Dental calculus (DC) is a common deposit in periodontitis patients. DC contains both microbial components and calcium phosphate crystals that induce an osteoclastogenic cytokines, such as IL-1β and IL-18, via the NLRP3 inflammasome in macrophages.
- dental calculus
- NLRP3 inflammasome
- IL-1β
- IL-18
- osteoclast
1. Introduction
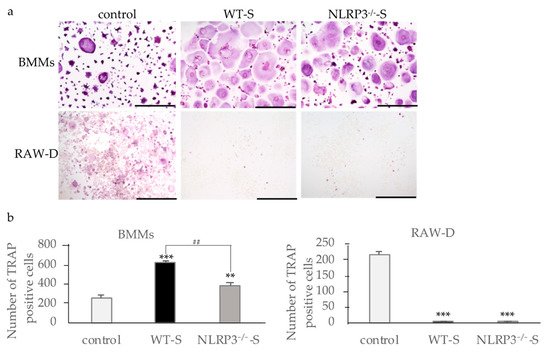
2. Effects of Culture Supernatants from Mouse Macrophages Stimulated with Dental Calculus on Osteoclast Formation
When bone marrow macrophages (BMMs) primed with M-CSF and RANKL were incubated with the supernatant from the NLRP3-deficient mouse macrophages stimulated with DC, osteoclast formation was promoted, suggesting the effects of pro-inflammatory cytokines in the supernatants (Figure 1a). The supernatant of WT mouse macrophages stimulated with DC induced higher numbers of osteoclasts than the supernatant from NLRP3-deficient mouse macrophages, suggesting the effect of another pro-inflammatory cytokine (Figure 1b). In contrast to the results of RANKL-primed BMMs, the supernatants from both WT and NLRP3-deficient mouse macrophages stimulated with DC suppressed osteoclast formation in RANKL-primed RAW-D cells (Figure 1a,b). Incubation of RANKL-primed RAW-D cells with the supernatant from unstimulated WT or NLRP3-deficient mouse macrophages did not affect the osteoclastogenesis.
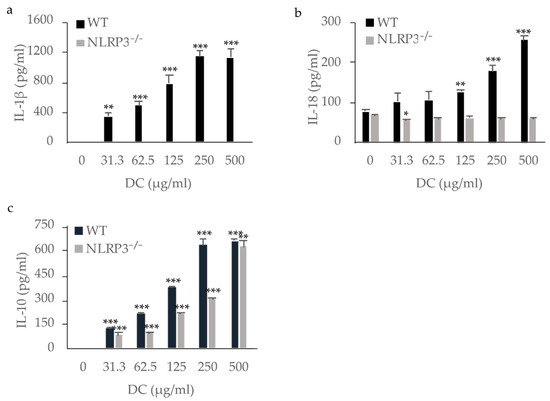
3. Cytokine Production by Macrophages Stimulated with DC
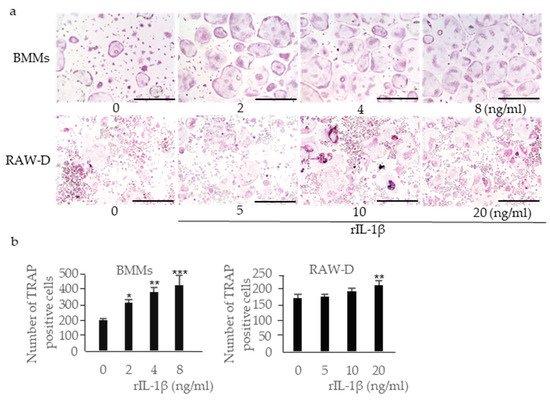
4. Effects of IL-1β, IL-18, and IL-10 on Osteoclast Formation

References
- Papapanou, P.N.; Sanz, M.; Buduneli, N.; Dietrich, T.; Feres, M.; Fine, D.H.; Flemmig, T.F.; Garcia, R.; Giannobile, W.V.; Graziani, F.; et al. Periodontitis: Consensus report of workgroup 2 of the 2017 world workshop on the classification of periodontal and peri-implant diseases and conditions. J. Periodontol. 2018, 89, S173–S182.
- Pihlstrom, B.L.; Michalowicz, B.S.; Johnson, N.W. Periodontal diseases. Lancet 2005, 366, 1809–1820.
- Ruscitti, P.; Cipriani, P.; Carubbi, F.; Liakouli, V.; Zazzeroni, F.; Di Benedetto, P.; Berardicurti, O.; Alesse, E.; Giacomelli, R. The role of IL-1β in the bone loss during rheumatic diseases. Mediat. Inflamm. 2015, 2015, 1–10.
- Zhu, H.; Lin, X.; Zheng, P.; Chen, H. Inflammatory cytokine levels in patients with periodontitis and/or coronary heart disease. Int. J. Clin. Exp. Pathol. 2015, 8, 2214–2220.
- Gamonal, J.; Acevedo, A.; Bascones, A.; Jorge, O.; Silva, A. Levels of interleukin-1β, -8, and -10 and RANTES in gingival crevicular fluid and cell populations in adult periodontitis patients and the effect of periodontal treatment. J. Periodontol. 2000, 71, 1535–1545.
- Pope, R.M.; Tschopp, J. The role of interleukin-1 and the inflammasome in gout: Implications for therapy. Arthritis Rheum. 2007, 56, 3183–3188.
- Sutterwala, F.S.; Haasken, S.; Cassel, S.L. Mechanism of NLRP3 inflammasome activation. Ann. N. Y. Acad. Sci. 2014, 1319, 82–95.
- Burns, K.; Martinon, F.; Tschopp, J. New insights into the mechanism of IL-1β maturation. Curr. Opin. Immunol. 2003, 15, 26–30.
- Kostura, M.J.; Tocci, M.J.; Limjuco, G.; Chin, J.; Cameron, P.; Hillman, A.G.; Chartrain, N.A.; Schmidt, J.A. Identification of a monocyte specific pre-interleukin 1β convertase activity. Proc. Natl. Acad. Sci. USA 1989, 86, 5227–5231.
- Latz, E.; Xiao, T.S.; Stutz, A. Activation and regulation of the inflammasomes. Nat. Rev. Immunol. 2013, 13, 397–411.
- Dinarello, C.A. Overview of the IL-1 family in innate inflammation and acquired immunity. Immunol. Rev. 2018, 281, 8–27.
- Thomassen, E.; Bird, T.A.; Renshaw, B.R.; Kennedy, M.K.; Sims, J.E. Binding of interleukin-18 to the interleukin-1 receptor homologous receptor IL-1Rrp1 leads to activation of signaling pathways similar to those used by interleukin-1. J. Interferon Cytokine Res. 1998, 18, 1077–1088.
- Okamura, H.; Tsutsui, H.; Komatsu, T.; Yutsudo, M.; Hakura, A.; Tanimoto, T.; Torigoe, K.; Okura, T.; Nukada, Y.; Hattori, K.; et al. Cloning of a new cytokine that induces IFN-γ production by T cells. Nature 1995, 378, 88–91.
- Dai, S.-M. Interleukin (IL) 18 stimulates osteoclast formation through synovial T cells in rheumatoid arthritis: Comparison with IL1β and tumour necrosis factor. Ann. Rheum. Dis. 2004, 63, 1379–1386.
- Casadio, R.; Frigimelica, E.; Bossù, P.; Neumann, D.; Martin, M.U.; Tagliabue, A.; Boraschi, D. Model of interaction of the IL-1 receptor accessory protein IL-1RAcP with the IL-1β/IL-1RIcomplex. FEBS Lett. 2001, 499, 65–68.
- Cayrol, C.; Girard, J.-P. The IL-1-like cytokine IL-33 is inactivated after maturation by caspase-1. Proc. Natl. Acad. Sci. USA 2009, 106, 9021–9026.
- Saleh, H.; Eeles, D.; Hodge, J.M.; Nicholson, G.C.; Gu, R.; Pompolo, S.; Gillespie, M.T.; Quinn, J.M.W. Interleukin-33, a target of parathyroid hormone and oncostatin M, increases osteoblastic matrix mineral deposition and inhibits osteoclast formation in vitro. Endocrinology 2011, 152, 1911–1922.
- Kiyomiya, H.; Ariyoshi, W.; Okinaga, T.; Kaneuji, T.; Mitsugi, S.; Sakurai, T.; Habu, M.; Yoshioka, I.; Tominaga, K.; Nishihara, T. IL-33 inhibits RANKL-induced osteoclast formation through the regulation of Blimp-1 and IRF-8 expression. Biochem. Biophys. Res. Commun. 2015, 460, 320–326.
- Roberts-Harry, E.A.; Clerehugh, V. Subgingival calculus: Where are we now? A comparative review. J. Dent. 2000, 28, 93–102.
- Jin, Y.; Yip, H.-K. Supragingival calculus: Formation and control. Crit. Rev. Oral Biol. Med. 2002, 13, 426–441.
- Abraham, J.; Grenón, M.; Sánchez, H.J.; Pérez, C.; Barrea, R. A case study of elemental and structural composition of DC during several stages of maturation using SRXRF. J. Biomed. Mater. Res. Part A 2005, 75A, 623–628.
- Montenegro Raudales, J.L.; Yoshimura, A.; Sm, Z.; Kaneko, T.; Ozaki, Y.; Ukai, T.; Miyazaki, T.; Latz, E.; Hara, Y. DC stimulates interleukin-1β secretion by activating NLRP3 inflammasome in human and mouse phagocytes. PLoS ONE 2016, 11, e0162865.
- Akcalı, A.; Lang, N.P. DC: The calcified biofilm and its role in disease development. Periodontology 2000 2018, 76, 109–115.
- Fons-Badal, C.; Fons-Font, A.; Labaig-Rueda, C.; Fernanda Solá-Ruiz, M.; Selva-Otaolaurruchi, E.; Agustín-Panadero, R. Analysis of predisposing factors for rapid DC formation. J. Clin. Med. 2020, 9, 858.
- Kitaura, H.; Tatamiya, M.; Nagata, N.; Fujimura, Y.; Eguchi, T.; Yoshida, N.; Nakayama, K. IL-18 induces apoptosis of adherent bone marrow cells in TNF-α mediated osteoclast formation in synergy with IL-12. Immunol. Lett. 2006, 107, 22–31.
- Evans, K.E.; Fox, S.W. Interleukin-10 inhibits osteoclastogenesis by reducing NFATc1 expression and preventing its translocation to the nucleus. BMC Cell Biol. 2007, 8, 4.
