD) | 1220–2600 | ||||
| Dunaliella salina | Nitrogen Phosphorus heavy metal: Ni | [4,6,8,32,33] | |||
| Zn | 65% | 40% 90% 80% |
[40] | [63] | Sodium ions (Na+ |
| Nannochloropsis oculata | Ammonium and Nitrogen Organic carbon Iron |
~100% 40% >90% |
[41] | [64] | ||||||||||||||||||
| Parachlorella kessleri | Benzene and Xylenes Toluene Ethylbenzene |
40% 63% 30% |
[42] | [65] | ||||||||||||||||||
| Chlorella vulgaris | ( | C.v | ) | Neochloris oleoabundans | ( | N.o | ) | COD by ( | C.v | ) by ( | N.o | ) Ammonia by | C.v. | and | N.o | Phosphorus by | C.v. | and | N.o | 51%, 55% and 80% 63%, 47% and 72% (70–84%) (>84%), (>22%) and (<15%) |
[43] | [66] |
| Chlorella pyrenoidosa | Chromium Nickel |
11.24% 33.89% |
[44] | [67] | ||||||||||||||||||
| ) | 0–150,000 | |||||||||||||||||||||
| Total suspended solids (TSS) | 1.2–1000 | [4,6,8,20,32,33] | ||||||||||||||||||||
| Calcium ion (Ca2+ |
| ) | 0–74,000 |
| Cultivation System | Algae Species | Cultivation Condition | Type of Waste | Biomass Productivity g/(L.d). | Organic Removal | Biofuel Type | Refs. |
|---|---|---|---|---|---|---|---|
| Total polar compounds | |||||||
| 9.7–600 | [6,8,34] | ||||||
| Boron (B) | 5–95 | ||||||
| Total dissolved solids (TDS) | 100–400,000 | [8,34] | |||||
| Chlorine (Cl−) |
| Closed system (PBRs) | Scenedesmus acutus (UTEX B72) | Agriculture-grade urea, triple super phosphate (TSP), pot ash and Sprint 330 (iron chelate) | Flue gas | 0.15 | Sulfur, NOx | [45] | [68] | ||||||||
| Closed system 4-L cylindrical photobioreactor (PBR) | Mixed culture of | Chlorella vulgaris | , | Scenedesmus Obliquus, Botryococcus braunii | , | Botryococcus sudeticus | , and | Afrocarpus falcatus | pH = 7, Temp = 25 °C. | 0.15 | 21, 60, and 47% for protein, carbohydrate and DOC, respectively | [46] | [69] | ||
| 500 mL glass flasks | Dunaliella tertiolecta | pH—8.1, Temp = 24 °C, f/2 medium | Real PW | 0.0172 @ salinity 30 gTDS/L to 0.0098 @ 201 gTDS/L | Biodiesel | [47] | [61] | ||||||||
| 500 mL glass flasks | Cyanobacterium aponinum, Parachlorella kessleri | pH—8.1, Temp = 24 °C, f/2 medium | Real PW | 0.113 * | Biodiesel | [48] | [70] | ||||||||
| Synechococcus | sp., | Cyanobacterium aponinum and Phormidium | sp. | pH = (6–9), | BG-11 medium | NA | Biodiesel | [49] | [71] | ||||||
| Chlorella | sp. and | Scenedesmus | sp. | pH = 7.1 | 0.115 * | Chlorella | sp.: remove 92% of the TN and 73% of the TOC | [50] | [72] | ||||||
| Dunaliella salina | Salinity 52.7–63.3 g/L NaCl | Real produced water | NA | Aluminum, barium, copper, magnesium, manganese, nickel, and strontium | Biodiesel | [51] | [59] | ||||||||
| Horizontal laminar air flow chamber | Chlorella pyrenoidosa | T = 121 °C | Fogg’s Medium, slant culture | NA | Biofuel and bioplastic | ||||||||||
| 0–270,000 | |||||||||||||||
| BTEX; benzene (B), toluene (T), ethylbenzene (E), and xylenes (X) | 0.73–24.1 | [4,32] | |||||||||||||
| Magnesium (Mg2+) |
| 8–6000 | ||
| Total organic compound (TOC) | 0–1500 | [4,6,20,32,33] |
| Iron(II) (Fe2+ |
| ) | 0.1–1100 | |
| Total oil and grease | 2–565 | [34] |
| Barium ion (Ba2+) |
| 0–850 | ||
| Phenol | 0.009–23 | [6,32,34] |
| Potassium ion (K+ |
| ) | 24–4300 | |
| pH | 4.3–10 | [4] |
| Strontium ion ( |
| ) | 0–6250 | |
| Total organic acids | 0.001–10,000 | [4] |
| Aluminium (Al3+ |
| ) | 310–340 |
| Lithium (Li+ |
| [ | ||
| 44 | ||
| ] | ||
| [ | ||
| 67 | ||
| ] | ||
| ) | 3–40 | [4,32] |
| Lead (Pb) | 0.008–0.08 | |
| Bicarbonate (HCO−3 |
| ) | 0–15,000 | [8] |
| Arsenic (As) | 0.002–11 | |
| Sulfate (SO2−4 |
| ) | 0–15,000 | [4,6,8] |
| Manganese (Mn) | 0.004–175 | |
| Titanium (Ti) | 0.01–0.7 | [4,6] |
| Zinc (Zn) | 0.01–35 |
