Nanoemulsions, exhibiting droplet sizes of <200 nm, represent liquid-in-liquid dispersions that are kinetically stable. Water and oil are the two incompatible liquids most extensively applied in commercial environments. Because of their small size, characteristics such as visible transparency, high surface area per unit volume, sound stability and tunable rheology are often observed. Additionally, large-scale nanoemulsions’ preparation is easily achievable in industrial conditions. Therefore, nanoemulsions are especially suitable for commercial applications.
1. Introduction
Nanoemulsions, exhibiting droplet sizes of <200 nm, represent liquid-in-liquid dispersions that are kinetically stable. Water and oil are the two incompatible liquids most extensively applied in commercial environments. Because of their small size, characteristics such as visible transparency, high surface area per unit volume, sound stability and tunable rheology are often observed. Additionally, large-scale nanoemulsions’ preparation is easily achievable in industrial conditions. Therefore, nanoemulsions are especially suitable for commercial applications
[1][2][3][1,2,3].
Since the oil and water phases are distributed relatively spatially, simple nanoemulsions can be divided into oil-in-water (O/W) nanoemulsions denoting the dispersion of small oil droplets in an aqueous medium, and water-in-oil (W/O) nanoemulsions signifying small water droplets distributed in an oil medium
[3]. Additionally, utilizing a two-step procedure, it is also possible to produce two types of multiple nanoemulsions, namely water-in-oil-in-water (W/O/W) or oil-in-water-in-oil (O/W/O)
[4]. For instance, the preparation of W/O/W nanoemulsions is achieved by assimilating the oil phase comprising lipophilic surfactant with the water phase to form the initial W
1/O nanoemulsions, which are then homogenized with an additional water phase (W
2) comprising hydrophilic surfactant
[5].
The methods used for nanoemulsions’ preparation can be divided into two principal groups namely low-energy and high-energy techniques. When environmental factors (e.g., composition or temperature) or nanoemulsions’ compositions are modified, small droplets are generated, providing the basis necessary for the successful operation of the low-energy methods
[3][6][7][8][3,6,7,8]. High-energy methods usually consume significant energy (~10
8–10
10 W/kg) to form small droplets. Furthermore, in the utilization of high-energy methods, the oil and water phases are breached and blended using the powerful cavitational, shear and turbulent flow profiles created by the specifically designed devices
[9][10][9,10].
Nanoemulsions are thermo-dynamically unstable since the free energy required to separate the oil phases from the water phases is lower than what is necessary for emulsification. Therefore, nanoemulsions typically break down during storage due to various mechanisms, such as gravitational separation (creaming or sedimentation), flocculation, coalescence and Ostwald ripening
[11]. Moreover, various chemical and biochemical reactions such as flavor loss, biopolymer hydrolysis, color fading and lipid oxidation can adversely affect nanoemulsions, causing them to degrade during storage or lose their acceptable quality characteristics. Among the chemical deterioration phenomena mentioned above, lipid oxidation occurs the most frequently in nanoemulsions
[12].
For several commercial uses, it is crucial that nanoemulsions-based products remain physiochemically stable when exposed to unfavorable environmental conditions (including temperature, mechanical forces, and ionic strength) during their production, storage, transportation and application
[3][6][3,6]. The addition of suitable stabilizers, including emulsifiers, weighting agents, texture modifiers and ripening inhibitors can improve the physical stability of nanoemulsions
[6][13][6,13]. Given that, three methods are commonly used to improve the nanoemulsions’ chemical stability, including the manipulation of interfacial characteristics (e.g., thickness, charge, and chemical reactivity), the addition of chelating agents or antioxidants, as well as controlling environmental elements (e.g., temperature, light, pH, and oxygen levels)
[3][6][3,6].
So far, a number of food ingredients and additives, including bioactive lipids, vitamins, flavorings, acidulants, preservatives, colorings, antioxidants and so on, have been encapsulated by nanoemulsions and some of them are already available in the market
[1][3][14][1,3,14]. A larger droplet surface area, as well as a decline in particle size of the nanoemulsions may lead to increased functionality of the bioactive compounds contained within them. The majority of the bioactive compounds are characteristically lipophilic. Thus, O/W nanoemulsions are commonly used to improve the solubility and dispersibility of lipophilic substance in aqueous media, enhance stability, appearance, taste or texture, increase uptake absorption and bioavailability, and reduce the off-flavor (such as bitterness or astringency)
[14][15][16][14,15,16].
2. Preparation
A number of methods were developed to facilitate nanoemulsions, which include high-energy as well as low-energy techniques
[17]. Selecting an appropriate method for the preparation of nanoemulsions rely on the characteristics of the compounds needing homogenization (specifically the surfactant and oil phases), as well as the required physicochemical attributes and operational qualities of the ultimate product (including rheological, optical, release, and stability properties)
[6]. Understanding the various fabrication methods is crucial for relevant personnel to choose the most suitable preparation technique and fabricate nanoemulsions for special application.
2.1. Low-Energy Methods
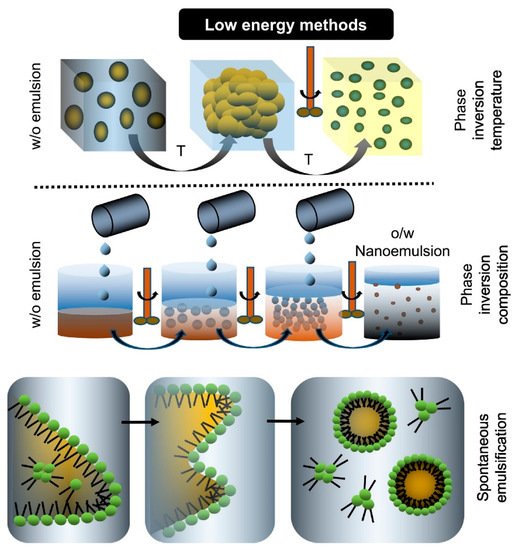
Low-energy methods are denoted by changes in environmental conditions, as well as the composition of the mixture influencing the development of oil nanodroplets within the mixed systems containing surfactants, oil, and water. The most frequently used low-energy techniques are spontaneous emulsion (SE), emulsion phase inversion (EPI) (including phase inversion composition (PIC), and phase inversion temperature (PIT))
[8][18][8,18]. The principles of the characteristic low-energy techniques used to O/W nanoemulsions were shown in
Figure 1.
Figure 1. Schematic depiction of the characteristic low-energy techniques used to create O/W nanoemulsions, including phase inversion temperature (PIT), phase inversion composition (PIC) and spontaneous emulsion (SE)
[19].
The low-energy methods for nanoemulsions’ preparation and their application in encapsulation of bioactive compounds are summarized in
Table 1.
Table 1. Examples of application of low-energy methods for nanoemulsions’ preparation.
|
| Emulsification Method |
|
| Optimal Processing Conditions |
|
| Bioactive Compound Encapsulated |
|
| Droplet Diameter (nm) |
|
| Reference |
|
Figure 2. Schematic portrayal of high-energy techniques utilized for the preparation of O/W nanoemulsions. (A) traditional high-speed mixers are usually employed to form a coarse O/W emulsions before emulsification by (B) high-pressure homogenization (HPH), (C) ultrasonic homogenization (USH), (D) high-pressure microfluidic homogenization (HPMH)
[10].
The high-energy methods for nanoemulsions’ preparation and their application in encapsulation of bioactive compounds are summarized in
Table 2.
Table 2. Examples of application of high-energy methods for nanoemulsions’ preparation.
|
| Emulsification Method |
|
| Optimal Processing Conditions |
|
| Bioactive Compound Encapsulated |
|
| Droplet Diameter (nm) |
|
| Reference |
|
|
| SE |
|
| (1) titration of organic phase into aqueous phase, (2) constant stirring, 600 rpm, (3) room temperature |
|
| Peppermint |
| essential |
|
|
| RSE |
|
| 24000 rpm/25 min |
| oil |
|
| docosahexaenoic acid |
| ≈50 |
|
| 87 |
[20]
|
|
| [ | 35 |
| (1) titration of organic phase into the aqueous phase, (2) constant stirring, 1000 rpm/10 min, (3) room temperature |
|
HPH |
| Citrus oil |
|
|
| 800 bar/8 cycles |
| 10–30 |
|
[21]
|
] |
|
| [ | 46] |
| docosahexaenoic acid |
|
| 11.17 |
|
[35]
|
[46]
|
| (1) titration of organic phase into aqueous phase, (2) constant stirring, 750 rpm, (3) room temperature |
|
| Citrus oil |
|
| ≈100 |
|
[22]
|
| |
| 103 M Pa/10 cycles |
|
| pepper extract |
|
| 132 ± 2.0-145 ± 1.0 |
|
[36]
|
[47]
|
| (1) titration of organic phase into aqueous phase, (2) constant stirring, 600 rpm/15 min, (3) room temperature |
|
| Cinnamaldehyde |
|
| <100 |
|
| |
| 60 MPa/3 cycles |
|
| curcumin |
[23]
|
|
| 203.6-260.6 |
|
| [37]
|
[48]
|
| (1) stirred, 1000 rpm/1 h, (2) room temperature |
|
| Capsaicin |
|
| 13–14 |
|
|
| |
| 40 kpsi/10 cycles |
|
| fish oil[24] |
|
|
|
| 89.7 ± 27.7 |
|
[38]
|
[49]
|
| (1) deprotonated eugenol in hot alkaline added to surfactant mixtures, (2) the mixtures were acidified to pH 7.0, stirred, 600 rpm |
|
| Eugenol |
|
|
| HPMH |
|
| 137.9 MPa/10 cycles |
|
| ≈ 109–139 |
|
| rosemary essential oil |
|
[25]
|
|
| 2.88 |
|
| [39]
|
[50]
|
| PIC |
|
| (1) mixed oil and surfactant, (2) oil phase added to aqueous phase, (3) phase inversion occurred at a certain oil-to water ratio, (4) stirred, 30 min |
|
|
| 1000 bar/5 cycles |
|
| docosahexaenoic acid |
| Docosahexaenoic acid |
| Eicosapentaenoic acid |
|
| 148 |
| <200 |
|
[40]
|
[51
[26]
|
] |
|
| (1) aqueous phase (water, glycerol) added to organic phase (sunflower oil, polysorbate 80, curcumin), (2) stirred, 300 rpm/30 min |
|
| Curcumin |
|
| ≈200 |
|
[27][28]
|
[27,28]
|
|
| (1) mixed organic phase and aqueous phase, (2) continuing stirred, (3) ambient temperature |
|
| Essential |
| Oils |
| Blend* |
|
| 29.55–37.12 |
|
[29]
|
|
| PIT |
|
| (1) all components were stirred, 30 min, (2) heated to 15 °C above the PIT, (3) the temperature was reduced to the PIT |
|
| Cinnamon oil |
|
| 101 |
|
[30][31]
|
[30,31]
|
|
| (1) coarse emulsions were heated, 21–98 °C /0–3 h, (2) immediately quenching in ice/water with hand shaking |
|
| Lemon oil |
|
| ≈100 |
|
[32]
|
|
| (1) mixing all components, (2) 3 temperature cycles (90–60–90–60–90–75 °C) |
|
| Curcuminoids |
|
| 20–100 |
|
[33]
|
Note: essential oils blend* containing cape jasmine absolute, wan saw long oil, lemongrass oil and basil oil.
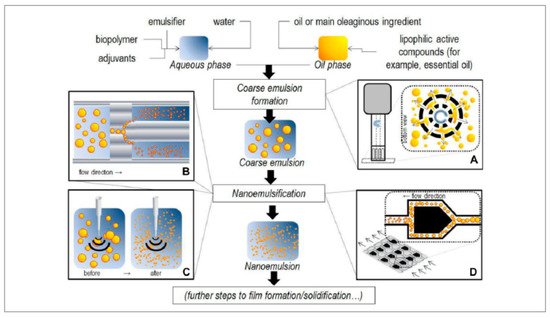
2.2. High-Energy Methods
High-energy methods, denote mechanical techniques, employing mechanical equipment to separate the dispersed phase into droplets inside the continuous phase to generate forces that are highly disruptive
[10]. Since high-energy methods permit the utilization of non-toxic/natural emulsifiers at lower concentration levels, they are more appropriate for food-related nanoemulsions preparation, while they are also expedient for production at an industrial scale and the necessary equipment is available commercially
[14]. Usually, two steps are involved when producing O/W nanoemulsions using high-energy methods. Firstly, the coarse O/W emulsions were formed by mixing of the components using a high-speed mixer or stirrer. Later on, the coarse emulsions are exposed to disruptive forces to facilitate a reduction in the droplet diameter to 200~500 nm
[10]. Based on the devices used, high-energy methods include rotor-stator emulsification (RSE), high-pressure homogenization (HPH), high-pressure microfluidic homogenization (HPMH) and ultrasonic homogenization (USH)
[9][34][9,45]. The principles of high-energy techniques used to create O/W nanoemulsions were shown in
Figure 2.

|
|
|
|
|
|
350 bar/5 cycles |
|
|
|
| curcumin |
|
| 275.5 |
|
[41]
|
[52]
|
|
| 13 kpsi/1 cycle |
|
| fish oil |
|
| <160 |
|
[42]
|
[53]
|
|
| USH |
|
| 350 W/5 min |
|
| Resveratrol |
|
| 20.41 ± 3.41 |
|
[43]
|
[54]
|
|
| resveratrol cyclodextrin inclusion complex |
|
| 24.48 ± 5.70 |
|
| |
| 20.5 kHz/400 W for 15 min |
|
| thymus daenensis oil |
|
| 171.88 ± 1.57 |
|
[44]
|
[55]
|
|
| Combined method |
|
| HPH (24,000 rpm/15 min) + HSP (800 bar/8 cycles) |
|
| docosahexaenoic acid |
|
| 11.31 |
|
[35]
|
[46]
|
3. Stability
3.1. Physical Stability
The undesirable molecular interactions at the oil-water interface as a result of the hydrophobic effect induces thermodynamic instability in nanoemulsions
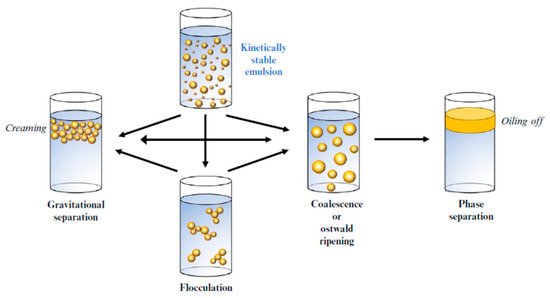
[45][59]. Nanoemulsions will eventually degrade as a result of several mechanisms such as flocculation, gravitational separation, coalescence, phase separation, and Ostwald ripening, as shown in
Figure 3 [3][6][3,6]. Understanding the essential mechanisms responsible for nanoemulsions’ instability is crucial in developing systems exhibiting adequate stability qualities.
Figure 3. Schematic representation of the mechanisms responsible for nanoemulsion physical instability (phase separation): gravitational separation, flocculation, coalescence and Ostwald ripening.
3.1.1. Gravitational Separation
The process in which nanoemulsion droplets move downward (sedimentation) or upward (creaming) due to their density exceeding or being lower than that of the liquid surrounding it, is known as gravitational separation. Water tends to move in a downward direction, while oil migrates upward since most liquid oils are less dense than water in a liquid state. Therefore, sedimentation is common in W/O nanoemulsions, while O/W exhibit more cases of creaming
[3][6][3,6]. An increase in droplet size, density contrast in conjunction with a decline in the aqueous phase viscosity, influence the motion speed of the droplets induced by gravitational separation
[6].
According to Arancibia et al., the inferior stability of nanoemulsions containing 15% avocado oil and 8% starch could be ascribed to gravitational separation of phases caused by fat droplet flocculation/coalescence and starch precipitation/aggregation
[46][64]. According to Chen et al., due to gravitational separation (sedimentation), pure cinnamaldehyde could not produce stable nano-emulsions. The oil droplets moved downwards in the pure cinnamaldehyde system due to the higher density of cinnamaldehyde (1050 kg m
-3) than that of room temperature water (997 kg m
-3)
[47][65].
3.1.2. Flocculation and Coalescence
The colloidal interaction between the droplets determines two types of droplet accumulation namely coalescence and flocculation
[6].
The process by which two or more droplets attract each other to form clusters is known as flocculation
[6]. As reported by Bai et al., utilizing amphiphilic polysaccharides above a certain level as emulsifiers could promote emulsion instability, which was attributed to exhausted flocculation mechanism, could diminish the stability of some nanoemulsion products in the long run
[48][66]. As reported by Li et al., D-limonene nanoemulsions became unstable and tended to flocculate and coalesce, which caused a variation in zeta-potential at the storage temperatures
[49][67]. As reported by Bai et al., a cream layer was evident for saponin-coated droplets when salt concentrations exceeded 300 mM, suggesting that the accumulation of droplets primarily resulted from flocculation instead of coalescence
[50][68].
The process by which a larger droplet is formed when several droplets collide and amalgamate is known as coalescence
[6]. As reported by Bai et al., rhamnolipids were able to stabilize O/W nanoemulsions. The droplets coated with rhamnolipids displayed stability following thermal treatments between 30 °C to 90 °C, with salt concentrations below 100 mM NaCl, pH levels between 5 and 9, as well as storage for at least two weeks at room temperature. However, nanoemulsions were unstable when subjected to storage conditions that were extremely acidic (pH 2–4) or in the presence of high ionic strength (200–500 mM NaCl). These results were ascribed to coalescence caused by a decline in the electrostatic repulsion among the droplets in the range and magnitude at high salt and low pH levels
[50][68]. Wooster et al. prepared triglyceride nanoemulsions by the HPMH method, using a combination of Span 80 and Tween 80 as emulsifiers and n-alcohol as a co-solvent. However, addition of an excess of n-alcohol led to nanoemulsion destabilization, coalescence was found to be the primary destabilization mechanism
[51][69]. According to Shu et al., nanoemulsions containing astaxanthin stabilized by saponins displayed an extremely high sensitivity and were predisposed to droplet coalescence when high salt concentrations were present. These results could be ascribed to a decline in the electrostatic repulsion among the negatively charged droplets facilitated by the presence of Na
+ cations, and inducing the coalescence and instability of the droplets
[52][70].
3.1.3. Ostwald Ripening
Driven by the curvature differences of the particles, dispersed phase molecules diffuse through the continuous phase causing the expansion of larger droplets and the shrinkage of smaller droplets in a process known as Ostwald ripening, which is the primary instability mechanism for such nanoemulsions. Ostwald ripening occurs when the dispersed phase solubility in large droplets (small curvature) is lower than in small droplets (large curvature), which prompts droplet growth due to the appearance of a concentration gradient
[53][71].
According to Ryu et al., nanoemulsions containing essential oil are especially susceptible to Ostwald ripening because of a significant oil phase solubility in the aqueous phase even though its predominantly hydrophobic nature
[54][72]. According to Walker et al., during or soon after homogenization, nanoemulsions containing thyme oil experienced swift droplet growth when low levels of fish oil (<75%) were present. The reason was that the relatively high water-solubility of thyme oil induced expeditious droplet growth as a result of Ostwald ripening
[55][73].
3.2. Chemical Stability
Various biochemical and chemical reactions such as flavor loss, lipid oxidation, biopolymer hydrolysis and color fading occur in nanoemulsions leading them to lose their favorable characteristics. Of these, lipid oxidation is considered as one of the most significant types of chemical degradation
[12]. The interfacial areas of nanoemulsions are relatively large, leading to accelerated lipid oxidation due to water-soluble pro-oxidants (e.g., transition metals) coming into contact with oil-soluble reactants (e.g., polyunsaturated lipids and hydroperoxides)
[56][74]. Park et al. suggested that the long-term stability of nanoemulsions might exhibit a more significant association with the chemical stability instead of the physical stability in the case of O/W nanoemulsions with retinol
[57][75].
3.3. Correlative Instability Mechanism
Each instability mechanism is always associated with others or they appear simultaneously
[8]. Powell et al. prepared nanoemulsions using Pluronic F68 and various oils generally utilized for pharmaceutical and cosmetic applications, and explored their stability mechanisms. The eventual destabilization appeared due to the rising of large drops which formed through coalescence and Ostwald ripening and coalescence were responsible for the formation of large drops, which rose to cause the ultimate destabilization
[58][76]. According to Chen et al., for pure cinnamaldehyde, a transparent layer was evident at the top of the nanoemulsion samples, showing that storage prompted the oil droplets to sink to the bottom of the test tube, and might be attributed to both the physical and chemical effects, such as chemical interaction, coalescence, Ostwald ripening, and sedimentation. In detail, cinnamaldehyde displays relatively high water-solubility, inducing droplet expansion via Ostwald ripening. Consequently, the mean particle size of the nanoemulsions expands, resulting in accelerated droplet coalescence and sedimentation
[47][65].
4. Nanoemulsion Stabilizer
In order to satisfy the specific requirements of commercial applications, nanoemulsions should be designed to improve their kinetic stability, which is achieved through meticulous structure and composition control. Particularly, it is critical to select adequate aqueous and oil phases, as well as the most suitable additives, such as emulsifier, weighting agent, texture modifier, and ripening inhibitor
[6]. The stabilization mechanism usually refers to the physicochemical properties of nanoemulsion such as composition, interfacial composition, electric charge, droplet size, physical state, aggregation state, rheology property and so on
[3][6][3,6].
For instance, apart from decreasing the droplet size, gravitational separation can also be reduced by adding thickeners to improve the viscosity of the aqueous phase, or adding weighting agents to decrease the density contrast. Droplet aggregation due to flocculation and coalescence can be restricted by making sure that the repellent interactions (e.g., steric and electrostatic) of droplets exceed their attractive interactions (e.g., hydrophobic, van der Waals, and depletion). This is often accomplished by changing the aqueous phase composition or the nature of the emulsifier used
[6]. The addition of ripening inhibitors or the utilization of an oil phase displaying low water-solubility, can restrict Ostwald ripening
[6][59][6,77].
5. Applications in Encapsulation of Bioactive Compounds
The use of many bioactive compounds, such as bioactive lipids, essential oils, flavor compounds, vitamins, polyphenols, carotenoids and so on in the food industry remain challenging due to their inadequate solubility in water as well as their stability in food preparations
[14][60][14,162]. Generally, nanoemulsions are usually designed to retain bioactive compounds during storage within a food product but control their release when they encounter specific environmental conditions, such as the mouth for flavors or the gastrointestinal tract for pharmaceuticals or nutraceuticals
[61][163]. Additionally, the delivery and slow release of hydrophobic bioactive compounds in O/W conventional emulsions or nanoemulsions are beneficial to enhance their bioaccessibility by improving their solubility and the incorporation of them into mixed micelles of the simulated gastrointestinal tract (GIT) system
[62][63][164,165].
5.1. Bioactive Lipids
Essential polyunsaturated fatty acids (PUFAs), especially omega-3 oils, e.g., eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), α-linoleic acid and α-linolenic acid, are the main bioactive lipids
[64][65][166,167]. PUFAs reportedly have substantial health benefits, such as the neuroplasticity of nerve membranes, nervous system activity, memory-related learning, cognitive development, synaptic transmission, and synaptogenesis
[66][168]. However, bioactive lipids are highly unstable against oxidation and presented low odor thresholds. Therefore, exceedingly low concentrations affected the sensory parameters. Encapsulation of bioactive lipids in nanoemulsions is beneficial in reducing autoxidation, displaying compatibility with various food products, enhancing functional properties, solubilizing flavor or volatile compounds in lipids, while masking bitter or astringent tastes
[67][169].
According to Zhang et al., stable DHA and EPA nanoemulsions were prepared by EPI method. Within 20 days, the best nanoemulsions have good physical stability under different storage conditions, and the retention rate of DHA/EPA can be stabilized at >60%
[26]. Dey et al., suggested that nanoemulsions demonstrated a considerably higher uptake rate of PUFAs in three small intestinal regions, compared to conventional emulsions. Meanwhile, nanoemulsions showed a stronger resistance to lipopolysaccharide-induced nitric oxide production in the peripheral blood mononuclear cells of rats
[38][49]. As reported by Karthik et al., DHA O/W nanoemulsions were fabricated by microfluidization using Tween 40, SC or SL as emulsifiers. Among them, Tween 40 stabilized nanoemulsions showed improved stability and extent of lipid digestibility
[40][51].
5.2. Essential Oils and Flavor Compounds
Essential oils, in the form of aromatic volatile liquids, as well as semi-liquids, are commonly derived from the seeds, flowers, leaves, buds, fruits, bark, resins, and roots of plants. The antimicrobial activity provided by essential oils is beneficial against several types of fungi, Gram-positive bacteria, and Gram-negative bacteria
[68][170]. The nature of essential oils, including hydrophobic, volatile and reactive, limits the incorporation of them into food matrices directly
[69][171]. The significant application challenges regarding the incorporation of essential oils into food could be negated by their encapsulation in nanoemulsions. Essential oils loaded within nanoemulsions can be formed by two methods.
One method is homogenizing essential oils in the aqueous solutions containing emlsifier. According to Xue et al. and Ma et al., nanoemulsions containing essential oils (e.g., thyme oil, eugenol) were fabricated by directly homogenizing the essential oils in the aqueous solutions containing CS and lecithin, indicating that the antimicrobial characteristics were similar or exceeded those of the free essential oils
[70][71][172,173]. As reported by Wang et al., eugenol (2-methoxy-4-(2-propenyl)-phenol), which forms a major part of clove essential oil, presents a wide range of antibacterial and antifungal activity. Eugenol-loaded nanoemulsions stabilized by the mixture of zein and SC were prepared by SE method. The results showed that the entrapment efficiency of eugenol was 84.24% when 1% (
v/
v) of eugenol and 2% (
m/
v) of SC/zein (1:1 mass ratio) were used. The obtained nanoemulsions demonstrated the most restricted size distribution, and retained remarkable stability throughout a storage period at ambient temperatures (22 °C, 30 days), while exhibiting exceptional redispersibility following freeze-drying or spray-drying
[25]. According to Sharma et al., a synergistic O/W nanoemulsion containing clove and lemongrass oil was developed and its potential as antifungal agents was explored. The antifungal activity of clove and lemongrass oil was enhanced by formulation in nanoemulsions
[72][174]. According to Moghimi et al., stable nanoemulsion of
Thymus daenensis oil was produced by utilizing lecithin and Tween 80 as emulsifiers, with droplet size of 171.88 ± 1.57 nm. The nanoemulsions showed stronger antibacterial activity than pure oil against
Acinetobacter baumannii, which is notoriously resistant to multiple drugs. This result is evidenced by the minimum inhibitory concentration of nanoemulsion and pure oil being 30–45 μg/mL and 62.5–87.5 μg/mL, respectively. Additionally, after incubation for 24 h, the nanoemulsions exhibited remarkable anti-biofilm activity at a sub-lethal dose (56.43% inhibition in 1/2 minimum inhibitory concentration)
[44][55].
Another method is to pre-dissolve essential oils in commonly used oils and then emulsified in the aqueous phase. As reported by Yang et al., nanoemulsions containing the mixtures of citrus oils (e.g., bergamot oil and sweet orange oil) and common triacylglycerol oils (e.g., corn oil and MCT oil) with different mixing ratios were produced. The subsequent results suggested that the stability of nanoemulsions that contain mixed oil significantly exceeded that of nanoemulsions containing pure citrus oil
[73][175]. As reported by Tian et al., it was difficult using pure cinnamaldehyde as the oil phase to form stable nanoemulsions. Stable nanoemulsions containing cinnamaldehyde were obtained with the addition of MCT. Additionally, nanoemulsions containing cinnamaldehyde and MCT could provide an enhanced long-term inhibition on the bacterial growth of
Escherichia coli compared with pure cinnamaldehyde
[23].
Recently, essential oil nanoemulsion-containing films have been produced and used for active food packaging. However, this aspect has been extensively described elsewhere
[10] and is not repeated in the present review.
5.3. Vitamins
As essential micronutrients, vitamins form a crucial part of human health. There are two types of vitamins: fat-soluble (lipophilic) and water-soluble (hydrophilic). Vitamins A, E, D, and K are grouped as the lipophilic vitamins, while vitamins B and C are hydrophilic.
Lipophilic vitamins are biologically sensitive material, displaying marginal chemical stability and water solubility
[74][176]. For instance, Vitamins A, and Vitamins E are easily oxidized, particularly when exposed to light, heat, light, and metal ions. Furthermore, visible and fluorescent light possessed the ability to alter the vitamin K structure dramatically. Vitamin D is the only element that was not significantly affected by the processing and storage environments
[75][177]. Nanoemulsions are usually fabricated to improve their chemical stability, solubility and oral bioavailability.
Park et al. examined the stability of O/W nanoemulsions containing retinol (vitamin A) when exposed to ultraviolet (UV) light and subjected to a storage period at varying temperatures (4 °C, 25 °C, and 40 °C). The results suggested that UV light reduced the residual retinol in the emulsion systems that utilized low oil concentrations during preparation compared to bulk oil. However, oil concentrations exceeding 10 wt% caused residual retinol levels to be higher than in the bulk oil due to elevated emulsion turbidity
[57][75]. According to Ji et al., the vitamin A palmitate retention ration in the nanoemulsion exceeded 93% following a storage period of three months at room temperature
[76][108]. As reported by Kadappan et al., the nanoemulsions-based delivery system increased in vitro bioaccessibility of vitamin D
3 by 3.94 folds, as evidenced by the significantly higher concentration of vitamin D
3 in micelles. An animal study showed that the nanoemulsions significantly increased the serum 25(OH)D
3 by 73%
[77][178]. According to Schoener et al., the type of carrier oil (corn oil, fish oil and flaxseed oil) significantly affected the preparation, simulated gastrointestinal performance and stability of vitamins D
3 nanoemulsions stabilized by pea protein. The results showed that the three lipids were all digested in the small intestinal simulation model within the first few minutes. Additionally, for the different carrier oils, both the digestion rate in simulated gastrointestinal and vitamin bioaccessibility declined in the following order: corn oil > fish oil ≈ flaxseed oil
[78][104]. As reported by Lv et al., nanoemulsions containing vitamin E were fabricated by dual-channel microfluidizer, using corn oil as a carrier oil and using QS as an emulsifier. The optimized nanoemulsions resulted in a relatively high vitamin bioaccessibility (53.9%)
[79][179]. According to Moradi et al., the cellular uptake of a-tocopherol in nanoemulsions displayed a rise of up to 12 times higher than microsized a-tocopherol
[80][112]. As reported by Campani et al., nanoemulsions containing vitamin K
1 have been prepared to overcome some difficulties associated to the incorporation of semi-solid vitamin K
1 into food formulations. The results showed that nanoemulsions could offer an option for the commercial development of a liquid and aqueous formulation to deliver vitamin K
1 [81][180].
5.4. Phenolic Compounds
Phenolic compounds displaying significant antioxidant properties can be employed in biological preparations and various food products such as anti-microbial, anti-atherogenic, anti-inflammatory, anti-thrombotic, and anti-allergenic agents
[82][181]. Phenolic compounds are classified into lipophilic and hydrophilic compounds.
Lipophilic phenolic compounds were usually encapsulated by O/W nanoemulsions. O/W nano-emulsification, can reportedly improve the bioavailability of lipophilic phenolic compounds due to higher absorption, solubility, and permeation into the body, as well as the safeguarding of the lipophilic phenolic compounds in nanoemulsions within food preparations
[67][169]. According to Kumar et al., curcumin nanoemulsions with sodium caseinate were prepared. The cellular uptake of curcumin was improved by nanoemulsification because that the slow release of curcumin in the intestine is beneficial to incorporate it into mixed micelles of the bile salts or phospholipids
[83][101]. Zheng et al. prepared curcumin nanoemulsions by three different methods (e.g., pH-driven, conventional, and heat-driven) and compared them with three curcumin supplements that are currently widely available. The results showed that the bioaccessibility of all curcumin obtained nanoemulsions compared well to even the most superior commercial formulation. Additionally, the nanoemulsions produced using the pH-driven technique denoted the highest concentrations of curcumin in the mixed micelles phase following exposure to a simulation of a gastrointestinal tract
[84][182]. Sugasini et al. prepared a phospholipid-stabilized nanoemulsion containing curcumin and carrier oil (sunflower oil, coconut oil, or linseed oil) and explored the possibility of nanoemulsions to enhancing the curcumin bioavailability and DHA levels in rats. The results indicated the presence of high DHA levels in tissue and serum lipids, as well as elevated curcumin levels in the serum, heart, liver, and brain of rats given feed nanoemulsions containing linseed oil and curcumin
[85][183]. According to Silva et al., compared with WPI-nanoemulsions, nanoemulsions stabilized by WPI-chitosan mixture showed the improved apparent permeability coefficient of curcumin via Caco-2 cells, as well as the improved bioaccessibility and antioxidant ability
[86][124]. According to Singh et al., the rate and extent of bioavailability of t-resveratrol was significantly enhanced by loading in nanoemulsions rather than that of free t-resveratrol. Alongside this, the results of an in situ single pass intestinal perfusion study showed a remarkable enhancement in the absorptivity and permeability parameters of nanoemulsions
[87][184]. Son et al. prepared quercetin-loaded O/W nanoemulsions containing Tween 80, caprylic/capric triglyceride (Captex
® 355), soy lecithin, and sodium alginate using the SE method. The nanoemulsion polydispersity index and particle size were <0.47 and 207–289 nm, respectively. The nanoemulsions were stable at pH levels ranging from 6.5–9.0 during a storage period of three months at 21 °C and 37 °C. Additionally, in rats that received a diet high in cholesterol, the nanoemulsion containing quercetin displayed a more substantial efficacy in decreasing the level of serum and hepatic cholesterol, with higher release of bile acid into feces, compared to free quercetin
[88][185]. As reported by Carli et al., nanoemulsion-encapsulated quercetin was created with the EIP method and using two separate surfactants, namely Brij 30, and Tween 80. Nanoemulsions were obtained with mean particle size of 180–200 nm. The retention of quercetin was around 70% in nanoemulsions that contained 0.30% quercetin (w/w) and were stored for 90 d. Additionally, the incorporation of quercetin-loaded nanoemulsions in chicken patés can improve their oxidative stability in a considerably more efficient manner than synthetic antioxidants. Sensory information suggested that the quercetin encapsulation in nanoemulsions enhances consumer acceptability of the products
[89][186].
Hydrophilic phenolics or the mixture of hydrophilic and lipophilic phenolics were usually encapsulated by W/O nanoemulsions. According to Rabelo et al., stable W/O nanoemulsions containing açaí berry extracts (ABE, rich in anthocyanins) were successfully formulated. All W/O nanoemulsions containing different concentrations of ABE exhibited high antioxidant activity and retention rates of anthocyanins after 30 days of storage. When 2% of anthocyanins was encapsulated in a 30 wt% ϕd (weight fraction of the dispersed phase) W/O nanoemulsions, they had an estimated half-life of 385 days
[90][187]. Moreover, hydrophilic phenolics can also be encapsulated by O/W nanoemulsions. As reported by Peng et al., The O/W tea polyphenols (TP) nanoemulsion were prepared with polysorbate 80 and corn oil using the HPH method. The TP nanoemulsions with particle sizes of 99.42 ± 1.25 nm were stable during a 20-day storage period at 4 °C, 25 °C, or 40 °C. The results of in vitro assay of the simulated digestion model displayed a higher degree of bioaccessibility with regard to (−)-epigallocatechin gallate (EGCG), while (−)-epicatechin (EC), (−)-epigallocatechin (EGC), and (−)-gallocatechin gallate (GCG) exhibited lower bioaccessibility in the nanoemulsions compared to the aqueous solutions
[91][188].
5.5. Carotenoids
Carotenoids represent natural lipophilic pigments that provide various health advantages such as safeguarding the eyes and reducing certain cancers. Increasing carotenoid bioavailability can be achieved when they are ingested with dietary lipids since the micelles derived from digested products are beneficial to solubilization and transportation of carotenoids to the epithelial cells
[67][92][93][169,189,190]. Encapsulation of hydrophobic carotenoids into O/W nnaoemulsions could protect them from external stress factors. Additionally, the bioavailability of carotenoids can be increased after nano-emulsification.
As reported by Fan et al., O/W nanoemulsions containing β-carotene (BC) were prepared using WPI and WPI-dextran as emulsifiers. Following a 30-day storage period at 25 °C and 50 °C, the highest BC retention rate was evident in nanoemulsions that were stabilized with WPI-DT (5 kDa) conjugate due to the relatively high scavenging ability of diphenyl-1-picryl-hydrazil (DPPH). Additionally, the encapsulation in nanoemulsions stabilized by WPI-dextran (70 kDa) significantly impeded the lipolysis and release of BC
[94][126]. According to Yi et al., BC retention of lactalbumin-catechin conjugate-stabilized nanoemulsions was significantly greater than that of lactalbumin-stabilized ones, which was attributed to the increased radical-scavenging and binding ability with free metal ion of lactalbumin after grafting with catechin
[95][131]. Meng et al. prepared nanoemulsions containing TP and BC and found that the addition of TP was effective in enhancing the oral bioavailability and storage stability of BC. During storage at varying temperatures of 4 °C, 25 °C, and 35 °C, the stability and the BC retention of nanoemulsions containing TP and BC was higher than those of nanoemulsions containing only BC. Additionally, as shown by the in vitro simulated digestion assay and the in vivo absorption study, comparing with nanoemulsions containing only BC, the nanoemulsions containing TP and BC exhibited the higher recovery rates of BC at digestion phases I and II and the higher conversion efficiency of BC to vitamin A
[96][191]. As reported by Sotomayor-Gerding et al., carotenoid (astaxanthin or lycopene) nanoemulsions were obtained by the HPH method. Nanoemulsions were stable to environmental conditions and storage time. The nanoemulsion oxidative stability was improved by trolox and the stability of lycopene nanoemulsions was improved by the synergistic effect of trolox and butylated hydroxytoluene (BHT). Additionally, carotenoid nanoemulsions were partially (66%) digested and highly bioaccessible (70–93%)
[97][192]. As reported by Liu et al., the bioaccessibility of astaxanthin in nanoemulsions containing different carrier oils (olive oil, flaxseed oil and corn oil) was much higher than that in nanoemulsions containing no lipid, due to that the hydrophobic carotenoids could be solubilized by the mixed micelles formed from the carrier oils. The final free fatty acid release, as well as the bioaccessibility of astaxanthin exhibited a decrease in the following order: olive oil > flaxseed oil > corn oil
[98][193]. As reported by Shen et al., the nanoemulsions stabilized with WPI had the highest cellular uptake of astaxanthin, followed, in order, by PWP, WPI–lecithin mixture, PWP–lecithin mixture (5.05 ± 0.1%), lecithin, and Tween 20
[99][152].