Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Vivi Li and Version 1 by Tjalf Ziemssen.
As more treatment options emerge that have a significant impact on the peripheral immune system, the evaluation of lymphocyte count, and that of specific lymphocyte subsets, become more important in the treatment selection and management of patients with multiple sclerosis (MS).
- multiple sclerosis
- lymphocyte counts
- mechanism of action
- adverse event
1. Introduction
As more treatment options emerge that have a significant impact on the peripheral immune system, the evaluation of lymphocyte count, and that of specific lymphocyte subsets, become more important in the treatment selection and management of patients with multiple sclerosis (MS) [1,2][1][2]. A greater understanding of the underlying pathophysiological mechanisms of MS has led to the development of therapeutics that address the cell count, migration, or functional state of lymphocytes. Though helpful in combatting the disease, changes in lymphocyte physiology can also be treatment-limiting. In addition, the measurement of peripheral lymphocyte counts appears to be important for treatment sequencing and planning of wash-out periods [3]. Pharmacological effects on lymphocytes in the peripheral blood can serve as markers of patient compliance and can also assist in understanding the mechanism of action of MS therapies [4,5][4][5].
Peripheral blood lymphocytes are frequently monitored in clinical practice as blood is easily accessible [6]: lymphocytes continuously enter and exit the lymphoid and non-lymphoid organs via the blood [7]. The assessment of lymphocyte subsets in the blood may provide useful information on immune system status [8]. The measurement of physiological parameters of lymphocyte subsets has been used for some time to assist the selection of treatment regimens in specific diseases, e.g., human immunodeficiency virus (HIV) infection [9]. However, blood lymphocytes can also be influenced by many conditions other than a disease or its treatment, including stress, smoking, sports, and aging [8]. The extent of variation caused by these different factors can easily obscure alterations that have diagnostic value in pathogenic conditions.
2. Disease-Modifying Drugs and Their Effects on Lymphocyte Count
2.1. Mechanism of Action: Immunomodulation
2.1.1. Glatiramer Acetate
General Facts and Clinical Trial Data
Glatiramer acetate (GA) was the first DMT for MS successfully evaluated in humans (1977) and was approved by the US Food and Drug Administration in December 1996 and by the European Medicines Agency in 2001 for daily (20 mg/day) or triweekly (40 mg) subcutaneous application in patients with MS. Initially developed as a chemical and immunological analog of the major myelin antigen (myelin basic protein, MBP) to induce experimental autoimmune encephalopathy (EAE), GA did not work as intended. Instead of promoting encephalitic changes, GA was revealed as an efficient suppressor of encephalitic modulation. This effect and could even prevent EAE, which should normally be induced by myelin antigens such as GA [34][10]. Across five randomized controlled clinical trials, GA 20 mg has consistently demonstrated efficacy in reducing the annualized relapse rate (ARR 29%) and MRI disease activity (33% reduction in the total number of enhancing lesions) and slowing of disability progression in patients with relapsing-remitting MS (RRMS) [35,36][11][12]. Due to its favorable and well-characterized safety profile, GA is still often prescribed in patients with mild or moderate forms of MS.
Mechanism of Action and Impact on Lymphocyte Count
GA cross-reacts with MBP in a humoral and cellular respect and serves as an altered peptide ligand that promotes regulatory T cells instead of stimulating autoimmune T cell reactivity [34,37][10][13]. The immunological effect underlies a strong and effective binding of MHCII molecules on antigen-presenting cells (APC). They compete with MBP and other myelin proteins for binding sites [38,39][14][15]. This binding effectively replaces MBP, proteolipid protein (PLP), and MOG-derived peptides on their MHCII binding sites. This results in an altered T cell response, leading to suppression of myelin reactive T cells [39,40][15][16] and the emergence of regulatory Th2 cells, which are able to recognize GA as well as MBP to cross the blood-brain barrier and secrete anti-inflammatory cytokines [41,42][17][18]. These GA-specific Th2 cells additionally secrete high amounts of brain-derived neurotrophic factor (BDNF), which promotes neuroprotective effects [43][19]. Furthermore, GA functionally inactivates T cells by antagonism on the T cell receptor and can induce regulatory CD4+, CD25+ cells by activating the regulatory pathway protein FOXP3 (Figure 31B) [44][20].
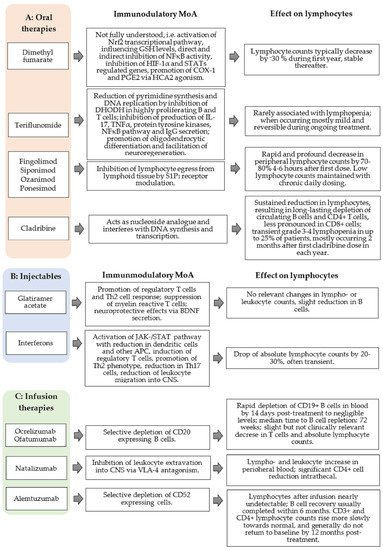

Figure 1. Association of proposed mechanism of action (MoA) of disease-modifying therapies (DMTs) and effects on lymphocytes. Categorized: oral therapies (A), injectables (B), and infusion therapies (C). CNS, central nervous system; COX-1, Cyclooxygenase-1; GSH, DHODH, dihydroorotate dehydrogenase; Glutathione; HCA2, hydroxy-carboxylic acid receptor 2, HIF-1α, hypoxia-inducible factor -1α; IL, interleukin; JAK/STAT, Janus kinases/signal transducer and activator of transcription proteins; nrf2, nuclear factor erythroid-derived 2-like 2; TNF-α, tumor necrosis factor-α; PGE2, prostaglandin E2; S1P, sphingosine-1-phosphate; Th1/2/17, T helper 1/2/17 cells; VLA, very late antigen.
While the total number of T cells in the blood compartment remains stable, studies have shown that GA treatment is associated with a reduction of B cells, plasma blasts, memory B cells, and a shift from pro- to anti-inflammatory B cell phenotypes [45][21]. This may be driven by the cross-reactivity of B cell receptors for GA with antigens that are expressed in MS lesions [45][21]. In contrast with interferon beta, GA is only associated with leukopenia or leukocytosis in exceptional cases [46][22].
Recommended Monitoring
Considering these rare cases of lymphopenia/leukopenia but also leukocytosis and thrombocytopenia, a regular check of blood counts should be done at least tri-monthly in the course of the first year of therapy (Table 21 and Table 32). Subsequently, laboratory intervals can be increased to once or twice per year in the case of normal blood counts. The risk of severe GA-associated infections is low and not clinically meaningfully increased (1–2%) [47][23].
Although there are no convincing study results regarding immune responses following vaccinations, GA treatment is not considered to limit immune responses [1]. Verifying sufficient vaccination response via titer recording should be considered. Patients receiving GA should not be vaccinated with attenuated vaccines.
2.1.2. Interferons
General Facts and Clinical Trial Data
Interferons are a family of cytokines and physiologically function as signaling proteins. Since 1993 (US) and 1995 (EU), respectively, interferon-type beta (IFN-β) has played a role in the disease-modifying treatment of MS. Within the scope of the PRISMS study, subcutaneous (three times a week) application of INF-β-1a showed a risk reduction for relapses of 27% (22 µg, three times a week) and 33% (44 µg, three times a week) in a dose-related manner. Furthermore, it proved an effective treatment for RRMS in terms of defined disability and all MRI outcome measures [48][24]. Today there are various preparations that differ by mode and frequency of administration. In addition to RRMS, interferon is approved for the treatment of clinically isolated syndrome (CIS) and immunomodulation during pregnancy and breastfeeding [49,50][25][26].
Mechanism of Action and Impact on Lymphocyte Count
The effects of interferons are complex and, even today, are not completely understood. Activation of the JAK-/STAT-pathway via binding of the IFNAR-2 receptor is an established mechanism of action that leads to the expression of various genes (e.g., MX protein, beta2-microglobulin, 2′/5′-oligoadenylate synthetase, neopterin) [51][27]. The activation of the signal transduction by INF-β results in an antiviral, immunomodulatory, and antiproliferative effect [52][28].
With respect to the immunomodulatory impact, the following underlying mechanisms are considered (Figure 31B):
- (a)
-
IFN-β leads to a reduction of dendritic cells and down-regulates the antigen presentation by APCs in peripheral blood and in the CNS by microglia and monocytes.
- (b)
-
The expression of toll-like receptor (TLR) 3, TLR7, and myeloid differentiation primary response 88 (MyD88) on dendritic cells increases, which leads to an altered immune response.
- (c)
-
INF-β induces CD4+, CD8+, CD25+, FOXP3+, and FOXA1+ T cells (regulatory T cells). A reduced inflammatory T cell response is observed by inhibiting the stimulation and activation of T cells (e.g., by modulation of co-stimulating molecules on dendritic cells), inhibition of the expression of MHCII molecules, and co-stimulating factors like CD80 and CD28 on APC [53,54][29][30].
- (d)
-
The secretion of cytokines and chemokines is altered during IFN-β treatment (interleukin (IL)-10 and IL-4 increased; IL-2 and TNFα decreased). The differentiation of CD4+ cells shift from Th1 to a Th2 phenotype; thereby, resulting in a less pro-inflammatory but more anti-inflammatory cytokine milieu [55][31].
- (e)
-
The number of Th17 cells also decreases, leading to a reduction of IL-17 release and induction of apoptosis of autoreactive T cells [56,57][32][33].
- (f)
-
The effects on cytokines, chemokines, matrix metalloproteinase, and adhesion molecules (especially very late antigen [VLA]-4 on T cells) result in a reduced leukocyte migration via the blood-brain barrier into the CNS [53,58,59][29][34][35].IFN-β-1a treatment results in selective, time-dependent effects on many cell populations in peripheral blood [60][36]. The IFN-β-promotes down-regulation of pro-inflammatory CD4+, CD8+ memory T cells, and memory B cells accompanied by an increase in regulatory T cells [52,53,58,61][28][29][34][37].The majority of patients treated with IFN-β exhibit a fall in absolute lymphocyte counts of approximately 20–30% compared to the baseline value. About 15% of interferon-treated patients develop lymphocyte decreases below the lower limit of normal, 3.5% below 0.8 GPt/L, and about 1% of patients below 0.5 GPt/L [62][38]. The drop in lymphocyte count is often transient and recovers to normal levels within months. During a study evaluating the dynamics of lymphopenia during IFN-β treatment, onset of cytopenia occurred within the first 6 months of therapy in at least two-thirds of patients [62][38]. The majority of events were mild and generally resolved within 3–4 months while continuing therapy. Dose reductions were uncommon, and only a small proportion of patients (6 of 727; 0.8%) discontinued treatment after approximately 2 years because of hematological abnormalities when receiving the highest dose of INF-β-1a (44 μg three times weekly).
Recommended Monitoring
The rate of severe infections during IFN-β treatment does not seem to be significantly increased [1]. On the contrary, IFN-β has clear antiviral effects. There are no data available with respect to the duration of lymphocyte recovery in the case of lymphopenia. However, if repopulation has not occurred long after treatment discontinuation, hematological diseases should be excluded.A regular check of blood counts including, leukocyte and lymphocyte counts, should be done at least tri-monthly in the course of the first year of therapy (Table 21 and Table 32). Subsequently, laboratory intervals can be increased to once or twice a year in the case of normal blood count levels.Table 1. Recommended lymphocyte thresholds for disease-modifying therapies.Drug Name Recommendations for Lymphocyte Cut-Off Values Ig, immunoglobulin; VLA, very late antigen.Table 2. Recommended monitoring of lymphocyte counts for disease-modifying therapies.Months of Treatment Drug Name Predose 1 2 3 4 5 6 7 8 9 10 11 12 Post Month 12 Oral therapies Dimethyl fumarate Complete blood count every 6–8 weeks in first year of treatment, subsequently every 3–6 months, discontinuation of therapy in case of leukopenia of <3.0 GPt/L or lymphopenia of <0.5 GPt/L, in case of grade 2 lymphopenia (0.5–0.8 GPt/L) continuous control of blood counts and high vigilance for opportunistic infections Oral therapies Dimethyl
fumarateX X X X Teriflunomide X X X every 3–6 months Regular check of blood counts every second month in the first six months, subsequently every three months in the case of normal lymphocyte and leukocyte counts; therapy discontinuation in case of lymphocyte decrease < 0.5 GPt/L Teriflunomide X X X X X X every 3 months Fingolimod
Siponimod
Ozanimod
PonesimodRegular check of blood counts 4 weeks after starting therapy, subsequently in case of normal lymphocyte and leukocyte counts, every 3–6 months; in case of repeated peripheral lymphopenia < 0.2 GPt/L, therapy discontinuation until lymphocyte counts reach levels > 0.6 GPt/L Fingolimod
Siponimod
Ozanimod
PonesimodX a,b X X X X X every 3–6 months Cladribine Regular complete blood count prior to cladribine intake and 2 and 6 months after start of treatment in each treatment year, in case of lymphocytopenia < 0.8 GPt/L, the next cladribine pulse must not be started and active monitoring is required until values increase again; in case of not reaching a lymphocyte count of at least 0.8 GPt/L within 18 months after cladribine start, continuation is not recommended Cladribine X c X X X before initiating treatment in year 2,
2 and 6 months after start of treatment cycle in each year dInjectables Glatiramer
acetateRegular check of blood counts at least 3 monthly in first year of therapy, subsequently once or twice a year; in case of lymphopenia < 0.5 GPt/L discontinuation of therapy Injectables Glatiramer
acetateX X X X X once or twice a year Interferons InterferonsRegular check of blood counts at least 3 monthly in first year of therapy, subsequently once or twice a year; in case of lymphopenia < 0.5 GPt/L discontinuation of therapy X X X X X once or twice a year Infusion therapies Ocrelizumab
OfatumumabRegular check of blood counts 3 monthly, including status of peripheral T and B cell subtypes as well as immunoglobulin levels, relevant humoral immunoglobulin deficiency (Ig < 3 g/L), and significant decrease of CD4+ T cells (<0.250 GPt/L) should be ruled out Infusion
TherapiesOcrelizumab
OfatumumabX X X X X every 3 months Natalizumab Regular check of blood counts every 3–6 months, peripheral increase of absolute leukocyte and lymphocyte count can serve as a biomarker, indicating sufficient VLA-4 antagonism Natalizumab X a X X X X every 3–6 months Alemtuzumab AlemtuzumabRegular complete blood count monthly in the course of at least 48 months after last alemtuzumab application X X X X X X X X X X X X X monthly for at least 48 months after last application CBC: complete blood count; WBC: white blood cells. a Washout period following previous treatment must be sufficient for lymphocyte count recovery. b Recent CBC (within the last 6 months) or after prior therapy discontinuation before treatment initiation. c Lymphocyte counts must be normal before initiating cladribine in year 1 and ≥0.8 GPt/L before initiating cladribine in year 2. If recovery takes >6 months, do not administer further cladribine therapy. If lymphocytes < 0.2 GPt/L, consider anti-herpes prophylaxis during time of grade 4 lymphopenia. If lymphocytes < 0.5GPt/L/L, actively monitor for signs/symptoms suggestive of infection, particularly herpes zoster. If such signs and symptoms occur, initiate anti-infectives as clinically indicated. Consider interruption or delay of cladribine until proper resolution of infection. d If lymphocytes < 0.5 GPt/L, actively monitor until values increase again (treatment course in year 2 may be delayed for ≤6 months to allow for lymphocyte recovery. “X” marks the time of monitoring.Although data regarding immune responses following vaccinations are lacking, it is not thought that humoral or cellular immune response to vaccination during IFN-β treatment might be impaired [63,64][39][40]. Verifying sufficient vaccination response via titer recording should be considered. Vaccinating patients during IFN-β treatment with attenuated live vaccines (e.g., varicella-zoster virus [VZV]) should be carefully considered.2.1.3. Dimethyl Fumarate
General Facts and Clinical Trial Data
Dimethyl fumarate (DMF) has been used to treat psoriasis since 1994. In 2013, it was finally licensed as an oral first-line treatment for MS, after two phase III clinical trials, DEFINE and CONFIRM, demonstrated clinical efficacy of DMF in RRMS by reducing the ARR and the mean number of new or enlarging MRI lesions throughout the course of the study [65,66,67][41][42][43]. DMF-treated patients receive two single daily doses of 240 mg. Common side effects are gastrointestinal complaints and intermittent flushing.Mechanism of Action and Impact on Lymphocyte Count
Although the precise mechanism of action is not completely characterized, there are currently at least five main mechanisms for the general action of DMF and its active metabolite, monomethylfumarate (MMF), that have been described so far (Figure 31A). These include:- (a)
-
The activation of the nuclear factor erythroid-derived 2-like 2 (Nrf2) transcriptional pathway, which mediates the regulation of cellular antioxidant responses and stimulation of cytoprotective and anti-inflammatory factors such as heme oxygenase-1 (HO-1) [68,69][44][45];
- (b)
-
The regulation of cellular responses to oxidative stress via binding of DMF/MMF to thiol groups of glutathione (GSH) and therefore influencing intracellular GSH levels [70,71][46][47];
- (c)
-
The direct and indirect inhibition of NF-κB activity by DMF leading to altered cytokine production by APC, to the inhibition of Th1/Th17 responses and promotion of Th2 responses [72,73][48][49];
- (d)
-
The modulation of oxidative stress-sensitive transcription factors, hypoxia-inducible factor-1α (HIF-1α), and STATs by DMF mediating the inhibition of their regulated genes [70,74,75][46][50][51];
- (e)
-
Agonism of the hydroxy-carboxylic acid receptor 2 (HCA2) by MMF promoting the formation of cyclooxygenase-1 (COX-1) and prostaglandin E2 (PGE2) and the inhibition of neutrophil recruitment [76,77][52][53].In both the DEFINE and CONFIRM studies, lymphocyte counts in DMF-treated patients declined by approximately 30% during the first year of treatment and remained stable thereafter [78][54]. Grade 3 lymphopenia, with <0.5 GPt/L, was seen in about 6% of the patients receiving DMF [67,79][43][55].Unlike other DMTs in MS, a DMF-driven fall in lymphocyte count does not appear rapidly; however, it is often present within the first six months of DMF intake [46][22]. Despite the short pharmacological half-life of DMF, after therapy cessation, full lymphocyte regeneration takes several weeks or months in the majority of patients. The precise mechanism of fumaric acid-promoted lymphopenia is still unknown, but until now, apoptotic processes and depletion of lymphocytes have been assumed [80][56]. Among others, in vitro studies have shown that DMF induces T cell apoptosis with a preferential effect on memory T cells. Furthermore, DMF induces concentration-dependent apoptosis of B cells from healthy controls, with B cells of MS patients appearing to be more vulnerable [80][56]. During early DMF treatment, the dynamics of lymphocyte subsets change in the following way: B cell counts initially experience the greatest rate and proportion of decline, detected as early as four weeks after treatment initiation. By week eight, reduced circulating numbers of CD4+ and CD8+ T and NK cells can be observed. Consistent with the pattern in B cells, the decline in NK cell counts appears to stabilize after 12 weeks, remaining below normal, whereas CD4+ and CD8+ T cells counts continue to decline from baseline to week 24, whereby CD8+ T cells have the greatest median percentage reduction [81][57]. Overall, a significant reduction in the absolute counts of functional subsets can be observed at week 24, with the greatest median percentage reduction from baseline in T and B cell memory populations and the least effect on naive T and B cell subsets [81][57]. As studies show, early absolute lymphocyte count drop is associated with later development of severe, prolonged lymphopenia (<0.5 GPt/L for >6 months) while on treatment [66,67,82][42][43][58]. Known risk factors of this phenomenon are older age (>55 years), lower baseline absolute lymphocyte count, and recent natalizumab treatment [65][41].
Recommended Monitoring
After starting DMF treatment, a complete blood count should be performed every 6–8 weeks, as lymphocyte decline can be expected during the first weeks (Table 32). Less frequent monitoring of blood count is then needed in a 3–6 month interval, as lymphocyte counts normally remain stable after month 12. In the case of leukopenia of <3.0 GPt/L or lymphopenia of <0.5 GPt/L, DMF therapy should be discontinued (Table 21). In the case of grade 2 lymphopenia (0.5–0.8 GPt/L), continuous monitoring of blood counts and high vigilance for opportunistic infections, especially the development of PML, are required [1].Although approval studies showed no increased risk of infection during DMF therapy in general, real-world data presented several cases of PML when taking fumaric acid derivates. Up to 11/2015 four PML cases, after long-duration DMF treatment, appeared without previous immunosuppression or other crucial immuno-compromising factors. As all of these patients were 50–70 years old, there seems to be an age-dependent effect. Furthermore, DMT-associated lymphopenia appears to develop predominantly in elderly patients. The role of lymphopenia in DMF-associated PML is not yet fully understood. While three of the DMF-treated patients with PML had lymphopenia < 0.5 GPt/L intermediately, one DMF-associated PML case only developed enduring grade 2 lymphopenia of about 0.6 GPt/L [83][59]. Although the causal relationship between DMF and the development of PML is not completely understood, continuous lymphopenia is a well-known risk factor for PML in general [83][59]. Nonetheless, it appears that PML can occur during DMF therapy even with moderate lymphopenia [84][60], requiring careful and frequent monitoring of blood counts.Limitations of vaccination in DMF-treated patients have not yet been evaluated [1]. However, the risk of attenuated live vaccines during DMF treatment should be thoughtfully weighed. To this date, there are no data for increased malignancy risk during DMF long-term therapy.2.2. Mechanism of Action: Target Lymphocyte Proliferation
Teriflunomide
General Facts and Clinical Trial Data
Teriflunomide is an active metabolite of the prodrug leflunomide, which has been used in the treatment of rheumatoid arthritis as a DMT since 1988. In 2012, it was approved for the treatment of RRMS in the US (7 and 14 mg daily) and in 2013, in Europe (14 mg daily) [85][61] after efficacy and safety of teriflunomide were confirmed in the phase II trial, TEMSO and phase III clinical trials, TOWER and TENERE. Treatment significantly reduced ARR in MS patients by about 34% compared with placebo [86,87][62][63]. The simple application scheme of a once-daily oral intake led to a broad acceptance and compliance in patients, although typical adverse events like hair thinning, arthralgia, paresthesia, and persistent gastrointestinal complaints including nausea, diarrhea, or elevation of transaminases could be limiting.Mechanism of Action and Impact on Lymphocyte Count
Teriflunomide reversibly inhibits the dihydroorotate dehydrogenase (DHODH)—a mitochondrial enzyme involved in de novo pyrimidine synthesis and DNA replication of highly proliferating T and B lymphocytes (Figure 31A). By reducing pyrimidine de novo synthesis, the proliferation of activated B and T cells declines without prompting cell death. As resting T lymphocytes use nucleotides from degraded DNA and RNA, their survival does not depend on an intact DHODH function. The immunological protection against pathogens is thus ensured, whereas the damaging proliferation of activated autoimmunity driving B and T cells is reduced. In this way, a shift to regulatory T cell subtypes and a reduction in clonal diversity in the CD4+ T cell repertoire can be observed [88][64]. As teriflunomide crosses the blood-brain barrier [88][64], it might also be able to reduce microglia proliferation and induce the production of anti-inflammatory interleukins like IL-10 by microglia, which has been shown in vitro studies [89][65].Besides the anti-proliferating effect, both leflunomide and teriflunomide inhibit the production of IL-17, TNFα, protein tyrosine kinases, the NFκB-pathway, and the immunoglobulin G (IgG) secretion of activated B cells [85,90,91][61][66][67]. Furthermore, teriflunomide induces apoptosis of Epstein-Barr virus (EBV)-transformed B cells [92][68] and seems to reduce glutamate levels and endotoxicity in the CNS [93][69]. Another favorable effect is the promotion of oligodendrocytic differentiation, the amelioration of axonopathy by attenuating CD8+ T cell cytotoxicity and supporting the proliferation of regulatory CD8+ T cells in the CNS [94[70][71],95], which facilitates neuroregeneration.The effect on circulating lymphocytes appears within the first six weeks of treatment [96][72]. The selective and reversible inhibition of mitochondrial DHODH results from targeted inhibition of proliferating lymphocytes in a decline of CD4+ and CD8+ T cells, memory B cells, and NK cells [97][73]. However, teriflunomide is rarely associated with lymphopenia and neutropenia. When it occurs, it is mostly mild and reversible during ongoing therapy or after discontinuation. In the patient populations of the TEMSO, TOWER, and TENERE studies, there was an overall decline in absolute lymphocyte counts from week 0 (1.89 GPt/L) to week 24 (1.67 GPt/L), remaining stable thereafter [98][74]. Mean counts generally remained above the lower limits of normal; however, grade 1 and 2 lymphopenia occurred in 7.3% and 2.2% of patients, respectively. No cases of grade 3 or 4 lymphopenia were reported in the pooled core studies [98][74]. The median treatment duration with teriflunomide prior to the development of lymphopenia was 17.9 weeks for grade 1 and 20.4 weeks for grade 2. The prevalence of lymphopenia during teriflunomide declined over time (up to 10.7 years follow-up); most events occurred in the first year of treatment. The median time to recovery from grade 1 lymphopenia during teriflunomide treatment was 10.6–11.1 weeks, and for patients with grade 2 lymphopenia, 16.6–49.9 weeks [98][74]. 2.3% of patients exposed to teriflunomide had grade 1 lymphopenia lasting longer than six months. The duration of grade 2 lymphopenia, however, did not persist for longer than six months.Recommended Monitoring
A complete blood count should be done every second month in the course of the first six months after starting teriflunomide therapy (Table 32). Subsequently, laboratory intervals can be increased to every three months in the case of normal lymphocyte and leukocyte counts. In the event of a lymphocyte decrease to <0.5 GPt/L, teriflunomide therapy should be discontinued (Table 21). In the rare case of critical lymphopenia and/or opportunistic infections during teriflunomide therapy, an accelerated elimination should be achieved with the oral administration of a bile acid sequestrant (e.g., 8 g cholestyramine three times daily for 11 days) as teriflunomide serum levels are detectable up to two years after discontinuation due to enterohepatic recirculation. The rate of severe infections with teriflunomide therapy is 1.4% (7 mg) and 2.2% (14 mg), respectively, versus 2.1% for placebo [96][72]. Patients treated with teriflunomide were able to mount sufficient immune response to vaccines, which, however, tended to be weaker than those in placebo-treated patients [99][75]. Immunization via live attenuated vaccines should be avoided.2.3. Mechanism of Action: Target Lymphocyte Migration
2.3.1. Sphingosine-1-Phosphate Receptor Modulation
General Facts and Clinical Trial Data
Fingolimod was the first sphingosine-1-phosphate (S1P)-receptor modulating agent approved in the USA in 2010 for relapsing MS after two phase III trials (FREEDOMS and TRANSFORMS) demonstrated potent efficacy, safety, and tolerability. Whereas fingolimod, as an unselective S1P receptor antagonist binds to four of the five known S1P receptors (S1PR1–5) and therefore exhibits a higher risk for adverse events (bradyarrhythmia, atrioventricular blocking, macular edema), second generation agents siponimod and ozanimod demonstrate favorable selectivity towards S1P1 receptors. A further S1PR1-selective agent, ponesimod, was recently approved in 2021. Siponimod is the first potential oral therapy for secondary progressive (SP) MS as the phase III trial EXPAND demonstrated a significant reduction in disability progression in SPMS patients compared with placebo [100][76]. Ozanimod and ponesimod both broaden the therapy range for active relapsing MS.Mechanism of Action and Impact on Lymphocyte Counts
Fingolimod, siponimod, ozanimod, and ponesimod are structural analogs of natural sphingosine phosphate [101][77]. In a phosphorylated state, fingolimod binds to four of the five known S1P receptors (S1PR1 and S1PR3–5) [102,103][78][79]. Siponimod, ozanimod, and ponesimod exhibit selective affinity for type 1 and 5 of the S1P receptors, leading to a lower risk of adverse events, such as bradycardia and vasoconstriction, mainly induced by S1PR3 activation.Binding with high affinity to S1PR1 expressed on lymphocytes, lymphocyte egress from lymphoid tissues into the peripheral compartment is inhibited by all approved S1P receptor modulators, preventing the infiltration of auto-aggressive lymphocytes into the CNS (Figure 31A) [104,105,106][80][81][82]. Initial receptor activation is, paradoxically, followed by S1PR1 functional antagonism. Accordingly, receptors are internalized and degraded, thus rendering lymphocytes unresponsive to the normal S1P gradient, which represents the obligatory signal that would ordinarily allow them to egress from lymphoid tissues [105,107,108][81][83][84]. Additionally, binding to S1P receptors expressed in the CNS (S1PR1/5) promotes a modulating effect on neurogenesis, neural function, and migration [109,110][85][86]. Fingolimod binds to S1PR1/3 on smooth muscle and endothelial cells, which influences vascular homeostasis and vascular permeability. Furthermore, fingolimod induces a negative chronotropic effect via S1PR on atrial myocytes [111,112][87][88].As S1PR modulators inhibit CCR7+ lymphocyte egress from secondary lymphoid organs, resulting in a profound decrease in naive and central memory T cells and memory B cells in the periphery [113,114][89][90]. Treatment with fingolimod significantly decreases the absolute numbers of all major lymphocyte subsets, except for NK cells. The reduction is most pronounced within T helper and B cell populations [115][91]. Dramatic reductions within the naïve and central memory T cell populations can be found [115][91]; the reduction is less pronounced among effector memory cells. The number of regulatory T cells (Tregs) also decreases, but to a lesser extent than other T cell populations, resulting in a relative preservation of Tregs with a memory phenotype [115][91]. In summary, within T cells, naïve and central memory cells are most profoundly affected by a fingolimod-induced reduction, whereas memory Tregs are relatively preserved.A dose-dependent decrease in total peripheral lymphocytes by 70–80% can be observed, and most fingolimod-treated patients reach grade 2–4 lymphopenia after starting therapy. Grade 4 lymphopenia is a common adverse event occurring in 15–20% of patients [116,117][92][93]. In a German and Swedish cohort of fingolimod-treated patients with a low baseline lymphocyte count, women with a low body mass index were suggested to have a higher risk of lymphopenia [116][92]. A history of treatment with any IFN-β was significantly more frequent in patients who experienced lymphopenia [118][94]. This is because the IFN-β family influences the production of cytokines by lymphocytes and are considered to be related to myelosuppressive activities [119][95]. A study by Ohtani et al. showed that a low lymphocyte count at baseline and a treatment history of any IFN-β therapy is associated with grade 4 lymphopenia during fingolimod treatment [118][94]. Lymphocytes and their subsets return to the normal range around 1–2 months after treatment discontinuation [120][96]. Different studies discuss efficacy depending on T and B cell decreases during fingolimod therapy. Current real-world data show a wide range of peripheral lymphocyte counts during treatment, depending on the individual distribution of CD4+ and CD8+ T cells, CD19+ B cells, and NK cells. While peripheral CD4+ T cells and CD19+ B cells are markedly reduced by S1PR1 therapy, CD8+ T cells and NK cells are less affected and less relevant to variations in lymphocyte counts in individual patients [21][97].It is assumed that withdrawal of fingolimod results in overexpression of lymphocytic S1PR1 leading to lymphocyte egress from lymph nodes and promoting disease rebound after treatment discontinuation [121][98]. Autopsy results from a patient who died after severe rebound relapse revealed increased S1PR1 immunoreactivity on hypertrophic astrocytes in tumefactive plaques, indicating that the withdrawal of fingolimod results in astrocytic overexpression of S1PR1 [122,123][99][100]. Due to the increased risk of more intense lymphopenia during fingolimod therapy, different treatment regimen alternatives have been assessed. However, the change from conventional therapy to intermittent dosing carries a risk of rebound, and the efficacy of an alternate-day fingolimod administration was not effective enough to inhibit disease activity [124,125][101][102].Siponimod leads to a dose-dependent reduction of peripheral lymphocytes to 20–30% of baseline (median nadir approximately 0.56 GPt/L), with a recovery to the normal range within 10 days in 90% of patients after treatment discontinuation [126][103]. However, in some patients, lymphocyte recovery can take up to 3–4 weeks. In the pivotal phase III EXPAND study, grade 4 lymphopenia was observed in 1% of patients [126][103].There are insufficient real-world data of lymphocyte count during ozanimod treatment. Combining data from the RADIANCE and SUNBEAM trials enabled a comparison of ozanimod to fingolimod, and analysis showed a higher absolute mean lymphocyte count (difference in means 0.4 GPt/L) during ozanimod treatment compared with fingolimod treatment [127][104]. During the RADIANCE study, ozanimod treatment led to dose-dependent suppression of absolute lymphocyte counts to <0.2 GPt/L in four participants (3.3%). These reductions were transient and not associated with infections or treatment discontinuation [128][105]. Early clinical studies of ponesimod therapy show an overall reduction of absolute lymphocyte count, compared to baseline, of about −1.3 GPt/L. Ponesimod treatment led to a marked reduction in overall T and B cell counts. Specifically, the number of CD4+ cells showed a significant drop, whereas CD8+ and NK cells were less affected [129][106]. Similar to siponimod and ozanimod, reliable real-world data for ponesimod are not yet available due to recent regulatory approval.Taken together, data on studies of siponimod, ozanimod, and ponesimod show a lower risk of higher-grade lymphopenia than for fingolimod, and this might be considered when selecting treatment alternatives where the desire is for fewer side effects.Recommended Monitoring
Before starting treatment with S1PR-modulators, chronic active infections should be excluded. Specifically, VZV status should be defined, and the evaluation of hepatitis B, C, and HIV should be considered. In the absence of VZV antibodies, patients should be immunized with VZV vaccine prior to treatment, which can be started four weeks after vaccination at the earliest.Four weeks after the commencement of S1PR-modulators, a complete blood count should be performed (Table 32). Subsequent laboratory intervals can be increased to 3–6 months in the case of normal lymphocyte and leukocyte counts. In the case of peripheral lymphopenia < 0.2 GPt/L (confirmed by a second test after two weeks), S1PR therapy should be discontinued until lymphocyte counts reach levels > 0.6 GPt/L (Table 21).In the case of acute infection, diagnostic and therapeutic measures should be adopted immediately, especially concerning viral herpetic infections (e.g., VZV infection or reactivation, Herpes simplex virus (HSV)-encephalitis), mycotic (e.g., cryptococcal meningitis), or bacterial infections (e.g., atypical mycobacteria). A higher risk of infections can be assumed considering the underlying mechanism of action. However, trial results suggest that for S1PR modulators, there is no direct correlation between absolute peripheral lymphocyte count and the likelihood of infective complications [117][93].The risk of PML during S1P receptor modulator therapy is lower than that for natalizumab [130][107]. In most of the known cases, a ‘carry over’ mechanism following prior natalizumab therapy is assumed. There was no correlation to peripheral lymphopenia [130][107]. Frequent MRIs should be performed to assess the potential risk of PML, in addition to standard MRI MS monitoring. Regular evaluation of JCV-serostatus should be considered.Efficacy of vaccination can be limited during, and up to two months after, therapy discontinuation. Immunization with live, attenuated vaccines should be avoided during S1PR modulator therapy.2.3.2. Natalizumab
General Facts and Clinical Trial Data
Natalizumab was the first monoclonal antibody approved for the treatment of RRMS in 2004. The efficacy of natalizumab (300 mg i.v. every four weeks) has been demonstrated in two phase III trials (AFFIRM, SENTINEL) [131][108]. Despite the significant reduction in both relapse rate and the number of new T2 or gadolinium-enhancing MRI lesions [131[108][109],132], it did not achieve inhibition of disability progression [133][110]. After temporary withdrawal because of an accumulation of PML cases, natalizumab is available for the treatment of highly active RRMS, with consideration of PML predisposing risk factors.Mechanism of Action and Impact on Lymphocyte Counts
Natalizumab is a humanized recombinant IgG antibody that impairs leukocyte extravasation into the CNS and intestinal tract by blocking the alpha-4 subunit of the integrin molecules on leukocytes [134][111]. By inhibiting the interaction with the endothelial vascular cell adhesion molecule (VCAM)1 lymphocytes are not able to cross the blood-brain barrier, and inflammation in the CNS compartment is reduced (Figure 31C) [135][112]. As natalizumab is an IgG4 antibody, the binding does not result in lysis or destruction of the target cells, e.g., by complement factors [136][113].In the brain tissue compartment, however, natalizumab leads to a significant reduction in CD4+ cells in cerebrospinal fluid, resulting in a reduction of the CD4+/CD8+ ratio [137,138][114][115] that is detectable up to 6 months after treatment cessation. In addition to inhibiting the migration of CD4+ cells into the CNS, natalizumab has other anti-inflammatory effects. These include a significant decrease of APC and dendritic cells in the perivascular space as well as the down-regulation of surface expression markers MHCII, which might also contribute to the long-lasting effect on CD4+ cell counts in the CNS [139][116]. Unlike other known DMTs, the administration of natalizumab leads to an increase in CD4+, CD8+ T cells, CD19+ B cells, and NK cells in serum without relevant effects on the CD4+/CD8+ ratio in peripheral blood, but with a reduction of this ratio in the CNS [140,141][117][118]. The increase of absolute lymphocyte counts in serum results from an increased release of CD34+ promotor cells from the bone marrow, on the one hand, and the impaired lymphocytes egress from the vessel into the brain tissue, on the other. The natalizumab-induced increase of peripheral lymphocytes stabilizes 3–6 months after starting treatment [140][117] and lasts up to 6 months after discontinuation [142][119].Recommended Monitoring
During natalizumab therapy, a complete blood count should be performed every 3–6 months (Table 21 and Table 32). The peripheral increase of absolute leukocyte and lymphocyte count serves as a robust biomarker, indicating a sufficient VLA-4 antagonism [143,144][120][121]. In the case of clinical or subclinical disease activity, a lack of increase in lymphocyte cell count may indicate the appearance of neutralizing antibodies against natalizumab.The altered immune-cellular milieu observed in the CNS up to 6 months after stopping natalizumab should be considered when changing therapy regimen, especially when switching to an immunodepletion therapy mechanism. However, severe rebound disease activity is a known phenomenon, especially longer than 3 months after natalizumab discontinuation. Taking into consideration the well-known dynamics of CNS cell changes after interruption of natalizumab therapy, the interval preceding immunodepletion therapy should be as long as possible but as short as necessary. Frequent MRI to assist individual decision-making for lumbar puncture can help detect the early return of disease progression versus PML in this wash-out period. In this context, vanishing lymphocytosis prior to the end of natalizumab therapy should raise awareness of neutralizing antibodies as a possible cause of sudden disease progression.Although two studies did not show significant differences in the vaccine-specific antibody responses to several types of immunization, three other studies revealed restricted immune response following influenza vaccination in natalizumab-treated patients compared to healthy controls [145,146][122][123]. Immunization via live, attenuated vaccines should be avoided during natalizumab treatment.2.4. Lysis of Specific Lymphocytes Subsets
2.4.1. B Cell Depletion
General Facts and Clinical Trial Data
We have learned that many of the underlying inflammatory processes in MS pathology appear to be B cell-mediated, evidenced by the development of oligoclonal bands in the CSF, the role of antigen presentation, antibody production, pathogenic cytokine release, and the formation of meningeal ectopic lymphoid tissues. These findings suggest that B cell depletion could be an effective treatment strategy for MS [147][124]. Ocrelizumab is a half-yearly intravenous humanized anti-CD20 monoclonal IgG1 antibody, which is approved for the treatment of active relapsing MS or primary progressive MS (PPMS). In clinical trials, ocrelizumab significantly reduced ARR relative to IFN-β-1a in RRMS patients and decreased the risk of disability progression relative to placebo in patients with PPMS (OPERA I + II, ORATORIO) [148,149][125][126]. Since the beginning of 2021, subcutaneous monthly ofatumumab—a humanized anti-CD20 monoclonal IgG1 antibody—has complemented the range of B cell-depleting therapies for MS.Mechanism of Action and Impact on Lymphocyte Count
Ocrelizumab binds to the surface CD20-molecules and selectively depletes CD20-expressing B cells through antibody-dependent cell-mediated cytotoxicity, antibody-dependent cellular phagocytosis, complement-dependent cytotoxicity, and apoptosis (Figure 31C) [150,151][127][128]. The resulting decrease in the number and function of B cells promotes the chief immunomodulatory effect of ocrelizumab. However, additional, poorly understood mechanisms may also contribute to its clinical benefits [150][127]. As CD20 is expressed on pre-, mature, and memory B cells, but not on lymphoid stem cells, pre-existing humoral immunity due to plasma cells is preserved during ocrelizumab therapy [150][127]. Furthermore, innate [150][127] and adaptive [152][129] immunity remain unaffected after B cell depletion.
